Assessment of Biophysical Properties of Faecal Pellets from Channel Catfish (Ictalurus punctatus) and Bighead Carp (Aristichthys nobilis)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design and Faeces Sample Collection
2.2. Settling Velocity of Faecal Pellets
2.3. Water Stability and Nutrient Leaching of Faecal Pellets
2.4. Statistical Analysis
3. Results
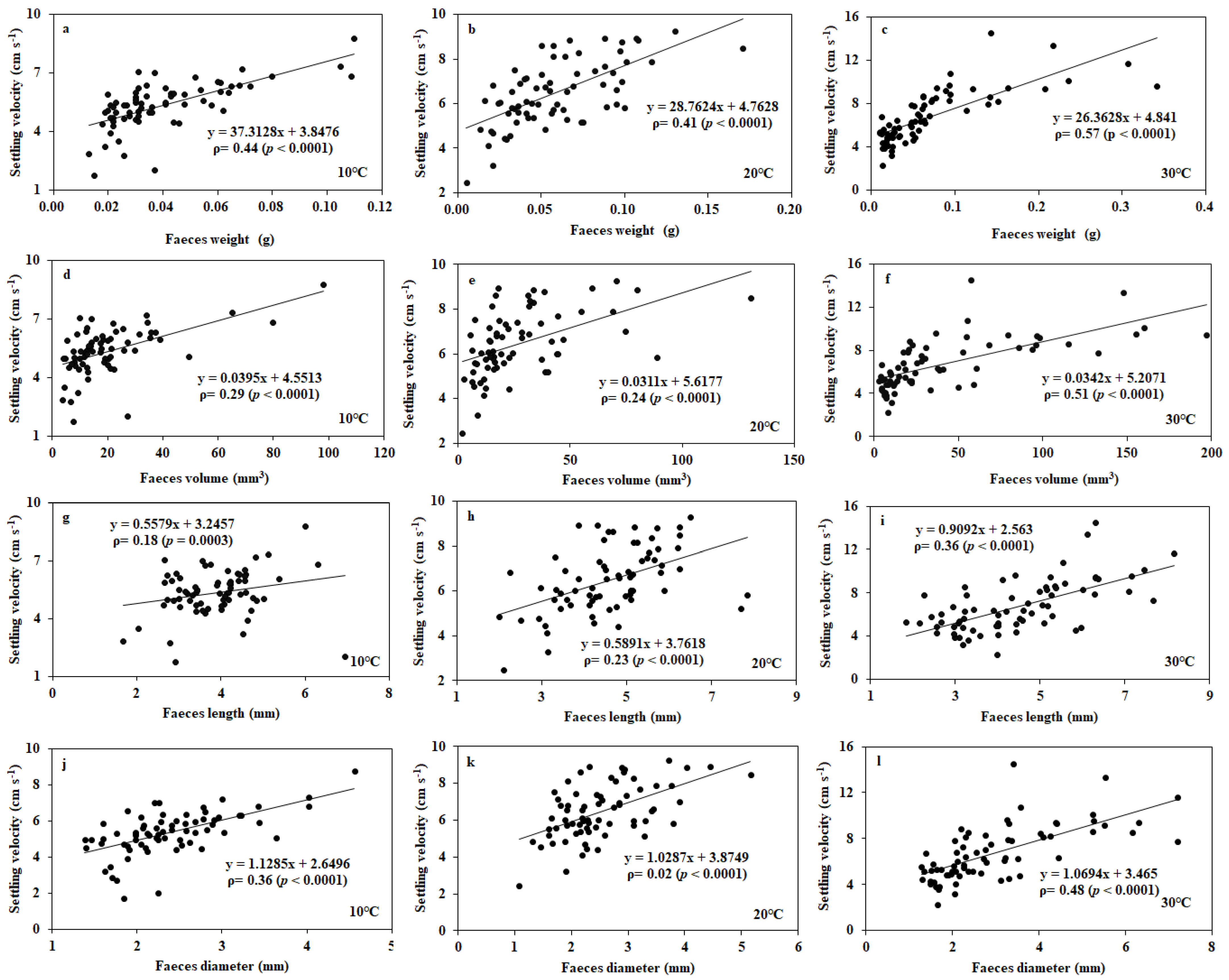
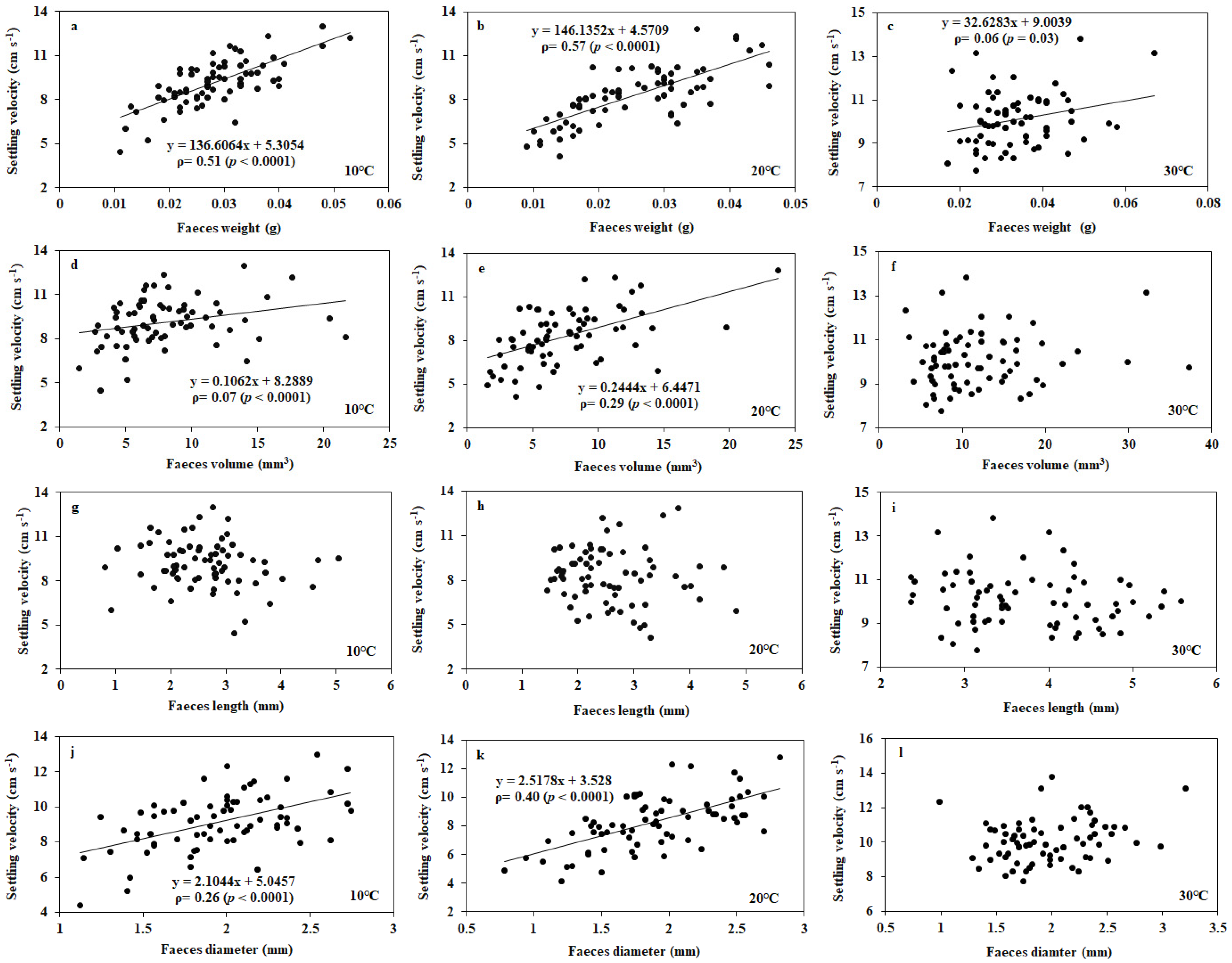
3.1. Settling Velocity of Faecal Pellets

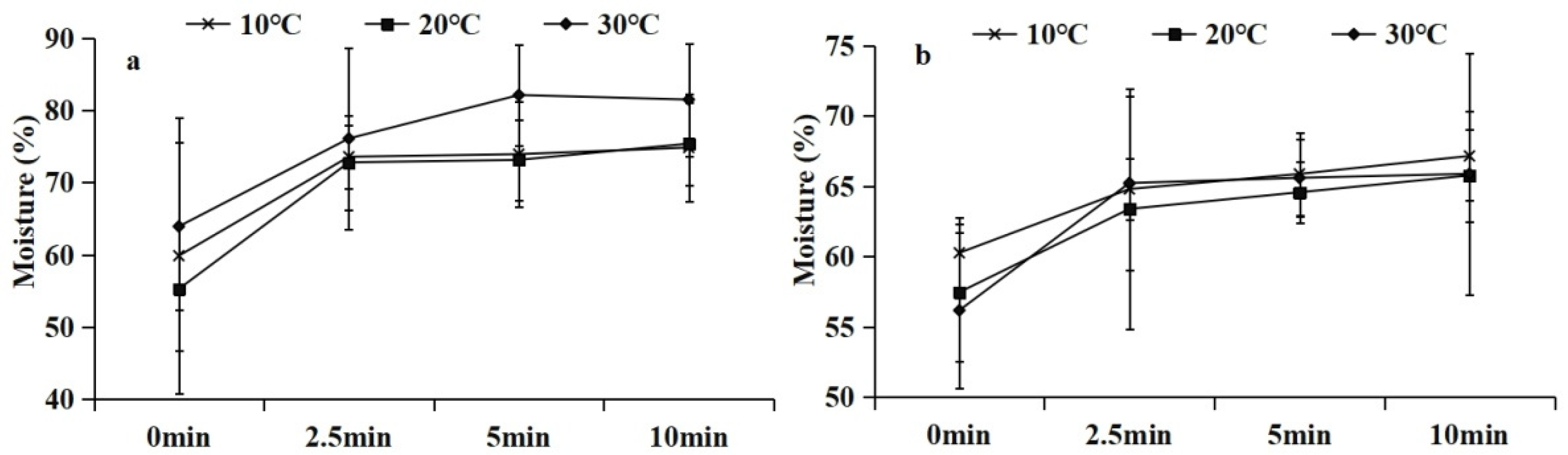
3.2. Moisture of Immersed Faecal Pellets
3.3. Release of Nutrients from Faecal Pellets
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zeng, Q.; Gu, X.; Chen, X.; Mao, Z. The impact of Chinese mitten crab culture on water quality, sediment and the pelagic and macrobenthic community in the reclamation area of Guchenghu Lake. Fish. Sci. 2013, 79, 689–697. [Google Scholar] [CrossRef]
- Wang, Q.D.; Li, Z.; Gui, J.F.; Liu, J.S.; Ye, S.W.; Yuan, J.; De Silva, S.S. Paradigm changes in freshwater aquaculture practices in china: Moving towards achieving environmental integrity and sustainability. Ambio 2017, 47, 410–426. [Google Scholar] [CrossRef]
- Wang, X.X.; Olsen, L.M.; Reitan, K.I.; Olsen, Y. Discharge of nutrient wastes from salmon farms: Environmental effects, and potential for integrated multi-trophic aquaculture. Aquac. Environ. Interact. 2012, 2, 267–283. [Google Scholar] [CrossRef]
- Amirkolaie, A.K. Environmental impact of nutrient discharged by aquaculture waste water on Haraz the river. J. Fish. Aquat. Sci. 2008, 3, 275–279. [Google Scholar] [CrossRef]
- Verdegem, M.C.J. Nutrient discharge from aquaculture operations in function of system design and production environment. Rev. Aquac. 2013, 5, 158–171. [Google Scholar] [CrossRef]
- Zhang, Y.; Bleeker, A.; Liu, J.G. Nutrient discharge from China’s aquaculture industry and associated environmental impacts. Environ. Res. Lett. 2015, 10, S109. [Google Scholar] [CrossRef]
- Chen, Y.S.; Beveridge, M.C.M.; Telfer, T.C.; Roy, W.J. Nutrient leaching and settling rate characteristics of the faeces of Atlantic salmon (Salmo salar L.) and the implications for modelling of solid waste dispersion. J. Appl. Ichthyol. 2003, 19, 114–117. [Google Scholar] [CrossRef]
- Porrello, S.; Tomassetti, P.; Manzueto, L.; Finoia, M.G.; Persia, E.; Mercatali, I.; Stipa, P. The influence of marine cages on the sediment chemistry in the Western Mediterranean Sea. Aquaculture 2005, 249, 145–158. [Google Scholar] [CrossRef]
- Holmer, M.; Thorsen, S.W.; Carlsson, M.S.; Kjerulf, P.J. Pelagic and benthic nutrient regeneration processes in mussel cultures (Mytilus edulis) in a eutrophic coastal area (Skive Fjord, Denmark). Estuaries Coasts 2015, 38, 1629–1641. [Google Scholar] [CrossRef]
- Chopin, T.; Buschmann, A.H.; Halling, C.; Troell, M.; Kautsky, N.; Neori, A.; Kraemer, G.P.; Zertuche-Gonzalez, J.A.; Yarish, C.; Neefus, C. Integrating seaweeds into marine aquaculture systems: A key towards sustainability. J. Phycol. 2001, 37, 975–986. [Google Scholar] [CrossRef]
- Cheshuk, B.W.; Purser, G.J.; Quintana, R. Integrated open-water mussel (Mytilus planulatus) and Atlantic salmon (Salmo salar) culture in Tasmania, Australia. Aquaculture 2003, 218, 357–378. [Google Scholar] [CrossRef]
- Neori, A.; Chopin, T.; Troell, M.; Buschmann, A.; Kraemer, G.P.; Halling, C.; Shpigel, M.; Yarish, C. Integrated aquaculture: Rationale, evolution and state of the art emphasizing seaweed biofiltration in modern mariculture. Aquaculture 2004, 231, 361–391. [Google Scholar] [CrossRef]
- Troell, M.; Joyce, A.; Chopin, T.; Neori, A.; Buschmann, A.; Fang, J. Ecological engineering in aquaculture-potential for integrated multi-trophic aquaculture in marine offshore systems. Aquaculture 2009, 297, 1–9. [Google Scholar] [CrossRef]
- Wang, Q.D.; Cheng, L.; Liu, J.S.; Li, Z.J.; Xie, S.Q.; De Silva, S.S. Freshwater aquaculture in PR China: Trends and prospects. Rev. Aquac. 2015, 7, 283–302. [Google Scholar] [CrossRef]
- Castro, J.L.R.; Flores, R.G.; Guerra, P.T.; Martínez, J.G.S.; Guzmán, G.A.; Osti, J.L.; Pfeiffer, C.R.; Hernández, N.I.D.L.C. Spatial and seasonal variations on Henneguya exilis prevalence on cage intensive cultured channel catfish (Ictalurus punctatus), in tamaulipas, Mexico. Lat. Am. J. Aquat. Res. 2013, 41, 194–198. [Google Scholar] [CrossRef]
- Kleinholz, C.; Luker, G.; Tang, L. A comparison of channel catfish, Ictalurus punctatus, production in single and mixed cohort culture during two production seasons. J. Appl. Aquac. 2005, 17, 85–96. [Google Scholar] [CrossRef]
- FBMA (Fisheries Bureau of Ministry of Agriculture). China Fishery Statistics. 2021. Available online: https://www.yearbookchina.com/navibooklist-n3022013079-1.html (accessed on 1 June 2021).
- Soes, D.M.; Leuven, R.S.E.W.; Matthews, J.; Broeckx, P.B.; Haenen, O.L.M.; Engelsma, M.Y. A Risk Analysis of Bigheaded Carp (Hypophthalmichthys sp.) in the Netherlands; Bureau Waardenburg bv: Culemborg, The Netherlands, 2011; pp. 14–15. [Google Scholar]
- Freeze, M.; Kelly, A.; Engle, C.; Armstrong, M.; Mitchell, A. Early governmental research and promotion of silver carp and bighead carp culture in the United States. In Proceedings of the American Fisheries Society 143rd Annual Meeting, Little Rock, AR, USA, 8–14 September 2013. [Google Scholar]
- Magill, S.H.; Thetmeyer, H.; Cromey, C.J. Settling velocity of faecal pellets of gilthead sea bream (Sparus aurata L.) and sea bass (Dicentrarchus labrax L.) and sensitivity analysis using measured data in a deposition model. Aquaculture 2006, 251, 295–305. [Google Scholar] [CrossRef]
- Akhan, S.; Gedik, K. The nutrient releases from sea bass (Dicentrarchus labrax Linnaeus,1758) faeces and food in estuarine Black Sea condition. J. Food Agric. Environ. 2011, 9, 738–740. [Google Scholar] [CrossRef]
- Reid, G.K.; Liutkus, M.; Robinson, S.M.C.; Chopin, T.R.; Blair, T.; Lander, T.; Mullen, J.; Page, F.; Moccia, R.D. A review of the biophysical properties of salmonid faeces: Implications for aquaculture waste dispersal models and integrated multi-trophic aquaculture. Aquac. Res. 2009, 40, 257–273. [Google Scholar] [CrossRef]
- Hakanson, L.; Wrvik, A.; Makinene, T.; Molleg, B. Basic Concepts Concerning Assessments of Environmental Effects of Marine Fish Farms; Nordic Council of Ministers: Copenhagen, Denmark, 1988; p. 103. [Google Scholar]
- Schneider, O.; Amirkolaie, A.K.; Vera-Cartas, J.; Eding, E.H.; Schrama, J.W. Digestibility, faeces recovery, and related carbon, nitrogen and phosphorus balances of five feed ingredients evaluated as fishmeal alternatives in Nile tilapia, Oreochromis niloticus L. Aquac. Res. 2004, 35, 1370–1379. [Google Scholar] [CrossRef]
- Amirkolaie, A.K. Reduction in the environmental impact of waste discharged by fish farms through feed and feeding. Rev. Aquac. 2011, 3, 19–26. [Google Scholar] [CrossRef]
- Lee, S.; Hartstein, N.D.; Wong, K.Y.; Jeffs, A. Assessment of the production and dispersal of faecal waste from the sea-cage aquaculture of spiny lobsters. Aquac. Res. 2014, 47, 1569–1583. [Google Scholar] [CrossRef]
- Chen, Y.S.; Beveridge, M.C.M.; Telfer, T.C. Settling rate characteristics and nutrient content of the faeces of Atlantic salmon, Salmo salar L. and the implications for modelling of solid waste dispersion. Aquac. Res. 1999, 30, 395–398. [Google Scholar] [CrossRef]
- Fernandez-Jover, D.; Sanchez-Jerez, P.; Bayle-Sempere, J.; Carratala, A.; Leon, V.M. Addition of dissolved nitrogen and dissolved organic carbon from wild fish faeces and food around Mediterranean fish farms: Implications for waste-dispersal models. J. Exp. Mar. Biol. Ecol. 2007, 340, 160–168. [Google Scholar] [CrossRef]
- Piedecausa, M.A.; Aguado-Giménez, F.; García-García, B.; Ballester, G.; Telfer, T. Settling velocity and total ammonia nitrogen leaching from commercial feed and faecal pellets of gilthead seabream (Sparus aurata L. 1758) and seabass (Dicentrarchus labrax L. 1758). Aquac. Res. 2009, 40, 1703–1714. [Google Scholar] [CrossRef]
- Liu, X.D.; Wang, J.Y.; Zhang, L.M.; Wang, S.X.; Huang, B.S. Anesthetic MS-222 and the influence factors on its anesthetic effects. Fish. Sci. Technol. Inf. 2009, 36, 56–59. (In Chinese) [Google Scholar] [CrossRef]
- Zhang, M.Q.; Wang, K.Y.; Wang, J.; Chen, D.F.; Huang, L.Y. The anesthetic effect of MS-222 on channel catfish. Fish. Sci. Technol. Inf. 2014, 41, 21–25+31. (In Chinese) [Google Scholar] [CrossRef]
- Wong, K.B.; Piedrahita, R.H. Settling velocity characterization of aquacultural solids. Aquac. Eng. 2000, 21, 233–246. [Google Scholar] [CrossRef]
- Cromey, C.J.; Nickell, T.D.; Treasurer, J.; Black, K.D.; Inall, M. Modelling the impact of cod (Gadus morhua L.) farming in the marine environment-CODMOD. Aquaculture 2009, 289, 42–53. [Google Scholar] [CrossRef]
- Pérez, Ó.; Almansa, E.; Riera, R.; Rodriguez, M.; Ramos, E.; Costa, J.; Monterroso, Ó. Food and faeces settling velocities of meagre (Argyrosomus regius) and its application for modelling waste dispersion from sea cage aquaculture. Aquaculture 2014, 420, 171–179. [Google Scholar] [CrossRef]
- Celis-Guerrero, L.E.; García-Carreño, F.L.; del Toro, M.A.N. Characterization of proteases in the digestive system of spiny lobster (Panulirus interruptus). Mar. Biotechnol. 2004, 6, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Tlusty, M.F.; Snook, K.; Pepper, V.A.; Anderson, M.R. The potential for soluble and transport loss of particulate aquaculture wastes. Aquac. Res. 2000, 31, 745–755. [Google Scholar] [CrossRef]
- Curran, K.J.; Hill, P.S.; Milligan, T.G. Time variation of floc properties in a settling column. J. Sea Res. 2003, 49, 1–9. [Google Scholar] [CrossRef]
- Vita, R.; Marín, A.; Madrid, J.A.; Jiménez-Brinquis, B.; Cesar, A.; Marín-Guirao, L. Effects of wild fishes on waste exportation from a Mediterranean fish farm. Mar. Ecol. Prog. Ser. 2004, 277, 253–261. [Google Scholar] [CrossRef]
- Pillay, T.V.R. Aquaculture and Environment; Black Well Publishing: Oxford, UK, 2004; pp. 73–74. [Google Scholar]
- Brooks, K.M.; Mahnken, C.V.W. Interactions of Atlantic salmon in the Pacific northwest environment: II. Organic wastes. Fish. Res. 2003, 62, 255–293. [Google Scholar] [CrossRef]
- Austreng, E. Digestibility determination in fish using chromic oxide marking and analysis of contents from different segments of the gastrointestinal tract. Aquaculture 1978, 13, 265–272. [Google Scholar] [CrossRef]
- Windell, J.T.; Foltz, J.W.; Sarokon, J.A. Methods of fecal collection and nutrient leaching in digestibility studies. Progress. Fish-Cult. 1978, 40, 51–55. [Google Scholar] [CrossRef]
- Lefebvre, S.; Barille, L.; Clerc, M. Pacific oyster (Crassostrea gigas) feeding responses to fish farm-effluent. Aquaculture 2000, 187, 185–198. [Google Scholar] [CrossRef]
- Gao, Q.; Shin, P.; Lin, G.; Chen, S.; Cheung, S. Stable isotope and fatty acid evidence for uptake of organic waste by green-lipped mussels Perna viridis in a polyculture fish farm system. Mar. Ecol. Prog. Ser. 2006, 317, 273–283. [Google Scholar] [CrossRef]
- MacDonald, B.A.; Robinson, S.M.C.; Barrington, K.A. Feeding activity of mussels (Mytilus edulis) held in the field at an integrated multi-trophic aquaculture (IMTA) site (Salmo salar) and exposed to fish food in the laboratory. Aquaculture 2011, 314, 244–251. [Google Scholar] [CrossRef]
- Ajie, G.S.; Prihatiningtyas, E. Nutrients removal from integrated multi-trophic aquaculture (IMTA) water using waste stabilization ponds (WSP). IOP Conf. Ser. Earth Environ. Sci. 2022, 976, 012029. [Google Scholar] [CrossRef]
- Kerrigan, D.; Suckling, C.C. A meta-analysis of integrated multitrophic aquaculture: Extractive species growth is most successful within close proximity to open-water fish farms. Rev. Aquac. 2018, 10, 560–572. [Google Scholar] [CrossRef]
- Kibria, G.; Nugegoda, D.; Fairclough, R.; Lam, P. The nutrient content and the release of nutrients from fish food and faeces. Hydrobiologia 1997, 357, 165–171. [Google Scholar] [CrossRef]
- Garcia-Ruiz, R.; Hall, G.H. Phosphorus fractionation and mobility in the food and faeces of hatchery reared rainbow trout (Onchorhynchus mykiss). Aquaculture 1996, 145, 183–193. [Google Scholar] [CrossRef]
- Kristiansen, G.; Hessen, D.O. Nitrogen and phosphorus excretion from the noble crayfish, Astacus astacus L. in relation to food type and temperature. Aquaculture 1992, 102, 245–264. [Google Scholar] [CrossRef]
| Fish Species and Water Temperatures | Immersion Time | |||
|---|---|---|---|---|
| 0 min | 2.5 min | 5 min | 10 min | |
| Channel catfish | ||||
| 10 °C (n = 16) | 342.27 ± 12.15 | 329.57 ± 19.12 (3.71%) | 322.24 ± 4.42 (5.85%) | 314.44 ± 7.49 (8.13%) |
| 20 °C (n = 16) | 382.78 ± 3.80 | 378.97 ± 5.96 (0.99%) | 362.28 ± 8.84 (5.36%) | 349.60 ±11.98 (8.67%) |
| 30 °C (n = 16) | 375.92 ± 6.52 | 368.24 ± 3.47 (2.04%) | 363.28 ± 7.69 (3.36%) | 330.29 ± 11.01 (12.14%) |
| Bighead carp | ||||
| 10 °C (n = 20) | 63.38 ± 0.75 | 63.26 ± 0.78 (0.19%) | 59.89 ± 2.34 (5.50%) | 59.76 ± 0.83 (5.70%) |
| 20 °C (n = 20) | 65.41 ± 0.45 | 64.04 ± 0.67 (2.10%) | 61.71 ± 1.40 (5.66%) | 60.68 ± 0.18 (7.23%) |
| 30 °C (n = 20) | 58.05 ± 0.37 | 57.73 ± 0.39 (0.55%) | 56.79 ± 0.57 (2.18%) | 56.59 ± 0.85 (2.52%) |
| Fish Species and Water Temperatures | Immersion Time | |||
|---|---|---|---|---|
| 0 min | 2.5 min | 5 min | 10 min | |
| Channel catfish | ||||
| 10 °C (n = 16) | 16.71 ± 0.63 | 16.67 ± 0.13 (0.25%) | 15.87 ± 0.34 (5.01%) | 15.43 ± 0.69 (7.66%) |
| 20 °C (n = 16) | 24.27 ± 0.36 | 23.24 ± 1.43 (4.25%) | 20.58 ± 0.86 (15.20%) | 19.18 ± 0.28 (20.97%) |
| 30 °C (n = 16) | 26.10 ± 1.76 | 21.72 ± 0.82 (16.79%) | 21.67 ± 0.12 (16.98%) | 17.72 ± 2.36 (32.11%) |
| Bighead carp | ||||
| 10 °C (n = 20) | 7.67 ± 0.10 | 7.40 ± 0.11 (3.54%) | 7.40 ± 0.07 (3.46%) | 7.24 ± 0.06 (5.66%) |
| 20 °C (n = 20) | 7.60 ± 0.09 | 7.51 ± 0.08 (1.17%) | 7.46 ± 0.06 (1.86%) | 7.32 ± 0.03 (3.67%) |
| 30 °C (n = 20) | 7.59 ± 0.33 | 7.28 ± 0.04 (4.08%) | 7.27 ± 0.07 (4.27%) | 7.18 ± 0.06 (5.41%) |
| Fish Species and Water Temperatures | Immersion Time | |||
|---|---|---|---|---|
| 0 min | 2.5 min | 5 min | 10 min | |
| Channel catfish | ||||
| 10 °C (n = 16) | 25.36 ± 0.04 | 24.88 ± 0.05 (1.88%) | 21.86 ± 0.11 (13.79%) | 17.87 ± 0.04 (29.53%) |
| 20 °C (n = 16) | 23.69 ± 0.03 | 21.77 ± 0.05 (8.09%) | 18.51 ± 0.01 (21.87%) | 15.03 ± 0.05 (36.54%) |
| 30 °C (n = 16) | 20.93 ± 0.06 | 18.28 ± 0.07 (12.67%) | 15.80 ± 0.03 (24.53%) | 14.59 ± 0.08 (30.29%) |
| Bighead carp | ||||
| 10 °C (n = 20) | 6.22 ± 0.03 | 6.10 ± 0.01 (1.89%) | 5.75 ± 0.03 (7.59%) | 5.68 ± 0.04 (8.71%) |
| 20 °C (n = 20) | 5.67 ± 0.01 | 5.47 ± 0.03 (3.52%) | 5.43 ± 0.03 (4.28%) | 5.43 ± 0.00 (4.32%) |
| 30 °C (n = 20) | 5.68 ± 0.02 | 5.57 ± 0.02 (1.99%) | 5.49 ± 0.00 (3.29%) | 5.47 ± 0.03 (3.75%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yuan, T.; Wang, Q.; Li, S.; Huang, G.; Zhang, T.; Li, Z.; Liu, J. Assessment of Biophysical Properties of Faecal Pellets from Channel Catfish (Ictalurus punctatus) and Bighead Carp (Aristichthys nobilis). Sustainability 2022, 14, 7201. https://doi.org/10.3390/su14127201
Yuan T, Wang Q, Li S, Huang G, Zhang T, Li Z, Liu J. Assessment of Biophysical Properties of Faecal Pellets from Channel Catfish (Ictalurus punctatus) and Bighead Carp (Aristichthys nobilis). Sustainability. 2022; 14(12):7201. https://doi.org/10.3390/su14127201
Chicago/Turabian StyleYuan, Ting, Qidong Wang, Shiqi Li, Geng Huang, Tanglin Zhang, Zhongjie Li, and Jiashou Liu. 2022. "Assessment of Biophysical Properties of Faecal Pellets from Channel Catfish (Ictalurus punctatus) and Bighead Carp (Aristichthys nobilis)" Sustainability 14, no. 12: 7201. https://doi.org/10.3390/su14127201
APA StyleYuan, T., Wang, Q., Li, S., Huang, G., Zhang, T., Li, Z., & Liu, J. (2022). Assessment of Biophysical Properties of Faecal Pellets from Channel Catfish (Ictalurus punctatus) and Bighead Carp (Aristichthys nobilis). Sustainability, 14(12), 7201. https://doi.org/10.3390/su14127201






