Influence of Rice Husk Biochar and Lime in Reducing Phosphorus Application Rate in Acid Soil: A Field Trial with Maize
Abstract
:1. Introduction
2. Materials and Methods
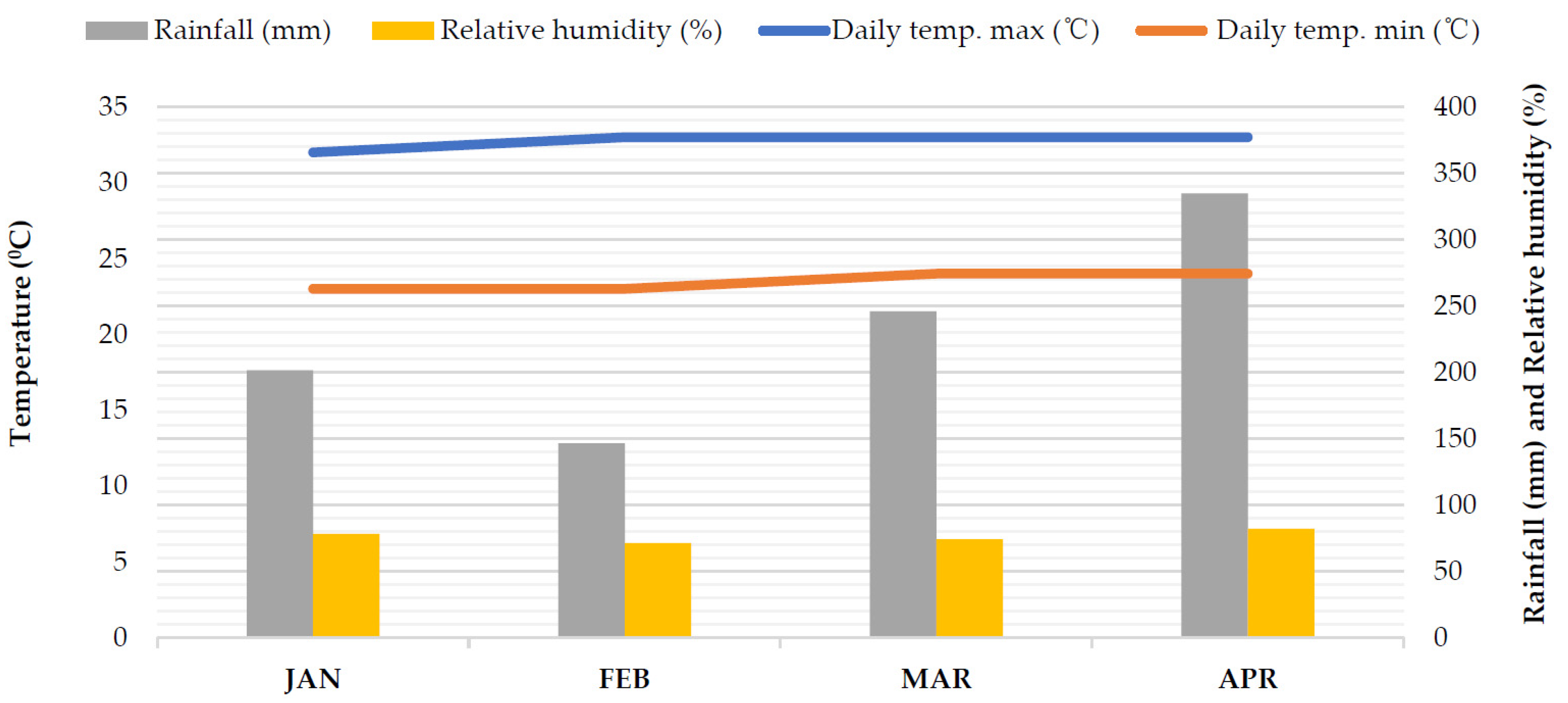
2.1. Experimental Site
2.2. Soil Properties
2.3. Characteristics of Rice Husk Biochar (RHB)
2.4. Experimental Layout and Treatment
2.5. Agronomic Practice
2.6. Plant Analysis
2.7. Determination of Protein Content
2.8. Soil CO2 Gas Measurement
2.9. Statistical Analysis
3. Results
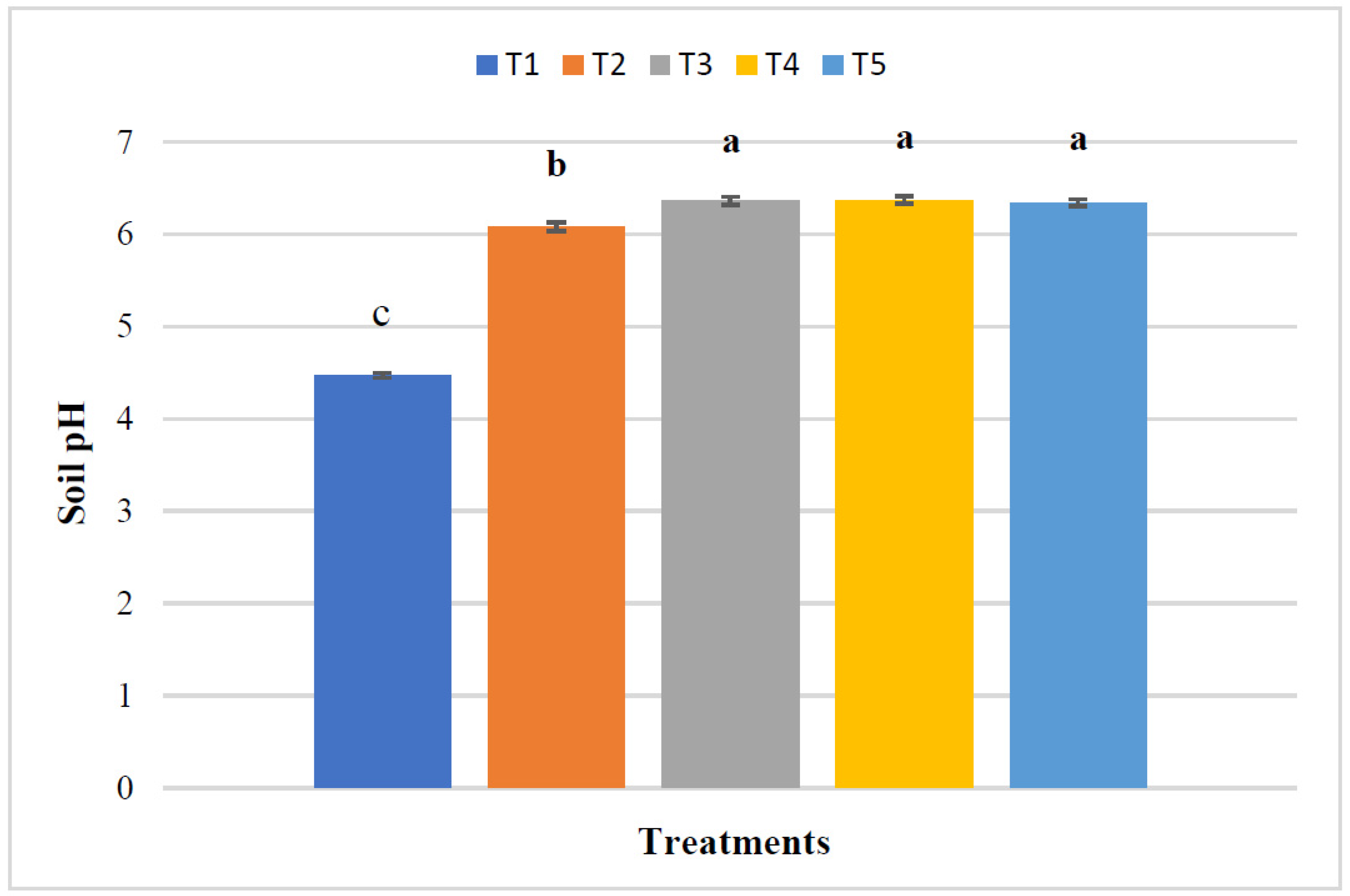
3.1. Effect of RHB, Lime, and Varying Doses of Phosphorus on Soil Properties at Harvest
3.2. Effect of RHB, Lime, and Varying Doses of Phosphorus on the Growth and Yield Components of Maize
3.3. Effect of RHB, Lime, and Varying Doses of Phosphorus on Concentration and Nutrient Uptake by Maize
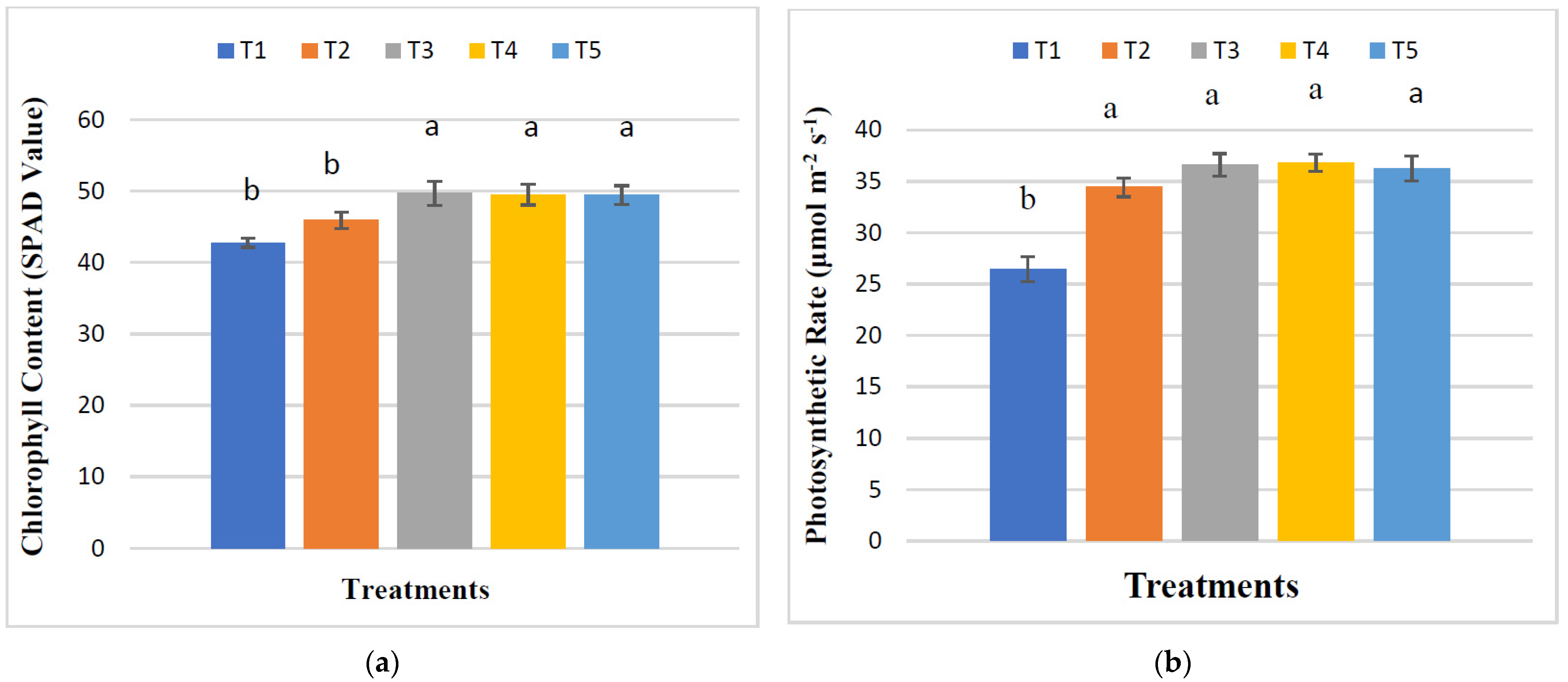
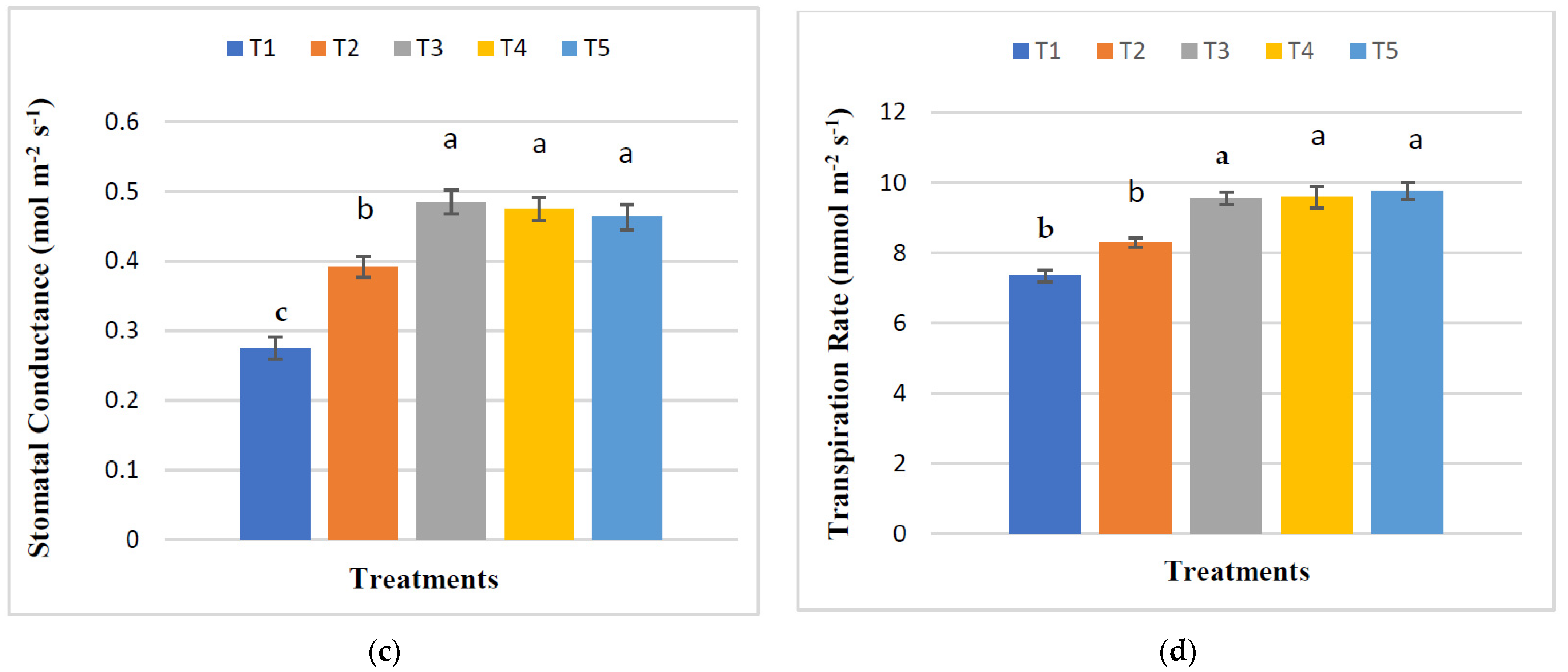
3.4. Effect of RHB, Lime, and Varying Doses of Phosphorus on the Physiological Parameters of Maize
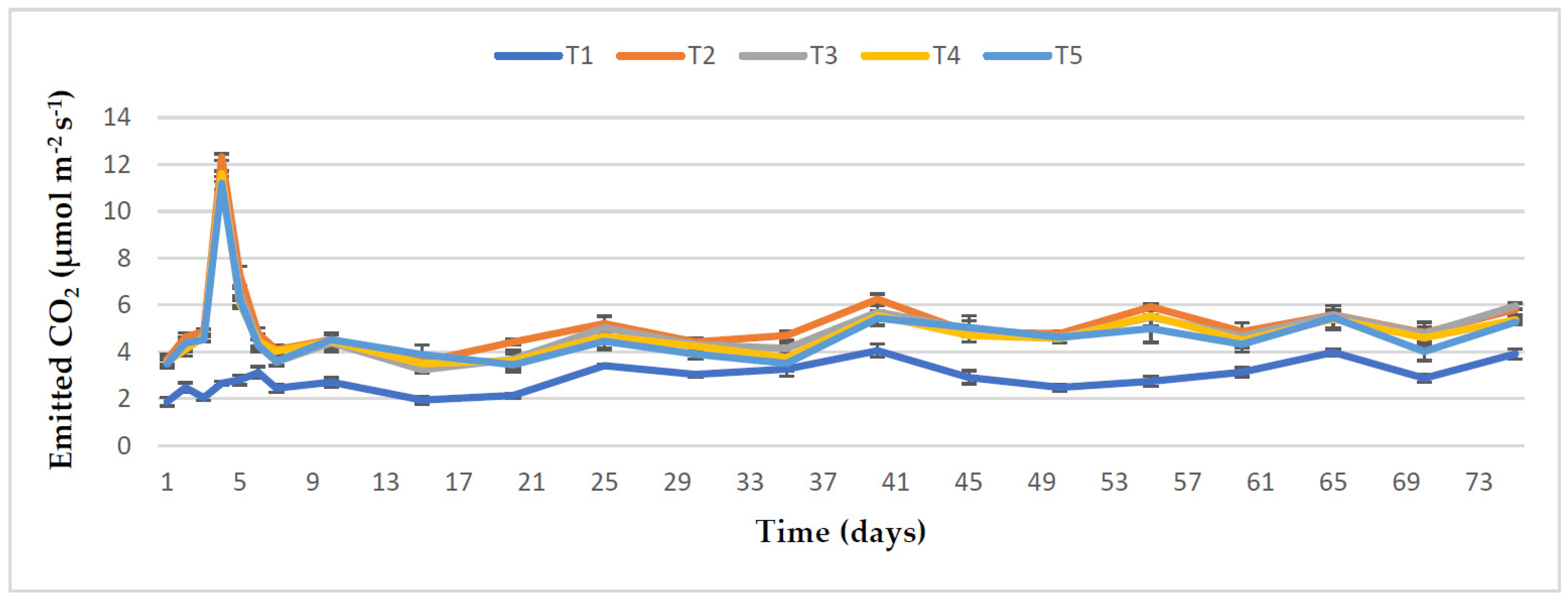
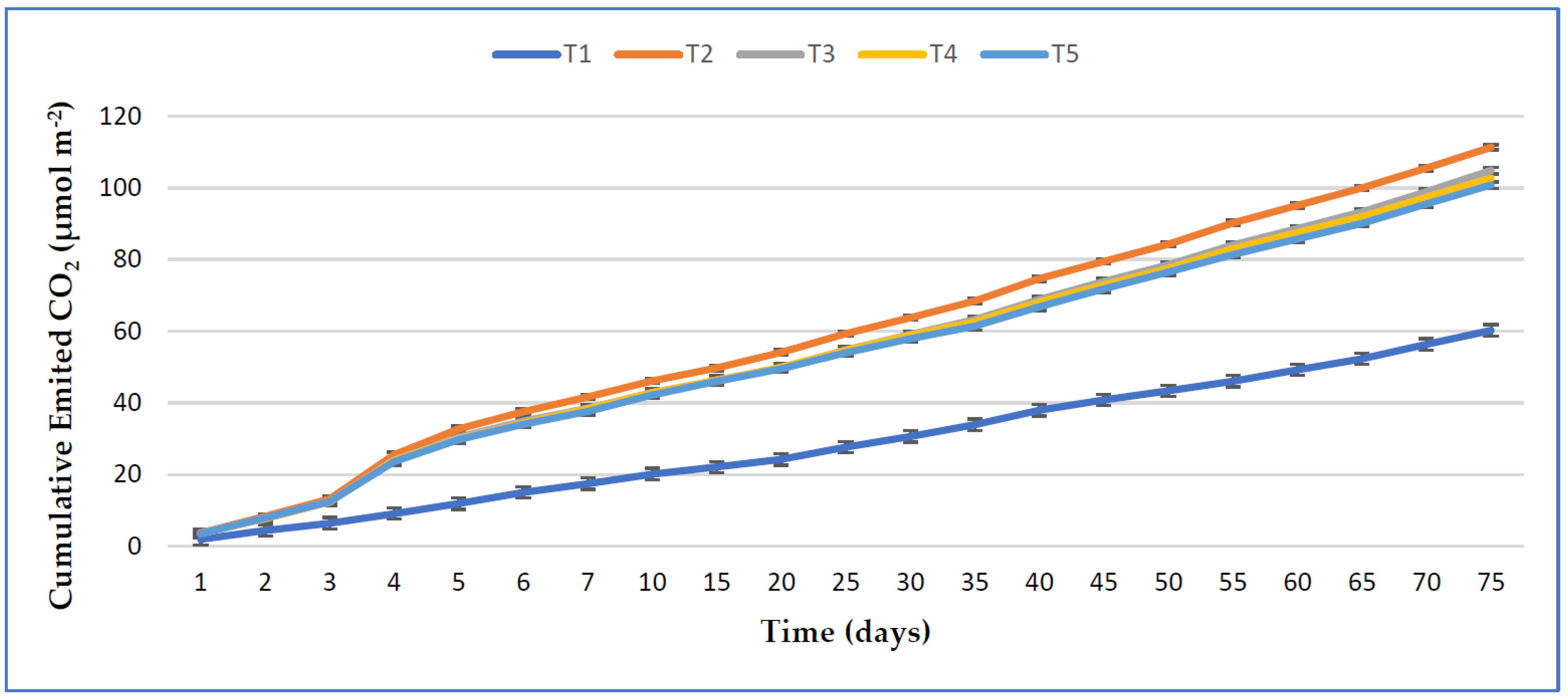
3.5. Effect of RHB, Lime, and Varying Doses of Phosphorus on Soil CO2 Emission from the Maize Field
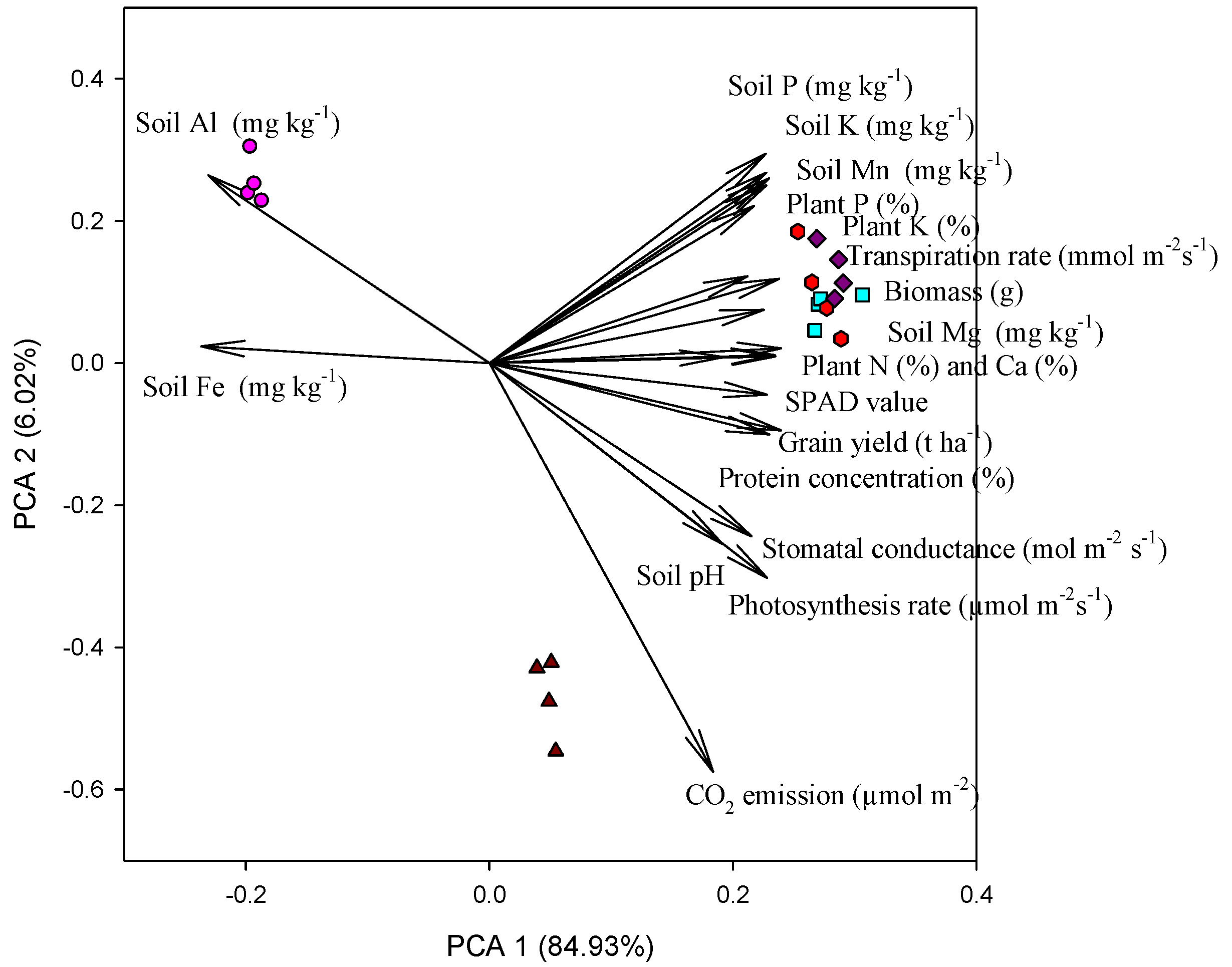
3.6. Principal Component Analysis
4. Discussion
4.1. Impact of RHB, Lime, and Different Doses of Phosphorus on Soil Nutrients
4.2. Impact of RHB, Lime, and Different Doses of Phosphorus on Plant Growth Variables
4.3. Impact of RHB, Lime, and Different Doses of Phosphorus on Plant Nutrient Concentration and Grain Uptake
4.4. Impact of RHB, Lime, and Different Rates of Phosphorus on Soil CO2 Emission
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Reddy, Y.R.; Ravi, D.; Reddy, C.H.R.; Prasad, K.V.S.V.; Zaidi, P.H.; Vinayan, M.T.; Blümmel, M. A note on the correlations between maize grain and maize stover quantitative and qualitative traits and the implications for whole maize plant optimization. Field Crops Res. 2013, 153, 63–69. [Google Scholar] [CrossRef]
- von Uexküll, H.R.; Mutert, E. Global extent, development and economic impact of acid soils. Plant Soil 1995, 171, 1–15. [Google Scholar] [CrossRef]
- Guo, J.; Liu, X.; Zhang, Y.; Shen, J.; Han, W.; Zhang, W.; Christie, P.; Goulding, K.W.T.; Vitousek, P.M.; Zhang, F. Significant acidification in major Chinese croplands. Science 2010, 327, 1008–1010. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryan, P.R. Assessing the role of genetics for improving the yield of Australia’s major grain crops on acid soils. Crop Pasture Sci. 2018, 69, 242–264. [Google Scholar] [CrossRef]
- Abreha, K.; Heluf, G.; Tekalign, M.; Kindie, T. Wheat crop response to liming materials and N and P fertilizers in acidic soils of Tsegede highlands, northern Ethiopia. Agric. For. Fish. 2013, 2, 126–135. [Google Scholar] [CrossRef]
- Butterly, C.R.; Costello, B.; Lauricella, D.; Sale, P.; Li, G.; Tang, C. Alkalinity movement down acid soil columns was faster when lime and plant residues were combined than when either was applied separately. Eur. J. Soil Sci. Adv. 2021, 72, 313–325. [Google Scholar] [CrossRef]
- Wang, M.; Xian-Jun, J. Effects of applying lime and calcium montmorillonite on nitrification dynamics in acidic soil. J. Agric. Resour. Environ. 2017, 34, 47–53. [Google Scholar] [CrossRef]
- Huber, C.; Baier, R.; Göttlein, A.; Weis, W. Changes in soil, seepage water and needle chemistry between 1984 and 2004 after liming an N-saturated Norway spruce stand at the Höglwald, Germany. For. Ecol. Manag. 2006, 233, 11–20. [Google Scholar] [CrossRef]
- Wu, S.; Zhang, Y.; Tan, Q.; Sun, X.; Wei, W.; Hu, C. Science of the Total Environment Biochar is superior to lime in improving acidic soil properties and fruit quality of Satsuma mandarin. Sci. Total Environ. 2020, 714, 136722. [Google Scholar] [CrossRef]
- Lustosa Carvalho, M.; Tuzzin de Moraes, M.; Cerri, C.E.P.; Cherubin, M.R. Biochar amendment enhances water retention in a tropical sandy soil. Agriculture 2020, 10, 62. [Google Scholar] [CrossRef] [Green Version]
- Uslu, O.S.; Babur, E.; Alma, M.H.; Solaiman, Z.M. Walnut Shell Biochar Increases Seed Germination and Early Growth of Seedlings of Fodder Crops. Agriculture 2020, 10, 427. [Google Scholar] [CrossRef]
- Dai, Z.; Zhang, X.; Tang, C.; Muhammad, N.; Wu, J.; Brookes, P.C.; Xu, J. Potential role of biochars in decreasing soil acidification—A critical review. Sci. Total Environ. 2017, 581–582, 601–611. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.H.; Xu, R.K. The amelioration effects of low temperature biochar generated from nine crop residues on an acidic Ultisol. Soil Use Manag. 2011, 27, 110–115. [Google Scholar] [CrossRef]
- Mia, S.; Dijkstra, F.A.; Singh, B. Aging induced changes in biochar’s functionality and adsorption behavior for phosphate and ammonium. Environ. Sci. Technol. 2017, 51, 8359–8367. [Google Scholar] [CrossRef] [PubMed]
- Silber, A.; Levkovitch, I.; Graber, E.R. pH-Dependent Mineral Release and Surface Properties of Cornstraw Biochar: Agronomic Implications. Environ. Sci. Technol. 2010, 44, 9318–9323. [Google Scholar] [CrossRef] [PubMed]
- Agegnehu, G.; Srivastava, A.K.; Bird, M.I. The role of biochar and biochar-compost in improving soil quality and crop performance: A review. Appl. Soil Ecol. 2017, 119, 156–170. [Google Scholar] [CrossRef]
- Palansooriya, K.N.; Ok, Y.S.; Awad, Y.M.; Lee, S.S.; Sung, J.-K.; Koutsospyros, A.; Moon, D.H. Impacts of biochar application on upland agriculture: A review. J. Environ. Manag. 2019, 234, 52–64. [Google Scholar] [CrossRef]
- Yu, H.; Zou, W.; Chen, J.; Chen, H.; Yu, Z.; Huang, J.; Tang, H.; Wei, X.; Gao, B. Biochar amendment improves crop production in problem soils: A review. J. Environ. Manag. 2019, 232, 8–21. [Google Scholar] [CrossRef]
- Sarfraz, R.; Hussain, A.; Sabir, A.; Ben Fekih, I.; Ditta, A.; Xing, S. Role of biochar and plant growth promoting rhizobacteria to enhance soil carbon sequestration-a review. Environ. Monit. Assess. 2019, 191, 251. [Google Scholar] [CrossRef]
- Solaiman, Z.M.; Shafi, M.I.; Beamont, E.; Anawar, H.M. Poultry litter biochar increases mycorrhizal colonisation, soil fertility and cucumber yield in a fertigation system on sandy soil. Agriculture 2020, 10, 480. [Google Scholar] [CrossRef]
- Liu, L.Y.; Tan, Z.X.; Gong, H.B.; Huang, Q.Y. Migration and transformation mechanisms of nutrient elements (N, P, K) within biochar in straw-biochar-soil-plant systems: A review. ACS Sustain. Chem. Eng. 2019, 7, 22–32. [Google Scholar] [CrossRef]
- Lin, Q.; Zhang, L.; Riaz, M.; Zhang, M.; Xia, H.; Lv, B.; Jiang, C. Assessing the potential of biochar and aged biochar to alleviate aluminum toxicity in an acid soil for achieving cabbage productivity. Ecotoxicol. Environ. Saf. 2018, 161, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Gul, S.; Whalen, J.K. Biochemical cycling of nitrogen and phosphorus in biochar-amended soils. Soil Biol. Biochem. 2016, 103, 1–15. [Google Scholar] [CrossRef]
- Zhao, Y.H.; Zhao, L.; Mei, Y.Y.; Li, F.Y.; Cao, X.D. Release of nutrients and heavy metals from biochar-amended soil under environmentally relevant conditions. Environ. Sci. Pollut. Res. 2018, 25, 2517–2527. [Google Scholar] [CrossRef] [PubMed]
- Mete, F.Z.; Mia, S.; Dijkstra, F.A.; Abuyusuf, M.; Hossain, A.S.M.I. Synergistic effects of biochar and NPK fertilizer on soybean yield in an alkaline soil. Pedosphere 2015, 25, 713–719. [Google Scholar] [CrossRef]
- Kemmitt, S.J.; Wright, D.; Goulding, K.W.T.; Jones, D.L. pH regulation of carbon and nitrogen dynamics in two agricultural soils. Soil Biol. Biochem. 2006, 38, 898–911. [Google Scholar] [CrossRef]
- Kirkham, J.; Rowe, B.; Doyle, R. Persistent improvements in the structure and hydraulic conductivity of a ferrosol due to liming. Aust. J. Soil Res. 2007, 45, 218–223. [Google Scholar] [CrossRef]
- Gomes, E.A.; Lana, U.G.P.; Quensen, J.F.; de Sousa, S.M.; Oliveira, C.A.; Guo, J.; Guimarães, L.J.; Tiedje, J.M. Root-associated microbiome of maize genotypes with contrasting phosphorus use efficiency. Phytobiomes 2018, 2, 129–137. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.Z.; Chen, C.R.; Gray, E.M.; Boyd, S.E.; Yang, H.; Zhang, D.K. Roles of biochar in improving phosphorus availability in soils: A phosphate adsorbent and a source of available phosphorus. Geoderma 2016, 276, 1–6. [Google Scholar] [CrossRef]
- Hiemstra, T.; Mia, S.; Duhaut, P.B.; Molleman, B. Natural and pyrogenic humic acids at goethite and natural oxide surfaces interacting with phosphate. Environ. Sci. Technol. 2013, 47, 9182–9189. [Google Scholar] [CrossRef]
- Mia, S.; Singh, B.; Dijkstra, F.A. Chemically oxidized biochar increases ammonium-15 N recovery and phosphorus uptake in a grassland. Biol. Fertil. Soils 2019, 55, 577–588. [Google Scholar] [CrossRef]
- Chintala, R.; Mollinedo, J.; Schumacher, T.E.; Malo, D.D.; Julson, J.L. Effect of Biochar on Chemical Properties of Acidic Soil. Arch. Agron. Soil Sci. 2014, 60, 393–404. [Google Scholar] [CrossRef]
- Lehmann, J.; da Silva, J.P.; Steiner, C.; Nehls, T.; Zech, W.; Glaser, B. Nutrient availability and leaching in an archaeological Anthrosol and a Ferralsol of the Central Amazon basin: Fertilizer, manure and charcoal amendments. Plant Soil 2003, 249, 343–357. [Google Scholar] [CrossRef]
- Mosharrof, M.; Uddin, M.K.; Sulaiman, M.F.; Mia, S.; Shamsuzzaman, S.M.; Haque, A.N.A. Combined Application of Rice Husk Biochar and Lime Increases Phosphorus Availability and Maize Yield in an Acidic Soil. Agriculture 2021, 11, 793. [Google Scholar] [CrossRef]
- Bouyoucos, G.J. Hydrometer method improved for making particle size analysis of soils. Agron. J. 1962, 54, 464–465. [Google Scholar] [CrossRef]
- Benton, J.J. Laboratory Guide for Conducting Soil Tests and Plant Analysis; CRC Press: Boca Raton, FL, USA, 2001; ISBN 9780429132117. [Google Scholar]
- Schollenberger, C.J.; Simon, R.H. Determination of exchange capacity and exchangeable bases in soil-ammonium acetate method. Soil Sci. 1945, 59, 13–24. [Google Scholar] [CrossRef]
- Elisa, A.A.; Ninomiya, S.; Shamshuddin, J.; Roslan, I. Alleviating aluminum toxicity in an acid sulfate soil from Peninsular Malaysia by calcium silicate application. Solid Earth 2016, 7, 367–374. [Google Scholar] [CrossRef] [Green Version]
- Tan, K.H. Soil and plant test. In Soil Sampling, Preparation, and Analysis, 2nd ed.; Tan, K.H., Ed.; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2005; pp. 98–134. [Google Scholar]
- Bray, R.H.; Kurtz, L.T. Determination of total, organic, and available forms of phosphorus in soils. Soil Sci. 1945, 59, 39–46. [Google Scholar] [CrossRef]
- Ahmedna, M.; Marshall, W.E.; Rao, R.M. Production of granular activated carbon from select agricultural by-products and evaluation of their physical, chemical, and adsorption properties. Bioresour. Technol. 1998, 71, 113–123. [Google Scholar] [CrossRef]
- Cottenie, A. Soil testing and plant testing as a basis of fertilizer recommendation. FAO Soil Bull. 1980, 38, 70–73. [Google Scholar]
- Pedram, K. Genetic Potential of Selected Sweet Corn Inbred Lines and Analysis of Their Combining Ability Assisted by Microsatellite DNA Markers. Ph.D. Thesis, Universiti Putra Malaysia, Seri Kembangan, Malaysia, 2012. [Google Scholar]
- Mosharrof, M.; Uddin, M.K.; Jusop, S.; Sulaiman, M.F.; Shamsuzzaman, S.M.; Haque, A.N.A. Changes in Acidic Soil Chemical Properties and Carbon Dioxide Emission Due to Biochar and Lime Treatments. Agriculture 2021, 11, 219. [Google Scholar] [CrossRef]
- Lija, M.; Ahmed, O.H.; Susilawati, K. Maize (Zea mays L.) nutrient use efficiency as affected by formulated fertilizer with Clinoptilolite Zeolite. Emir. J. Food Agric. 2014, 26, 284–292. [Google Scholar] [CrossRef]
- Fageria, N.K.; Baligar, V.C.; Jones, C. Growth and Mineral Nutrition of Field Crops, 2nd ed.; Marcel Dekker, Inc.: New York, NY, USA, 1997; p. 494. [Google Scholar]
- Yuan, Z.; Cao, Q.; Zhang, K.; Ata-Ul-Karim, S.T.; Tian, Y.; Zhu, Y.; Cao, W.; Liu, X. Optimal leaf positions for SPAD meter measurement in rice. Front. Plant Sci. 2016, 7, 719. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gezahegn, A.M. Integrated Nutrient Management for Maize-Soybean Cropping System. Ph.D. Thesis, Universiti Putra Malaysia, Seri Kembangan, Malaysia, 2016. [Google Scholar]
- Iqbal, J.; Hu, R.G.; Feng, M.; Lin, S.; Malghani, S.; Ali, I.M. Microbial biomass, and dissolved organic carbon and nitrogen strongly affect soil respiration in different land uses: A case study at Three Gorges Reservoir Area, South China. Agric. Ecosyst. Environ. 2010, 137, 294–307. [Google Scholar] [CrossRef]
- Shen, Y.; Zhu, L.; Cheng, H.; Yue, S.; Li, S. Effects of biochar application on CO2 emissions from a cultivated soil under semiarid climate conditions in Northwest China. Sustainability 2017, 9, 1482. [Google Scholar] [CrossRef] [Green Version]
- Masud, M.M.; Abdulaha-Al Baquyb, M.; Akhtera, S.; Sena, R.; Barmana, A.; Khatuna, M.R. Liming effects of poultry litter derived biochar on soil acidity amelioration and maize growth. Ecotoxicol. Environ. Saf. 2020, 202, 110865. [Google Scholar] [CrossRef]
- Shi, R.Y.; Ni, N.; Nkoh, J.N.; Li, J.Y.; Xu, R.K.; Qian, W. Beneficial dual role of biochars in inhibiting soil acidification resulting from nitrification. Chemosphere 2019, 234, 43–51. [Google Scholar] [CrossRef]
- Eduah, J.O.; Nartey, E.K.; Abekoe, M.K.; Henriksen, S.W.; Andersen, M.N. Mechanism of orthophosphate (PO4-P) adsorption onto different biochars. Environ. Technol. Innov. 2020, 17, 100572. [Google Scholar] [CrossRef]
- Panhwar, Q.A.; Naher, U.A.; Shamshuddin, J.; Ismail, M.R. Effects of biochar and ground magnesium limestone application, with or without bio-Fertilizer addition, on biochemical properties of an acid sulfate soil and rice yield. Agronomy 2020, 10, 1100. [Google Scholar] [CrossRef]
- Islam, M.R.; Akter, A.; Hoque, M.A.; Farzana, S.; Uddin, S.; Talukder, M.M.H.; Alsanie, W.F.; Gaber, A.; Hossain, M.A. Lime and Organic Manure Amendment: A Potential Approach for Sustaining Crop Productivity of the T. Aman-Maize-Fallow Cropping Pattern in Acidic Piedmont Soils. Sustainability 2021, 13, 9808. [Google Scholar] [CrossRef]
- Islam, M.R.; Jahan, R.; Uddin, S.; Harine, I.J.; Hoque, M.A.; Hassan, S.; Hassan, M.M.; Hossain, M.A. Lime and Organic Manure Amendment Enhances Crop Productivity of Wheat–Mungbean–T. Aman Cropping Pattern in Acidic Piedmont Soils. Agronomy 2021, 11, 1595. [Google Scholar] [CrossRef]
- Dari, B.; Nair, V.D.; Harris, W.G.; Nair, P.K.R.; Sollenberger, L.; Mylavarapu, R. Relative influence of soil vs. biochar properties on soil phosphorus retention. Geoderma 2016, 280, 82–87. [Google Scholar] [CrossRef] [Green Version]
- Piash, M.I.; Iwabuchi, K.; Itoh, T.; Uemura, K. Release of essential plant nutrients from manure and wood-based biochars. Geoderma 2021, 397, 115100. [Google Scholar] [CrossRef]
- Pandit, N.R.; Mulder, J.; Hale, S.E.; Martinsen, V.; Schmidt, H.P.; Cornelissen, G. Biochar improves maize growth by alleviation of nutrient stress in a moderately acidic low-input Nepalese soil. Sci. Total Environ. 2018, 625, 1380–1389. [Google Scholar] [CrossRef]
- Rabileh, M.A.; Shamshuddin, J.; Panhwar, Q.A.; Rosenani, A.B.; Anuar, A.R. Effects of biochar and/or dolomitic limestone application on the properties of Ultisol cropped to maize under glasshouse conditions. Can. J. Soil Sci. 2015, 95, 37–47. [Google Scholar] [CrossRef]
- Mia, S.; Dijkstra, F.; Singh, B. Long-term aging of biochar: A molecular understanding with agricultural and environmental implications. Adv. Agron. 2017, 141, 1–51. [Google Scholar] [CrossRef]
- Cheng, C.H.; Lehmann, J.; Thies, J.E.; Burton, S.D.; Engelhard, M.H. Oxidation of black carbon by biotic and abiotic processes. Org. Geochem. 2006, 37, 1477–1488. [Google Scholar] [CrossRef]
- Shetty, R.; Vidya, C.S.N.; Prakash, N.B.; Lux, A.; Vaculík, M. Aluminum toxicity in plants and its possible mitigation in acid soils by biochar: A review. Sci. Total Environ. 2021, 765, 142744. [Google Scholar] [CrossRef]
- Ch’ng, H.Y.; Ahmed, O.H.; Majid, N.M.A.; Jalloh, M.B. Improving soil phosphorus availability and yield of Zea mays L. using biochar and compost derived from agro-industrial waste. Ital. J. Agron. 2019, 14, 1107. [Google Scholar] [CrossRef] [Green Version]
- Sasmita, K.D.; Iswandi, A.; Syaiful, A.; Sudirman, Y.; Gunawan, D. Application of biochar and organic fertilizer on acid soil as growing medium for Cacao (Theobroma cacao L.) seedlings. Int. J. Sci. Basic Appl. Res. 2017, 36, 261–273. [Google Scholar]
- Fageria, N.K.; Baligar, V.C. Ameliorating soil acidity of tropical Oxisols by liming for sustainable crop production. Adv. Agron. 2008, 99, 345–399. [Google Scholar] [CrossRef]
- Akinrinde, E.A. Strategies for improving crops’ use-effciencies of fertilizer nutrients in sustainable agricultural systems. Pak. J. Nutr. 2006, 5, 185–193. [Google Scholar] [CrossRef] [Green Version]
- Steiner, C.; Teixeira, W.G.; Lehmann, J.; Nehls, T.; Vasconcelos de Macêdo, J.L.; Blum, W.E.H.; Zech, W. Long-term effect of manure, charcoal and mineral fertilization on crop production and fertility on highly weathered central amazonian upland soil. Plant Soil 2007, 291, 275–290. [Google Scholar] [CrossRef] [Green Version]
- Ndor, E.; Jayeoba, J.O.; Asadu, C.L.A.; Iheshiulo, E.M.-A. Growth, nutrient uptake and dry matter yield of maize (Zea mays L.) grown in soil amended with rice husk and sawdust biochar. Int. J. Sci. Res. Agric. Sci. 2016, 3, 99–103. [Google Scholar] [CrossRef]
- Gandahi, A.W.; Baloch, F.B.; Sarki, M.S.; Gandahi, R.; Lashari, M.S. Impact of Rice Husk Biochar and Macronutrient Fertilizer on Fodder Maize and Soil Properties. Int. J. Biosci. 2015, 7, 12–21. [Google Scholar] [CrossRef]
- Nekesa, A.O.; Okaebo, J.R.; Otheno, C.O.; Thuita, M.N.; Kipsat, M.; Batiano, A.; Sanginga, N.; Kimettu, J.; Vanlauwe, B. The potential of Minjingu phosphate rock from Tanzania as a liming material: Effect on maize and bean intercrop on acid soils of western Kenya. Afr. Crop Sci. Conf. Proc. 2005, 7, 1121–1128. [Google Scholar]
- Liu, B.; Yue, Y.M.; Li, R.; Shen, W.J.; Wang, K.L. Plant leaf chlorophyll content retrieval based on a field imaging spectroscopy system. Sensors 2014, 14, 19910–19925. [Google Scholar] [CrossRef] [Green Version]
- Parthasarathi, T.; Vanitha, K.; Lakshamanakumar, P.; Kalaiyarasi, D. Aerobic rice-mitigating water stress for the future climate change. Int. J. Agron. Plant Prod. 2012, 3, 241–254. [Google Scholar]
- Anjum, S.A.; Xie, X.Y.; Wang, L.C.; Saleem, M.F.; Man, C.; Lei, W. Morphological, physiological and biochemical responses of plants to drought stress. Afr. J. Agric. Res. 2011, 6, 2026–2032. [Google Scholar] [CrossRef]
- Panhwar, Q.; Naher, U.; Radziah, O.; Shamshuddin, J.; Razi, I.M. Bio-fertilizer, ground magnesium limestone and basalt applications may improve chemical properties of Malaysian acid sulfate soils and rice growth. Pedosphere 2014, 24, 827–835. [Google Scholar] [CrossRef]
- Cao, Y.; Ma, Y.; Guo, D.; Wang, Q.; Wang, G. Chemical properties and microbial responses to biochar and compost amendments in the soil under continuous watermelon cropping. Plant Soil Environ. 2017, 63, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Moreno-Cornejo, J.; Zornoza, R.; Faz, A. Carbon and nitrogen mineralization during decomposition of crop residues in a calcareous soil. Geoderma 2014, 230–231, 58–63. [Google Scholar] [CrossRef]
- El-Naggar, A.H.; Usman, A.R.A.; Al-Omran, A.; Yong, S.O.; Ahmad, M.; Al-Wabel, M.I. Carbon mineralization and nutrient availability in calcareous sandy soils amended with woody waste biochar. Chemosphere 2015, 138, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Madiba, O.F.; Solaiman, Z.M.; Carson, J.K.; Murphy, D.V. Biochar increases availability and uptake of phosphorus to wheat under leaching conditions. Biol. Fertil. Soils 2016, 52, 439–446. [Google Scholar] [CrossRef]
- Islam, M.R.; Bilkis, S.; Hoque, T.S.; Uddin, S.; Jahiruddin, M.; Rahman, M.M.; Rahman, M.M.; Alhomrani, M.; Gaber, A.; Hossain, M.A. Mineralization of Farm Manures and Slurries for Successive Release of Carbon and Nitrogen in Incubated Soils Varying in Moisture Status under Controlled Laboratory Conditions. Agriculture 2021, 11, 846. [Google Scholar] [CrossRef]
- Reed, E.Y.; Chadwick, D.R.; Hill, P.W.; Jones, D.L. A critical comparison of the impact of biochar and wood ash on soil organic matter cycling and grassland productivity. Soil Biol. Biochem. 2017, 110, 134–142. [Google Scholar] [CrossRef] [Green Version]
- Ameloot, N.; Sleutel, S.; Case, S.D.C.; Alberti, G.; McNamara, N.P.; Zavalloni, C.; Vervisch, B.; DelleVedove, G.; De Neve, S. C mineralization and microbial activity in four biochar field experiments several years after incorporation. Soil Biol. Biochem. 2014, 78, 195–203. [Google Scholar] [CrossRef]
- Case, S.D.C.; Mcnamara, N.P.; Reay, D.S.; Whitaker, J. Can biochar reduce soil greenhouse gas emissions from a Miscanthus bioenergy crop? GCB Bioenergy 2014, 6, 76–89. [Google Scholar] [CrossRef] [Green Version]
- West, T.O.; McBride, A.C. The contribution of agricultural lime to carbon dioxide emissions in the United States: Dissolution, transport, and net emissions. Agric. Ecosyst. Environ. 2005, 108, 145–154. [Google Scholar] [CrossRef]
- Brassard, P.; Godbout, S.; Raghavan, V. Soil biochar amendment as a climate change mitigation tool: Key parameters and mechanisms involved. J. Environ. Manag. 2016, 181, 484–497. [Google Scholar] [CrossRef]
- Fang, Y.Y.; Singh, B.P.; Luo, Y.; Boersma, M.; Van Zwieten, L. Biochar carbon dynamics in physically separated fractions and microbial use efficiency in contrasting soils under temperate pastures. Soil Biol. Biochem. 2018, 116, 399–409. [Google Scholar] [CrossRef]
- Fang, Y.; Singh, B.P.; Singh, B. Temperature sensitivity of biochar and native carbon mineralization in biochar-amended soils. Agric. Ecosyst. Environ. 2014, 191, 158–167. [Google Scholar] [CrossRef]
| Treatment | P (mg kg−1) | K (cmolc kg−1) | Ca (cmolc kg−1) | Mg (cmolc kg−1) | Al (cmolc kg−1) | Fe (mg kg−1) | Mn (mg kg−1) |
|---|---|---|---|---|---|---|---|
| T1 | 4.37b ± 0.24 | 0.25b ± 0.026 | 0.93c ± 0.019 | 0.36c ± 0.022 | 2.64a ± 0.019 | 122.40a ± 1.71 | 2.54b ± 0.074 |
| T2 | 6.89b ± 0.60 | 0.42b ± 0.040 | 1.61b ± 0.075 | 0.91b ± 0.033 | 0.55b ± 0.037 | 94.33b ± 1.78 | 2.86b ± 0.052 |
| T3 | 17.11a ± 0.66 | 1.31a ± 0.056 | 3.32a ± 0.058 | 1.47a ± 0.047 | 0.08c ± 0.016 | 72.61c ± 2.34 | 4.84a ± 0.099 |
| T4 | 16.22a ± 0.73 | 1.32a ± 0.049 | 3.32a ± 0.056 | 1.46a ± 0.054 | 0.07c ± 0.018 | 72.97c ± 1.46 | 4.81a ± 0.074 |
| T5 | 15.39a ± 0.45 | 1.31a ± 0.056 | 3.31a ± 0.069 | 1.48a ± 0.056 | 0.09c ± 0.017 | 71.50c ± 2.25 | 4.84a ± 0.089 |
| Treatment | Plant Height (cm) | Stem Diameter (cm) | Dry Biomass (g) | Fresh Cob Weight (g) | No. of Grain Cob−1 | Cob Length (cm) | Cob Yield (t ha−1) |
|---|---|---|---|---|---|---|---|
| T1 | 190.30c ± 2.45 | 2.10c ± 0.024 | 42.30c ± 1.61 | 221.85c ± 2.17 | 443c ± 3.42 | 18.55c ± 0.427 | 11.83c ± 0.116 |
| T2 | 216.48b ± 1.70 | 2.36b ± 0.022 | 60.48b ± 1.03 | 307.63b ± 4.00 | 514b ± 3.28 | 23.25b ± 0.477 | 16.41b ± 0.213 |
| T3 | 233.85a ± 1.52 | 2.79a ± 0.018 | 92.03a ± 1.55 | 360.13a ± 2.13 | 644a ± 3.97 | 24.88a ± 0.246 | 19.21a ± 0.114 |
| T4 | 232.05a ± 2.07 | 2.81a ± 0.017 | 93.79a ± 1.43 | 357.05a ± 3.27 | 643a ± 3.38 | 24.35ab ± 0.247 | 19.04a ± 0.176 |
| T5 | 232.36a ± 2.63 | 2.80a ± 0.017 | 90.71a ± 1.68 | 357.50a ± 1.69 | 635a ± 3.59 | 24.23ab ± 0.232 | 19.07a ± 0.091 |
| Treatment | N (%) | P (%) | K (%) | Ca (%) | Mg (%) |
|---|---|---|---|---|---|
| T1 | 0.29c ± 0.004 | 0.085d ± 0.006 | 0.106b ± 0.004 | 0.135c ± 0.01 | 0.063b ± 0.009 |
| T2 | 0.36b ± 0.006 | 0.105c ± 0.003 | 0.12b ± 0.004 | 0.168b ± 0.009 | 0.096a ± 0.005 |
| T3 | 0.44a ± 0.009 | 0.175a ± 0.003 | 0.177a ± 0.002 | 0.208a ± 0.009 | 0.108a ± 0.009 |
| T4 | 0.45a ± 0.008 | 0.15b ± 0.004 | 0.167a ± 0.002 | 0.225a ± 0.006 | 0.105a ± 0.006 |
| T5 | 0.42a ± 0.014 | 0.14b ± 0.004 | 0.176a ± 0.002 | 0.235a ± 0.012 | 0.115a ± 0.006 |
| Total Uptake (kg ha−1) | ||||||
|---|---|---|---|---|---|---|
| Treatment | N | P | K | Ca | Mg | Protein Content in Maize (%) |
| T1 | 34.15c ± 0.74 | 10.07d ± 0.66 | 12.54c ± 0.44 | 15.96c ± 1.17 | 7.37c ± 0.96 | 1.81c ± 0.03 |
| T2 | 59.72b ± 1.21 | 17.39c ± 0.47 | 19.39b ± 0.60 | 27.50b ± 1.53 | 15.97b ± 0.58 | 2.28b ± 0.04 |
| T3 | 84.79a ± 1.54 | 33.42a ± 0.56 | 34.17a ± 0.80 | 39.83a ± 1.49 | 20.67ab ± 1.72 | 2.76a ± 0.05 |
| T4 | 86.06a ± 1.01 | 28.39b ± 0.43 | 31.87a ± 0.97 | 42.83a ± 1.13 | 20.02ab ± 1.38 | 2.83a ± 0.04 |
| T5 | 80.70a ± 2.75 | 26.66b ± 0.76 | 34.11a ± 0.51 | 44.81a ± 2.35 | 21.95a ± 1.24 | 2.65a ± 0.08 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mosharrof, M.; Uddin, M.K.; Mia, S.; Sulaiman, M.F.; Shamsuzzaman, S.M.; Haque, A.N.A. Influence of Rice Husk Biochar and Lime in Reducing Phosphorus Application Rate in Acid Soil: A Field Trial with Maize. Sustainability 2022, 14, 7418. https://doi.org/10.3390/su14127418
Mosharrof M, Uddin MK, Mia S, Sulaiman MF, Shamsuzzaman SM, Haque ANA. Influence of Rice Husk Biochar and Lime in Reducing Phosphorus Application Rate in Acid Soil: A Field Trial with Maize. Sustainability. 2022; 14(12):7418. https://doi.org/10.3390/su14127418
Chicago/Turabian StyleMosharrof, Mehnaz, Md. Kamal Uddin, Shamim Mia, Muhammad Firdaus Sulaiman, Shordar M. Shamsuzzaman, and Ahmad Numery Ashfaqul Haque. 2022. "Influence of Rice Husk Biochar and Lime in Reducing Phosphorus Application Rate in Acid Soil: A Field Trial with Maize" Sustainability 14, no. 12: 7418. https://doi.org/10.3390/su14127418
APA StyleMosharrof, M., Uddin, M. K., Mia, S., Sulaiman, M. F., Shamsuzzaman, S. M., & Haque, A. N. A. (2022). Influence of Rice Husk Biochar and Lime in Reducing Phosphorus Application Rate in Acid Soil: A Field Trial with Maize. Sustainability, 14(12), 7418. https://doi.org/10.3390/su14127418









