A Data Fusion Model to Merge the Spectra Data of Intact and Powdered Cayenne Pepper for the Fast Inspection of Antioxidant Properties
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cayenne Pepper Samples
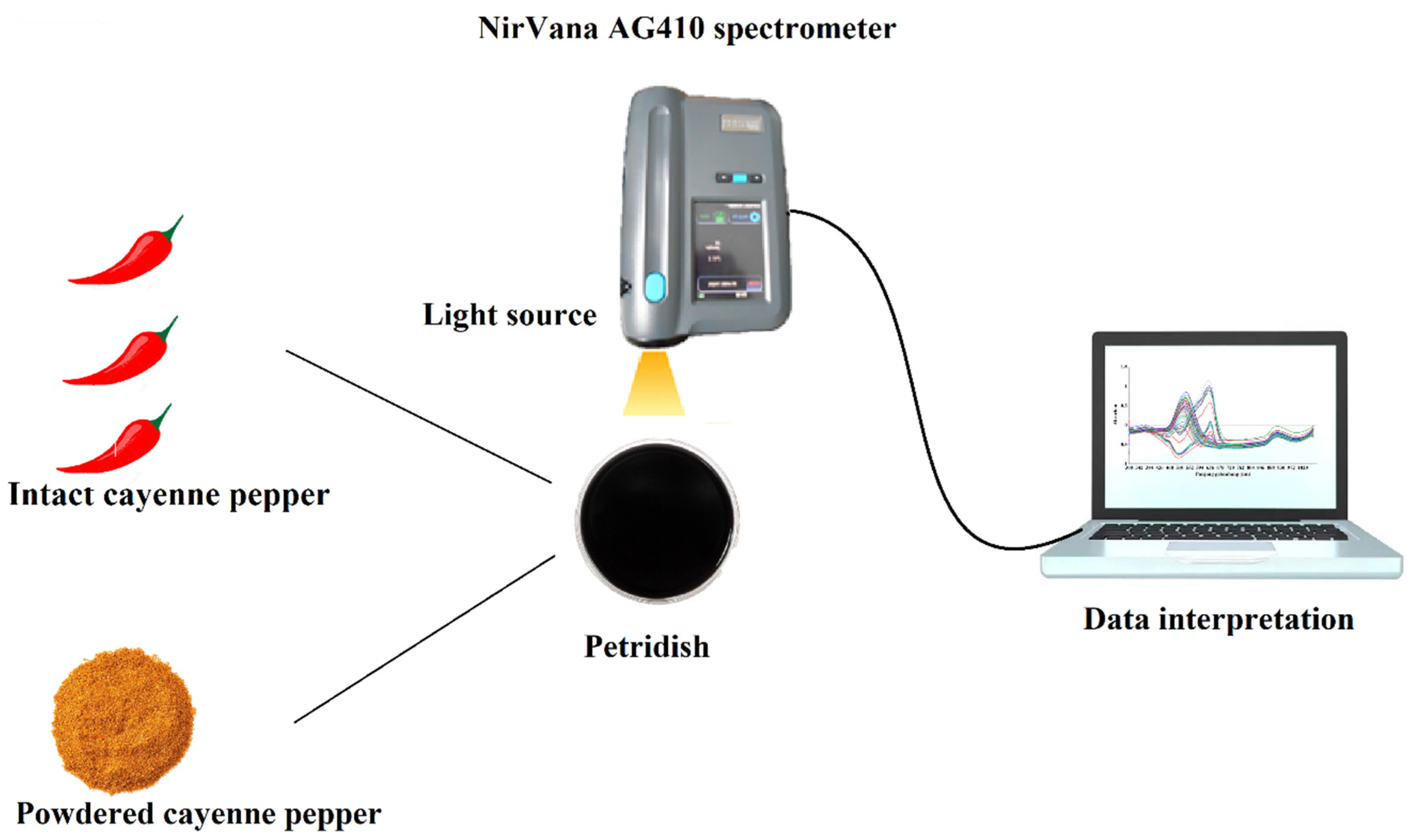
2.2. Spectra Measurement
2.3. Chemical Measurements Using Antioxidant DPPH Method
2.4. Data Analysis
3. Results and Discussion
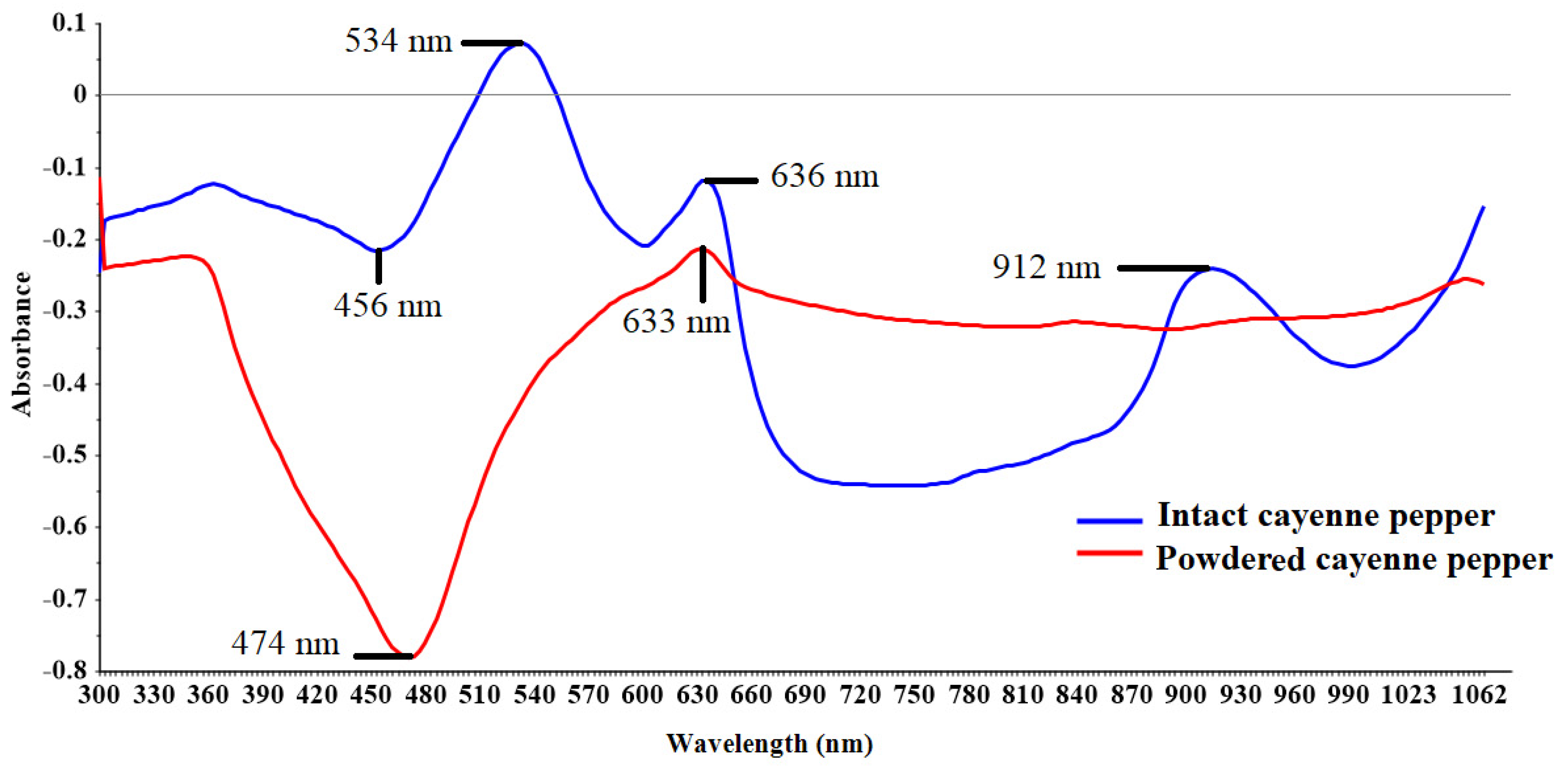
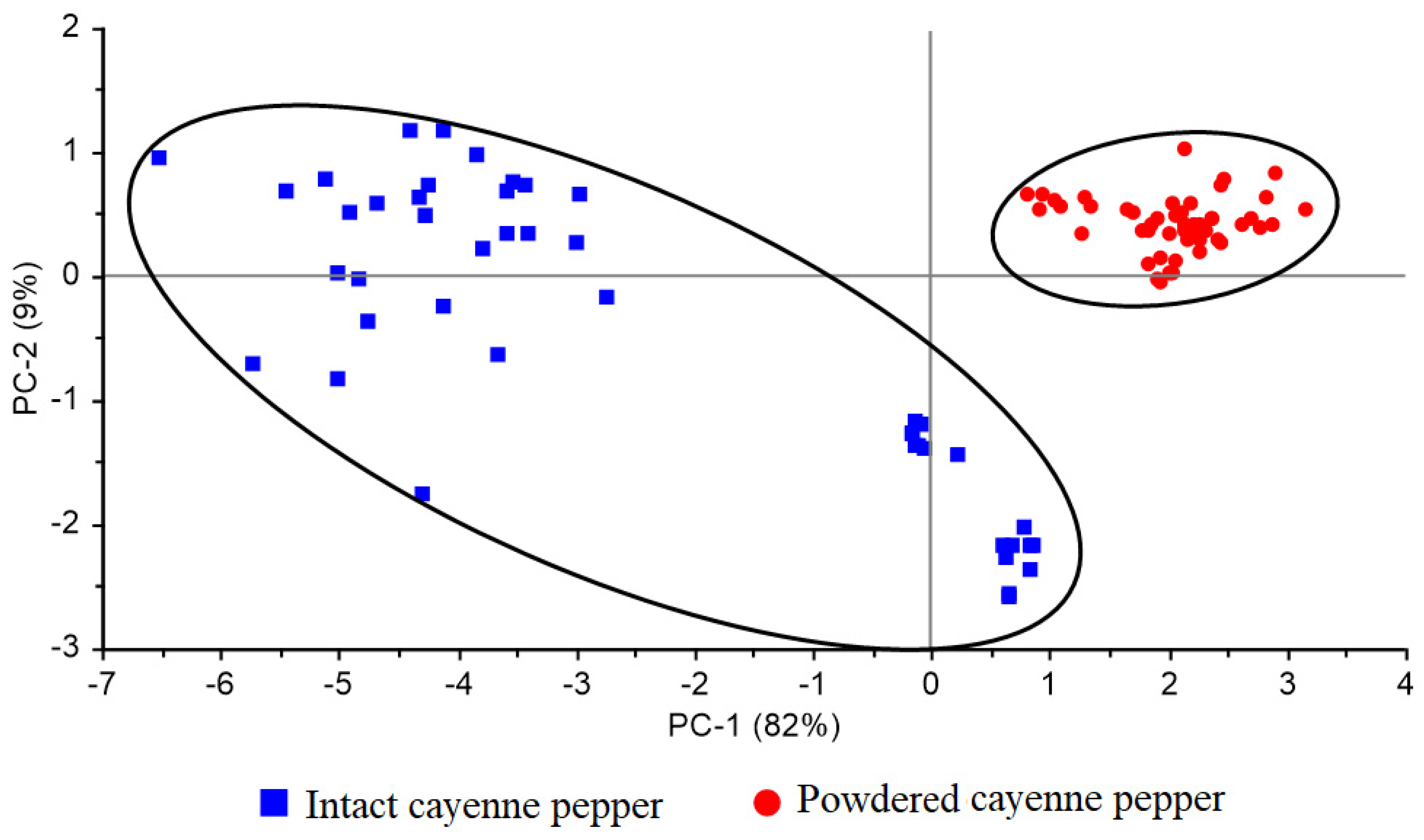
3.1. Spectral Information
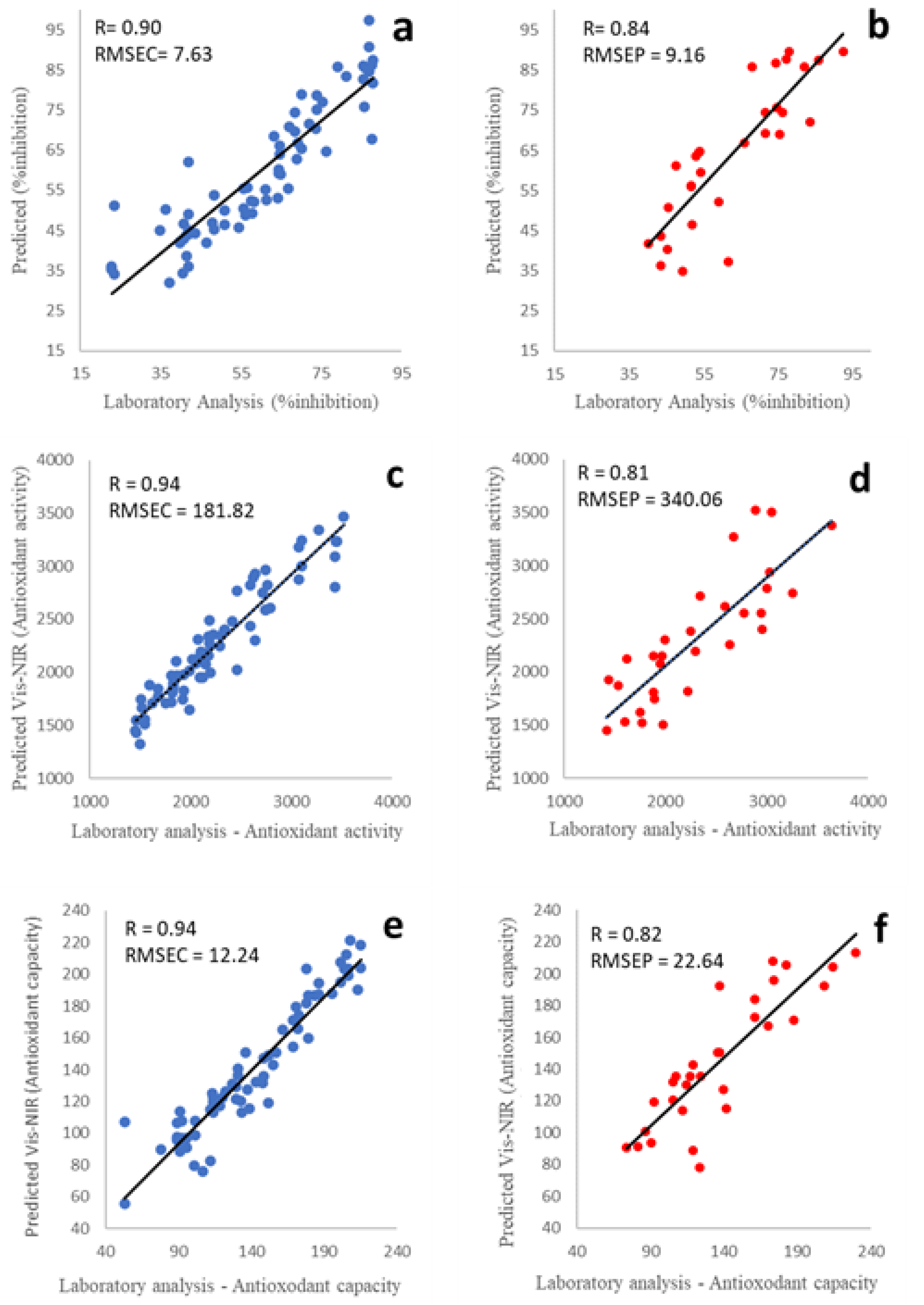
3.2. Calibration Model Development for Antioxidant Properties Prediction
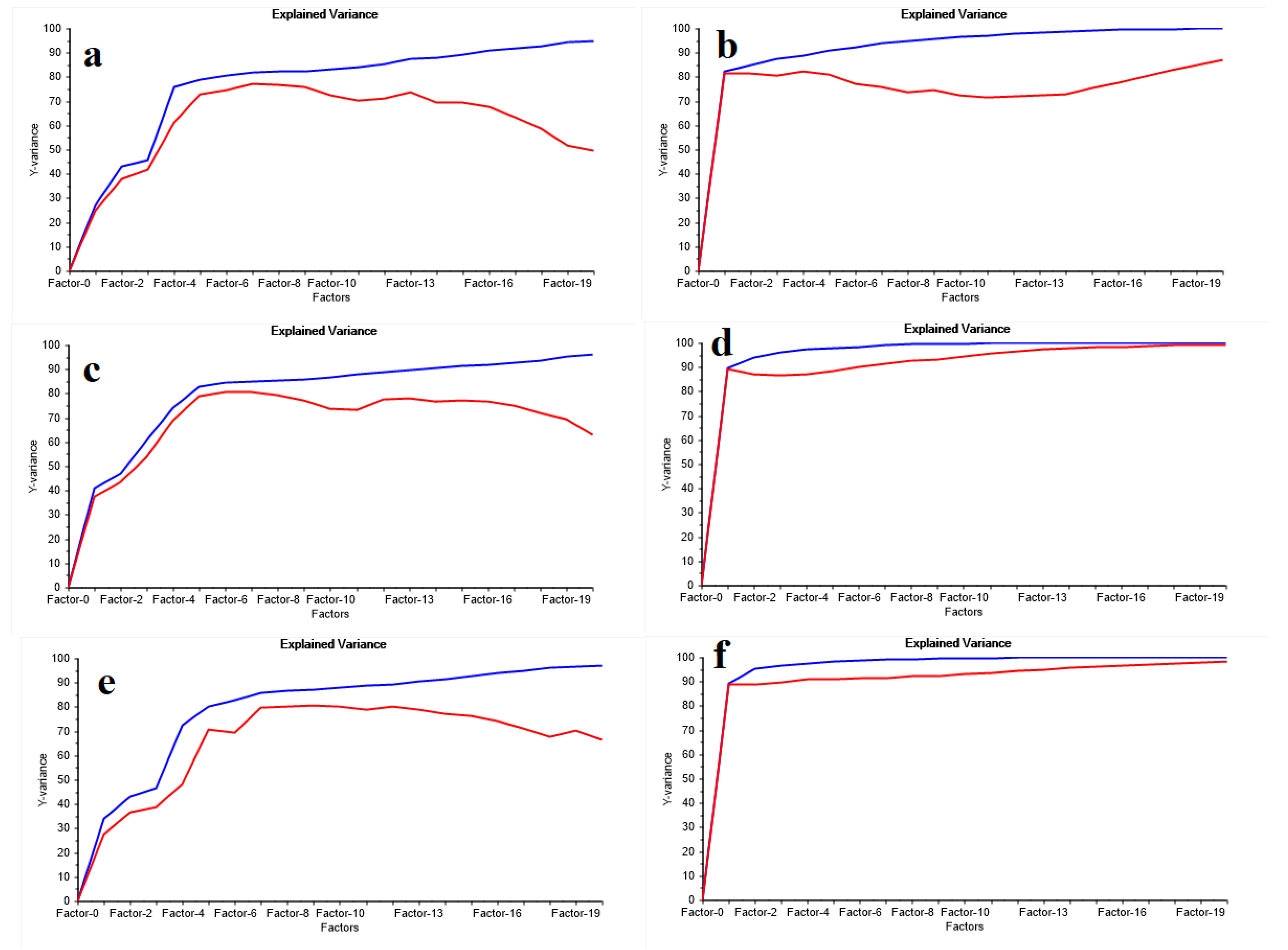
3.3. The Variance of the Antioxidant Properties Prediction
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Swallah, M.S.; Sun, H.; Affoh, R.; Fu, H.; Yu, H. Antioxidant potential overviews of secondary metabolites (polyphenols) in fruits. Int. J. Food Sci. 2020, 2020, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, A.R.; Leonardi, B.; Miron, D.; Schapoval, E.; de Oliveira, J.R.; Gosmann, G. Antioxidant and anti-inflammatory properties of Capsicum baccatum: From traditional use to scientific approach. J. Ethnopharmacol. 2012, 139, 228–233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, Z.; El-Obeid, T.; Riley, M.; Li, M.; Page, A.; Liu, J. High Chili intake and cognitive function among 4582 adults: An open cohort study over 15 years. Nutrients 2019, 11, 1183. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adebiyi, O.E.; Olayemi, F.O.; Ning-Hua, T.; Guang-Zhi, Z. In vitro antioxidant activity, total phenolic and flavonoid contents of ethanol extract of stem and leaf of Grewia carpinifolia. Beni-Suef Univ. J. Basic Appl. Sci. 2017, 6, 10–14. [Google Scholar] [CrossRef]
- Apak, R.; Gorinstein, S.; Böhm, V.; Schaich, K.M.; Özyürek, M.; Güçlü, K. Methods of measurement and evaluation of natural antioxidant capacity/activity (IUPAC technical report). Pure Appl. Chem. 2013, 85, 957–998. [Google Scholar] [CrossRef] [Green Version]
- Lim, Y.Y.; Murtijaya, J. Antioxidant properties of Phyllanthus amarus extracts as affected by different drying methods. LWT-Food Sci. Technol. 2007, 40, 1664–1669. [Google Scholar] [CrossRef]
- Paixão, N.; Perestrelo, R.; Marques, J.C.; Câmara, J.S. Relationship between antioxidant capacity and total phenolic content of red, rosé and white wines. Food Chem. 2007, 105, 204–214. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Bostic, T.R.; Gu, L. Antioxidant capacities, procyanidins and pigments in avocados of different strains and cultivars. Food Chem. 2010, 122, 1193–1198. [Google Scholar] [CrossRef]
- Shahidi, F.; Zhong, Y. Measurement of antioxidant activity. J. Funct. Foods 2015, 18, 757–781. [Google Scholar] [CrossRef]
- Polinar, Y.Q.; Yaptenco, K.F.; Peralta, E.K.; Agravante, J.U. Near-infrared spectroscopy for non-destructive prediction of maturity and eating quality of ‘carabao’ mango (Mangifera indica L.) fruit. Agric. Eng. Int. CIGR J. 2019, 21, 209–219. [Google Scholar]
- Sohaib Ali Shah, S.; Zeb, A.; Qureshi, W.S.; Arslan, M.; Ullah Malik, A.; Alasmary, W.; Alanazi, E. Towards fruit maturity estimation using NIR spectroscopy. Infrared Phys. Technol. 2020, 111, 103479. [Google Scholar] [CrossRef]
- Walsh, K.B.; Blasco, J.; Zude-Sasse, M.; Sun, X. Visible-NIR ‘point’ spectroscopy in postharvest fruit and vegetable assessment: The science behind three decades of commercial use. Postharvest Biol. Technol. 2020, 168, 111246. [Google Scholar] [CrossRef]
- Chen, W.; Li, H.; Zhang, F.; Xiao, W.; Zhang, R.; Chen, Z.; Du, Y. Handheld short-wavelength NIR spectroscopy for rapid determination of sugars and carbohydrate in fresh juice with Sampling Error Profile Analysis. Infrared Phys. Technol. 2021, 115, 103732. [Google Scholar] [CrossRef]
- Hayati, R.; Zulfahrizal, Z.; Munawar, A.A. Robust prediction performance of inner quality attributes in intact cocoa beans using near infrared spectroscopy and multivariate analysis. Heliyon 2021, 7, 1–7. [Google Scholar] [CrossRef]
- Mishra, P.; Woltering, E.; El Harchioui, N. Improved prediction of ‘Kent’ mango firmness during ripening by near-infrared spectroscopy supported by interval partial least square regression. Infrared Phys. Technol. 2020, 110, 103459. [Google Scholar] [CrossRef]
- Posom, J.; Klaprachan, J.; Rattanasopa, K.; Sirisomboon, P.; Saengprachatanarug, K.; Wongpichet, S. Predicting marian plum fruit quality without environmental condition impact by handheld visible-near-infrared spectroscopy. ACS Omega 2020, 5, 27909–27921. [Google Scholar] [CrossRef] [PubMed]
- Kljusurić, J.G.; Mihalev, K.; Bečić, I.; Polović, I.; Georgieva, M.; Djaković, S.; Kurtanjek, Ž. Near-infrared spectroscopic analysis of total phenolic content and antioxidant activity of berry fruits. Food Technol. Biotechnol. 2016, 54, 236–242. [Google Scholar] [CrossRef]
- Alfieri, M.; Cabassi, G.; Habyarimana, E.; Quaranta, F.; Balconi, C.; Redaelli, R. Discrimination and prediction of polyphenolic compounds and total antioxidant capacity in sorghum grains. J. Near Infrared Spectrosc. 2019, 27, 46–53. [Google Scholar] [CrossRef]
- Carbas, B.; Machado, N.; Oppolzer, D.; Queiroz, M.; Brites, C.; Rosa, E.A.S.; Barros, A.I.R.N.A. Prediction of phytochemical composition, in vitro antioxidant activity and individual phenolic compounds of common beans using MIR and NIR spectroscopy. Food Bioprocess Technol. 2020, 13, 962–977. [Google Scholar] [CrossRef]
- Calegari, M.A.; Ayres, B.B.; dos Santos Tonial, L.M.; de Alencar, S.M.; Oldoni, T.L.C. Fourier transform near infrared spectroscopy as a tool for predicting antioxidant activity of propolis. J. King Saud Univ.-Sci. 2020, 32, 784–790. [Google Scholar] [CrossRef]
- Fu, H.; Hu, O.; Xu, L.; Fan, Y.; Shi, Q.; Guo, X.; Lan, W.; Yang, T.; Xie, S.; She, Y. Simultaneous recognition of species, quality grades, and multivariate calibration of antioxidant activities for 12 famous green teas using mid- and near-infrared spectroscopy coupled with chemometrics. J. Anal. Methods Chem. 2019, 2019, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Xia, F.; Li, C.; Zhao, N.; Li, H.; Chang, Q.; Liu, X.; Liao, Y.; Pan, R. Rapid determination of active compounds and antioxidant activity of okra seeds using Fourier transform near infrared (FT-NIR) spectroscopy. Molecules 2018, 23, 550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiang, J.; Cen, H.; Zhang, C.; Lyu, X.; Weng, H.; Xu, H.; He, Y. Nondestructive quality assessment of chili peppers using near-infrared hyperspectral imaging combined with multivariate analysis. Postharvest Biol. Technol. 2018, 146, 147–154. [Google Scholar] [CrossRef]
- Rohaeti, E.; Muzayanah, K.; Septaningsih, D.A.; Rafi, M. Fast analytical method for authentication of chili powder from synthetic dyes using uv-vis spectroscopy in combination with chemometrics. Indones. J. Chem. 2019, 19, 668–674. [Google Scholar] [CrossRef]
- Li, J.; Zhang, H.; Zhan, B.; Wang, Z.; Jiang, Y. Determination of SSC in pears by establishing the multi-cultivar models based on visible-NIR spectroscopy. Infrared Phys. Technol. 2019, 102, 1–10. [Google Scholar] [CrossRef]
- Kusumiyati; Hadiwijaya, Y.; Putri, I.E.; Munawar, A.A. Multi-product calibration model for soluble solids and water content quantification in Cucurbitaceae family, using visible/near-infrared spectroscopy. Heliyon 2021, 7, e07677. [Google Scholar] [CrossRef] [PubMed]
- Kusumiyati, K.; Hadiwijaya, Y.; Putri, I.E.; Munawar, A.A. Enhanced visible/near-infrared spectroscopic data for prediction of quality attributes in Cucurbitaceae commodities. Data Brief 2021, 39, 107458. [Google Scholar] [CrossRef] [PubMed]
- Rambo, M.K.D.; Ferreira, M.M.C.; Amorim, E.P. Multi-product calibration models using NIR spectroscopy. Chemom. Intell. Lab. Syst. 2016, 151, 108–114. [Google Scholar] [CrossRef]
- González-zamora, A.; Sierra-campos, E.; Luna-ortega, J.G.; Pérez-morales, R.; Carlos, J.; Ortiz, R.; García-hernández, J.L. Characterization of different capsicum varieties by evaluation of their capsaicinoids content by high performance liquid chromatography, determination of pungency and effect of high temperature. Molecules 2013, 18, 13471–13486. [Google Scholar] [CrossRef]
- Thapa, B.; Skalko-basnet, N.; Takano, A.; Masuda, K.; Basnet, P. High-performance liquid chromatography analysis of capsaicin content in 16 capsicum fruits from nepal. J. Med. Food 2009, 12, 908–913. [Google Scholar] [CrossRef]
- Fan, S.; Wang, Q.; Tian, X.; Yang, G.; Xia, Y.; Li, J.; Huang, W. Non-destructive evaluation of soluble solids content of apples using a developed portable Vis/NIR device. Biosyst. Eng. 2020, 193, 138–148. [Google Scholar] [CrossRef]
- Zhang, Q.; Li, Q.; Zhang, G. Rapid determination of leaf water content using Vis/NIR spectroscopy analysis with wavelength selection. Spectrosc. Int. J. 2012, 27, 93–105. [Google Scholar] [CrossRef]
- Pérez-gálvez, A.; Viera, I.; Roca, M. Carotenoids and chlorophylls as antioxidants. Antioxidants 2020, 9, 505. [Google Scholar] [CrossRef] [PubMed]
- Pola, W.; Sugaya, S.; Photchanachai, S. Influence of postharvest temperatures on carotenoid biosynthesis and phytochemicals in mature green chili (Capsicum annuum L.). Antioxidants 2020, 9, 203. [Google Scholar] [CrossRef] [Green Version]
- Windig, W.; Shaver, J.; Bro, R. Loopy MSC: A simple way to improve multiplicative scatter correction. Appl. Spectrosc. 2008, 62, 1153–1159. [Google Scholar] [CrossRef]
- Saganowska, P.; Wesolowski, M. Principal component and cluster analyses as supporting tools for co-crystals detection. J. Therm. Anal. Calorim. 2017, 130, 45–55. [Google Scholar] [CrossRef] [Green Version]
- Hadiwijaya, Y.; Putri, I.E.; Mubarok, S.; Hamdani, J.S. Rapid and non-destructive prediction of total soluble solids of guava fruits at various storage periods using handheld near-infrared instrument. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2020; pp. 1–7. [Google Scholar]
- Wang, J.; Wang, J.; Chen, Z.; Han, D. Development of multi-cultivar models for predicting the soluble solid content and firmness of European pear (Pyrus communis L.) using portable vis–NIR spectroscopy. Postharvest Biol. Technol. 2017, 129, 143–151. [Google Scholar] [CrossRef]
- Lu, X.; Wang, J.; Al-Qadiri, H.M.; Ross, C.F.; Powers, J.R.; Tang, J.; Rasco, B.A. Determination of total phenolic content and antioxidant capacity of onion (Allium cepa) and shallot (Allium oschaninii) using infrared spectroscopy. Food Chem. 2011, 129, 637–644. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhang, L.; Li, Y.; Wang, J. Orthogonal signal correction used for noise elimination of open path Fourier transform infrared spectra. J. Environ. Sci. Health Part A Toxic/Hazard. Subst. Environ. Eng. 2005, 40, 1069–1079. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, S.; Kumar, R.; Devanathan, A.; Acharya, R.; Rai, A.K. Multivariate methods for analysis of environmental reference materials using laser-induced breakdown spectroscopy. Anal. Chem. Res. 2017, 12, 10–16. [Google Scholar] [CrossRef]
- Agelet, L.E.; Hurburgh, C.R. A tutorial on near infrared spectroscopy and its calibration. Crit. Rev. Anal. Chem. 2010, 40, 246–260. [Google Scholar] [CrossRef]
- Qu, H.B.; Ou, D.L.; Cheng, Y.Y. Background correction in near-infrared spectra of plant extracts by orthogonal signal correction. J. Zhejiang Univ. Sci. B 2005, 6, 838–843. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Parameters | Spectra Correction Methods | PCs | Rcal | RMSEC | Rcv | RMSECV | Rpred | RMSEP |
|---|---|---|---|---|---|---|---|---|
| %Inhibition | Original | 7 | 0.90 | 7.79 | 0.87 | 8.96 | 0.83 | 9.45 |
| MSC | 6 | 0.88 | 8.49 | 0.84 | 9.80 | 0.79 | 10.36 | |
| SNV | 7 | 0.87 | 8.73 | 0.83 | 10.13 | 0.81 | 10.03 | |
| AN | 6 | 0.88 | 8.27 | 0.84 | 10.05 | 0.77 | 10.81 | |
| MN | 8 | 0.90 | 7.85 | 0.86 | 9.13 | 0.80 | 10.24 | |
| Dg1 | 8 | 0.91 | 7.35 | 0.83 | 10.16 | 0.81 | 9.91 | |
| Dg2 | 3 | 0.76 | 11.69 | 0.76 | 13.84 | 0.58 | 13.96 | |
| DT | 7 | 0.90 | 7.92 | 0.86 | 9.24 | 0.82 | 9.75 | |
| SGS | 7 | 0.90 | 7.80 | 0.87 | 9.02 | 0.83 | 9.45 | |
| OSC | 1 | 0.90 | 7.63 | 0.90 | 7.79 | 0.84 | 9.16 | |
| Antioxidant activity | Original | 6 | 0.91 | 220.91 | 0.89 | 251.24 | 0.83 | 325.91 |
| MSC | 7 | 0.89 | 250.24 | 0.84 | 304.43 | 0.80 | 350.24 | |
| SNV | 5 | 0.88 | 253.65 | 0.85 | 292.81 | 0.74 | 391.74 | |
| AN | 5 | 0.88 | 259.20 | 0.86 | 278.64 | 0.69 | 423.18 | |
| MN | 7 | 0.90 | 236.95 | 0.86 | 282.36 | 0.76 | 375.58 | |
| Dg1 | 7 | 0.91 | 228.23 | 0.86 | 288.48 | 0.80 | 345.58 | |
| Dg2 | 5 | 0.88 | 254.56 | 0.72 | 381.40 | 0.64 | 452.79 | |
| DT | 6 | 0.88 | 252.26 | 0.85 | 294.65 | 0.78 | 369.78 | |
| SGS | 6 | 0.91 | 221.45 | 0.88 | 253.11 | 0.83 | 325.79 | |
| OSC | 1 | 0.94 | 181.82 | 0.94 | 186.57 | 0.81 | 340.06 | |
| Antioxidant capacity | Original | 7 | 0.92 | 16.73 | 0.88 | 19.64 | 0.86 | 19.78 |
| MSC | 8 | 0.92 | 16.73 | 0.89 | 19.87 | 0.87 | 19.95 | |
| SNV | 7 | 0.91 | 18.13 | 0.86 | 21.92 | 0.85 | 20.51 | |
| AN | 7 | 0.93 | 15.41 | 0.90 | 18.73 | 0.72 | 27.73 | |
| MN | 8 | 0.93 | 14.89 | 0.91 | 17.92 | 0.60 | 31.72 | |
| Dg1 | 9 | 0.94 | 13.57 | 0.89 | 19.47 | 0.83 | 21.83 | |
| Dg2 | 3 | 0.76 | 28.55 | 0.66 | 33.08 | 0.61 | 31.43 | |
| DT | 6 | 0.92 | 16.65 | 0.91 | 18.94 | 0.87 | 19.55 | |
| SGS | 7 | 0.92 | 16.76 | 0.89 | 19.76 | 0.86 | 19.76 | |
| OSC | 1 | 0.94 | 14.42 | 0.93 | 14.80 | 0.82 | 22.64 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kusumiyati, K.; Putri, I.E.; Munawar, A.A.; Suhandy, D. A Data Fusion Model to Merge the Spectra Data of Intact and Powdered Cayenne Pepper for the Fast Inspection of Antioxidant Properties. Sustainability 2022, 14, 201. https://doi.org/10.3390/su14010201
Kusumiyati K, Putri IE, Munawar AA, Suhandy D. A Data Fusion Model to Merge the Spectra Data of Intact and Powdered Cayenne Pepper for the Fast Inspection of Antioxidant Properties. Sustainability. 2022; 14(1):201. https://doi.org/10.3390/su14010201
Chicago/Turabian StyleKusumiyati, Kusumiyati, Ine Elisa Putri, Agus Arip Munawar, and Diding Suhandy. 2022. "A Data Fusion Model to Merge the Spectra Data of Intact and Powdered Cayenne Pepper for the Fast Inspection of Antioxidant Properties" Sustainability 14, no. 1: 201. https://doi.org/10.3390/su14010201
APA StyleKusumiyati, K., Putri, I. E., Munawar, A. A., & Suhandy, D. (2022). A Data Fusion Model to Merge the Spectra Data of Intact and Powdered Cayenne Pepper for the Fast Inspection of Antioxidant Properties. Sustainability, 14(1), 201. https://doi.org/10.3390/su14010201







