Litterfall and Associated Macrozoobenthic of Restored Mangrove Forests in Abandoned Aquaculture Ponds
Abstract
:1. Introduction
2. Materials and Methods
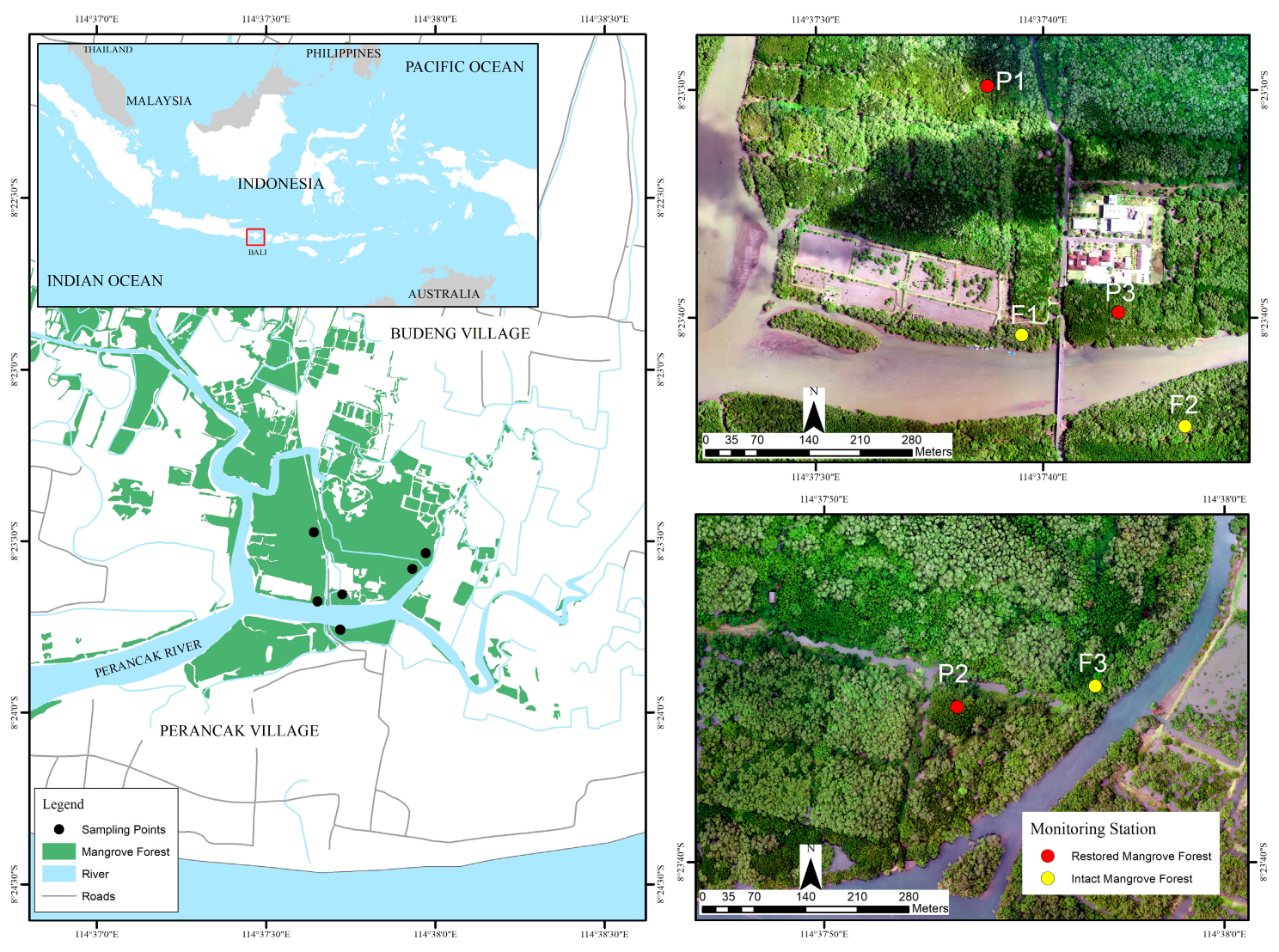
2.1. Site Description
2.2. Plant Litterfall Production
2.3. Leaf Litter Decay and Decomposition
2.4. Macrozoobenthos Sampling and Biodiversity Indices Assessment
- (a)
- The diversity index (H′) for each site was calculated using the following formula:where H′ is the Shannon–Wiener diversity index, Pi is the proportion (ni/N) of individuals of one particular species found (ni) divided by the total number of individuals found (N), and S is the number of species.
- (b)
- The similarity index was calculated as follows:where E is the similarity index (evenness), and H max = logS.
- (c)
- The dominance index reflects the central level of dominance (mastery) in a community. The dominance index was calculated using the following formula:where D is Simpson’s dominance index.
2.5. Data Analysis
3. Results
3.1. Plant Litter Production
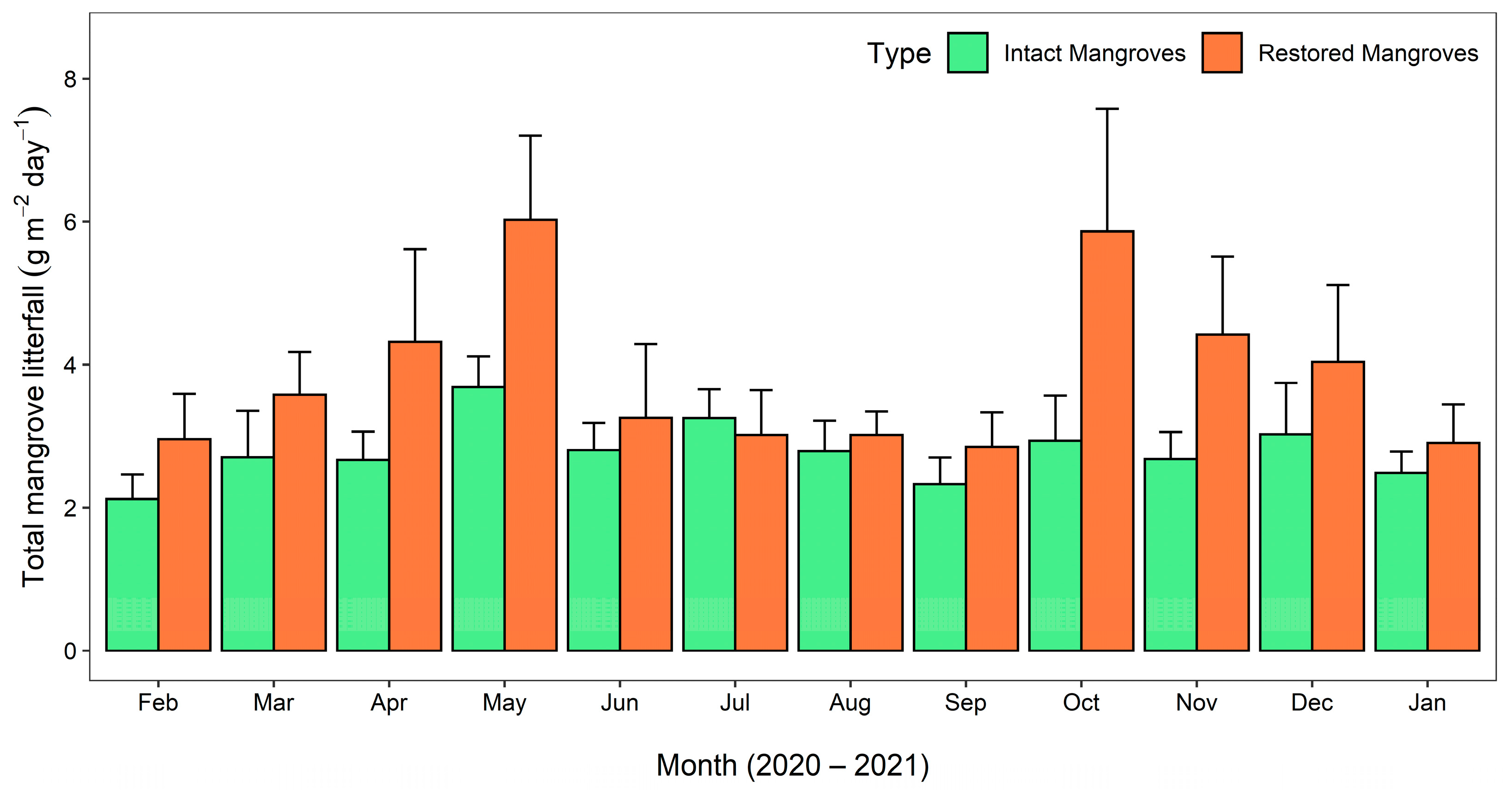
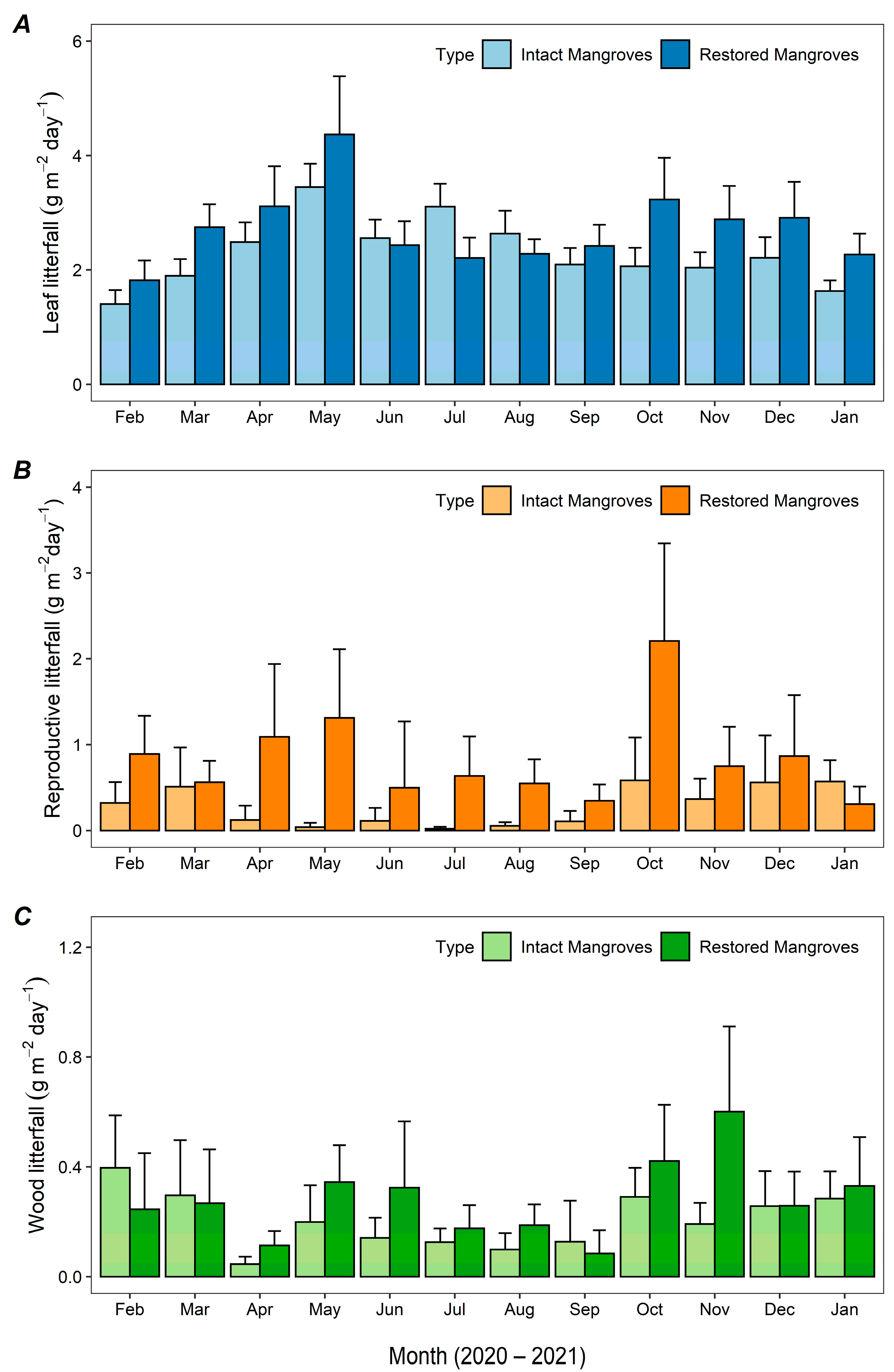
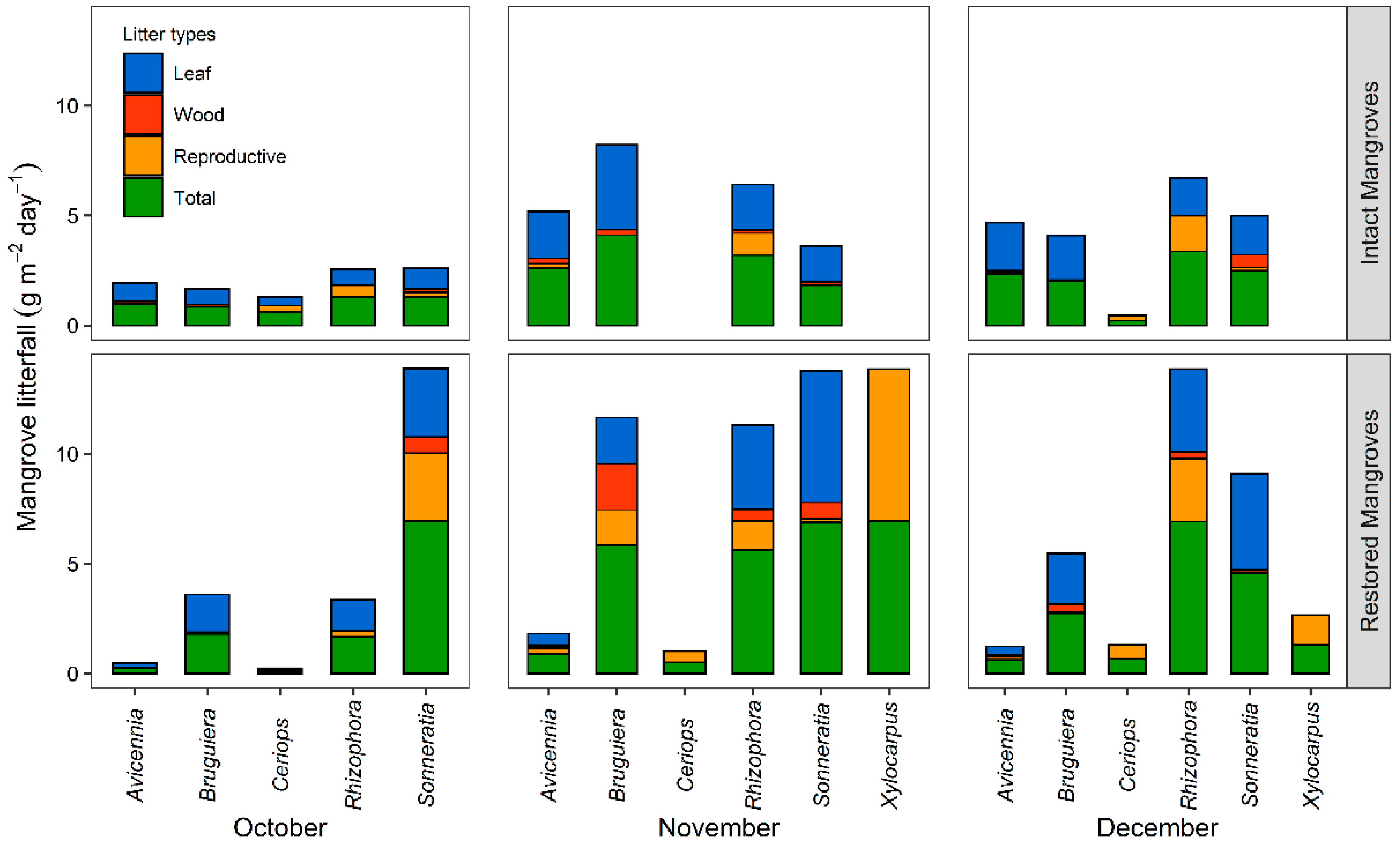
3.1.1. Monthly Mangrove Litterfall Production
3.1.2. Annual Mangrove Litterfall Production
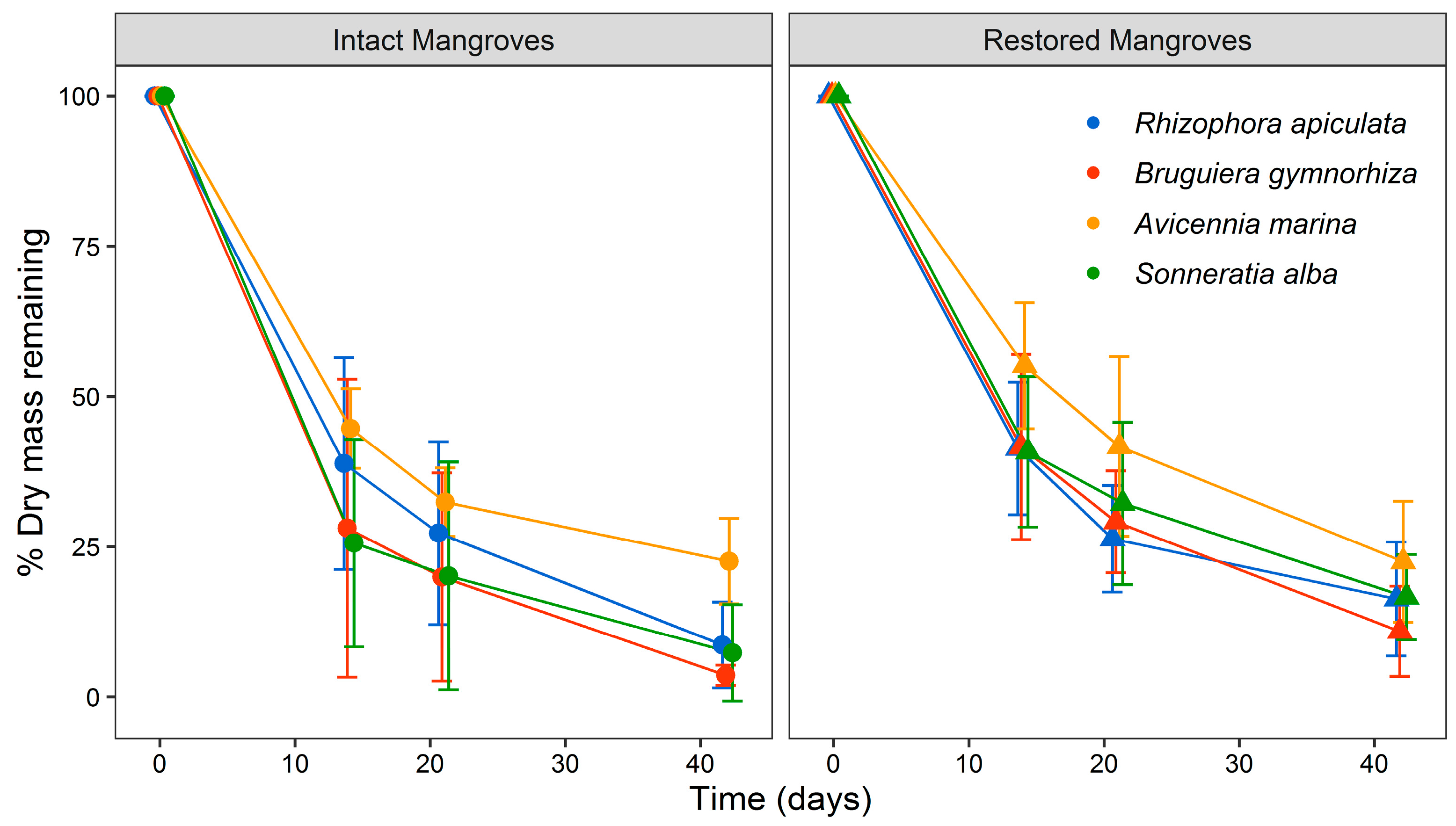
3.2. Leaf Litter Decay and Decomposition
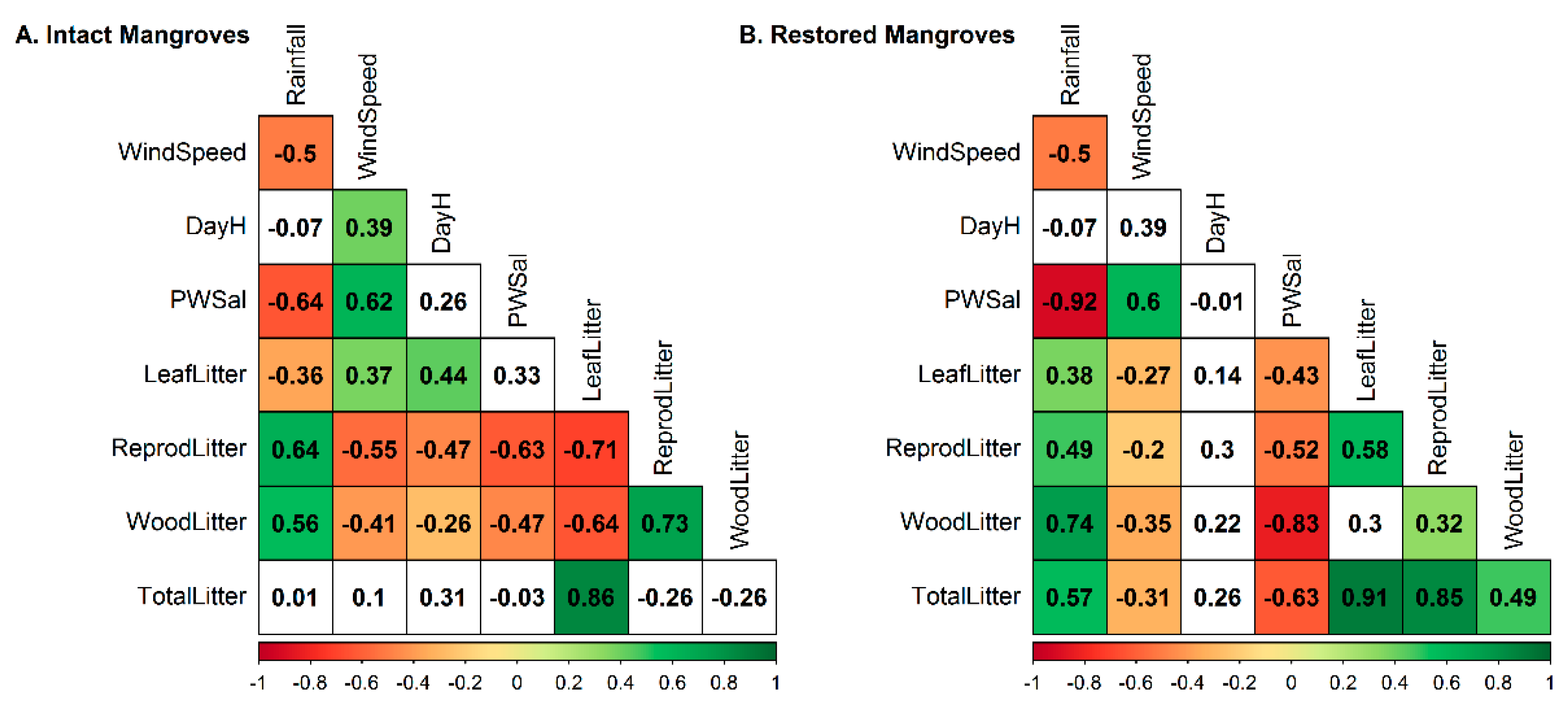
3.3. Climate Data
3.4. Macrozoobenthic Community Structure
3.4.1. Environmental Conditions during Macrozoobenthos Sampling
3.4.2. Macrozoobenthos Abundance, Diversity, Similarity, and Dominance Indices
4. Discussion
4.1. Mangrove Litterfall and Its Affecting Factors
4.2. Leaf Decomposition and Associated Macrozoobenthos
4.3. Implications for Mangrove Conservation and Restoration
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bunting, P.; Rosenqvist, A.; Lucas, R.; Rebelo, L.-M.; Hilarides, L.; Thomas, N.; Hardy, A.; Itoh, T.; Shimada, M.; Finlayson, C. The Global Mangrove Watch—A New 2010 Global Baseline of Mangrove Extent. Remote Sens. 2018, 10, 1669. [Google Scholar] [CrossRef] [Green Version]
- Krauss, K.W.; McKee, K.L.; Lovelock, C.E.; Cahoon, D.R.; Saintilan, N.; Reef, R.; Chen, L. How Mangrove Forests Adjust to Rising Sea Level. New Phytol. 2014, 202, 19–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Donato, D.C.; Kauffman, J.B.; Murdiyarso, D.; Kurnianto, S.; Stidham, M.; Kanninen, M. Mangroves among the Most Carbon-Rich Forests in the Tropics. Nat. Geosci. 2011, 4, 293–297. [Google Scholar] [CrossRef]
- Zeng, Y.; Friess, D.A.; Sarira, T.V.; Siman, K.; Koh, L.P. Global Potential and Limits of Mangrove Blue Carbon for Climate Change Mitigation. Curr. Biol. 2021, 31, 1737–1743.e3. [Google Scholar] [CrossRef] [PubMed]
- Alongi, D.M. Carbon Cycling and Storage in Mangrove Forests. Annu. Rev. Mar. Sci. 2014, 6, 195–219. [Google Scholar] [CrossRef] [PubMed]
- Duarte, C.M.; Losada, I.J.; Hendriks, I.E.; Mazarrasa, I.; Marbà, N. The Role of Coastal Plant Communities for Climate Change Mitigation and Adaptation. Nat. Clim. Chang. 2013, 3, 961–968. [Google Scholar] [CrossRef] [Green Version]
- Andreetta, A.; Fusi, M.; Cameldi, I.; Cimò, F.; Carnicelli, S.; Cannicci, S. Mangrove Carbon Sink. Do Burrowing Crabs Contribute to Sediment Carbon Storage? Evidence from a Kenyan Mangrove System. J. Sea Res. 2014, 85, 524–533. [Google Scholar] [CrossRef] [Green Version]
- Bouillon, S.; Connolly, R.M. Carbon Exchange Among Tropical Coastal Ecosystems. In Ecological Connectivity among Tropical Coastal Ecosystems; Springer: Dordrecht, The Netherlands, 2009; pp. 45–70. [Google Scholar]
- Riascos, J.M.; Blanco-Libreros, J.F. Pervasively High Mangrove Productivity in a Major Tropical Delta throughout an ENSO Cycle (Southern Caribbean, Colombia). Estuar. Coast. Shelf Sci. 2019, 227, 106301. [Google Scholar] [CrossRef]
- Bouillon, S.; Borges, A.V.; Castañeda-Moya, E.; Diele, K.; Dittmar, T.; Duke, N.C.; Kristensen, E.; Lee, S.Y.; Marchand, C.; Middelburg, J.J.; et al. Mangrove Production and Carbon Sinks: A Revision of Global Budget Estimates. Glob. Biogeochem. Cycles 2008, 22, GB2013. [Google Scholar] [CrossRef] [Green Version]
- Nga, B.T.; Tinh, H.Q.; Tam, D.T.; Scheffer, M.; Roijackers, R. Young Mangrove Stands Produce a Large and High Quality Litter Input to Aquatic Systems. Wetl. Ecol. Manag. 2005, 13, 569–576. [Google Scholar] [CrossRef] [Green Version]
- Kamal, A.H.M.; Hoque, M.M.; Idris, M.H.; Ahmed, O.H.; Bhuiyan, M.K.A.; Billah, M.M.; Hoque, M.N.; Rosli, Z. Decay of Rhizophora apiculata (Blume) and Xylocarpus granatum (Koenig) Detrital Sources in the Sarawak Mangrove, Malaysia. J. For. Res. 2020, 31, 613–623. [Google Scholar] [CrossRef]
- Azad, M.S.; Kamruzzaman, M.; Ahmed, S.; Kanzaki, M. Litterfall Assessment and Reproductive Phenology Observation in the Sundarbans, Bangladesh: A Comparative Study among Three Mangrove Species. Trees For. People 2021, 4, 100068. [Google Scholar] [CrossRef]
- Allison, S.D.; Vitousek, P.M. Responses of Extracellular Enzymes to Simple and Complex Nutrient Inputs. Soil Biol. Biochem. 2005, 37, 937–944. [Google Scholar] [CrossRef]
- Prescott, C.E. Litter Decomposition: What Controls It and How Can We Alter It to Sequester More Carbon in Forest Soils? Biogeochemistry 2010, 101, 133–149. [Google Scholar] [CrossRef]
- Alongi, D.M. Carbon Sequestration in Mangrove Forests. Carbon Manag. 2012, 3, 313–322. [Google Scholar] [CrossRef]
- Atwood, T.B.; Connolly, R.M.; Almahasheer, H.; Carnell, P.E.; Duarte, C.M.; Ewers Lewis, C.J.; Irigoien, X.; Kelleway, J.J.; Lavery, P.S.; Macreadie, P.I.; et al. Global Patterns in Mangrove Soil Carbon Stocks and Losses. Nat. Clim. Chang. 2017, 7, 523–528. [Google Scholar] [CrossRef]
- Chen, G.; Azkab, M.H.; Chmura, G.L.; Chen, S.; Sastrosuwondo, P.; Ma, Z.; Dharmawan, I.W.E.; Yin, X.; Chen, B. Mangroves as a Major Source of Soil Carbon Storage in Adjacent Seagrass Meadows. Sci. Rep. 2017, 7, 42406. [Google Scholar] [CrossRef]
- Richards, D.R.; Friess, D.A. Rates and Drivers of Mangrove Deforestation in Southeast Asia, 2000–2012. Proc. Natl. Acad. Sci. USA 2016, 113, 344–349. [Google Scholar] [CrossRef] [Green Version]
- Sidik, F.; Lovelock, C.E. CO2 Efflux from Shrimp Ponds in Indonesia. PLoS ONE 2013, 8, e66329. [Google Scholar]
- Ilman, M.; Dargusch, P.; Dart, P.; Onrizal, P. A Historical Analysis of the Drivers of Loss and Degradation of Indonesia’s Mangroves. Land Use Policy 2016, 54, 448–459. [Google Scholar] [CrossRef]
- Arifanti, V.B.; Kauffman, J.B.; Hadriyanto, D.; Murdiyarso, D.; Diana, R. Carbon Dynamics and Land Use Carbon Footprints in Mangrove-Converted Aquaculture: The Case of the Mahakam Delta, Indonesia. For. Ecol. Manag. 2019, 432, 17–29. [Google Scholar] [CrossRef]
- Murdiyarso, D. Climate and Development-the Challenges in Delivering the Promises: An Editorial Essay. Wiley Interdiscip. Rev. Clim. Chang. 2010, 1, 765–769. [Google Scholar] [CrossRef]
- Murdiyarso, D.; Purbopuspito, J.; Kauffman, J.B.; Warren, M.W.; Sasmito, S.D.; Donato, D.C.; Manuri, S.; Krisnawati, H.; Taberima, S.; Kurnianto, S. The Potential of Indonesian Mangrove Forests for Global Climate Change Mitigation. Nat. Clim. Chang. 2015, 5, 1089–1092. [Google Scholar] [CrossRef]
- Sanderman, J.; Hengl, T.; Fiske, G.; Solvik, K.; Adame, M.F.; Benson, L.; Bukoski, J.J.; Carnell, P.; Cifuentes-Jara, M.; Donato, D.; et al. A Global Map of Mangrove Forest Soil Carbon at 30 m Spatial Resolution. Environ. Res. Lett. 2018, 13, 055002. [Google Scholar] [CrossRef]
- Duncan, C.; Primavera, J.H.; Pettorelli, N.; Thompson, J.R.; Loma, R.J.A.; Koldewey, H.J. Rehabilitating Mangrove Ecosystem Services: A Case Study on the Relative Benefits of Abandoned Pond Reversion from Panay Island, Philippines. Mar. Pollut. Bull. 2016, 109, 772–782. [Google Scholar] [CrossRef]
- Lee, S.Y.; Hamilton, S.; Barbier, E.B.; Primavera, J.; Lewis, R.R. Better Restoration Policies Are Needed to Conserve Mangrove Ecosystems. Nat. Ecol. Evol. 2019, 3, 870–872. [Google Scholar] [CrossRef]
- van Bijsterveldt, C.E.J.; Debrot, A.O.; Bouma, T.J.; Maulana, M.B.; Pribadi, R.; Schop, J.; Tonneijck, F.H.; van Wesenbeeck, B.K. To Plant or Not to Plant: When Can Planting Facilitate Mangrove Restoration? Front. Environ. Sci. 2022, 9, 762. [Google Scholar] [CrossRef]
- Sidik, F.; Pradisty, N.A.; Widagti, N. Restored Mangrove Forests in Perancak Estuary, Bali; Institute for Marine Research and Observation: Bali, Indonesia, 2021; ISBN 978-623-97774-0-1. [Google Scholar]
- Proisy, C.; Viennois, G.; Sidik, F.; Andayani, A.; Enright, J.A.; Guitet, S.; Gusmawati, N.; Lemonnier, H.; Muthusankar, G.; Olagoke, A.; et al. Monitoring Mangrove Forests after Aquaculture Abandonment Using Time Series of Very High Spatial Resolution Satellite Images: A Case Study from the Perancak Estuary, Bali, Indonesia. Mar. Pollut. Bull. 2018, 131, 61–71. [Google Scholar] [CrossRef] [Green Version]
- Gusmawati, N.; Soulard, B.; Selmaoui-Folcher, N.; Proisy, C.; Mustafa, A.; Le Gendre, R.; Laugier, T.; Lemonnier, H. Surveying Shrimp Aquaculture Pond Activity Using Multitemporal VHSR Satellite Images-Case Study from the Perancak Estuary, Bali, Indonesia. Mar. Pollut. Bull. 2018, 131, 49–60. [Google Scholar] [CrossRef] [Green Version]
- Sidik, F.; Adame, M.F.; Lovelock, C.E. Carbon Sequestration and Fluxes of Restored Mangroves in Abandoned Aquaculture Ponds. J. Indian Ocean Reg. 2019, 15, 177–192. [Google Scholar] [CrossRef]
- Sidik, F.; Kusuma, D.W.; Priyono, B.; Proisy, C.; Lovelock, C.E. Managing Sediment Dynamics through Reintroduction of Tidal Flow for Mangrove Restoration in Abandoned Aquaculture Ponds. In Dynamic Sedimentary Environments of Mangrove Coasts; Elsevier: Amsterdam, The Netherlands, 2021; pp. 563–582. [Google Scholar]
- Bosire, J.O.; Dahdouh-Guebas, F.; Walton, M.; Crona, B.I.; Lewis, R.R.; Field, C.; Kairo, J.G.; Koedam, N. Functionality of Restored Mangroves: A Review. Aquat. Bot. 2008, 89, 251–259. [Google Scholar] [CrossRef] [Green Version]
- Matsui, N.; Morimune, K.; Meepol, W.; Chukwamdee, J. Ten Year Evaluation of Carbon Stock in Mangrove Plantation Reforested from an Abandoned Shrimp Pond. Forests 2012, 3, 431–444. [Google Scholar] [CrossRef] [Green Version]
- Macklin, P.A.; Suryaputra, I.G.N.A.; Maher, D.T.; Sidik, F.; Santos, I.R. Carbon Dioxide Dynamics in a Tropical Estuary over Seasonal and Rain-Event Time Scales. Cont. Shelf Res. 2020, 206, 104196. [Google Scholar] [CrossRef]
- Ruslisan, R.; Kamal, M.; Sidik, F. Monitoring the Restored Mangrove Condition at Perancak Estuary, Jembrana, Bali, Indonesia from 2001 to 2015. IOP Conf. Ser. Earth Environ. Sci. 2018, 123, 012022. [Google Scholar] [CrossRef]
- Nugraha, M.A.R. Analisis Komposisi Jenis Mangrove Pada Mangrove Ditanam Dan Mangrove Alami Menggunakan Citra Satelit Resolusi Tinggi Worldview-3 Di Estuari Perancak. Bachelor’s Thesis, Universitas Brawijaya, East Java, Indonesia, 2019. [Google Scholar]
- Kauffman, J.B.; Donato, D.C. Protocols for the Measurement, Monitoring and Reporting of Structure, Biomass, and Carbon Stocks in Mangrove Forests; CIFOR: Bogor, Indonesia, 2012. [Google Scholar]
- Zhao, X.; Rivera-Monroy, V.H.; Wang, H.; Xue, Z.G.; Tsai, C.-F.; Willson, C.S.; Castañeda-Moya, E.; Twilley, R.R. Modeling Soil Porewater Salinity in Mangrove Forests (Everglades, Florida, USA) Impacted by Hydrological Restoration and a Warming Climate. Ecol. Model. 2020, 436, 109292. [Google Scholar] [CrossRef]
- Pradisty, N.A.; Amir, A.A.; Zimmer, M. Plant Species-and Stage-Specific Differences in Microbial Decay of Mangrove Leaf Litter: The Older the Better? Oecologia 2021, 195, 843–858. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.S. Energy Storage and the Balance of Producers and Decomposers in Ecological Systems. Ecology 1963, 44, 322–331. [Google Scholar] [CrossRef] [Green Version]
- Basyuni, M.; Bimantara, Y.; Cuc, N.T.K.; Balke, T.; Vovides, A.G. Macrozoobenthic Community Assemblage as Key Indicator for Mangrove Restoration Success in North Sumatra and Aceh, Indonesia. Restor. Ecol. 2022, e13614, early view. [Google Scholar] [CrossRef]
- Basyuni, M.; Gultom, K.; Fitri, A.; Susetya, I.E.; Wati, R.; Slamet, B.; Sulistiyono, N.; Yusriani, E.; Balke, T.; Bunting, P. Diversity and Habitat Characteristics of Macrozoobenthos in the Mangrove Forest of Lubuk Kertang Village, North Sumatra, Indonesia. Biodiversitas J. Biol. Divers. 2018, 19, 311–317. [Google Scholar] [CrossRef]
- APHA. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; Rice, E.W., Baird, R.B., Eaton, A.D., Clesceri, L.S., Eds.; American Public Health Association: Washington, DC, USA, 2012; ISBN 9780875530130 0875530133. [Google Scholar]
- Krebs, C. Ecological Methodology; Pearson Higher Education: Harlow, UK, 1998. [Google Scholar]
- Sidik, F.; Kadarisman, H.; Widagti, N. Buku Panduan Mangrove Estuari Perancak. BROL KKP. 2018. Available online: https://www.researchgate.net/profile/Frida-Sidik/publication/333310292_Buku_panduan_mangrove_Estuari_Perancak/links/5ce63d9b458515712ebb92f8/Buku-panduan-mangrove-Estuari-Perancak.pdf (accessed on 1 March 2022).
- Ahmed, S.; Kamruzzaman, M. Species-Specific Biomass and Carbon Flux in Sundarbans Mangrove Forest, Bangladesh: Response to Stand and Weather Variables. Biomass Bioenergy 2021, 153, 106215. [Google Scholar] [CrossRef]
- Berger, U.; Hildenbrandt, H.; Grimm, V. Age-Related Decline in Forest Production: Modelling the Effects of Growth Limitation, Neighbourhood Competition and Self-Thinning. J. Ecol. 2004, 92, 846–853. [Google Scholar] [CrossRef]
- Komiyama, A.; Ong, J.E.; Poungparn, S. Allometry, Biomass, and Productivity of Mangrove Forests: A Review. Aquat. Bot. 2008, 89, 128–137. [Google Scholar] [CrossRef]
- Wang’ondu, V.W.; Bosire, J.O.; Kairo, J.G.; Kinyamario, J.I.; Mwaura, F.B.; Dahdouh-Guebas, F.; Koedam, N. Litter Fall Dynamics of Restored Mangroves (Rhizophora mucronata Lamk. and Sonneratia alba Sm.) in Kenya. Restor. Ecol. 2014, 22, 824–831. [Google Scholar] [CrossRef]
- Sukardjo, S.; Alongi, D.M.; Kusmana, C. Rapid Litter Production and Accumulation in Bornean Mangrove Forests. Ecosphere 2013, 4, 1–7. [Google Scholar] [CrossRef]
- Parkinson, R.W.; Perez-Bedmar, M.; Santangelo, J.A. Red Mangrove (Rhizophora mangle L.) Litter Fall Response to Selective Pruning (Indian River Lagoon, Florida, U.S.A.). In Diversity and Function in Mangrove Ecosystems; Springer: Dordrecht, The Netherlands, 1999; pp. 63–76. [Google Scholar]
- Hoque, M.M.; Mustafa Kamal, A.H.; Idris, M.H.; Haruna Ahmed, O.; Rafiqul Hoque, A.T.M.; Masum Billah, M. Litterfall Production in a Tropical Mangrove of Sarawak, Malaysia. Zool. Ecol. 2015, 25, 157–165. [Google Scholar] [CrossRef]
- Sukardjo, S.; Yamada, I. Biomass and Productivity of a Rhizophora mucronata Lamarck Plantation in Tritih, Central Java, Indonesia. For. Ecol. Manag. 1992, 49, 195–209. [Google Scholar] [CrossRef]
- Dharmawan, I.W.E.; Guangcheng, C.; Pramudji; Bin, C. Spatial and Seasonal Variation of Mangrove Litter Production in Bitung, Indonesia. IOP Conf. Ser. Earth Environ. Sci. 2019, 278, 012015. [Google Scholar] [CrossRef] [Green Version]
- Peel, J.R.; Golubov, J.; Mandujano, M.C.; López-Portillo, J. Phenology and Floral Synchrony of Rhizophora mangle along a Natural Salinity Gradient. Biotropica 2019, 51, 355–363. [Google Scholar] [CrossRef]
- Sharma, S.; Hoque, A.T.M.R.; Analuddin, K.; Hagihara, A. Litterfall Dynamics in an Overcrowded Mangrove Kandelia obovata (S., L.) Yong Stand over Five Years. Estuar. Coast. Shelf Sci. 2012, 98, 31–41. [Google Scholar] [CrossRef]
- Azad, M.S.; Kamruzzaman, M.; Paul, S.K.; Ahmed, S.; Kanzaki, M. Vegetative and Reproductive Phenology of the Mangrove Xylocarpus mekongensis Pierre in the Sundarbans, Bangladesh: Relationship with Climatic Variables. Reg. Stud. Mar. Sci. 2020, 38, 101359. [Google Scholar] [CrossRef]
- Ouyang, X.; Lee, S.Y.; Connolly, R.M. The Role of Root Decomposition in Global Mangrove and Saltmarsh Carbon Budgets. Earth Sci. Rev. 2017, 166, 53–63. [Google Scholar] [CrossRef] [Green Version]
- Sanders, C.J.; Maher, D.T.; Tait, D.R.; Williams, D.; Holloway, C.; Sippo, J.Z.; Santos, I.R. Are Global Mangrove Carbon Stocks Driven by Rainfall? J. Geophys. Res. Biogeosci. 2016, 121, 2600–2609. [Google Scholar] [CrossRef]
- Zhang, H.; Yuan, W.; Dong, W.; Liu, S. Seasonal Patterns of Litterfall in Forest Ecosystem Worldwide. Ecol. Complex. 2014, 20, 240–247. [Google Scholar] [CrossRef]
- Komiyama, A.; Poungparn, S.; Umnouysin, S.; Rodtassana, C.; Pravinvongvuthi, T.; Noda, T.; Kato, S. Occurrence of Seasonal Water Replacement in Mangrove Soil and the Trunk Growth Response of Avicennia alba Related to Salinity Changes in a Tropical Monsoon Climate. Ecol. Res. 2019, 34, 428–439. [Google Scholar] [CrossRef]
- Souza, S.R.; Veloso, M.D.M.; Espírito-Santo, M.M.; Silva, J.O.; Sánchez-Azofeifa, A.; Souza e Brito, B.G.; Fernandes, G.W. Litterfall Dynamics along a Successional Gradient in a Brazilian Tropical Dry Forest. For. Ecosyst. 2019, 6, 35. [Google Scholar] [CrossRef] [Green Version]
- Aké-Castillo, J.A.; Vázquez, G.; López-Portillo, J. Litterfall and Decomposition of Rhizophora mangle L. in a Coastal Lagoon in the Southern Gulf of Mexico. Hydrobiologia 2006, 559, 101–111. [Google Scholar] [CrossRef]
- Alongi, D.M. The Energetics of Mangrove Forests; Alongi, D.M., Ed.; Springer: Dordrecht, The Netherlands, 2009. [Google Scholar]
- Duke, N.C.; Ball, M.C.; Ellison, J.C. Factors Influencing Biodiversity and Distributional Gradients in Mangroves. Glob. Ecol. Biogeogr. Lett. 1998, 7, 27–47. [Google Scholar] [CrossRef] [Green Version]
- McKee, K.L. Seedling Recruitment Patterns in a Belizean Mangrove Forest: Effects of Establishment Ability and Physico-Chemical Factors. Oecologia 1995, 101, 448–460. [Google Scholar] [CrossRef]
- Donnelly, M.; Walters, L. Trapping of Rhizophora mangle Propagules by Coexisting Early Successional Species. Estuaries Coasts 2014, 37, 1562–1571. [Google Scholar] [CrossRef]
- McKee, K.L.; Rooth, J.E.; Feller, I.C. Mangrove Recruitment after Forest Disturbance Is Facilitated by Herbaceous Species in the Caribbean. Ecol. Appl. 2007, 17, 1678–1693. [Google Scholar] [CrossRef]
- Tomlinson, P.B. The Botany of Mangroves; Cambridge University Press: Cambridge, UK, 2016; ISBN 9781139946575. [Google Scholar]
- Clarke, P.J.; Kerrigan, R.A.; Westphal, C.J. Dispersal Potential and Early Growth in 14 Tropical Mangroves: Do Early Life History Traits Correlate with Patterns of Adult Distribution? J. Ecol. 2001, 89, 648–659. [Google Scholar] [CrossRef] [Green Version]
- Van der Stocken, T.; Wee, A.K.S.; De Ryck, D.J.R.; Vanschoenwinkel, B.; Friess, D.A.; Dahdouh-Guebas, F.; Simard, M.; Koedam, N.; Webb, E.L. A General Framework for Propagule Dispersal in Mangroves. Biol. Rev. 2019, 94, 1547–1575. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gillis, L.; Zimmer, M.; Bouma, T. Mangrove Leaf Transportation: Do Mimic Avicennia and Rhizophora Roots Retain or Donate Leaves? Mar. Ecol. Prog. Ser. 2016, 551, 107–115. [Google Scholar] [CrossRef]
- Balke, T.; Friess, D.A. Geomorphic Knowledge for Mangrove Restoration: A Pan-Tropical Categorization. Earth Surf. Process. Landforms 2016, 41, 231–239. [Google Scholar] [CrossRef]
- Hossain, M. Hoque Litter Production and Decomposition in Mangroves—A Review. Indian J. For. 2008, 32, 227–238. [Google Scholar]
- Middleton, B.A.; McKee, K.L. Degradation of Mangrove Tissues and Implications for Peat Formation in Belizean Island Forests. J. Ecol. 2001, 89, 818–828. [Google Scholar] [CrossRef]
- Valiela, I.; Teal, J.M.; Allen, S.D.; Van Etten, R.; Goehringer, D.; Volkmann, S. Decomposition in Salt Marsh Ecosystems: The Phases and Major Factors Affecting Disappearance of above-Ground Organic Matter. J. Exp. Mar. Biol. Ecol. 1985, 89, 29–54. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; D’Odorico, P. Decomposition and Mineralization. In Encyclopedia of Ecology; Jørgensen, S.E., Fath, B.D., Eds.; Academic Press: Oxford, UK, 2008; pp. 838–844. ISBN 978-0-08-045405-4. [Google Scholar]
- Li, T.; Ye, Y. Dynamics of Decomposition and Nutrient Release of Leaf Litter in Kandelia obovata Mangrove Forests with Different Ages in Jiulongjiang Estuary, China. Ecol. Eng. 2014, 73, 454–460. [Google Scholar] [CrossRef]
- Zimmer, M. Detritus. In Encyclopedia of Ecology, 2nd ed.; Elsevier: Oxford, UK, 2019; pp. 292–301. ISBN 978-0-444-64130-4. [Google Scholar]
- Bosire, J.O.; Dahdouh-Guebas, F.; Kairo, J.G.; Kazungu, J.; Dehairs, F.; Koedam, N. Litter Degradation and CN Dynamics in Reforested Mangrove Plantations at Gazi Bay, Kenya. Biol. Conserv. 2005, 126, 287–295. [Google Scholar] [CrossRef] [Green Version]
- Luo, L.; Gu, J.-D. Influence of Macrofaunal Burrows on Extracellular Enzyme Activity and Microbial Abundance in Subtropical Mangrove Sediment. Microb. Ecol. 2018, 76, 92–101. [Google Scholar] [CrossRef]
- Salmo, S.G.; Tibbetts, I.; Duke, N.C. Colonization and Shift of Mollusc Assemblages as a Restoration Indicator in Planted Mangroves in the Philippines. Biodivers. Conserv. 2017, 26, 865–881. [Google Scholar] [CrossRef]
- Ariyanto, D.; Bengen, D.G.; Prartono, T.; Wardiatno, Y. The Association of Cassidula nucleus (Gmelin 1791) and Cassidula angulifera (Petit 1841) with Mangrove in Banggi Coast, Central Java, Indonesia. Aquac. Aquar. Conserv. Legis. 2018, 11, 348–361. [Google Scholar]
- Zvonareva, S.S.; Kantor, Y.I.; Nguyen, T.T.H.; Britayev, T.A. Diversity and Long-Term Dynamics of the Macrobenthos in a Mangrove Plantation and Two Natural Associations of Khanh Hoa Province, Vietnam. Biol. Bull. 2020, 47, 1115–1125. [Google Scholar] [CrossRef]
- Arbi, I.; Liu, S.; Zhang, J.; Wu, Y.; Huang, X. Detection of Terrigenous and Marine Organic Matter Flow into a Eutrophic Semi-Enclosed Bay by Δ13C and Δ15N of Intertidal Macrobenthos and Basal Food Sources. Sci. Total Environ. 2018, 613–614, 847–860. [Google Scholar] [CrossRef]
- Leung, J.Y.S.; Tam, N.F.Y. Influence of Plantation of an Exotic Mangrove Species, Sonneratia caseolaris (L.) Engl., on Macrobenthic Infaunal Community in Futian Mangrove National Nature Reserve, China. J. Exp. Mar. Biol. Ecol. 2013, 448, 1–9. [Google Scholar] [CrossRef]
- Arifanti, V.B.; Sidik, F.; Mulyanto, B.; Susilowati, A.; Wahyuni, T.; Subarno, S.; Yulianti, Y.; Yuniarti, N.; Aminah, A.; Suita, E.; et al. Challenges and Strategies for Sustainable Mangrove Management in Indonesia: A Review. Forests 2022, 13, 695. [Google Scholar] [CrossRef]
- Kamal, M.; Sidik, F.; Prananda, A.R.A.; Mahardhika, S.A. Mapping Leaf Area Index of Restored Mangroves Using WorldView-2 Imagery in Perancak Estuary, Bali, Indonesia. Remote Sens. Appl. Soc. Environ. 2021, 23, 100567. [Google Scholar] [CrossRef]
- Friess, D.; Yando, E.; Alemu, J.; Wong, L.-W.; Soto, S.; Bhatia, N. Ecosystem Services and Disservices of Mangrove Forests and Salt Marshes. Oceanogr. Mar. Biol. 2020, 58, 107–142. [Google Scholar]
- Friesen, S.D.; Dunn, C.; Freeman, C. Decomposition as a Regulator of Carbon Accretion in Mangroves: A Review. Ecol. Eng. 2018, 114, 173–178. [Google Scholar] [CrossRef] [Green Version]
- Lewis, R.R.; Brown, B. Ecological Mangrove Rehabilitation: A Field Manual for Practitioners; Mangrove Action Project, Canadian International Development Agency and OXFAM: Seattle, WA, USA, 2014. [Google Scholar]
| Plot | Forest Type | Abundance (ind/ha) | Basal Area (m2/ha) | Species Richness (S) | Diversity (H′) | Similarity (e) | Dominance (D) |
|---|---|---|---|---|---|---|---|
| F1 | Intact forest | 1233.77 | 22.61 | 19 | 1.51 | 1.09 | 0.34 |
| F2 | 1753.25 | 16.62 | 27 | 1.53 | 0.95 | 0.27 | |
| F3 | 519.48 | 13.88 | 8 | 0.48 | 0.34 | 0.34 | |
| P1 | Restored mangroves | 1623.38 | 29.14 | 25 | 0.93 | 0.29 | 0.55 |
| P2 | 909.09 | 18.85 | 14 | 1.53 | 0.95 | 0.27 | |
| P3 | 844.16 | 16.43 | 13 | 1.24 | 0.77 | 0.28 |
| Litterfall Types | Annual Litterfall Production (Mg ha−1 year−1) | |
|---|---|---|
| Intact Mangroves | Restored Mangroves | |
| Leaf | 8.38 ± 0.39 | 9.90 ± 0.60 |
| Reproductive | 1.01 ± 0.29 | 2.98 ± 0.62 |
| Wood | 0.75 ± 0.13 | 1.01 ± 0.18 |
| Total | 10.18 ± 0.48 | 13.96 ± 1.02 |
| Species | Forest Type | R2 | Intercept | Decay Rate (day−1) | t0.50 (days) | t0.95 (days) | t0.99 (days) |
|---|---|---|---|---|---|---|---|
| Rhizophora apiculata | Intact | 0.96 | 0.0594 | 0.059 | 12 | 51 | 84 |
| Restored | 0.96 | 0.0509 | 0.051 | 14 | 59 | 98 | |
| Bruguiera gymnorhiza | Intact | 0.87 | 0.0789 | 0.079 | 9 | 38 | 63 |
| Restored | 1.00 | 0.0589 | 0.059 | 12 | 51 | 85 | |
| Avicennia marina | Intact | 0.91 | 0.0362 | 0.036 | 19 | 83 | 138 |
| Restored | 0.99 | 0.0391 | 0.039 | 18 | 77 | 128 | |
| Sonneratia alba | Intact | 0.94 | 0.0808 | 0.081 | 9 | 37 | 62 |
| Restored | 0.93 | 0.0443 | 0.044 | 16 | 68 | 113 |
| Family | Genus | Species | Habitat | Number of Species (Individual) | |
|---|---|---|---|---|---|
| Intact | Restored | ||||
| Crabs | |||||
| Ocypodidae | Tubuca | T. flammula | Epifauna, infauna | 12 | 1 |
| Sesarmidae | Parasesarma | P. eumolpe | Epifauna, infauna | 7 | 5 |
| Snails | |||||
| Assimineidae | Optediceros | O. breviculum | Brackish water, epifauna | 9 | nf |
| Cerithidae | Cerithidea | C. quoyii | Brackish water, epifauna | 14 | 21 |
| Ellobidae | Cassidula | C. angulifera | Epifauna | 89 | 16 |
| Cassidula | C. aurisfelis | Brackish water, epifauna | 55 | 28 | |
| Cassidula | C. nucleus | Brackish water, epifauna | 138 | 129 | |
| Cassidula | C. sowerbyana | Brackish water, epifauna | 4 | 2 | |
| Cassidula | C. sulculosa | Brackish water, epifauna | 11 | 23 | |
| Cassidula | C. vespertiolinis | Brackish water | nf | 22 | |
| Littorinidae | Littoraria | L. articulata | Epifauna | 3 | 1 |
| Littoraria | L. carinifera | Epifauna | 1 | 3 | |
| Littoraria | L. melanostoma | Epifauna | 1 | nf | |
| Littoraria | L. intermedia | Epifauna | 1 | nf | |
| Littoraria | L. scabra | Brackish water, epifauna | 12 | 3 | |
| Muricidae | Chicoreus | C. capucinus | Arboreal, Epifauna | 1 | nf |
| Neritidae | Nerita | N. baltaeta | Epifauna | 1 | 2 |
| Placunidae | Placuna | P. ephippium | Arboreal | 1 | nf |
| Potamididae | Terebralia | T. sulcata | Epifauna | 12 | 6 |
| Total | 372 | 263 | |||
| Index | Intact Mangroves | Restored Mangroves |
|---|---|---|
| Abundance (K) | 44.53 ± 30.9 | 45.17 ± 20.54 |
| Diversity (H′) | 1.48 ± 0.15 | 1.36 ± 0.31 |
| Similarity (E) | 0.71 ± 0.08 | 0.62 ± 0.19 |
| Dominance (D) | 0.27 ± 0.03 | 0.48 ± 0.10 |
| Sites and Countries | Mangrove Species | Forest Type | Total Litterfall Production (Mg ha−1 y−1) | References |
|---|---|---|---|---|
| Southwest Florida, USA | Rhizophora mangle dominated | Intact | 3.73 | [53] |
| Selective pruning | 3.05 | |||
| Atrato River Delta, Southern Caribbean, Colombia | Rhizophora mangle dominated | Intact | 20 | [9] |
| Gazi Bay, Kenya | Rhizophora mucronata and Sonneratia alba | Planted (5–13 years) | 5.22–10.15 | [51] |
| Rhizophora mucronata and Sonneratia alba | Intact | 8.36–10.15 | ||
| Tam Giang Lagoon, Vietnam | Rhizophora apiculata | Planted (7–24 years) | 8.86–14.16 | [11] |
| Sibuti mangrove forest, Sarawak, Malaysia | Mixed species | Intact | 16.41 | [54] |
| Tritih, Central Java, Indonesia | Rhizophora mucronata | Planted (7 years) | 7.06–10.40 | [55] |
| Kema-Bitung, North Sulawesi, Indonesia | Mixed species | Disturbed | 6.66–8.93 | [56] |
| Apar-Adang Nature Reserve, East Kalimantan, Indonesia | Mixed species | Intact | 23.7 | [52] |
| Perancak Estuary, Bali, Indonesia | Mixed species | Intact | 10.18 | This study |
| Rhizophora spp. dominated | Planted (≥14 years in 2021) | 13.96 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pradisty, N.A.; Sidik, F.; Bimantara, Y.; Susetya, I.E.; Basyuni, M. Litterfall and Associated Macrozoobenthic of Restored Mangrove Forests in Abandoned Aquaculture Ponds. Sustainability 2022, 14, 8082. https://doi.org/10.3390/su14138082
Pradisty NA, Sidik F, Bimantara Y, Susetya IE, Basyuni M. Litterfall and Associated Macrozoobenthic of Restored Mangrove Forests in Abandoned Aquaculture Ponds. Sustainability. 2022; 14(13):8082. https://doi.org/10.3390/su14138082
Chicago/Turabian StylePradisty, Novia Arinda, Frida Sidik, Yuntha Bimantara, Ipanna Enggar Susetya, and Mohammad Basyuni. 2022. "Litterfall and Associated Macrozoobenthic of Restored Mangrove Forests in Abandoned Aquaculture Ponds" Sustainability 14, no. 13: 8082. https://doi.org/10.3390/su14138082
APA StylePradisty, N. A., Sidik, F., Bimantara, Y., Susetya, I. E., & Basyuni, M. (2022). Litterfall and Associated Macrozoobenthic of Restored Mangrove Forests in Abandoned Aquaculture Ponds. Sustainability, 14(13), 8082. https://doi.org/10.3390/su14138082









