Abstract
Climate change has a profound impact on the conservation and management of the Picea species, and establishing more nature reserves would be an effective way to conserve wild species in general. Based on a novel computational method using ecological niche modeling to predict the potential geographical distribution of species and a spatial decision support system, the planning process could predict the future distribution of the Picea species and thus select appropriate nature reserves. In this research, we utilized systematic conservation planning to define priority conservation areas for the Picea species in China according to future climate predictions. We hypothesized that: (1) the distribution of the Picea species could be changed under predicted climate conditions in China; (2) the current national nature reserves had sufficient capacity to conserve Picea species under predicted climate conditions in China; and (3) there were still deficiencies in the planned conservation for the Picea species based on predicted climate predictions in China. The results of a spatial analysis showed that the predicted climate would have an impact on the area of distribution of the Picea species. Current nature reserves have a strong potential to conserve the Picea species. However, the conservation of the Picea species in the existing nature reserves was not adequate. There were still many Picea specimens outside the reserve that would be threatened. This research systematically improved the research on the Picea species, and it also scientifically identified the suitable growth and conserved areas of the Picea species in China to provide an empirical basis for the conservation and management of the Picea species.
1. Introduction
Species have been reacting to environmental changes by adapting, migrating [1], or even becoming extinct [2,3]. In the same way, conservation management has been highly affected by climate change [4]. Preserving endangered plant species in the face of climate change is both imperative and urgent [5]. However, the issue of integrating climate change into biodiversity conservation planning of endangered plant species is still a challenge [6].
Picea species, as common species in high-altitude areas [7], have been objects of study by several researchers [8,9]. Examples include the growth of P. mariana forests in northern Canada [10], and the cultivation of P. abies L. Karst in Slovakia under climate change [11]. However, previous studies have primarily focused on the research of a specific Picea species [8,12] or on the biological characteristics of the Picea species from a micro perspective [13]. There has been a lack of research on the macro-distribution pattern of the Picea species under the influence of climate change. However, there are substantial conservation values concerning the Picea species as a resource, specifically in China [14]. These species have a high economic value and show a strong adaptation capacity [15]. Moreover, they are also highly effective in conserving water [16]. The Picea species have been relevant for the afforestation efforts in their distribution areas [17]. Some of these species show high tolerance to drought [18], including the severely endangered species P. meyeri [19]. The Picea species are an important dominant coniferous species in the temperate regions of north-central China [20]. The natural forests of the Picea species grow slowly [21]. China has the greatest amount of diversity within the Picea genus [17], holding about 19 different species, and could serve as the distribution center of plant material. Of the total of species that grow in China, 11 have been listed as endangered or in need of conservation (IUCN, 2021; NAEP and IBCAS, 1987; http://www.iplant.cn (accessed on 1 December 2021)) [22,23]. If the Picea species is not properly monitored and conserved, it could result in the extinction of a natural forest resource and, thus, damage biodiversity [24].
Previous studies have shown that nature reserves play an important role in conserving endangered plant species through in situ and ex situ conservation [25,26,27]. Establishing more nature reserves is a direct and effective way of biodiversity conservation [28]. Nature reserves refer to representative natural ecosystems and natural concentrated distribution areas of rare and endangered wild animals and plants [29]. These ones may also include areas designated by law for the special conservation and management of the land, body of water, or coastal regions where conserved species or objects, such as natural relics with special significance, are located [29]. National nature reserves have been effective for the conservation of biodiversity, the construction of ecological security barriers [30], the safety and stability of natural ecosystems [31], and improvements in the quality of ecological environments. The establishment of national nature reserves has been suggested as the most effective method by which to conserve rare and endangered wild plants [32].
Novel computational methods have been developed based on prediction algorithms that use ecological niche modeling to predict the potential geographical distribution of species [33]. Predicting future species distributions and selecting appropriate nature reserves requires the use of species distribution model (SDM) programs that are based on spatial analysis. Spatial decision support systems provide a framework for the integration and analysis of spatial, economic, technical, and other disproportionate data in the planning process. The flexible and effective spatial analysis does not rely on a single model or tool. Based on our research, data have been expressed more effectively using spatial decision-making. In this study, we used systematic conservation planning to define priority conservation areas (PCAs) for the Picea species in China under predicted climate conditions, and we tested the following three hypotheses: (1) the species distribution could change for the Picea species in China under predicted climate conditions; (2) the current national nature reserves had sufficient capacity to conserve the Picea species in China under predicted climate conditions; and (3) conservation deficiencies exist for the Picea species in China under predicted climate conditions.
2. Material and Methods
2.1. Data of Species, Climate and National Nature Reserves
The species within the Picea genus that were considered in this study are: P. brachytyla, P. brachytyla var. comparata, P. likiangensis, P. likiangensis var. rubescens, and P. likiangensis var. linzhiensis. Their wood has economic value and can be used as industrial raw materials. They are suitable as the main afforestation trees in plateau areas, and they are endemic to China as endangered conserved species. Due to the scattered wild resources of these Picea species and their variants, the quantity of goods they provide has been limited. From the perspective of production and marketing, the output of these five Picea species’ wild products has been declining, and cultivated products have been increasing. Therefore, the cultivation of these five Picea species could still be the main source of commercial Picea species in the future. This fact provides an extra reason for reinforcing the conservation plans.
The MaxEnt model was used to predict the potential distribution areas of the studied species and discuss the environmental factors shaping their distribution patterns. The grid cells of the area in which Picea species appear were primarily obtained using the following two methods: (1) Field survey data that were recorded from 2020 to 2021 using a handheld Global Positioning System (GPS) and (2) a variety of online databases, including the Global Biodiversity Information Facility (GBIF; www.gbif.org (accessed on 1 December 2021)), LIFEMAPPER (www.lifemapper.com (accessed on 1 December 2021)), and the Chinese Virtual Herbarium (CVH; www.cvh.org.cn (accessed on 1 December 2021)). The number of grid cells indicated the size of area. A larger number of grid cells indicated a larger area. There were 775 sample data of the two species and the three varieties, of which 258 corresponded to P. brachytyla, 137 to P. brachytyla var. comparata, 187 to P. likiangensis, 141 to P. likiagensis var. rubescens and 52 to P. likiagensis var. linzhiensis.
Previous studies have focused on species classification [34,35], growth [36], tree ring records [37], somatic embryogenesis and plant regeneration, as well as the methods of intraspecific differentiation and expansion [38], the impact of the climate on species growth [39], the relationship between insect species [40], and their trait variation [41]. Thus far, there has been little research concerning Picea distribution and conservation.
Climate-related data were obtained from WorldClim (https://worldclim.org/ (accessed on 1 December 2021)). The predicted climate scenarios involved three sets of predicted climate models (MIROC6, MIROC-ES2L, and MRI-ESM2-0) of Shared Socioeconomic Pathways (SSPS) (245 and 585) for 5-arc-min over 20-year periods (2081–2100). The ssp2-4.5 [42] is a moderate socioeconomic development pathway that has been updated based on an RCP4.5 scenario with a targeted radiative forcing of 4.5 Wm−2 at the end of twenty-first century; it is also referred to as an intermediate development pathway; while ssp5-8.5, updated from RCP8.5, is referred as a high development pathway. This one includes an emission scenario with targeted radiative forcing 8.5 Wm−2 at the end of the twenty-first century [43]. The ssp2-4.5 poses moderate challenges to mitigation and adaptation, and ssp5-8.5 poses high challenges to mitigation and low challenges to adaptation [44]. One of the main simulation sets applied by the climate model was the predicted climate scenario, in which the model was provided a common set of predicted greenhouse gases and other parameters for its predictions. Currently, scientists have developed a new set of emission scenarios driven by different socioeconomic assumptions, namely, SSPS, which includes ssp2-4.5 and ssp5-8.5. In addition, 5-arc-min represented spatial resolution or minutes of a degree of longitude and latitude. We use the data of the weighted average values of the three climate models of each Picea species under different scenarios as climate variables [45,46,47]. Eight climate variables with large contribution values in the MaxEnt model were used: Bio1 (annual mean temperature), Bio4 (temperature seasonality), Bio5 (max temperature of warmest month), Bio6 (min temperature of coldest month), Bio12 (annual precipitation), Bio13 (precipitation of wettest month), Bio14 (precipitation of driest month), and Bio15 (precipitation seasonality (coefficient of variation)).
By the end of 2019, China established 2750 nature reserves for conservation [48] (among these, there were 489 national nature reserves), covering a total area of approximately 147 million km2 and representing approximately 14.86% of the land area (http://www.nrchina.org/ (accessed on 5 December 2021)). In this study, the data on China’s national nature reserves were obtained from the Resource and Environmental Science and Data Center (https://www.resdc.cn/Default.aspx (accessed on 5 December 2021)). The data from the world database of conserved areas (WDPA) were used to identify the nature reserves (IUCN I–VI) in China that were suitable for further analysis. Finally, we conducted a cross-analysis between the results of SDM and the boundary of the reserve to obtain the distribution of Picea species in 250 nature reserves to evaluate the capacity of national nature reserves to conserve Chinese Picea species under predicted climate conditions.
2.2. Species Distribution Modeling (SDM)
MaxEnt (v.3.3.3; http://www.cs.princeton.edu/~schapire/maxent/ (accessed on 15 December 2021)) was used to model the current and predicted distribution of Picea species [49]. All the pixels were regarded as possible distribution spaces of maximum entropy [50]. The probability of the occurrence of potential distribution of Picea species was interpreted as habitat quality, and the distribution pixels of the Picea species records collected from the field surveys were treated as sample occurrences. MaxEnt cell values of 1 were the highest habitat quality scores, and values close to 0 were the lowest. MaxEnt was used to estimate the function of habitat quality with maximum entropy, and the model of geographic locations of Picea species was based on environmental variables. Furthermore, the potential areas of distribution were determined by comparison to the areas where the climate conditions of the research region were similar. Therefore, the computed results reflected the quality of habitat.
The GPS positions (longitude and latitude) were used for the grid cell inputs. Of these locations, 75% were used for model training, and 25% were used for testing. The maximum number of background grid cells was 10,000, and auto features were used. Other values were maintained as default. The jackknife test was used to analyze the importance of different environmental factors using MaxEnt. Receiver-operating characteristic (ROC) curves of each value of the prediction results were a potential threshold, and the corresponding sensitivity and specificity were then obtained by calculation. The precision of the model was evaluated by calculating the area under the ROC Curve (AUC). Higher AUC values indicated that there was a stronger relationship between the 19 bioclimatic variables [51]. This ensured better predictions of species distribution. The model was graded as follows: poor (AUC < 0.8), fair (0.8 < AUC < 0.9), good (0.9 < AUC < 0.95), and very good (0.95 < AUC < 1.0; [52]). The value range of the final potential distribution of Picea species from 0 to 1 was divided into five categories to denote potential habitat quality: highest (>0.8), high (0.6–0.8), good (0.4–0.6), moderate (0.2–0.4), and least (<0.2).
2.3. Systematic Conservation Planning
The details of Zonation software and its algorithm have been fully described in previous studies [53,54,55,56]. First, we planned to model priority conservation areas (PCAs) for the studied plant material using the conservation planning software Zonation (http://cbig.it.helsinki.fi/software/ (accessed on 15 December 2021)). We used it to build the distribution map and provided the software with target PCAs with high-priority rankings for species-richness conservation. The highest PCAs of Picea species were confirmed by identifying the top-ranking cells after the Zonation computation [57]. We minimized the geographic distance between the potential habitats of five Picea species and varieties and then considered the influence of geographic distance when selecting potential sites for reserves. Using the original core-area cell removal rule, we set spatial priorities and computed the marginal loss of each cell, which we then used to determine if a conservation goal had been reached. The attainment of goals was determined based on a provided “conservation proportion” of distributions for all the species with high priority rankings [58]. The predicted habitat distribution maps of Picea species, as assessed by MaxEnt for each pixel (namely, the input layers for Zonation), were used in the Zonation algorithm to simulate PCAs. Five Picea species and varieties were weighted equally in our analysis as we assumed that all the species had equal conservation values and that they should all be included in the prioritization solution. The warp factor was set equal to 200 [58,59]. To emphasize areas with higher model certainty, we established the default Zonation criterion (=1) as the uncertainty parameter [60,61,62,63]. We established all the other settings as described by Moilanen et al. [60] and Faleiro et al. [57]. The objective of this study was to conserve 30% of the region that we selected in northeastern China in accordance with Target Four of the Global Strategy for Plant Conservation (GSPC; http://www.cbd.int/gspc/ (accessed on 15 December 2021); [64]). We used the entire land area of China as the ecological region of this research. We selected 30% of this ecological region, namely, the provided conservation proportion of distributions for species with high priority (Zonation conservation values ≥ 0.7), as PCAs for Picea species. Lastly, we used ArcGIS 10.4 (ESRI, Redlands, CA, USA) to compute the area and distribution of the PCAs. Habitat suitability results were expressed in probability, ranging from 0–1 [65]. Using the “Reclassify” tool of ArcMap 10.3, the obtained probability results were divided into two levels, of which 0–0.7 was inappropriate and 0.7–1 was appropriate and high potential [66,67].
2.4. Gap Analysis
According to the international biodiversity conservation roadmap, 30% of the territory should be protected by 2030 [68]. Honeck et al. (2020) [69] also showed that when the priority area is 30% (70–100%), it is the best high priority and zonation’s outputs. Based on these, we retained 30% of the PCAs of Picea species in China under predicted climate conditions. Therefore, we regarded the potential distribution area of Picea species as the ecological region using MaxEnt to make full use of the land and adhere to the sustainable development strategy of China. Moreover, the threshold value of PCAs was extracted as the standard of conservation for each population and not vulnerable to the simulated predicted climate change. The terrestrial ecological region had to be considered in relation to the different national conditions. In this way, we were able to evaluate the efficacy of the existing national nature reserves and take appropriate measures to plan the construction of new conservation areas. We then analyzed the important existing national nature reserves for Picea species through computation of the PCAs.
Considering that the target Picea species included two species and three varieties, niche overlapping could occur. The potential distribution area maps of five Picea species were used for spatial decision analysis and the potential distribution probability of these five Picea species under different climate models. We selected nature reserves with high distribution probability (probability > 0.7) [67] under current and predicted climates to analyze whether there was niche overlapping.
3. Results
3.1. Changes in the Species Distribution of Picea Species
The AUC values predicted by MaxEnt based on several potential Picea species distribution variables were greater than 0.9, indicating that the MaxEnt model had good prediction results for the potential distribution area of the Picea species. The SDM and the results of the spatial analysis (Figure 1) showed that, as compared to the current distribution map (b), the species distribution area of the Picea species in China would change under the two predicated climate models ssp2-4.5 (c) and ssp5-8.5 (d). A comparison of the current (b) and predicted climate models indicated that there was little change in the distribution area of the Picea species under the ssp2-4.5 (c) scenario. The overall terrain of China is characterized by a stepped distribution of high in the West and low in the East [70]. When combined with the PCA distribution area map of the Picea species under the current climate (Figure 1b) and ssp5-8.5 predicated climate (Figure 1d), the PCA of the Picea species showed a norward trend, while the distribution area at high latitude and in western areas was increasing. The area of distribution of the Picea species showed a trend of moving to high latitude and altitude under the predicted ssp5-8.5 (d) climate model (Figure 1).
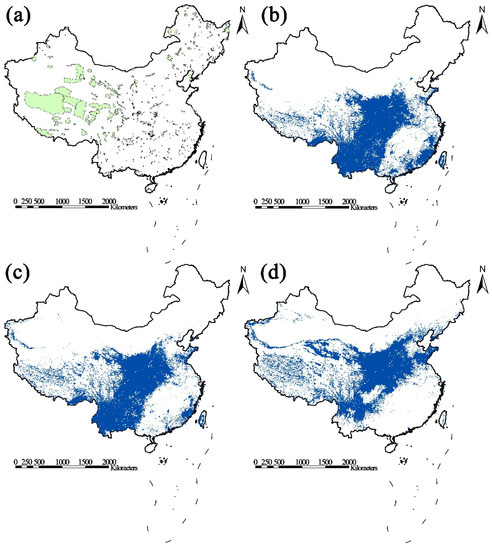
Figure 1.
(a) Distribution map of China’s nature reserves. (b) Current distribution map of the Picea species. (c) The area of distribution of the Picea species in the climate priority conservation area under the predicted ssp2-4.5 scenario mode. (d) The area of distribution of the Picea species in the climate priority conserved areas of the Picea species under the predicted ssp5-8.5 scenario mode.
In the future scenarios ssp2-4.5, the distribution areas of Picea species that correspond to Qiangtang, Sanjiangyuan and the Qaidam Haloxylon Forest, Hoh Xil, and Qilian Mountain Nature Reserves in Gansu Province showed a tendency of increasing; whereas The Xishuangbanna and Dongzhai Nature Reserves showed an opposite trend (Table 1). Under the future ssp5-8.5 scenario, the reserves with a significant increase in the area of the Picea species included Qiangtang; Sanjiangyuan; Lop Nur Wild Camel; Hoh Xil; Altun Mountain; Qilian Mountain in Gansu Province; Qaidam Haloxylon ammodendron Forest and Annan Dam Wild Camel; Xishuangbanna Nature Reserve, a rare and unique fish nature reserve in the upper reaches of the Yangtze River; Yarlung Zangbo Grand Canyon; Dawei Mountain in Yunnan; Wuyi Mountain in Fujian; Huanglian Mountain; Nanling; Gaoligong Mountain; Nangunhe River; Leigong Mountain, and Huping Mountain (Table 1) (See Table S2 for the complete table).

Table 1.
Under the emission scenarios of ssp2-4.5 and ssp5-8.5, the distribution area of the studied Picea species PCAs in the national nature reserves.
3.2. Ability of the National Nature Reserves to Conserve Picea Species
Based on the regional analytical results of our spatial analysis, the total number of distribution grid cells in the nature reserves was 1748. Under the ssp2-4.5 emission scenario, there were 2221 grid cells in China’s nature reserve, whereas the amount increased to 3648 under the spp5-8.5 scenario (Table 1). The results showed that the conservation capacity of Picea species in the National Nature Reserve under ssp5-8.5 was higher than the one obtained under a low emission scenario. In the low-emission scenario, climate change had a high positive impact on the Picea species in the Qiangtang, Sanjiangyuan, Qaidam Haloxylon Forest, Hoh Xil, and Qilian Mountain Nature Reserves (Table 1). In the high-emission scenario, climate change had a high positive impact on the Qiangtang, Sanjiangyuan, Lop Nur Wild Camel, Hoh Xil, Arkin Mountain, Qilian Mountain in Gansu Province, Qaidam Haloxylon Forest, and Annan Dam Wild Camel Nature Reserves (Table 1).
3.3. Conservation Gap
The predicted environmental conditions appeared to increase the distribution area of the Picea species outside some of the current national reserves. Based on the results of regional statistical analysis, we obtained the number of the studied species distributed outside China’s land nature reserves under the current climate conditions and in two emission scenarios in the future (Figure 1). The total number of grid cells in the PCAs of the Picea species was 32,507. A total of 30,724 grid cells of the Picea species, and a total of 30,724 grid cells of the Picea species were located outside the nature reserves under the current climate conditions (Figure 1b). In the predicted low-emission scenario, 30,252 grid cells were distributed outside China’s nature reserves (Figure 1c). In the predicted high-emission scenario, 28,846 Picea species grid cells were distributed across China (Figure 1d). Whether high or low emissions, most Picea species were distributed outside the nature reserve under the predicted climate conditions (Figure 1).
The sum of grid cells in and outside the nature reserve was not equal to the total number of grid cells. The error was due to a small number of nature reserves not being distributed entirely in China or the Picea species being distributed at the boundary of the nature reserve.
The results of the spatial decision analysis showed that niche overlapping occurred between P. brachytyla and P. brachytyla var. comparata in Gongga Mountain and Xiaojin Siguniang mountain. P. likiangensis, P. likiangensis var. rubescens, and P. likiangensis var. linzhiensis had niche overlapping in Dupangling, Yongzhou, upstream of Yalu River, and Yangmingshan. P. likiangensis and P. likiangensis var. rubescens had niche overlapping in Ordos relic gull (Table 2).

Table 2.
Niche overlap analysis of five Picea species.
4. Discussion
4.1. Distribution of the Picea Species
The results of the spatial analysis showed that the predicted climate would have a significant impact on the distribution area of the Picea species. This could be due to the change in temperature and precipitation caused by climate changes, which affect the habitat of the Picea species [71]. In particular, the change in the area of distribution in the nature reserves under the predicted ssp2-4.5 scenario increased the Picea species. The nature reserves with an obvious trend were concentrated in the high-altitude Qinghai Tibet Plateau in northwest China. This increase was significantly higher when the ss5-8.5 scenario was assessed. Changes in extreme temperature and precipitation caused by human activities would lead to a reduction in seasonal and perennial ice and snow ranges and an increase in sea levels [72]. Drought caused by warming has significantly affected forest dynamics in most regions of the northern hemisphere [73]. Factors such as temperature and precipitation have affected the range of distribution of the Picea species [74]. Picea species have been resistant to shade and cold and often grow in higher altitude areas [75]. In this study, a lower impact on the distribution area of Picea species is predicted under a low-emission scenario in comparison with a high-emission scenario. These findings could be explained by the differences in the expected changes in temperature and precipitation between both scenarios, being lower in ssp2-4.5. In the high-emission scenario, the warming trend was more apparent [76], which led to the movement and expansion of the area of distribution from low latitudes and altitudes to high latitudes and altitudes. This trend was similar to the findings reported by previous research, which showed that the niche of this genus had a northward trend [77,78].
4.2. Conservation Ability of the National Nature Reserves
We used the Zonation conservation planning software (http://cbig.it.helsinki.fi/software/ (accessed on 1 December 2021)) to develop conservation plans for the Picea species. Zonation is typically used as a spatial conservation prioritization framework for the large-scale conservation planning of multiple biodiversity features (e.g., species), but we adopted the use of the Zonation algorithm to establish the conservation areas for Picea species across large space–time scales. The highest priorities for conservation, namely conserved areas of genetic diversity, were confirmed by identifying the top-ranking cells after computation.
Conserved areas, such as nature reserves, have been effective in the prevention of habitat destruction and the conservation of ecosystems [79]. This study showed that some priority conservation areas of the Picea species coincided with the areas of distribution of the nature reserves, which showed that national nature reserves had sufficient capacity to conserve the Picea species, indicating the necessity of establishing new nature reserves. In the low-emission scenario, the PCA distribution area of the current Picea species increased, and the event was more significant in the high-emission scenario. It indicated that in the predicted high-emission scenario, the conservation capacity of national nature reserves for Picea species was better than that in the low-emission scenario. Studies have shown that nature reserves promote the increase in forest coverage and areas most suitable for a species [80]. The nature reserves had sufficient capacity to conserve water, reduce grazing intensity and human disturbance factors, and conserve grassland vegetation in fragile habitats [81]. The forests in the nature reserve had significant ecological benefits and were highly efficient at maintaining the ecological security of their immediate and surrounding areas [82].
4.3. Conservation Gap Analysis
More efforts will be necessary to expand the scope of conservation based on increasing nature reserve areas. A significant number of the Picea species are located outside the reserves, and these are under even more threats. Xu et al. (2019) [48] proposed the establishment of a national park administration to separate management and supervision and establish spatial planning for the diversity of national representative species, ecosystems, and natural landscapes. This strategy could promote a balance between conservation planning and management, similar to the objective of this study. More nature reserves should be established in the areas that are suitable for the Picea species by constructing a reserve network to conserve the biodiversity of the Picea species. Local species and ecosystems should be conserved, and the focus should not only encompass areas with the highest biodiversity [83]. Environmental degradation and difficult natural regeneration should be avoided since excessive deforestation leads to sporadic growth. The spatial decision support system effectively and directly analyzed the characteristics of the Picea species distribution in China under predicted climate conditions, assisted in finding the relationship between Picea species priority conservation areas and nature reserves, and provided a reliable reference for the predicted system conservation planning for this valuable genus.
In some nature reserves under current and predicted scenarios (Table 2), niche overlapping occurred among the Picea species and their variants. This was attributed to the similar niche between the variant and the original, so niche overlapping could occur in environments with limited resources [84]. In the management of nature reserves, we should consider the different degrees of niche overlapping in different nature reserves.
Our research had certain limitations. For example, regarding the spatial decision support system, our research had insufficient data and could only conduct general research. Therefore, we need to gather more data for future research [85]. In addition, the difference between the data and the models involved in the various processes to be integrated also affected the spatial decision system. A decentralized, bottom-up forest planning approach that consistently allows the cells to coevolve until the plans of all periods within the planning horizon are achieved [86] would resolve this issue. Nowakowska et al. (2020a) [87] and Berezovska et al. (2021) [88] studied the pathogens transmitted in soil and found that soil pathogens had a negative impact on the growth of trees. Nowakowska et al. (2020b) [89] researched Norwegian spruce trees using bark beetles (IPS typographus L.) in Polish forest areas to show that insects could also threaten the growth of spruce trees. We only considered the environmental impacts at a macro level, not the micro level. Even with these limitations, however, our study provided significant results.
5. Conclusions
This study examined five Picea species varieties to comprehensively predict the distribution of the Picea species under the influence of predicted climate conditions in China. The research also showed the conservation capacity of nature reserves for the Picea species. This research contributed to previous literature on the Picea species and the conservation capacity of nature reserves under climate change conditions. The Picea species has been a focus of research in botany. This research simulated the potential geographical distribution areas of the Picea species based on a spatial decision support system, which indicated scientific zoning for the suitable growth and conserved areas of the Picea species in China, and provided a scientific basis for the biodiversity conservation and management of the Picea species.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/su14127406/s1, Table S1: Five Picea species data from field survey and Chinese Virtual Herbarium (CVH; www.cvh.org.cn); Table S2: The predicted distribution area of Picea species under the current climate and the future climate of the two emission scenarios.
Author Contributions
Conceptualization, J.-Z.W.; supervision, C.-J.W. and J.-Z.W.; reviewing and Editing, J.-Z.W.; data collection, Q.W.; writing-original draft preparation, Q.W.; methodology, Q.W.; software, Q.W.; Editing, C.-J.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (Nos. 31800449 and 31800464) and Chun-Jing Wang was supported by “1000 Talent” programs of Qinghai Province.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the research.
Data Availability Statement
The grid cells of the area in which Picea species appears were primarily obtained from the Global Biodiversity Information Facility (GBIF; www.gbif.org (accessed on 1 December 2021)), LIFEMAPPER (www.lifemapper.com (accessed on 1 December 2021)), and the Chinese Virtual Herbarium (CVH; www.cvh.org.cn (accessed on 1 December 2021)). Climate-related data were obtained from WorldClim (https://worldclim.org/ (accessed on 1 December 2021)). Data of China’s national nature reserves were obtained from the Resource and Environmental Science and Data Center (https://www.resdc.cn/Default.aspx (accessed on 5 December 2021)).
Acknowledgments
We thank the Chinese Virtual Herbarium and Maixiu forest farm for our work.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Parmesan, C. Ecological and evolutionary responses to recent climate change. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 637–669. [Google Scholar] [CrossRef]
- Pinedo-Alvarez, C.; Renteria-Villalobos, M.; Aguilar-SotoJose, V.; Vega-Mares, J.H.; Melgoza-Castillo, A. Distribution dynamics of Picea chihuahuana Martínez populations under different climate change scenarios in Mexico. Glob. Ecol. Conserv. 2019, 17, e00559. [Google Scholar] [CrossRef]
- Root, T.L.; Price, J.T.; Hall, K.R.; Schneider, S.H.; Rosenzweig, C.; Pounds, J.A. Fingerprints of global warming on wild animals and plants. Nature 2003, 421, 57–60. [Google Scholar] [CrossRef] [PubMed]
- Hughes, L. Biological consequences of global warming: Is the signal already apparent? Trends Ecol. Evol. 2000, 15, 56–61. [Google Scholar] [CrossRef]
- Bothe, H.; Turnau, K.; Regvar, M. The potential role of arbuscular mycorrhizal fungi in protecting endangered plants and habitats. Mycorrhiza 2010, 20, 445–457. [Google Scholar] [CrossRef] [PubMed]
- Burgman, M.A. Are listed threatened plant species actually at risk? Aust. J. Bot. 2002, 50, 275. [Google Scholar] [CrossRef]
- Nosova, M.B.; Severova, E.E.; Volkova, O.A.; Kosenko, J.V. Representation of Picea pollen in modern and surface samples from Central European Russia. Veg. Hist. Archaeobot. 2015, 24, 319–330. [Google Scholar] [CrossRef]
- Karvonen, P.; Szmidt, A.E.; Savolainen, O. Length variation in the internal transcribed spacers of ribosomal DNA in Picea abies and related species. Theor. Appl. Genet. 1994, 89, 969–974. [Google Scholar] [CrossRef]
- Birks, H.H.; Giesecke, T.; Hewitt, G.M.; Tzedakis, C.; Bakke, J.; Birks, H.J.B. Comment on “Glacial Survival of Boreal Trees in Northern Scandinavia”. Science 2012, 338, 742. [Google Scholar] [CrossRef]
- Groot, A.; Saucierb, J.P. Volume increment efficiency of Picea mariana in northern Ontario, Canada. For. Ecol. Manag. 2008, 255, 1647–1653. [Google Scholar] [CrossRef]
- Büntgen, U.; Frank, D.C.; Kaczka, R.J.; Verstege, A.; Zwijacz-Kozica, T.; Esper, J. Growth responses to climate in a multi-species tree-ring network in the Western Carpathian Tatra Mountains, Poland and Slovakia. Tree Physiol. 2007, 27, 689–702. [Google Scholar] [CrossRef] [PubMed]
- Klymiuk, A.A.; Stockey, R.A. A Lower Cretaceous (Valanginian) seed cone provides the earliest fossil record for Picea (Pinaceae). Am. J. Bot. 2012, 99, 1069–1082. [Google Scholar] [CrossRef] [PubMed]
- Van de Ven, W.T.G.; McNicol, R.J. Microsatellites as DNA markers in Sitka spruce. Theor. Appl. Genet. 1996, 93, 613–617. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.Y.; Nan, Z.R.; Cheng, G.D.; Zhang, J.H.; Feng, Z.D. GIS-assisted modelling of the spatial distribution of Qinghai spruce (Picea crassifolia) in the Qilian Mountains, northwestern China based on biophysical parameters. Ecol. Model. 2006, 191, 487–500. [Google Scholar] [CrossRef]
- Wang, J.; Ma, J.; OuYang, F.; Wang, J.H.; Song, L.; Kong, L.S.; Zhang, H.G. Instrinsic relationship among needle morphology, anatomy, gas exchanges and tree growth across 17 Picea species. New For. 2021, 52, 509–535. [Google Scholar] [CrossRef]
- Wang, Y.J.; Lu, R.J.; Gao, S.Y.; Meng, H.W.; Gao, S.Y. Response to climate change of different tree species and NDVI variation since 1923 in the middle arid region of Ningxia, China. Sci. Cold Arid. Reg. 2014, 6, 30–36. [Google Scholar]
- Zhang, A.P. Dynamic Reconstruction of Historical Distribution and Prediction of Future Distribution of Picea Species in China. Master’s Thesis, University of Chinese Academy of Sciences, Beijing, China, 2018. [Google Scholar]
- Lv, D.; Zhang, H.B.; Zhan, M.; Zhao, H.; Li, B.X.; Yan, K.; Zhao, X.P. Adaptability of introduced 7 Picea species in arid and semi-arid region. J. Arid. Land Resour. Environ. 2019, 5, 5. [Google Scholar]
- Zou, C.J.; Han, S.J.; Xu, W.D.; Su, B.L. Formation, distribution, and nature of Picea mongolica in China. J. For. Res. 2001, 12, 187–191. [Google Scholar]
- Jiang, Y.; Zhang, W.T.; Wang, M.C.; Kang, M.Y.; Dong, M.Y. Radial Growth of Two Dominant Montane Conifer Tree Species in Response to Climate Change in North-Central China. PLoS ONE 2014, 9, e112537. [Google Scholar] [CrossRef]
- Zhao, W.L.; Gu, L.S.; Gu, L.J. Biomass Measures of Picea species and Seed Source. Xinjiang Agric. Sci. 2003, 40, 94–97. [Google Scholar]
- IUCN. The IUCN Red List of Threatened Species. Version 2021-3. 2021. Available online: https://www.iucnredlist.org (accessed on 1 December 2021).
- NAEP (National Administration of Environmental Protection); IBCAS (Institute of Botany, Chinese Academy of Sciences). List of Rare and Endangered Plants Protected in China; Science Press: Beijing, China, 1987. [Google Scholar]
- Brockerhoff, E.G.; Jactel, H.; Parrotta, J.A.; Quine, C.P.; Sayer, J. Plantation forests and biodiversity: Oxymoron or opportunity? Biodivers. Conserv. 2008, 17, 925–951. [Google Scholar] [CrossRef]
- Braverman, I. Conservation without nature: The trouble with in situ versus ex situ conservation. Geoforum 2013, 51, 47–57. [Google Scholar] [CrossRef]
- Volis, S.; Blecher, M. Quasi in situ: A bridge between ex situ and in situ conservation of plants. Biodivers. Conserv. 2010, 19, 2441–2454. [Google Scholar] [CrossRef]
- Wei, X.Z.; Jiang, M.X. Meta-analysis of genetic representativeness of plant populations under ex situ conservation in contrast to wild source populations. Conserv. Biol. 2020, 35, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, D.J.; Fa, J.E.; Oldfield, S.; Harrop, S.R. Bring the captive closer to the wild: Redefining the role of ex situ conservation. Oryx 2011, 46, 18–23. [Google Scholar] [CrossRef]
- Ni, T.L.; Li, X.Z.; Du, Z.X. Nature reserves and forest tourism. J. Shandong For. Sci. Technol. 2004, 5, 68. [Google Scholar]
- Pressey, R.L.; Humphries, C.J.; Margules, C.R.; Williams, P.H.; Vanewright, R.I. Beyond opportunism: Key principles for systematic reserve selection. Trends Ecol. Evol. 1993, 8, 124–128. [Google Scholar] [CrossRef]
- Voloscuk, I. Ecological stability in the Tatra mountains forests. Ekol. Bratisl. 1998, 17, 39–48. [Google Scholar]
- Yuan, H.; Zhang, Y.B.; Qin, H.N.; Liu, Y.; Yu, M. The in situ conservation of state key protected wild plants in national nature reserves in China. Biodivers. Sci. 2009, 17, 280. [Google Scholar]
- Abdel-Dayem, M.S.; Annajar, B.B.; Hanafi, H.A.; Obenauer, P.J. The potential distribution of Phlebotomus papatasi (Diptera: Psychodidae) in Libya based on ecological niche model. J. Med. Entomol. 2012, 49, 739–745. [Google Scholar] [CrossRef]
- Lyu, L.K.; Wang, D.L.; Li, L.; Zhu, Y.Y.; Jiang, D.C.; Liu, J.Q.; Xu, X.T. Polyphyly and species delimitation of Picea species brachytyla (Pinaceae) based on population genetic data. J. Syst. Evol. 2021, 59, 515–523. [Google Scholar] [CrossRef]
- Ru, D.F.; Mao, K.S.; Zhang, L.; Wang, X.J.; Lu, Z.Q.; Sun, Y.S. Genomic evidence for polyphyletic origins and interlineage gene flow within complex taxa: A case study of Picea species brachytyla in the Qinghai-Tibet Plateau. Mol. Ecol. 2016, 25, 2373–2386. [Google Scholar] [CrossRef] [PubMed]
- Yin, D.C.; Sun, M.; Zhang, W.G.; Yue, H.T.; Zhang, Y.; Tian, K.; Xiao, D.R.; Zhang, Y. Effects of Climate Warming on Radial Growth of Picea species brachytyla in Shangri-La, Southwestern China. J. Northeast. For. Univ. 2019, 47, 1–7. [Google Scholar]
- Li, Z.S.; Zhang, Q.B.; Ma, K.P. Tree-ring reconstruction of summer temperature for A.D. 1475-2003 in the central Hengduan Mountains, Northwestern Yunnan, China. Clim. Chang. 2012, 110, 455–467. [Google Scholar] [CrossRef]
- Li, L.; Abbott, R.J.; Liu, B.B.; Sun, Y.S.; Li, L.L.; Zou, J.B.; Wang, X.; Mie, H.; Liu, J.Q. Data from: Pliocene intraspecific divergence and Plio-Pleistocene range expansions within Picea species likiangensis (Lijiang Picea species), a dominant forest tree of the Qinghai-Tibet Plateau. Mol. Ecol. 2013, 27, 2280–2282. [Google Scholar]
- Zhang, M.; Shi, S.L.; Shi, C.M.; Bai, H.; Li, Z.S.; Peng, P.H. Radial growth responses of four typical coniferous species to climatic factors in the Western Sichuan Plateau, China. Chin. J. Ecol. 2021, 40, 1947–1957. [Google Scholar]
- Tang, X.Q.; Ren, Y.H.; Zang, J.C.; Zhang, Y.H. Biological Characteristics of Dioryctria abietella (Lepidoptera: Pyralidae) on Picea species likiangensis var. linzhiensis Trees. For. Res. 2019, 32, 60–64. [Google Scholar]
- Lin, L.; Luo, J. Variation in Traits of Picea species likiangensis var. linzhiensis Seedlings from Different Provenances. For. Res. 2014, 27, 557–561. [Google Scholar]
- Calvin, K.; Bond-Lamberty, B.; Clarke, L.; Edmonds, J.; Eom, J.; Hartin, C.; Kim, S.; Kyle, P.; Link, R.; Moss, R.; et al. The SSP4: A world of deepening inequality. Glob. Environ. Change 2017, 42, 284–296. [Google Scholar] [CrossRef]
- Kriegler, E.; Bauer, N.; Popp, A.; Humpenöder, F.; Leimbach, M.; Strefler, J.; Baumstark, L.; Bodirsky, B.L.; Hilaire, J.; Klein, D.; et al. Fossil-fueled development (SSP5): An energy and resource intensive scenario for the 21st century. Glob. Environ. Change 2017, 42, 297–315. [Google Scholar] [CrossRef]
- Hingmire, D.; Vellore, R.; Krishnan, R.; Singh, M.; Metya, A.; Gokul, T.; Ayantika, D.C. Climate change response in wintertime widespread fog conditions over the Indo-Gangetic Plains. Clim. Dyn. 2022, 58, 2745–2766. [Google Scholar] [CrossRef]
- Knutti, R.; Furrer, R.; Tebaldi, C.; Cermak, J.; Meehl, G.A. Challenges in Combining Projections from Multiple Climate Models. J. Clim. 2010, 23, 2739–2758. [Google Scholar] [CrossRef]
- Weiland, F.; Beek, L.; Weerts, A.H.; Bierkens, M.F.P. Extracting information from an ensemble of gcms to reliably assess future global runoff change. J. Hydrol. 2011, 412, 66–75. [Google Scholar]
- Massoud, E.C.; Espinoza, V.; Guan, B.; Waliser, D.E. Global Climate Model Ensemble Approaches for Future Projections of Atmospheric Rivers. Earth’s Future 2019, 7, 1136–1151. [Google Scholar] [CrossRef]
- Xu, W.; Pimm, S.L.; Du, A.; Su, Y.; Ouyang, Z. Transforming protected area management in china. Trends Ecol. Evol. 2019, 34, 762–766. [Google Scholar] [CrossRef]
- Kumar, P. Assessment of impact of climate change on rhododendrons in Sikkim Himalayas using maxent modelling: Limitations and challenges. Biodivers. Conserv. 2012, 21, 1251–1266. [Google Scholar] [CrossRef]
- Yang, X.Q.; Kushwaha, S.P.S.; Saran, S.; Xu, J.C.; Roy, P.S. Maxent modeling for predicting the potential distribution of medicinal plant, Justicia adhatoda L. in Lesser Himalayan foothills. Ecol. Eng. 2013, 51, 83–87. [Google Scholar] [CrossRef]
- Wan, J.Z.; Wang, C.J.; Han, S.J.; Yu, J.H. Planning the priority protected areas of endangered orchid species in northeastern China. Biodivers. Conserv. 2014, 23, 1395–1409. [Google Scholar] [CrossRef]
- Adhikari, D.; Barik, S.K.; Upadhaya, K. Habitat distribution modelling for reintroduction of Ilex khasiana Purk: A critically endangered tree species of northeastern India. Ecol. Eng. 2012, 40, 37–43. [Google Scholar] [CrossRef]
- Moilanen, A.; Franco, A.M.A.; Early, R.I.; Fox, R.; Wintle, B.; Thoms, C.D. Prioritizing multiple-use landscapes for conservation: Methods for large multi-species planning problems. Proc. R. Soc. B-Biol. Sci. 2005, 272, 1885–1891. [Google Scholar] [CrossRef]
- Moilanen, A.; Anderson, B.J.; Eigenbrod, F.; Heinemeyer, A.; Roy, D.B.; Gillings, S.; Armsworth, P.R.; Gaston, K.J.; Thomas, C.D. Balancing alternative land uses in conservation prioritization. Ecol. Appl. 2011, 21, 1419–1426. [Google Scholar] [CrossRef] [PubMed]
- Moilanen, A. Landscape Zonation, benefit functions and target-based planning: Unifying reserve selection strategies. Biol. Conserv. 2007, 134, 571–579. [Google Scholar] [CrossRef]
- Moilanen, A.; Kujala, H.; Leathwick, J. The Zonation Framework and Software for Conservation Prioritization; Oxford University Press: Oxford, UK, 2009. [Google Scholar]
- Faleiro, F.V.; Machado, R.B.; Loyola, R.D. Conservation priorities in the face of land-use and climate change. Biol. Conserv. 2013, 158, 248–257. [Google Scholar] [CrossRef]
- Klorvuttimontara, S.; McClean, C.J.; Hill, J.K. Evaluating the effectiveness of protected areas for conserving tropical forest butterflies of Thailand. Biol. Conserv. 2011, 144, 2534–2540. [Google Scholar] [CrossRef]
- Ballard, G.; Jongsomjit, D.; Veloz, S.D.; Ainley, D.G. Coexistence of mesopredators in an intact polar ocean ecosystem: The basis for defining a Ross Sea marine protected area. Biol. Conserv. 2012, 156, 72–82. [Google Scholar] [CrossRef]
- Moilanen, A.; Wilson, K.H.; Possingham, H.P. Spatial Conservation Prioritization: Quantitative Methods and Computational Tools; Oxford University Press: Oxford, UK, 2009; Volume 85, pp. 196–210. [Google Scholar]
- Moilanen, A.; Wintle, B.A. The Boundary-Quality Penalty: A Quantitative Method for Approximating Species Responses to Fragmentation in Reserve Selection. Conserv. Biol. 2007, 21, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Moilanen, A.; Wintle, B.A.; Elith, J.; Burgman, M. Uncertainty Analysis for Regional-Cale Reserve Selection. Conserv. Biol. 2007, 20, 1688–1697. [Google Scholar] [CrossRef] [PubMed]
- Moilanen, A.; Leathwick, J.; Elith, J. A method for spatial freshwater conservation prioritization. Freshw. Biol. 2008, 53, 577–592. [Google Scholar] [CrossRef]
- Williams, S.J.; Jones, J.P.; Clubbe, C. Why are some biodiversity policies implemented and others ignored? Lessons from the uptake of the global strategy for plant conservation by botanic gardens. Biol. Conserv. 2012, 21, 175–187. [Google Scholar] [CrossRef]
- Remya, K.; Ramachandran, A.; Jayakumar, S. Predicting the current and future suitable habitat distribution of Myristica dactyloides Gaertn. using MaxEnt model in the Eastern Ghats, India. Ecol. Eng. 2015, 82, 184–188. [Google Scholar] [CrossRef]
- Seidle, K.M.; Kiss, J.; Attanayake, A.U.; Devink, J.M.; Bedard-Haughn, A.; Westwood, R.; Lamb, E.G. Extent of Dakota skipper, Hesperia dacotae, distribution in Southeastern Saskatchewan, Canada. J. Insect Conserv. 2020, 24, 1073–1081. [Google Scholar] [CrossRef]
- Wang, C.; Lin, H.L.; Feng, Q.S.; Jin, C.Y.; Cao, A.C.; He, L. A New Strategy for the Prevention and Control of Eupatorium adenophorum under Climate Change in China. Sustainability 2017, 9, 2037. [Google Scholar] [CrossRef]
- Belote, R.T.; Barnett, K.; Dietz, M.S.; Burkle, L.; Jenkins, C.N.; Dreiss, L.; Aycrigg, J.L.; Aplet, G.H. Options for prioritizing sites for biodiversity conservation with implications for “30 by 30”. Biol. Conserv. 2021, 264, 109378. [Google Scholar] [CrossRef]
- Honeck, E.; Moilanen, A.; Guinaudeau, B.; Wyler, N.; Schlaepfer, M.A.; Martin, P.; Sanguet, A.; Urbina, L.; von Arx, B.; Massy, J.; et al. Implementing Green Infrastructure for the Spatial Planning of Peri-Urban Areas in Geneva, Switzerland. Sustainability 2020, 12, 1387. [Google Scholar] [CrossRef]
- Zhang, X.H.; Wang, Z.L.; Hou, F.H.; Yang, J.Y.; Guo, X.W. Terrain Evolution of China Seas and Land Since the Indo-China Movement and Characteristics of the Stepped Landform. Chin. J. Geophys. 2015, 58, 54–68. [Google Scholar]
- Sohar, K.; Altman, J.; Leheckova, E.; Dolezal, J. Growth-climate relationships of Himalayan conifers along elevational and latitudinal gradients. Int. J. Climatol. 2017, 37, 2593–2605. [Google Scholar] [CrossRef]
- Karl, T.R.; Trenberth, K.E. Modern global climate change. Science 2003, 302, 1719–1723. [Google Scholar] [CrossRef]
- Yu, L.; Huang, L.; Shao, X.M.; Xiao, F.J.; Wilmking, M.; Zhang, Y.X. Warming-Induced Decline of Picea species crassifolia Growth in the Qilian Mountains in Recent Decades. PLoS ONE 2015, 10, e0129959. [Google Scholar] [CrossRef]
- Miyazawa, K.; Lechowicz, M.J. Comparative Seedling Ecology of Eight North American Picea species (Picea species) Species in Relation to their Geographic Ranges. Ann. Bot. 2004, 94, 635–644. [Google Scholar] [CrossRef][Green Version]
- Huo, Y.X.; Gou, X.H.; Liu, W.H.; Li, J.B.; Zhang, F.; Fang, K.Y. Climate–growth relationships of Schrenk spruce (Picea schrenkiana) along an altitudinal gradient in the western Tianshan mountains, northwest China. Trees 2017, 31, 429–439. [Google Scholar] [CrossRef]
- Xu, K.; Wu, C.H.; Hu, B.X. Projected changes of temperature extremes over nine major basins in China based on the CMIP5 multimodel ensembles. Stoch. Environ. Res. Risk Assess. 2019, 33, 321–339. [Google Scholar] [CrossRef]
- Mckenney, D.W.; Pedlar, J.H.; Lawrence, K.; Campbell, K.; Hutchinson, M.F. Potential Impacts of Climate Change on the Distribution of North American Trees. Bioscience 2007, 57, 939–948. [Google Scholar] [CrossRef]
- Akyol, A.; Orucu, O.K.; Arslan, E.S. Habitat suitability mapping of stone pine (Pinus pinea L.) under the effects of climate change. Biologia 2020, 75, 2175–2187. [Google Scholar] [CrossRef]
- Clerici, N.; Bodini, A.; Eva, H.; Gregoire, J.M.; Dulieu, D.; Paolini, C. Increased isolation of two biosphere reserves and surrounding protected areas (WAP ecological complex, West Africa). J. Nat. Conserv. 2007, 15, 26–40. [Google Scholar] [CrossRef]
- Wang, C.L.; Zang, Z.H.; Qiu, Y.; Deng, S.Y.; Feng, C.Y.; Xie, Z.Q.; Xu, W.T.; Liu, L.; Chen, Q.S.; Shen, G.Z. The effectiveness of Shennongjia National Nature Reserve in conserving forests and habitat of Sichuan snub-nosed monkey. Biodivers. Sci. 2017, 25, 504–512. [Google Scholar] [CrossRef][Green Version]
- Ma, G.F.; Mansur, S.; Zhang, X. Water Conservation Function of Different Vegetation Types in the Upper Tailan River in Mount Tumor Nature Reserve. J. Soil Water Conserv. 2018, 32, 210–216. [Google Scholar]
- Chen, L.; Xie, G.D.; Zhang, C.S.; Gai, L.Q.; Pei, S.; Xu, Z.R. Typical forest ecosystem services in Baimaxueshan National Nature Reserve. Chin. J. Ecol. 2011, 30, 1781–1785. [Google Scholar]
- Wang, B.; Guan, W.B.; Wu, J.A.; Ma, K.M.; Wang, X.L.; Liu, G.H. A Method for Assessing Regional Ecological Security Pattern to Conserve Biodiversity–GAP Analysis. Res. Soil Water Conserv. 2006, 13, 192–196. [Google Scholar]
- Lu, Z.; Sun, Y.; Li, Y.; Yang, Y.; Liu, J.; Wang, G. Species delimitation and hybridisation history of a hazel species complex. Ann. Bot. 2021, 27, 875–886. [Google Scholar] [CrossRef]
- Girard, L.F.; Cerreta, M.; Toro, P.D. Towards a Local Comprehensive Productive Development Strategy: A Methodological Proposal for the Metropolitan City of Naples. Qual. Innov. Prosper. 2016, 21, 223. [Google Scholar] [CrossRef]
- Mathey, A.H.; Krcmar, E.; Dragicevic, S.; Vertinsky, I. An object-oriented cellular automata model for forest planning problems. Ecol. Model. 2008, 212, 359–371. [Google Scholar] [CrossRef]
- Nowakowska, J.A.; Stocki, M.; Stocka, N.; Lusarski, S.; Oszako, T. Interactions between phytophthora cactorum, armillaria gallica and betula pendula seedlings subjected to defoliation. Forests 2020, 11, 1107. [Google Scholar] [CrossRef]
- Berezovska, D.; Oszako, T.; Malewski, T.; Stocki, M.; Nowakowska, J.A. Effect of defoliation on the defense reactions of silver birch (Betula pendula) infected with phytophthora plurivora. Forests 2021, 12, 910. [Google Scholar] [CrossRef]
- Nowakowska, J.A.; Hsiang, T.; Patynek, P.; Stereńczak, K.; Oszako, T. Health Assessment and Genetic Structure of Monumental Norway Spruce Trees during A Bark Beetle (Ips typographus L.) Outbreak in the Biaowiea Forest District, Poland. Forests 2020, 11, 647. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).