Valorization of Coffee Silverskin through Subcritical Water Extraction: An Optimization Based on T-CQA Using Response Surface Methodology
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chemicals and Reagents
2.3. Extraction Procedure
2.4. Experimental Design
2.5. Statistical Analysis
2.6. Validation of the Optimized Conditions
2.7. Measurement of the Total Phenolic Content
2.8. Measurement of Antioxidant Activity
2.9. Quantitative Analysis of Total Chlorogenic Acid by HPLC-UV
3. Results
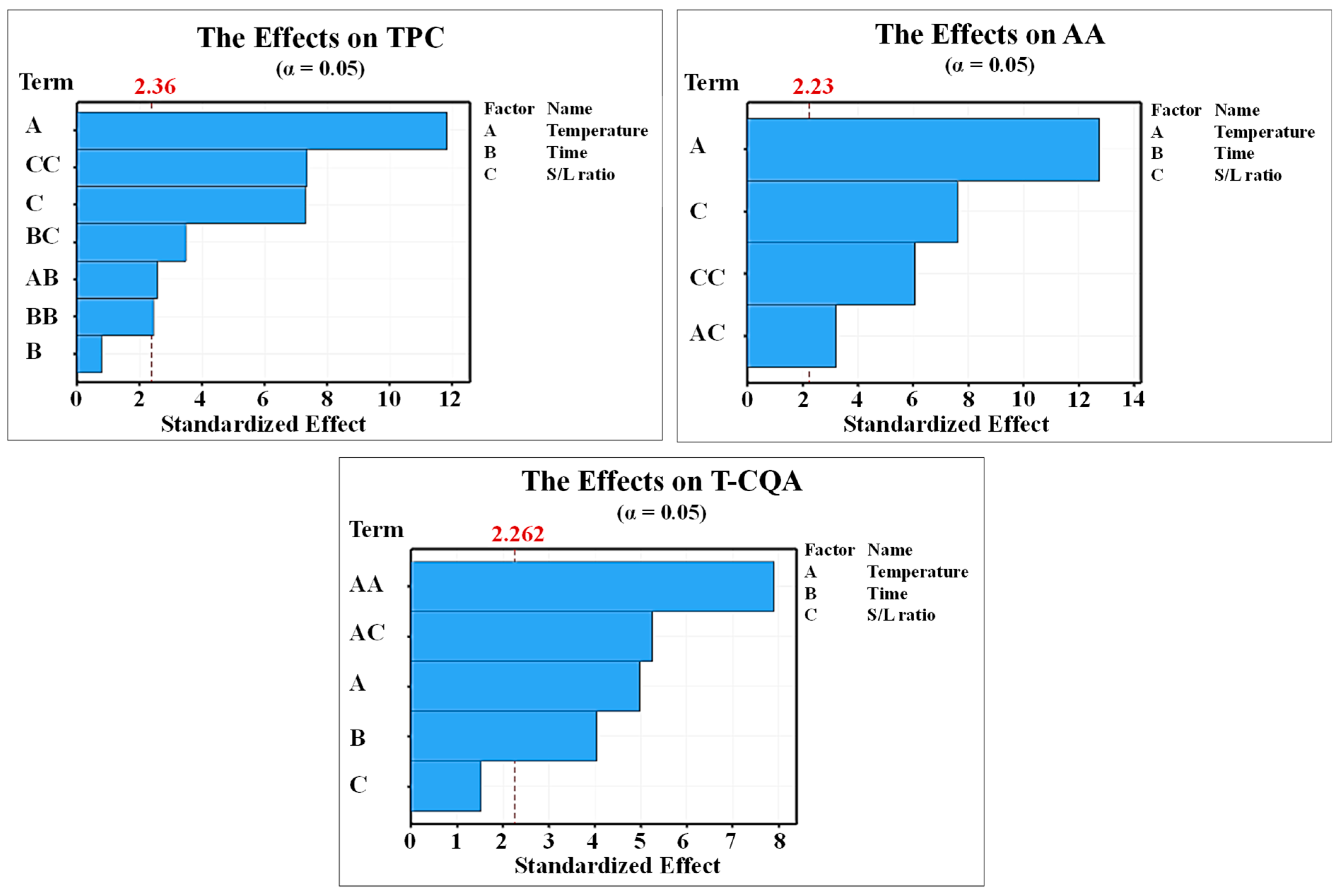
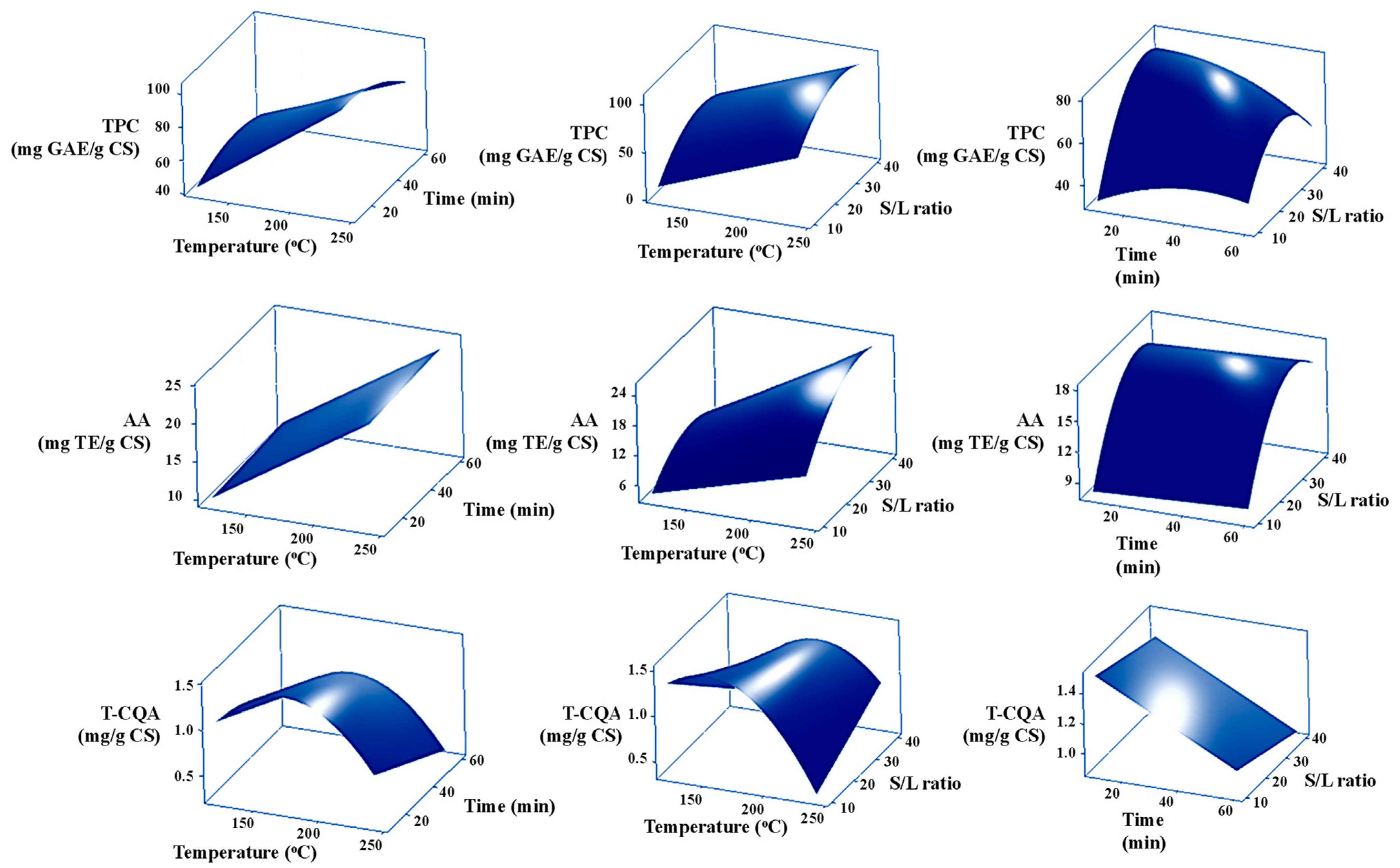
3.1. Effect of Extraction Conditions on the TPC
3.2. Effect of Extraction Conditions on the AA
3.3. Effect of Extraction Conditions on the T-CQA
3.4. Regression Modeling of the SWE Conditions on TPC, AA, and T-CQA
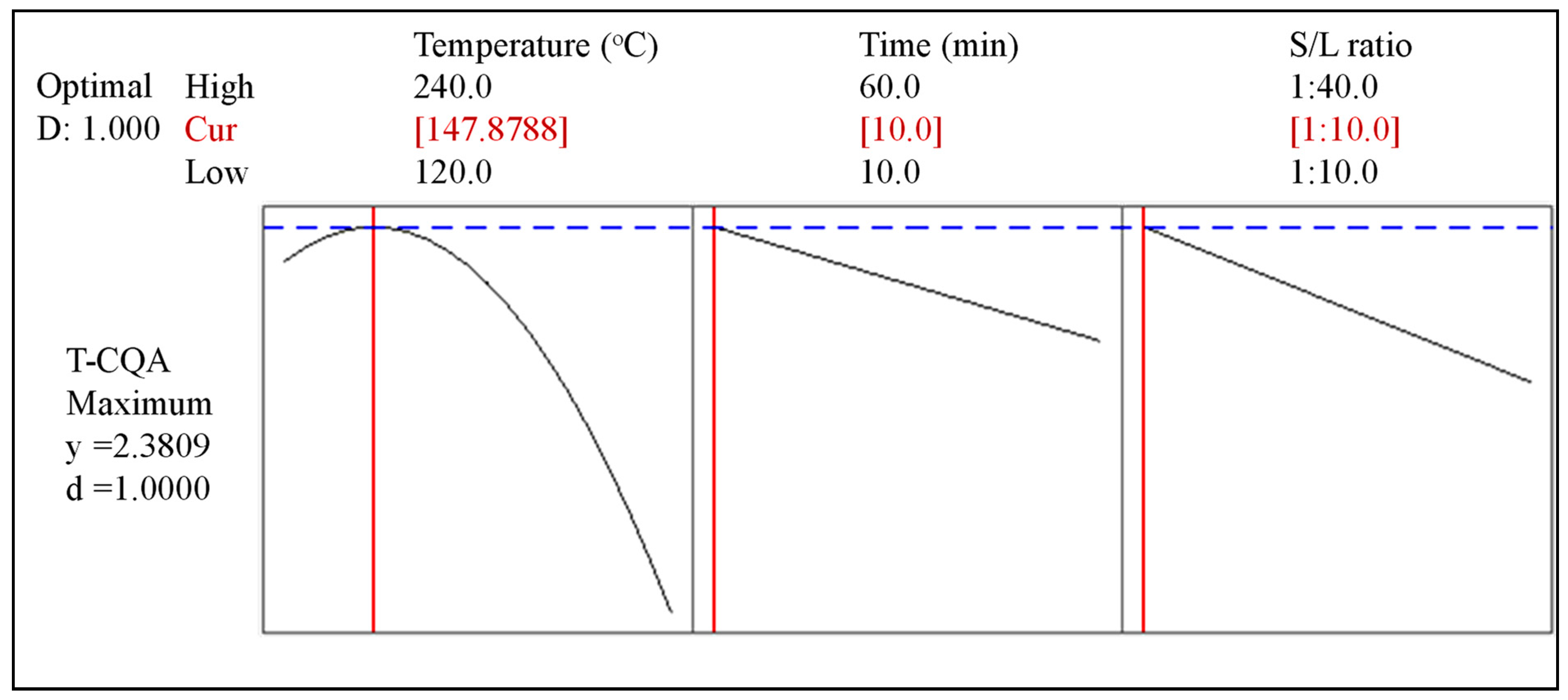
3.5. Optimal Extraction Conditions and Model Validation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- International Coffee Organization. 2021. Available online: https://www.ico.org/prices/new-consumption-table.pdf (accessed on 7 June 2022).
- International Coffee Organization. 2021. Available online: https://www.ico.org/prices/m1-exports.pdf (accessed on 28 November 2021).
- Mussatto, S.I.; Machado, E.M.S.; Martins, S.; Teixeira, J.A. Production, Composition, and Application of Coffee and Its Industrial Residues. Food Bioprocess Technol. 2011, 4, 661–672. [Google Scholar] [CrossRef] [Green Version]
- Jiménez-Zamora, A.; Pastoriza, S.; Rufián Henares, J. Revalorization of coffee by-products. Prebiotic, antimicrobial and antioxidant properties. LWT-Food Sci. Technol. 2015, 61, 12–18. [Google Scholar] [CrossRef]
- Janissen, B.; Huynh, T. Chemical composition and value-adding applications of coffee industry by-products: A review. Resour. Conserv. Recycl. 2017, 128, 110–117. [Google Scholar] [CrossRef]
- Manakitsomboon, H. Total Consumption of Coffee in Thailand from 1990 to 2019. 2020. Available online: https://www.statista.com/statistics/314998/thailand-total-coffee-consumption/ (accessed on 28 November 2021).
- Blinová, L.; Sirotiak, M.; Bartošová, A.; Soldán, M. Review: Utilization of waste from coffee production. Res. Pap. 2017, 25, 91–102. [Google Scholar] [CrossRef] [Green Version]
- Costa, A.S.G.; Alves, R.C.; Vinha, A.F.; Costa, E.; Costa, C.S.G.; Nunes, M.A.; Almeida, A.A.; Santos-Silva, A.; Oliveira, M.B.P.P. Nutritional, chemical and antioxidant/pro-oxidant profiles of silverskin, a coffee roasting by-product. Food Chem. 2018, 267, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Iriondo-dehond, A.; Iriondo-dehond, M.; Del Castillo, M.D. Applications of Compounds from Coffee Processing By-Products. Biomolecules 2020, 10, 1219. [Google Scholar] [CrossRef] [PubMed]
- Ballesteros, L.F.; Teixeira, J.A.; Mussatto, S.I. Chemical, Functional, and Structural Properties of Spent Coffee Grounds and Coffee Silverskin. Food Bioprocess Technol. 2014, 7, 3493–3503. [Google Scholar] [CrossRef] [Green Version]
- Costa, A.S.G.; Alves, R.C.; Vinha, A.F.; Barreira, S.V.P.; Nunes, M.A.; Cunha, L.M.; Oliveira, M.B.P.P. Optimization of antioxidants extraction from coffee silverskin, a roasting by-product, having in view a sustainable process. Ind. Crops Prod. 2014, 53, 350–357. [Google Scholar] [CrossRef]
- Bessada, S.M.F.F.; Alves, R.C.; Oliveira, M.B.P.P.P. Coffee silverskin: A review on potential cosmetic applications. Cosmetics 2018, 5, 5. [Google Scholar] [CrossRef] [Green Version]
- Alves, R.C.; Rodrigues, F.; Antónia Nunes, M.; Vinha, A.F.; Oliveira, M.B.P.P. Oliveira, State of the art in coffee processing by-products. In Handbook of Coffee Processing By-Products; Elsevier Inc.: Amsterdam, The Netherlands, 2017; ISBN 9780128112915. [Google Scholar]
- Bresciani, L.; Calani, L.; Bruni, R.; Brighenti, F.; Del Rio, D. Phenolic composition, caffeine content and antioxidant capacity of coffee silverskin. Food Res. Int. 2014, 61, 196–201. [Google Scholar] [CrossRef]
- Mesías, M.; Navarro, M.; Martínez-Saez, N.; Ullate, M.; del Castillo, M.D.; Morales, F.J. Antiglycative and carbonyl trapping properties of the water soluble fraction of coffee silverskin. Food Res. Int. 2014, 62, 1120–1126. [Google Scholar] [CrossRef] [Green Version]
- Narita, Y.; Inouye, K. Review on utilization and composition of coffee silverskin. Food Res. Int. 2014, 61, 16–22. [Google Scholar] [CrossRef] [Green Version]
- Ballesteros, L.F.; Teixeira, J.A.; Mussatto, S.I. Selection of the Solvent and Extraction Conditions for Maximum Recovery of Antioxidant Phenolic Compounds from Coffee Silverskin. Food Bioprocess Technol. 2014, 7, 1322–1332. [Google Scholar] [CrossRef] [Green Version]
- Narita, Y.; Inouye, K. High antioxidant activity of coffee silverskin extracts obtained by the treatment of coffee silverskin with subcritical water. Food Chem. 2012, 135, 943–949. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Borrelli, R.C.; Esposito, F.; Napolitano, A.; Ritieni, A.; Fogliano, V. Characterization of a New Potential Functional Ingredient: Coffee Silverskin. J. Agric. Food Chem. 2004, 52, 1338–1343. [Google Scholar] [CrossRef] [PubMed]
- Nzekoue, F.K.; Angeloni, S.; Navarini, L.; Angeloni, C.; Freschi, M.; Hrelia, S.; Vitali, L.A.; Sagratini, G.; Vittori, S.; Caprioli, G. Coffee silverskin extracts: Quantification of 30 bioactive compounds by a new HPLC-MS/MS method and evaluation of their antioxidant and antibacterial activities. Food Res. Int. 2020, 133, 109–128. [Google Scholar] [CrossRef] [PubMed]
- Cheng, B.; Furtado, A.; Smyth, H.E.; Henry, R.J. Influence of genotype and environment on coffee quality. Trends Food Sci. Technol. 2016, 57, 20–30. [Google Scholar] [CrossRef] [Green Version]
- Iriondo-Dehond, A.; Rios, M.B.; Herrera, T.; Rodriguez-Bertos, A.; Nuñez, F.; Andres, M.I.S.; Sanchez-Fortun, S.; Del Castillo, M.D. Coffee silverskin extract: Nutritional value, safety and effect on key biological functions. Nutrients 2019, 11, 2693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tajik, N.; Tajik, M.; Mack, I.; Enck, P. The potential effects of chlorogenic acid, the main phenolic components in coffee, on health: A comprehensive review of the literature. Eur. J. Nutr. 2017, 56, 2215–2244. [Google Scholar] [CrossRef] [PubMed]
- Silva, T.; Oliveira, C.; Borges, F. Caffeic acid derivatives, analogs and applications: A patent review (2009–2013). Expert Opin. Ther. Pat. 2014, 24, 1257–1270. [Google Scholar] [CrossRef] [PubMed]
- Iriondo-DeHond, A.; Martorell, P.; Genovés, S.; Ramón, D.; Stamatakis, K.; Fresno, M.; Molina, A.; Del Castillo, M.D. Coffee silverskin extract protects against accelerated aging caused by oxidative agents. Molecules 2016, 21, 721. [Google Scholar] [CrossRef] [Green Version]
- Fernandez-Gomez, B.; Lezama, A.; Amigo-Benavent, M.; Ullate, M.; Herrero, M.; Martín, M.Á.; Mesa, M.D.; del Castillo, M.D. Insights on the health benefits of the bioactive compounds of coffee silverskin extract. J. Funct. Foods 2016, 25, 197–207. [Google Scholar] [CrossRef] [Green Version]
- Castillo, M.; Fernandez-Gomez, B.; Martinez-Saez, N.; Iriondo-DeHond, A.; Martirosyan, D.; Mesa, M. Coffee Silverskin Extract for Aging and Chronic Diseases. In Functional Foods for Chronic Diseases; Createspace: Scotts Valley, CA, USA, 2016; pp. 386–409. ISBN 978-1536919431. [Google Scholar]
- Magnani, C.; Isaac, V.L.B.; Correa, M.A.; Salgado, H.R.N. Caffeic acid: A review of its potential use in medications and cosmetics. Anal. Methods 2014, 6, 3203–3210. [Google Scholar] [CrossRef]
- Zhang, Q.-W.; Lin, L.-G.; Ye, W.-C. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018, 13, 20. [Google Scholar] [CrossRef] [Green Version]
- Wen, L.; Zhang, Z.; Rai, D.; Sun, D.-W.; Tiwari, B. Ultrasound-assisted extraction (UAE) of bioactive compounds from coffee silverskin: Impact on phenolic content, antioxidant activity, and morphological characteristics. J. Food Process Eng. 2019, 42, e13191. [Google Scholar] [CrossRef]
- Barbosa-Pereira, L.; Guglielmetti, A.; Zeppa, G. Pulsed Electric Field Assisted Extraction of Bioactive Compounds from Cocoa Bean Shell and Coffee Silverskin. Food Bioprocess Technol. 2018, 11, 818–835. [Google Scholar] [CrossRef]
- Guglielmetti, A.; D’ignoti, V.; Ghirardello, D.; Belviso, S.; Zeppa, G. Optimization of ultrasound and microwave-assisted extraction of caffeoylquinic acids and caffeine from coffee silverskin using response surface methodology. Ital. J. Food Sci. 2017, 29, 409–423. [Google Scholar] [CrossRef]
- Özel, M.Z.; Göğüş, F. Subcritical Water as a Green Solvent for Plant Extraction. In Alternative Solvents for Natural Products Extraction; Chemat, F., Vian, M.A., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 73–89. ISBN 9783662436288. [Google Scholar]
- Conde, T.; Mussatto, S.I. Isolation of polyphenols from spent coffee grounds and silverskin by mild hydrothermal pretreatment. Prep. Biochem. Biotechnol. 2016, 46, 406–409. [Google Scholar] [CrossRef] [Green Version]
- Procentese, A.; Raganati, F.; Olivieri, G.; Russo, M.E.; Marzocchella, A. Combined antioxidant-biofuel production from coffee silverskin. Appl. Microbiol. Biotechnol. 2019, 103, 1021–1029. [Google Scholar] [CrossRef] [PubMed]
- Maran, J.P.; Manikandan, S.; Thirugnanasambandham, K.; Nivetha, C.V.; Dinesh, R. Box–Behnken design based statistical modeling for ultrasound-assisted extraction of corn silk polysaccharide. Carbohydr. Polym. 2013, 92, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Soraya, I.; Sulaiman, C.; Basri, M.; Reza, H.; Masoumi, F.; Chee, W.J.; Ashari, S.E. Effects of temperature, time, and solvent ratio on the extraction of phenolic compounds and the anti-radical activity of Clinacanthus nutans Lindau leaves by response surface methodology. Chem. Cent. J. 2017, 11, 54. [Google Scholar] [CrossRef]
- Mathews, P.G. Design of Experiments with MINITAB.; American Society for Quality (ASQ): Milwaukee, WI, USA, 2005; ISBN 9781628703368. [Google Scholar]
- Demirel, C.; Kabutey, A.; Herák, D.; Sedlaček, A.; Mizera, Č.; Dajbych, O. Using Box–Behnken Design Coupled with Response Surface Methodology for Optimizing Rapeseed Oil Expression Parameters under Heating and Freezing Conditions. Processes 2022, 10, 490. [Google Scholar] [CrossRef]
- Cha, J.; Kim, C.T.; Cho, Y.J. Optimizing extraction conditions for functional compounds from ginger (Zingiber officinale Roscoe) using response surface methodology. Food Sci. Biotechnol. 2020, 29, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Pittol, V.; Ortega, G.G.; Doneda, E.; Bianchi, S.E.; Santos, M.C.; Koetz, M.; Henriques, A.T.; Bassani, V.L. Box-Behnken Design for Extraction Optimization Followed by High Performance Countercurrent Chromatography: Production of a Flavonoid-enriched Fraction from Achyrocline Satureioides. Planta Med. 2020, 86, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Skrypnik, L.; Novikova, A. Response Surface Modeling and Optimization of Polyphenols Extraction from Apple Pomace Based on Nonionic Emulsifiers. Agronomy 2020, 10, 92. [Google Scholar] [CrossRef] [Green Version]
- Baba, S.A.; Malik, S.A. Determination of total phenolic and flavonoid content, antimicrobial and antioxidant activity of a root extract of Arisaema jacquemontii Blume. J. Taibah Univ. Sci. 2015, 9, 449–454. [Google Scholar] [CrossRef] [Green Version]
- Almeida, M.M.B.; de Sousa, P.H.M.; Arriaga, Â.M.C.; do Prado, G.M.; de Magalhães, C.E.C.; Maia, G.A.; de Lemos, T.L.G. Bioactive compounds and antioxidant activity of fresh exotic fruits from northeastern Brazil. Food Res. Int. 2011, 44, 2155–2159. [Google Scholar] [CrossRef] [Green Version]
- Craig, A.P.; Fields, C.; Liang, N.; Kitts, D.; Erickson, A. Performance review of a fast HPLC-UV method for the quantification of chlorogenic acids in green coffee bean extracts. Talanta 2016, 154, 481–485. [Google Scholar] [CrossRef] [Green Version]
- Murthy, P.S.; Naidu, M.M. Recovery of Phenolic Antioxidants and Functional Compounds from Coffee Industry By-Products. Food Bioprocess Technol. 2012, 5, 897–903. [Google Scholar] [CrossRef]
- Toschi, T.G.; Cardenia, V.; Bonaga, G.; Mandrioli, M.; Rodriguez-Estrada, M.T. Coffee silverskin: Characterization, possible uses, and safety aspects. J. Agric. Food Chem. 2014, 62, 10836–10844. [Google Scholar] [CrossRef]
- Khuwijitjaru, P.; Plernjit, J.; Suaylam, B.; Samuhaseneetoo, S.; Pongsawatmanit, R.; Adachi, S. Degradation kinetics of some phenolic compounds in subcritical water and radical scavenging activity of their degradation products. Can. J. Chem. Eng. 2014, 92, 810–815. [Google Scholar] [CrossRef]
- Khajeh, M. Optimization of process variables for essential oil components from Satureja hortensis by supercritical fluid extraction using Box-Behnken experimental design. J. Supercrit. Fluids 2011, 55, 944–948. [Google Scholar] [CrossRef]
- Mayanga-Torres, P.C.; Lachos-Perez, D.; Rezende, C.A.; Prado, J.M.; Ma, Z.; Tompsett, G.T.; Timko, M.T.; Forster-Carneiro, T. Valorization of coffee industry residues by subcritical water hydrolysis: Recovery of sugars and phenolic compounds. J. Supercrit. Fluids 2017, 120, 75–85. [Google Scholar] [CrossRef]
- Sato, T.; Takahata, T.; Honma, T.; Watanabe, M.; Wagatsuma, M.; Matsuda, S.; Smith, R.L.; Itoh, N. Hydrothermal Extraction of Antioxidant Compounds from Green Coffee Beans and Decomposition Kinetics of 3-O-Caffeoylquinic Acid. Ind. Eng. Chem. Res. 2018, 57, 7624–7632. [Google Scholar] [CrossRef]
- Lachos-Perez, D.; Brown, A.B.; Mudhoo, A.; Martinez, J.; Timko, M.T.; Rostagno, M.A.; Forster-Carneiro, T. Applications of subcritical and supercritical water conditions for extraction, hydrolysis, gasification, and carbonization of biomass: A critical review. Biofuel Res. J. 2017, 4, 611–626. [Google Scholar] [CrossRef]
- Pereira, D.M.; Valentão, P.; Pereira, J.A.; Andrade, P.B. Phenolics: From chemistry to biology. Molecules 2009, 14, 2202–2211. [Google Scholar] [CrossRef]
- Clarke, C.J.; Tu, W.-C.; Levers, O.; Bröhl, A.; Hallett, J.P. Green and Sustainable Solvents in Chemical Processes. Chem. Rev. 2018, 118, 747–800. [Google Scholar] [CrossRef]
- Carr, A.G.; Mammucari, R.; Foster, N.R. A review of subcritical water as a solvent and its utilisation for the processing of hydrophobic organic compounds. Chem. Eng. J. 2011, 172, 1–17. [Google Scholar] [CrossRef]
- Ko, M.J.; Cheigh, C.I.; Chung, M.S. Relationship analysis between flavonoids structure and subcritical water extraction (SWE). Food Chem. 2014, 143, 147–155. [Google Scholar] [CrossRef]
- Pinelo, M.; Rubilar, M.; Jerez, M.; Sineiro, J.; Núñez, M.J. Effect of solvent, temperature, and solvent-to-solid ratio on the total phenolic content and antiradical activity of extracts from different components of grape pomace. J. Agric. Food Chem. 2005, 53, 2111–2117. [Google Scholar] [CrossRef]

| Symbol | Factor | Coded and Uncoded Level | ||
|---|---|---|---|---|
| −1 | 0 | +1 | ||
| X1 | Temperature (°C) | 120 | 180 | 240 |
| X2 | Time (minutes) | 10 | 35 | 60 |
| X3 | Solid-to-liquid ratio (g/mL) | 10 | 25 | 40 |
| Run Order | Block | Independent Variables | Dependent Variables | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| X1 | X2 | X3 | Y1 | Y2 | Y3 | |||||
| Coded | Uncoded | Coded | Uncoded | Coded | Uncoded | |||||
| 1 | 1 | 1 | 240 | −1 | 10 | 0 | 1:25 | 93.83 ± 6.11 | 26.12 ± 3.27 | 0.99 ± 0.26 |
| 2 | 1 | 0 | 180 | 0 | 35 | 0 | 1:25 | 70.05 ± 5.06 | 17.54 ± 2.28 | 1.66 ± 0.19 |
| 3 | 1 | −1 | 120 | 0 | 35 | −1 | 1:10 | 23.70 ± 2.10 | 5.71 ± 1.90 | 2.01 ± 0.23 |
| 4 | 1 | 0 | 180 | −1 | 10 | 1 | 1:40 | 65.06± 4.58 | 14.89 ± 0.85 | 2.33 ± 0.32 |
| 5 | 1 | 0 | 180 | 0 | 35 | 0 | 1:25 | 69.96 ± 3.25 | 17.44 ± 1.66 | 1.81 ± 0.21 |
| 6 | 1 | 1 | 240 | 1 | 60 | 0 | 1:25 | 75.03 ± 3.82 | 21.52 ± 2.15 | 0.50 ± 0.13 |
| 7 | 1 | 1 | 240 | 0 | 35 | 1 | 1:40 | 91.97 ± 4.15 | 24.94 ± 3.87 | 0.99 ± 0.27 |
| 8 | 1 | 0 | 180 | 0 | 35 | 0 | 1:25 | 72.81 ± 3.25 | 17.42 ± 2.48 | 1.62 ± 0.19 |
| 9 | 1 | 1 | 240 | 0 | 35 | −1 | 1:10 | 89.29 ± 4.67 | 19.742 ± 2.87 | 0.48 ± 0.15 |
| 10 | 1 | 0 | 180 | −1 | 10 | −1 | 1:10 | 42.65 ± 3.00 | 9.98 ± 1.39 | 2.04 ± 0.32 |
| 11 | 1 | −1 | 120 | −1 | 10 | 0 | 1:25 | 19.01 ± 3.54 | 4.98 ± 1.28 | 1.66 ± 0.21 |
| 12 | 1 | 0 | 180 | 1 | 60 | −1 | 1:10 | 77.69 ± 9.24 | 18.44 ± 2.26 | 1.69 ± 0.19 |
| 13 | 1 | −1 | 120 | 1 | 60 | 0 | 1:25 | 24.00 ± 2.36 | 6.65 ± 1.93 | 1.25 ± 0.33 |
| 14 | 1 | 0 | 180 | 1 | 60 | 1 | 1:40 | 76.24 ± 3.38 | 19.20 ± 2.09 | 1.49 ± 0.21 |
| 15 | 1 | −1 | 120 | 0 | 35 | 1 | 1:40 | 19.09 ± 2.29 | 4.15 ± 0.43 | 0.61 ± 0.41 |
| Parameters | p-Values for Each Parameter in the Polynomial Regression Equations | ||
|---|---|---|---|
| TPC | AA | T-CQA | |
| Model | 0.000 | 0.000 | 0.000 |
| Linear | 0.000 | 0.000 | 0.001 |
| Temperature (X1) | 0.000 | 0.000 | 0.001 |
| Time (X2) | 0.457 | - | 0.003 |
| S/L Ratio (X3) | 0.000 | 0.000 | 0.160 |
| Square | 0.000 | 0.000 | 0.000 |
| Temperature × Temperature (X12) | - | - | 0.000 |
| Time × Time (X22) | 0.044 | - | - |
| S/L Ratio × S/L Ratio (X32) | 0.000 | 0.000 | - |
| 2-Way Interaction | 0.010 | 0.009 | 0.001 |
| Temperature × Time (X1X2) | 0.036 | - | - |
| Temperature × S/L Ratio (X1X3) | - | 0.009 | 0.001 |
| Time × S/L Ratio (X2X3) | 0.010 | - | - |
| Lack of Fit | 0.169 | 0.097 | 0.213 |
| Model Summary | |||
| R2 (adjusted) | 87.91% | 94.97% | 90.21% |
| R2 (predicted) | 84.94% | 90.07% | 78.07% |
| R2 | 89.64% | 96.41% | 93.70% |
| Responses | Polynomial Regression Equations | R2 | R2 (Adjusted) | Lack of Fit |
|---|---|---|---|---|
| TPC | Y1 = −126.7 + 0.5457 X1 + 1.989 X2 + 6.063 X3 − 0.01056 X22 − 0.0884 X32 − 0.00448 X1X2 − 0.02419 X2X3 | 89.64% | 87.91% | 0.169 |
| AA | Y2 = −10.28 + 0.0441 X1 + 0.805 X3 − 0.02022 X32 + 0.002586 X1X3 | 96.41% | 94.97% | 0.097 |
| T-CQA | Y3 = −1.019 + 0.05592 X1 − 0.01043 X2 − 0.1023 X3− 0.000207 X12 + 0.000532 X1X3 | 93.70% | 90.21% | 0.213 |
| T-CQA (mg CQA/g CS) | TPC (mg GAE/g CS) | AA (mg TE/g CS) | Yield (%) | |
|---|---|---|---|---|
| Predicted value | Experimental value | Experimental value | Experimental value | Experimental value |
| 2.38 | 2.70 ± 0.23 | 51.86 ± 5.98 | 13.72 ± 2.04 | 27.25 ± 1.57 |
| Raw Material | Extraction Method/Conditions | TPC | Antioxidant Activity | T-CQA (Based on 3-, 4-, and 5-CQA) | References |
|---|---|---|---|---|---|
| (Based on DPPH Assay) | |||||
| Coffee silverskin | Subcritical water | 51.86 mg GAE/g CS | 13.72 | 2.70 mg/g CS | This study |
| Ratio 1: 10 (w/v) | |||||
| Time 10 min | |||||
| Coffee silverskin | Solid–liquid extraction | 22 mg GAE/g CS | 11.5 mg TE/g CS | 1.5 mg 5-CQA/g CS | [18] |
| Subcritical water | |||||
| Ratio 1:50 (w/v) | |||||
| Temperature 80 °C | |||||
| Time 60 min | |||||
| Coffee silverskin | Solid–liquid extraction (Soxhlet) | 13.2 | 5.3 | 3 mg/g CS | [46] |
| 60% Isopropanol | |||||
| Ratio 1:10 (w/v) | |||||
| Temperature 27 °C | |||||
| Time 20 min | |||||
| Coffee silverskin | Solid–liquid extraction | 16.1 | n.a. | 4.3 mg/g CS | [14] |
| (Maceration) | |||||
| 1% formic acid | |||||
| Ratio 1:10 (w/v) | |||||
| Time 90 min | |||||
| Coffee silverskin | Solid–liquid extraction | 12.81 | 4.49 | n.a. | [17] |
| (Waterbath) | |||||
| 60% Ethanol | |||||
| Ratio 1:35 (w/v) | |||||
| Temperature 60–65 °C | |||||
| Time 30 min | |||||
| Coffee silverskin | Solid–liquid extraction | 22.2 | 13.9 | n.a. | [35] |
| Mild hydrothermal pretreatment | |||||
| Ratio 1:30 (w/v) | |||||
| Temperature 120 °C | |||||
| Time 20 min |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ginting, A.R.; Kit, T.; Mingvanish, W.; Thanasupsin, S.P. Valorization of Coffee Silverskin through Subcritical Water Extraction: An Optimization Based on T-CQA Using Response Surface Methodology. Sustainability 2022, 14, 8435. https://doi.org/10.3390/su14148435
Ginting AR, Kit T, Mingvanish W, Thanasupsin SP. Valorization of Coffee Silverskin through Subcritical Water Extraction: An Optimization Based on T-CQA Using Response Surface Methodology. Sustainability. 2022; 14(14):8435. https://doi.org/10.3390/su14148435
Chicago/Turabian StyleGinting, Agita Rachmala, Thavy Kit, Withawat Mingvanish, and Sudtida Pliankarom Thanasupsin. 2022. "Valorization of Coffee Silverskin through Subcritical Water Extraction: An Optimization Based on T-CQA Using Response Surface Methodology" Sustainability 14, no. 14: 8435. https://doi.org/10.3390/su14148435
APA StyleGinting, A. R., Kit, T., Mingvanish, W., & Thanasupsin, S. P. (2022). Valorization of Coffee Silverskin through Subcritical Water Extraction: An Optimization Based on T-CQA Using Response Surface Methodology. Sustainability, 14(14), 8435. https://doi.org/10.3390/su14148435







