Chromium Stress in Plants: Toxicity, Tolerance and Phytoremediation
Abstract
1. Introduction
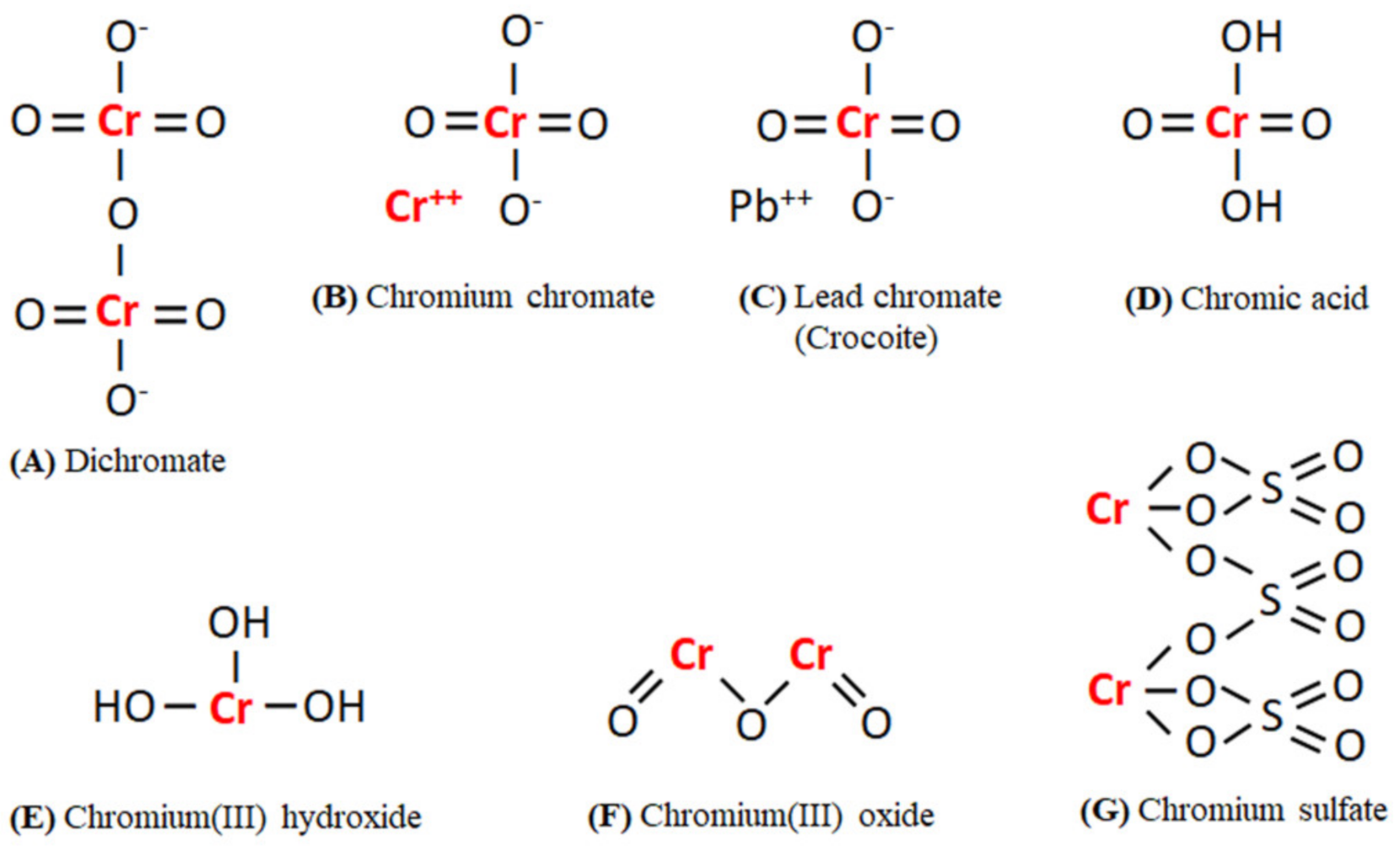
2. Cr Occurrence and Sources
3. The Effects of Cr on Plants
3.1. Effects on Seed Germination
3.2. Effects on Shoot Growth
3.3. Effects on Root Growth
3.4. Effects on Total Leaf Area
3.5. Effects on Grain Yield
3.6. Anatomical Changes
3.7. Physiological Changes
3.8. Effects on Nutrient Balance
3.9. Molecular Changes
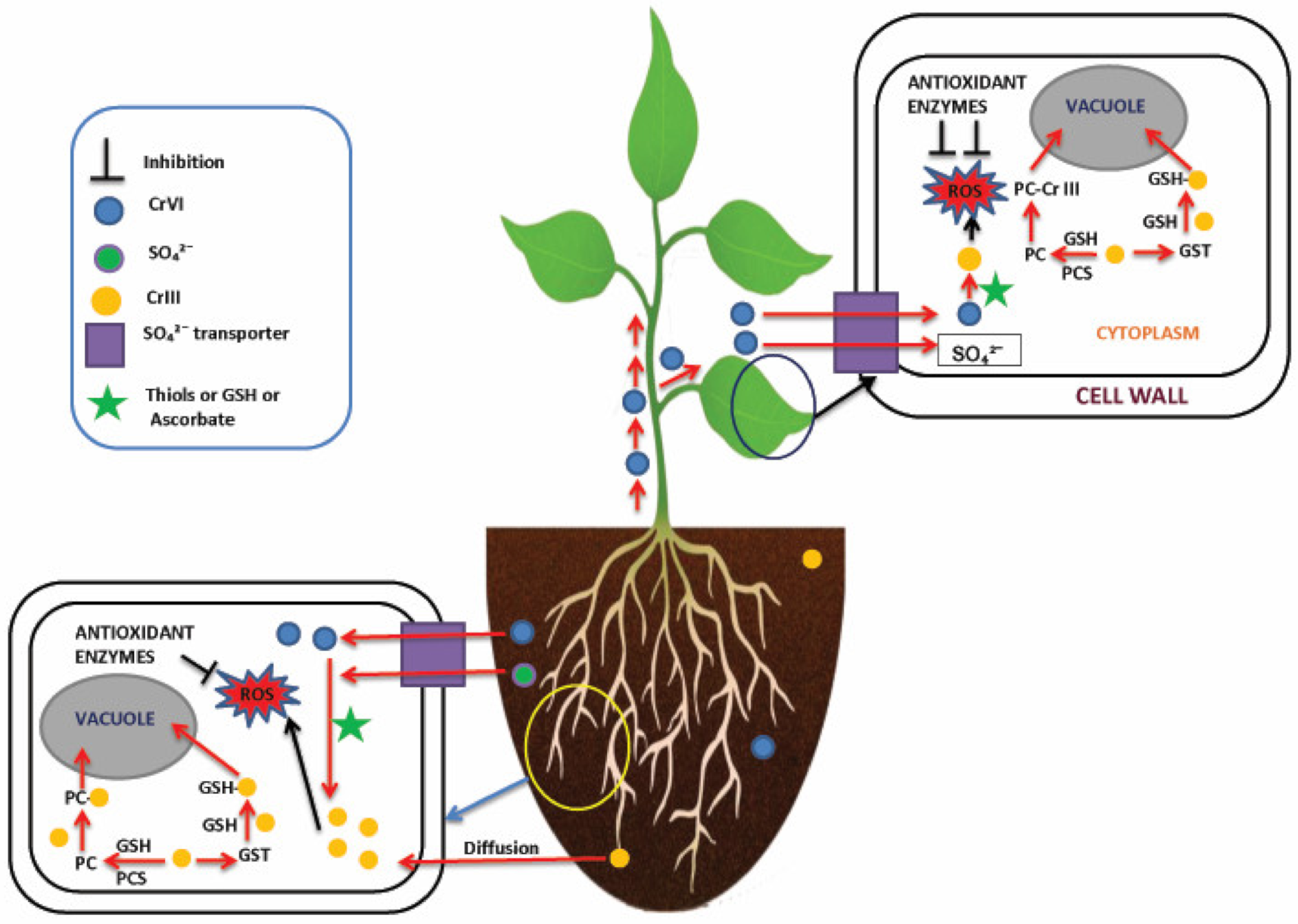
4. Cr Uptake and Translocation in Plants
5. Molecular Approaches of Cr Detoxification in Plants
5.1. Avoidance
5.2. Antioxidant Response
5.3. Compartmentalization and Sequestration
5.4. Reduction of Cr(VI) to Cr(III)
5.5. Detoxification Genes/Proteins in Plants
5.5.1. Phytochelatins
5.5.2. Metallothioneins
5.5.3. Glutathione-S-Transferases
6. Phytoremediation Approach for Cr Detoxification
6.1. Phytoremediation by Hyperaccumulating Plants
6.2. Cr Detoxification Using Anti-Oxidant Machinery and Other Innovative Strategies
6.3. Phytostabilization and Phytoextraction for Cr Tolerance
7. Concluding Remark and Future Prospect
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bhalerao, S.A.; Sharma, A.S. Chromium: As an environmental pollutant. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 732–746. [Google Scholar]
- World Health Organization. Chromium in Drinking-water (No. WHO/HEP/ECH/WSH/2020.3); World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- McNeill, L.; McLean, J.; Edwards, M.; Parks, J. State of the science of hexavalent chromium in drinking water. Water Res. 2012, 44, 5. [Google Scholar]
- Shrivastava, R.; Upreti, R.K.; Seth, P.K.; Chaturvedi, U.C. Effects of chromium on the immune sys-tem. FEMS Immunol. Med. Microbiol. 2002, 34, 1–7. [Google Scholar] [CrossRef]
- Batayneh, A.T. Toxic (aluminum, beryllium, boron, chromium and zinc) in groundwater: Health risk assessment. Int. J. Environ. Sci. Technol. 2012, 9, 153–162. [Google Scholar] [CrossRef]
- Amin, H.; Arain, B.A.; Amin, F.; Surhio, M.A. Phytotoxicity of chromium on germination, growth and biochemical at-tributes of Hibiscus esculentus L. Am. J. Plant Sci. 2013, 4, 41293. [Google Scholar] [CrossRef]
- Shahid, M.; Shamshad, S.; Rafiq, M.; Khalid, S.; Bibi, I.; Niazi, N.K.; Dumat, C.; Rashid, M.I. Chromium speciation, bioavailability, uptake, toxicity and detoxification in soil-plant system: A review. Chemosphere 2017, 178, 513–533. [Google Scholar] [CrossRef]
- Xu, Z.-R.; Cai, M.-L.; Chen, S.-H.; Huang, X.-Y.; Zhao, F.-J.; Wang, P. High-Affinity Sulfate Transporter Sultr1;2 Is a Major Transporter for Cr(VI) Uptake in Plants. Environ. Sci. Technol. 2021, 55, 1576–1584. [Google Scholar] [CrossRef]
- Singh, H.P.; Mahajan, P.; Kaur, S.; Batish, D.R.; Kohli, R.K. Chromium toxicity and tolerance in plants. Environ. Chem. Lett. 2013, 11, 229–254. [Google Scholar] [CrossRef]
- Srivastava, S.; Nigam, R.; Prakash, S.; Srivastava, M.M. Mobilization of Trivalent Chromium in Presence of Organic Acids: A Hydroponic Study of Wheat Plant (Triticum vulgare). Bull. Environ. Contam. Toxicol. 1999, 63, 524–530. [Google Scholar] [CrossRef]
- Hakeem, K.; Sabir, M.; Ozturk, M.; Mermut, A.R. (Eds.) Soil Remediation and Plants: Prospects and Challenges; Academic Press: Cambridge, MA, USA, 2014. [Google Scholar]
- Yan, X.; Wang, J.; Song, H.; Peng, Y.; Zuo, S.; Gao, T.; Duan, X.; Qin, D.; Dong, J. Evaluation of the phytoremediation potential of dominant plant species growing in a chromium salt–producing factory wasteland, China. Environ. Sci. Pollut. Res. 2020, 27, 7657–7671. [Google Scholar] [CrossRef]
- Banach, A.M.; Banach, K.; Stepniewska, Z. Phytoremediation as a promising technology for water and soil purification: Azollacaroliniana Willd as a case study. Acta Agrophys. 2012, 19, 241–252. [Google Scholar]
- Wu, S.; Hu, Y.; Zhang, X.; Sun, Y.; Wu, Z.; Li, T.; Lv, J.; Li, J.; Zhang, J.; Zheng, L.; et al. Chromium detoxification in arbuscular mycorrhizal symbiosis mediated by sulfur uptake and metabolism. Environ. Exp. Bot. 2018, 147, 43–52. [Google Scholar] [CrossRef]
- Sharma, P.; Tripathi, S.; Chaturvedi, P.; Chaurasia, D.; Chandra, R. Newly isolated Bacillus sp. PS-6 assisted phytoreme-diation of heavy metals using Phragmites communis: Potential application in wastewater treatment. Bioresour. Technol. 2021, 320, 124353. [Google Scholar] [CrossRef] [PubMed]
- Shanker, A.; Djanaguiraman, M.; Sudhagar, R.; Chandrashekar, C.; Pathmanabhan, G. Differential antioxidative response of ascorbate glutathione pathway enzymes and metabolites to chromium speciation stress in green gram ((L.) R.Wilczek. cv CO4) roots. Plant Sci. 2004, 166, 1035–1043. [Google Scholar] [CrossRef]
- Dubey, S.; Misra, P.; Dwivedi, S.; Chatterjee, S.; Bag, S.K.; Mantri, S.; Asif, M.H.; Rai, A.; Kumar, S.; Shri, M.; et al. Transcriptomic and metabolomic shifts in rice roots in response to Cr (VI) stress. BMC Genom. 2010, 11, 648. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.-Z.; Lin, Y.-J.; Zhang, Q. Metallothioneins enhance chromium detoxification through scavenging ROS and stimulating metal chelation in Oryza sativa. Chemosphere 2019, 220, 300–313. [Google Scholar] [CrossRef]
- Cassano, A.; Della-Pietra, L.; Drioli, E. Integrated membrane process for the recovery of chromium salts from tannery effluents. Ind. Eng. Chem. Res. 2007, 46, 6825–6830. [Google Scholar] [CrossRef]
- Aravindhan, R.; Madhan, B.; Rao, J.R.; Nair, B.U.; Ramasami, T. Bioaccumulation of Chromium from Tannery Wastewater: An Approach for Chrome Recovery and Reuse. Environ. Sci. Technol. 2004, 38, 300–306. [Google Scholar] [CrossRef]
- Kabata-Pendias, A. Trace Elements in Soils and Plants; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Alloway, B.J. (Ed.) Heavy Metals in Soils: Trace Metals and Metalloids in Soils and Their Bioavailability; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- CEPA. Canadian Soil Quality Guidelines for the Protection of Environmental and Human Health; CEPA: Strasbourg, France, 2007. [Google Scholar]
- Gonzalez, A.R.; Ndung, U.K.; Flegal, A.R. Natural Occurrence of Hexavalent Chromium in the Aromas Red Sands Aquifer, California. Environ. Sci. Technol. 2005, 39, 5505–5511. [Google Scholar] [CrossRef]
- Fantoni, D.; Brozzo, G.; Canepa, M.; Cipolli, F.; Marini, L.; Ottonello, G.; Zuccolini, M.V. Natural hexavalent chromium in groundwaters interacting with ophiolitic rocks. Environ. Earth Sci. 2002, 42, 871–882. [Google Scholar] [CrossRef]
- Robles-Camacho, J.; Armienta, M. Natural chromium contamination of groundwater at León Valley, México. J. Geochem. Explor. 2000, 68, 167–181. [Google Scholar] [CrossRef]
- Cooper, G. Oxidation and toxicity of chromium in ultramafic soils in Zimbabwe. Appl. Geochem. 2002, 17, 981–986. [Google Scholar] [CrossRef]
- Bourotte, C.; Bertolo, R.; Almodovar, M.; Hirata, R. Natural occurrence of hexavalent chromium in a sedimentary aquifer in Urânia, State of São Paulo, Brazil. An. Acad. Bras. Ciências 2009, 81, 227–242. [Google Scholar] [CrossRef]
- Saputro, S.; Yoshimura, K.; Matsuoka, S.; Takehara, K.; Narsito, K.; Aizawa, J.; Tennichi, Y. Speciation of dissolved chromium and the mechanisms controlling its concentration in natural water. Chem. Geol. 2014, 364, 33–41. [Google Scholar] [CrossRef]
- Chandra, P.; Kulshreshtha, K. Chromium Accumulation and Toxicity in Aquatic Vascular Plants. Bot. Rev. 2004, 70, 313–327. [Google Scholar] [CrossRef]
- Shanker, A.K.; Cervantes, C.; Loza-Tavera, H.; Avudainayagam, S. Chromium toxicity in plants. Environ. Int. 2005, 31, 739–753. [Google Scholar] [CrossRef]
- Akinci, I.E.; Akinci, S. Effect of chromium toxicity on germination and early seedling growth in melon (Cucumis melo L.). Afr. J. Biotechnol. 2010, 9, 4589–4594. [Google Scholar]
- Riaz, M.; Yasmeen, T.; Arif, M.S.; Ashraf, M.A.; Hussain, Q.; Shahzad, S.M.; Rizwan, M.; Mehmood, M.W.; Zia, A.; Mian, I.A.; et al. Variations in morphological and physiological traits of wheat regulated by chromium species in long-term tannery effluent irrigated soils. Chemosphere 2019, 222, 891–903. [Google Scholar] [CrossRef]
- Rout, G.R.; Samantaray, S.; Das, P. Effects of chromium and nickel on germination and growth in tolerant and non-tolerant populations of Echinochloacolona (L.) Link. Chemosphere 2000, 40, 855–859. [Google Scholar] [CrossRef]
- Bassi, M.; Corradi, M.G.; Realini, M. Effects of chromium (VI) on two freshwater plants, Lemna minor and Pistia stratiotes. 1. Morphological observations. Cytobiosis 1990, 62, 27–38. [Google Scholar]
- Corradi, M.; Bianchi, A.; Albasini, A. Chromium toxicity in Salvia sclarea—I. Effects of hexavalent chromium on seed germination and seedling development. Environ. Exp. Bot. 1993, 33, 405–413. [Google Scholar] [CrossRef]
- Ding, G.; Jin, Z.; Han, Y.; Sun, P.; Li, G.; Li, W. Mitigation of chromium toxicity in Arabidopsis thaliana by sulfur supple-mentation. Ecotoxicol. Environ. Saf. 2019, 182, 109379. [Google Scholar] [CrossRef]
- Fozia, A.; Muhammad, A.Z.; Muhammad, A.; Zafar, M.K. Effect of chromium on growth attributes in sunflower (Helianthus annuus L.). J. Environ. Sci. 2008, 20, 1475–1480. [Google Scholar] [CrossRef]
- Nematshahi, N.; Lahouti, M.; Ganjeali, A. Accumulation of chromium and its effect on growth of (Allium cepa cv. Hybrid). Eur. J. Exp. Biol. 2012, 2, 969–974. [Google Scholar]
- Shiyab, S. Morphophysiological Effects of Chromium in Sour Orange (Citrus aurantium L.). HortScience 2019, 54, 829–834. [Google Scholar] [CrossRef]
- Tang, J.; Xu, J.; Wu, Y.; Li, Y.; Tang, Q. Effects of high concentration of chromium stress on physiological and bio-chemical characters and accumulation of chromium in tea plant (Camellia sinensis L.). Afr. J. Biotechnol. 2012, 11, 2248–2255. [Google Scholar]
- Kakkalameli, S.B.; Daphedar, A.; Hulakoti, N.; Patil, B.N.; Taranath, T.C. Azollafiliculoides lam as a phytotool for re-mediation of heavy metals from sewage. Int. J. Pharm. 2018, 8, 282–287. [Google Scholar]
- Ghani, A.; Khan, I.; Umer, S.; Ahmed, I.; Mustafa, I.; Mohammad, N. Response of wheat (Triticumaestivum) to exog-enously applied chromium: Effect on growth, chlorophyll and mineral composition. J. Environ. Anal. Toxicol. 2015, 5. [Google Scholar] [CrossRef]
- Sundaramoorthy, P.; Chidambaram, A.; Ganesh, K.S.; Unnikannan, P.; Baskaran, L. Chromium stress in paddy: (i) Nutrient status of paddy under chromium stress; (ii) Phytoremediation of chromium by aquatic and terrestrial weeds. C. R. Biol. 2010, 333, 597–607. [Google Scholar] [CrossRef]
- Rodriguez, E.; Azevedo, R.; Fernandes, P.; Santos, C.A. Cr (VI) induces DNA damage, cell cycle arrest and polyploidization: A flow cytometric and comet assay study in Pisum sativum. Chem. Res. Toxicol. 2011, 24, 1040–1047. [Google Scholar] [CrossRef]
- Chatterjee, J.; Chatterjee, C. Phytotoxicity of cobalt, chromium and copper in cauliflower. Environ. Pollut. 2000, 109, 69–74. [Google Scholar] [CrossRef]
- Vernay, P.; Gauthier-Moussard, C.; Hitmi, A. Interaction of bioaccumulation of heavy metal chromium with water relation, mineral nutrition and photosynthesis in developed leaves of Loliumperenne L. Chemosphere 2007, 68, 1563–1575. [Google Scholar] [CrossRef]
- Radha, J.; Srivastava, S.; Madan, V. Influence of chromium on growth and cell division of sugarcane. Indian J. Plant Physiol. 2000, 5, 228–231. [Google Scholar]
- Poschenrieder, C.; Gunsé, B.; Barceló, J. Chromium-induced inhibition of ethylene evolution in bean (Phaseolus vulgaris) leaves. Physiol. Plant. 1993, 89, 404–408. [Google Scholar] [CrossRef]
- Buendía-González, L.; Orozco-Villafuerte, J.; Cruz-Sosa, F.; Barrera-Díaz, C.; Vernon-Carter, E. Prosopis laevigata a potential chromium (VI) and cadmium (II) hyperaccumulator desert plant. Bioresour. Technol. 2010, 101, 5862–5867. [Google Scholar] [CrossRef] [PubMed]
- Golovatyj, S.; Bogatyreva, E. Effect of levels of chromium content in a soil on its distribution in organs of corn plants. In Soil Research And Use of Fertilizers; BRISSA: Minsk, Belarus, 1999; pp. 197–204. [Google Scholar]
- Biacs, P.A.; Daood, H.G.; Kadar, I. Effect of Mo, Se, Zn, and Cr treatments on the yield, element concentration, and carotenoid content of carrot. J. Agric. Food Chem. 1995, 43, 589–591. [Google Scholar] [CrossRef]
- Ratheesh Chandra, P.; Abdussalam, A.; Nabeesa, S. Distribution of Bio-accumulated Cd and Cr in two Vigna species and the Associated Histological Variations. J. Stress Physiol. Biochem. 2010, 6, 4–12. [Google Scholar]
- Rai, V.; Mehrotra, S. Chromium-induced changes in ultramorphology and secondary metabolites of PhyllanthusamarusSchum&Thonn—An hepatoprotective plant. Environ. Monit. Assess. 2008, 147, 307–315. [Google Scholar] [CrossRef]
- Su, Y.; Han, F.X.; Sridhar, B.M.; Monts, D.L. Phytotoxicity and phytoaccumulation of trivalent and hexavalent chromium in brake fern. Environ. Toxicol. Chem. 2005, 24, 2019–2026. [Google Scholar] [CrossRef] [PubMed]
- Suseela, M.R.; Sinha, S.; Singh, S.; Saxena, R. Accumulation of Chromium and Scanning Electron Microscopic Studies in Scirpuslacustris L. Treated with Metal and Tannery Effluent. Bull. Environ. Contam. Toxicol. 2002, 68, 540–548. [Google Scholar] [CrossRef]
- Bianchi, A.; Corradi, M.G.; Tirillini, B.; Albasini, A. Effects of Hexavalent Chromium on Mentha aquatica L. J. Herbs Spices Med. Plants 1998, 5, 3–12. [Google Scholar] [CrossRef]
- Guilizzoni, P.; Adams, M.S.; MacGaffey, N. The effect of chromium on growth and photosynthesis of a submersed macrophyte, Myriophyllumspicatum. In Ecological Bulletins, Proceedings of the Third Oikos Conference, Copenhagen, Denmark, 30 November–2 December 1982; Publishing House of the Swedish Research Councils: Stockholm, Sweden, 1984; pp. 90–96. [Google Scholar]
- Subrahmanyam, D. Effects of chromium toxicity on leaf photosynthetic characteristics and oxidative changes in wheat (Triticum aestivum L.). Photosynthetica 2008, 46, 339–345. [Google Scholar] [CrossRef]
- Horcsik, Z.T.; Kovacs, L.; Laposi, R.; Meszaros, I.; Lakatos, G.; Garab, G. Effect of chromium on photosystem 2 in the unicellular green alga, Chlorella pyrenoidosa. Photosynthetica 2007, 45, 65–69. [Google Scholar] [CrossRef]
- Mathur, S.; Kalaji, H.M.; Jajoo, A. Investigation of deleterious effects of chromium phytotoxicity and photosynthesis in wheat plant. Photosynthetica 2016, 54, 185–192. [Google Scholar] [CrossRef]
- Hauschild, M. Putrescine (1,4-Diaminobutane) as an Indicator of Pollution-Induced Stress in Higher Plants: Barley and Rape Stressed with Cr(III) or Cr(VI). Ecotoxicol. Environ. Saf. 1993, 26, 228–247. [Google Scholar] [CrossRef]
- Zou, J.H.; Wang, M.; Jiang, W.S.; Liu, D.H. Effects of hexavalent chromium (VI) on root growth and cell division in root tip cells of Amaranthusviridis L. Pak. J. Bot. 2006, 38, 673. [Google Scholar]
- Wettlaufer, S.; Osmeloski, J.; Weinstein, L. Response of polyamines to heavy metal stress in oat seedlings. Environ. Toxicol. Chem. 1991, 10, 1083–1088. [Google Scholar] [CrossRef]
- Gopal, R.; Rizvi, A.H.; Nautiyal, N. Chromium Alters Iron Nutrition and Water Relations of Spinach. J. Plant Nutr. 2009, 32, 1551–1559. [Google Scholar] [CrossRef]
- Redondo-Gómez, S.; Mateos-Naranjo, E.; Vecino-Bueno, I.; Feldman, S.R. Accumulation and tolerance characteristics of chromium in a cordgrass Cr-hyperaccumulator, Spartinaargentinensis. J. Hazard. Mater. 2011, 185, 862–869. [Google Scholar] [CrossRef]
- Vázquez, M.D.; Poschenrieder, C.; Barceló, J. Chromium VI Induced Structural and Ultrastructural Changes in Bush Bean Plants (Phaseolus vulgaris L.). Ann. Bot. 1987, 59, 427–438. [Google Scholar] [CrossRef]
- Han, F.X.; Su, Y.; Sridhar, B.B.M.; Monts, D.L. Distribution, transformation and bioavailability of trivalent and hexavalent chromium in contaminated soil. Plant Soil 2004, 265, 243–252. [Google Scholar] [CrossRef]
- Gardea-Torresdey, J.; Peralta-Videa, J.; Montes, M.; de la Rosa, G.; Corral-Diaz, B. Bioaccumulation of cadmium, chromium and copper by Convolvulus arvensis L.: Impact on plant growth and uptake of nutritional elements. Bioresour. Technol. 2004, 92, 229–235. [Google Scholar] [CrossRef] [PubMed]
- Davies, F.T.; Puryear, J.D.; Newton, R.J.; Egilla, J.N.; SaraivaGrossi, J.A. Mycorrhizal fungi increase chromium uptake by sunflower plants: Influence on tissue mineral concentration, growth, and gas exchange. J. Plant Nutr. 2002, 25, 2389–2407. [Google Scholar] [CrossRef]
- Ahmad, M.; Wahid, A.; Ahmad, S.S.; Butt, Z.A.; Tariq, M. Ecophysiological responses of rice (Oryza sativa L.) to hexavalent chromium. Pak. J. Bot. 2011, 43, 2853–2859. [Google Scholar]
- Zeng, F.; Ali, S.; Qiu, B.; Wu, F.; Zhang, G. Effects of chromium stress on the subcellular distribution and chemical form of Ca, Mg, Fe, and Zn in two rice genotypes. J. Plant Nutr. Soil Sci. 2010, 173, 135–148. [Google Scholar] [CrossRef]
- Dube, B.; Tewari, K.; Chatterjee, J. Excess chromium alters uptake and translocation of certain nutrients in citrullus. Chemosphere 2003, 53, 1147–1153. [Google Scholar] [CrossRef]
- Mallick, S.; Sinam, G.; Mishra, R.K.; Sinha, S. Interactive effects of Cr and Fe treatments on plants growth, nutrition and oxidative status in Zea mays L. Ecotoxicol. Environ. Saf. 2010, 73, 987–995. [Google Scholar] [CrossRef]
- Fan, W.-J.; Feng, Y.-X.; Li, Y.-H.; Lin, Y.-J.; Yu, X.-Z. Unraveling genes promoting ROS metabolism in subcellular organelles of Oryza sativa in response to trivalent and hexavalent chromium. Sci. Total. Environ. 2020, 744, 140951. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Xu, L.; Wang, Y.; Shen, H.; Zhu, X.; Zhang, K.; Chen, Y.; Yu, R.; Limera, C.; Liu, L. Transcriptome-wide analysis of chromium-stress responsive microRNAs to explore miRNA-mediated regulatory networks in radish (Raphanus sativus L.). Sci. Rep. 2015, 5, 1–17. [Google Scholar] [CrossRef]
- Dubey, S.; Saxena, S.; Chauhan, A.S.; Mathur, P.; Rani, V.; Chakrabaroty, D. Identification and expression analysis of conserved microRNAs during short and prolonged chromium stress in rice (Oryza sativa). Environ. Sci. Pollut. Res. 2019, 27, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Babula, P.; Adam, V.; Opatrilova, R.; Zehnalek, J.; Havel, L.; Kizek, R. Uncommon heavy metals, metalloids and their plant toxicity: A review. Environ. Chem. Lett. 2008, 6, 189–213. [Google Scholar] [CrossRef]
- Srivastava, S.; Prakash, S.; Srivastava, M. Chromium mobilization and plant availability—The impact of organic complexing ligands. Plant Soil. 1999, 212, 201–206. [Google Scholar] [CrossRef]
- de Oliveira, L.M.; Gress, J.; De, J.; Rathinasabapathi, B.; Marchi, G.; Chen, Y.; Ma, L.Q. Sulfate and chromate increased each other’s uptake and translocation in As-hyperaccumulat or Pterisvittata. Chemosphere 2016, 147, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Zayed, A.; Lytle, C.M.; Qian, J.-H.; Terry, N. Chromium accumulation, translocation and chemical speciation in vegetable crops. Planta 1998, 206, 293–299. [Google Scholar] [CrossRef]
- Shanker, A.K.; Djanaguiraman, M.; Venkateswarlu, B. Chromium interactions in plants: Current status and future strategies. Metallomics 2009, 1, 375–383. [Google Scholar] [CrossRef]
- Murphy, T.F.; Kirkham, C.; Johnson, A.; Brauer, A.L.; Koszelak-Rosenblum, M.; Malkowski, M.G. Sulfate-binding protein, CysP, is a candidate vaccine antigen of Moraxella catarrhalis. Vaccine 2016, 34, 3855–3861. [Google Scholar] [CrossRef]
- Pootakham, W.; Gonzalez-Ballester, D.; Grossman, A.R. Identification and Regulation of Plasma Membrane Sulfate Transporters in Chlamydomonas. Plant Physiol. 2010, 153, 1653–1668. [Google Scholar] [CrossRef]
- Huffman, E.W.; Allaway, W.H. Chromium in plants. Distribution in tissues, organelles, and extracts and availability of bean leaf chromium to animals. J. Agric. Food Chem. 1973, 21, 982–986. [Google Scholar] [CrossRef]
- Caldelas, C.; Bort, J.; Febrero, A. Ultrastructure and subcellular distribution of Cr in Iris pseudacorus L. using TEM and X-ray microanalysis. Cell Biol. Toxicol. 2011, 28, 57–68. [Google Scholar] [CrossRef]
- López-Luna, J.; González-Chávez, M.; Esparza-García, F.; Rodríguez-Vázquez, R. Toxicity assessment of soil amended with tannery sludge, trivalent chromium and hexavalent chromium, using wheat, oat and sorghum plants. J. Hazard. Mater. 2009, 163, 829–834. [Google Scholar] [CrossRef]
- Cary, E.E.; Allaway, W.H.; Olson, O.E. Control of chromium concentrations in food plants. 1. Absorption and translocation of chromium by plants. J. Agric. Food Chem. 1977, 25, 300–304. [Google Scholar] [CrossRef]
- Marieschi, M.; Gorbi, G.; Zanni, C.; Sardella, A.; Torelli, A. Increase of chromium tolerance in Scenedesmusacutus after sulfur starvation: Chromium uptake and compartmentalization in two strains with different sensitivities to Cr(VI). Aquat. Toxicol. 2015, 167, 124–133. [Google Scholar] [CrossRef]
- Mangabeira, P.A.; Ferreira, A.S.; de Almeida, A.A.F.; Fernandes, V.F.; Lucena, E.; Souza, V.L.; dos Santos-Júnior, A.J.; Oliveira, A.H.; Grenier-Loustalot, M.F.; Barbier, F.; et al. Compartmentalization and ultrastructural alterations induced by chromium in aquatic macrophytes. Biometals 2011, 24, 1017–1026. [Google Scholar] [CrossRef]
- Miretzky, P.; Cirelli, A.F. Cr(VI) and Cr(III) removal from aqueous solution by raw and modified lignocellulosic materials: A review. J. Hazard. Mater. 2010, 180, 1–19. [Google Scholar] [CrossRef]
- Zeng, F.; Wu, X.; Qiu, B.; Wu, F.; Jiang, L.; Zhang, G. Physiological and proteomic alterations in rice (Oryza sativa L.) seedlings under hexavalent chromium stress. Planta 2014, 240, 291–308. [Google Scholar] [CrossRef]
- Costa, R.C.; Moura, F.C.; Oliveira, P.E.; Magalhães, F.; Ardisson, J.D.; Lago, R.M. Controlled reduction of red mud waste to produce active systems for environmental applications: Heterogeneous Fenton reaction and reduction of Cr(VI). Chemosphere 2010, 78, 1116–1120. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.K.; Dhote, M.; Kumar, P.; Sharma, J.; Chakrabarti, T.; Juwarkar, A.A. Differential antioxidative enzyme responses of Jatrophacurcas L. to chromium stress. J. Hazard. Mater. 2010, 180, 609–615. [Google Scholar] [CrossRef]
- Sinha, V.; Pakshirajan, K.; Chaturvedi, R. Chromium tolerance, bioaccumulation and localization in plants: An over-view. J. Environ. Manag. 2018, 206, 715–730. [Google Scholar] [CrossRef]
- Sharmin, S.A.; Alam, I.; Kim, K.H.; Kim, Y.G.; Kim, P.J.; Bahk, J.D.; Lee, B.H. Chromium-induced physiological and proteomic alterations in roots of Miscanthus sinensis. Plant Sci. 2012, 187, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Duan, C.-Q.; Zhang, X.-H.; Zhu, Y.-N.; Hu, C. Subcellular distribution of chromium in accumulating plant Leersiahexandra Swartz. Plant Soil 2009, 322, 187–195. [Google Scholar] [CrossRef]
- Lahouti, M.; Jamshidi, S.; Ejtehadi, H.; Rowshani, M.; Mahmoodzad, H. X-Ray Microanalysis and Ultrastructural Localization of Chromium in Raphanus sativus L. Int. J. Bot. 2008, 4, 340–343. [Google Scholar] [CrossRef]
- Huang, W.; Jiao, J.; Ru, M.; Bai, Z.; Yuan, H.; Bao, Z.; Liang, Z. Localization and Speciation of Chromium in CoptischinensisFranch. using Synchrotron Radiation X-ray Technology and Laser Ablation ICP-MS. Sci. Rep. 2018, 8, 8603. [Google Scholar] [CrossRef] [PubMed]
- Whitacre, D.M. Reviews of Environmental Contamination and Toxicology; Metzler, J.B., Ed.; Springer: New York, NY, USA, 2010; Volume 202. [Google Scholar]
- Soni, S.K.; Singh, R.; Awasthi, A.; Singh, M.; Kalra, A. In vitro Cr (VI) reduction by cell-free extracts of chro-mate-reducing bacteria isolated from tannery effluent irrigated soil. Environ. Sci. Pollut. 2013, 20, 1661–1674. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; McGrouther, K.; Chen, N.; Wu, W.; Wang, H. Subcellular Distribution of Metals within Brassica chinensis L. in Response to Elevated Lead and Chromium Stress. J. Agric. Food Chem. 2013, 61, 4715–4722. [Google Scholar] [CrossRef]
- Huda, A.N.; Haque, M.A.; Zaman, R.; Swaraz, A.M.; Kabir, A.H. Silicon ameliorates chromium toxicity through phytochelatin-mediated vacuolar sequestration in the roots of Oryza sativa (L.). Int. J. Phytoremediat. 2017, 19, 246–253. [Google Scholar] [CrossRef]
- Shanker, A.K.; Djanaguiraman, M.; Sudhagar, R.; Jayaram, K.; Pathmanabhan, G. Expression of metallothionein 3-like protein mRNA in sorghum cultivars under chromium (VI) stress. Curr. Sci. 2004, 86, 901–902. [Google Scholar]
- Tiwari, M.; Srivastava, S.; Singh, P.C.; Mishra, A.K.; Chakrabarty, D. Functional characterization of tau class gluta-thione-S-transferase in rice to provide tolerance against sheath blight disease. 3 Biotech 2020, 10, 1–7. [Google Scholar] [CrossRef]
- Kim, S.I.; Andaya, V.C.; Tai, T.H. Cold sensitivity in rice (Oryza sativa L.) is strongly correlated with a naturally oc-curring I99V mutation in the multifunctional glutathione transferase isoenzyme GSTZ2. Biochemical 2011, 435, 373–380. [Google Scholar] [CrossRef]
- Foyer, C.H.; Noctor, G. Oxidant and antioxidant signalling in plants: A re-evaluation of the concept of oxidative stress in a physiological context. Plant Cell Environ. 2005, 28, 1056–1071. [Google Scholar] [CrossRef]
- Tripathi, A.; Indoliya, Y.; Tiwari, M.; Tiwari, P.; Srivastava, D.; Verma, P.K.; Verma, S.; Gautam, N.; Chakrabarty, D. Transformed yeast (Schizosaccharomycespombe) overexpressing rice Tau class glutathione S-transferase (OsGSTU30 and OsGSTU41) shows enhanced resistance to hexavalent chromium. Metallomics 2014, 6, 1549–1557. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, D.; Verma, G.; Chauhan, A.S.; Pande, V.; Chakrabarty, D. Rice (Oryza sativa L.) tau class glutathione S-transferase (OsGSTU30) overexpression in Arabidopsis thaliana modulates a regulatory network leading to heavy metal and drought stress tolerance. Metalomics 2019, 11, 375–389. [Google Scholar] [CrossRef] [PubMed]
- Cervantes, C.; Campos-García, J.; Devars, S.; Gutiérrez-Corona, F.; Loza-Tavera, H.; Torres-Guzmán, J.C.; More-no-Sánchez, R. Interactions of chromium with microorganisms and plants. FEMS Microbiol. Rev. 2001, 25, 335–347. [Google Scholar] [CrossRef]
- Shahid, M.; Austruy, A.; Echevarria, G.; Arshad, M.; Sanaullah, M.; Aslam, M.; Nadeem, M.; Nasim, W.; Dumat, C. EDTA-Enhanced Phytoremediation of Heavy Metals: A Review. Soil Sediment Contam. Int. J. 2013, 23, 389–416. [Google Scholar] [CrossRef]
- Meng, F.; Gao, Y.; Feng, Q. Discovery and mechanism study of a novel chromium-accumulating plant, Lonicera japonica Thunb. Environ. Sci. Pollut. Res. 2019, 26, 13812–13817. [Google Scholar] [CrossRef] [PubMed]
- Kyzioł-Komosińska, J.; Augustynowicz, J.; Lasek, W.; Czupioł, J.; Ociński, D. Callitrichecophocarpa biomass as a potential low-cost biosorbent for trivalent chromium. J. Environ. Manag. 2018, 214, 295–304. [Google Scholar] [CrossRef]
- Eze, C.N.; Odoh, C.K.; Eze, E.A.; Orjiakor, P.I.; Enemuor, S.C.; Okobo, U.J. Chromium (III) and its effects on soil mi-crobial activities and phytoremediation potentials of Arachis hypogea and Vigna unguiculata. Afr. J. Biotechnol. 2018, 17, 1207–1214. [Google Scholar]
- Nayak, A.K.; Panda, S.S.; Basu, A.; Dhal, N.K. Enhancement of toxic Cr (VI), Fe, and other heavy metals phytoreme-diation by the synergistic combination of native Bacillus cereus strain and Vetiveriazizanioides L. Int. J. Phytoremediat. 2018, 20, 682–691. [Google Scholar] [CrossRef] [PubMed]
- Men, C.K.; Ghazi, R.M. Phytoremediation of chromium (VI) using Colocasia esculenta in laboratory scale constructed wetlands. J. Trop. Resour. Sustain. Sci. 2018, 6, 45–49. [Google Scholar]
- Ullah, R.; Hadi, F.; Ahmad, S.; Jan, A.U.; Rongliang, Q. Phytoremediation of Lead and Chromium Contaminated Soil Improves with the Endogenous Phenolics and Proline Production in Parthenium, Cannabis, Euphorbia, and Rumex Species. Water Air Soil Pollut. 2019, 230, 40. [Google Scholar] [CrossRef]
- Sajad, M.A.; Khan, M.S.; Bahadur, S.; Naeem, A.; Ali, H.; Batool, F.; Shuaib, M.; Batool, S. Evaluation of chromium phytoremediation potential of some plant species of Dir Lower, Khyber Pakhtunkhwa, Pakistan. Acta Ecol. Sin. 2020, 40, 158–165. [Google Scholar] [CrossRef]
- Singh, A.; Malaviya, P. Chromium phytoaccumulation and its impact on growth and photosynthetic pigments of Spirodelapolyrrhiza (L.) Schleid. on exposure to tannery effluent. Environ. Sustain. 2019, 2, 157–166. [Google Scholar] [CrossRef]
- Taufikurahman, T.; Pradisa, M.A.S.; Amalia, S.G.; Hutahaean, G.E.M. Phytoremediation of chromium (Cr) using Typha angustifolia L., Canna indica L. and Hydrocotyleumbellata L. in surface flow system of constructed wetland. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Ivano-Frankivsk, Ukraine, 21–22 October 2020; IOP Publishing: Bristol, UK, 2019; Volume 308, p. 012020. [Google Scholar]
- Rajendran, M.; An, W.-H.; Li, W.-C.; Perumal, V.; Wu, C.; Sahi, S.V.; Sarkar, S.K. Chromium detoxification mechanism induced growth and antioxidant responses in vetiver (Chrysopogonzizanioides(L.) Roberty). J. Cent. South. Univ. 2019, 26, 489–500. [Google Scholar] [CrossRef]
- Levizou, E.; Zanni, A.A.; Antoniadis, V. Varying concentrations of soil chromium (VI) for the exploration of tolerance thresholds and phytoremediation potential of the oregano (Origanum vulgare). Environ. Sci. Pollut. Res. 2018, 26, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Ram, B.K.; Han, Y.; Yang, G.; Ling, Q.; Dong, F. Effect of Hexavalent Chromium [Cr (VI)] on Phytoremediation Potential and Biochemical Response of Hybrid Napier Grass with and without EDTA Application. Plants 2019, 8, 515. [Google Scholar] [CrossRef]
- Afonso, T.F.; Demarco, C.F.; Pieniz, S.; Camargo, F.A.O.; Quadro, M.S.; Andreazza, R. Potential of SolanumviarumDunal in use for phytoremediation of heavy metals to mining areas, southern Brazil. Environ. Sci. Pollut. Res. 2019, 26, 24132–24142. [Google Scholar] [CrossRef]
- Patra, D.K.; Pradhan, C.; Kumar, J.; Patra, H.K. Assessment of chromium phytotoxicity, phytoremediation and tolerance potential of Sesbaniasesban and Brachiariamutica grown on chromite mine overburden dumps and garden soil. Chemosphere 2020, 252, 126553. [Google Scholar] [CrossRef] [PubMed]
- Cano-Ruiz, J.; Galea, M.R.; Amorós, M.; Alonso, J.; Mauri, P.; Lobo, M. Assessing Arundodonax L. in vitro-tolerance for phytoremediation purposes. Chemosphere 2020, 252, 126576. [Google Scholar] [CrossRef]
- Mohanty, M.; Patra, H.K. Phytoassessment of in situ weed diversity for their chromium distribution pattern and ac-cumulation indices of abundant weeds at South Kaliapani chromite mining area with their phytoremediation prospective. Ecotoxicol. Environ. Saf. 2020, 194, 110399. [Google Scholar] [CrossRef]
- Augustynowicz, J.; Sitek, E.; Bryniarski, T.; Baran, A.; Ostachowicz, B.; Urbańska-Stopa, M.; Szklarczyk, M. The use of Callitrichecophocarpa Sendtn. for the reclamation of Cr-contaminated freshwater habitat: Benefits and limitations. Environ. Sci. Pollut. Res. 2020, 27, 25510–25522. [Google Scholar] [CrossRef] [PubMed]
- Tabinda, A.B.; Irfan, R.; Yasar, A.; Iqbal, A.; Mahmood, A. Phytoremediation potential of Pistiastratiotes and Eichhornia crassipes to remove chromium and copper. Environ. Technol. 2018, 41, 1514–1519. [Google Scholar] [CrossRef]
- Dökmeci, A.H.; Adiloğlu, S. The Phytoremediation of Chromium from Soil Using Cirsium Vulgare and the Health Effects. Biosci. Biotechnol. Res. Asia 2020, 17, 535–541. [Google Scholar] [CrossRef]
- Haokip, N.; Gupta, A. Phytoremediation of chromium and manganese by Ipomoea aquaticaForssk. from aqueous medium containing chromium-manganese mixtures in microcosms and mesocosms. Water Environ. J. 2020. [Google Scholar] [CrossRef]
- Chen, J.-C.; Wang, K.-S.; Chen, H.; Lu, C.-Y.; Huang, L.-C.; Li, H.-C.; Peng, T.-H.; Chang, S.-H. Phytoremediation of Cr(III) by Ipomoneaaquatica (water spinach) from water in the presence of EDTA and chloride: Effects of Cr speciation. Bioresour. Technol. 2010, 101, 3033–3039. [Google Scholar] [CrossRef]
- Paisio, C.E.; Fernandez, M.; González, P.S.; Talano, M.A.; Medina, M.I.; Agostini, E. Simultaneous phytoremediation of chromium and phenol by Lemnaminuta Kunth: A promising biotechnological tool. Int. J. Environ. Sci. Technol. 2018, 15, 37–48. [Google Scholar] [CrossRef]
- Wang, C.; Tan, H.; Li, H.; Xie, Y.; Liu, H.; Xu, F.; Xu, H. Mechanism study of Chromium influenced soil remediated by an uptake-detoxification system using hyperaccumulator, resistant microbe consortium, and nano iron complex. Environ. Pollut. 2020, 257, 113558. [Google Scholar] [CrossRef] [PubMed]
- Salman, S.M.; Ali, A.; Khan, B.; Iqbal, M.; Alamzeb, M. Thermodynamic and kinetic insights into plant-mediated detoxification of lead, cadmium, and chromium from aqueous solutions by chemically modified Salvia moorcroftiana leaves. Environ. Sci. Pollut. Res. 2019, 26, 14339–14349. [Google Scholar] [CrossRef]
- Mondal, N.K.; Nayek, P. Hexavalent chromium accumulation kinetics and physiological responses exhibited by Eichhornia sp. and Pistia sp. Int. J. Environ. Sci. Technol. 2020, 17, 1397–1410. [Google Scholar] [CrossRef]
- Adejumo, S.A.; Tiwari, S.; Thul, S.; Sarangi, B.K. Evaluation of lead and chromium tolerance and accumulation level in Gomphrenacelosoides: A novel metal accumulator from lead acid battery waste contaminated site in Nigeria. Int. J. Phytoremediat. 2019, 21, 1341–1355. [Google Scholar] [CrossRef]
- Usman, K.; al Jabri, H.; Abu-Dieyeh, M.H.; Alsafran, M.H.S.A. Comparative Assessment of Toxic Metals Bioaccumulation and the Mechanisms of Chromium (Cr) Tolerance and Uptake in Calotropisprocera. Front. Plant Sci. 2020, 11, 883. [Google Scholar] [CrossRef]
- Tauqeer, H.M.; Hussain, S.; Abbas, F.; Iqbal, M. The potential of an energy crop “Conocarpus erectus” for lead phytoex-traction and phytostabilization of chromium, nickel, and cadmium: An excellent option for the management of multi-metal contaminated soils. Ecotoxicol. Environ. Saf. 2019, 173, 273–284. [Google Scholar] [CrossRef]
- Patra, D.K.; Grahacharya, A.; Pradhan, C.; Patra, H.K. Phytoremediation potential of coffee pod (Cassia tora): An in situ approach for attenuation of chromium from overburden soil of Sukinda Chromite Mine, India. Environ. Prog. Sustain. Energy 2021, 40, 13510. [Google Scholar] [CrossRef]
- Śliwa-Cebula, M.; Kaszycki, P.; Kaczmarczyk, A.; Nosek, M.; Lis-Krzyścin, A.; Miszalski, Z. The Common Ice Plant (Mesembryanthemum crystallinum L.)–Phytoremediation Potential for Cadmium and Chromate-Contaminated Soils. Plants 2020, 9, 1230. [Google Scholar] [CrossRef] [PubMed]
- Sallah-Ud-Din, R.; Farid, M.; Saeed, R.; Ali, S.; Rizwan, M.; Tauqeer, H.M.; Bukhari, S.A.H. Citric acid enhanced the antioxidant defense system and chromium uptake by Lemna minor L. grown in hydroponics under Cr stress. Environ. Sci. Pollut. Res. 2017, 24, 17669–17678. [Google Scholar] [CrossRef]
- Mongkhonsin, B.; Nakbanpote, W.; Nakai, I.; Hokura, A.; Jearanaikoon, N. Distribution and speciation of chromium ac-cumulated in Gynurapseudochina (L.) DC. Environ. Exp. Bot. 2011, 74, 56–64. [Google Scholar] [CrossRef]
- Ranieri, E.; Fratino, U.; Petruzzelli, D.; Borges, A.C. A comparison between Phragmites australis and Helianthus annuusin chromium phytoextraction. Water Air Soil Pollut. 2013, 224, 1–9. [Google Scholar] [CrossRef]
- Augustynowicz, J.; Wrobel, P.; Płachno, B.J.; Tylko, G.; Gajewski, Z.; Wegrzynek, D. Chromium distribution in shoots of macrophyte Callitrichecophocarpa Sendtn. Planta 2014, 239, 1233–1242. [Google Scholar] [CrossRef] [PubMed]
- Sinha, V.; Pakshirajan, K.; Chaturvedi, R. Chromium(VI) Accumulation and Tolerance by Tradescantia pallida: Biochemical and Antioxidant Study. Appl. Biochem. Biotechnol. 2014, 173, 2297–2306. [Google Scholar] [CrossRef]
- Christou, A.; Georgiadou, E.C.; Zissimos, A.M.; Christoforou, I.C.; Christofi, C.; Neocleous, D.; Dalias, P.; Torrado, S.O.; Argyraki, A.; Fotopoulos, V. Hexavalent chromium leads to differential hormetic or damaging effects in alfalfa (Medicago sativa L.) plants in a concentration-dependent manner by regulating nitro-oxidative and proline metabolism. Environ. Pollut. 2020, 267, 115379. [Google Scholar] [CrossRef]
- Zewge, F.; Woldemichael, D.; Leta, S. Potential of water hyacinth (Eichhornia crassipes (Mart.) Solms) for the removal of chromium from tannery effluent in constructed pond system. SINET Ethiop. J. Sci. 2011, 34, 49–62. [Google Scholar]
- Fibbi, D.; Doumett, S.; Lepri, L.; Checchini, L.; Gonnelli, C.; Coppini, E.; del Bubba, M. Distribution and mass balance of hexavalent and trivalent chromium in a subsurface, horizontal flow (SF-h) constructed wetland operating as post-treatment of textile wastewater for water reuse. J. Hazard. Mater. 2012, 199, 209–216. [Google Scholar] [CrossRef]
- Kalve, S.; Sarangi, B.K.; Pandey, R.A.; Chakrabarti, T. Arsenic and chromium hyperaccumulation by an ecotype of Pterisvittata–Prospective for phytoextraction from contaminated water and soil. Curr. Sci. 2011, 100, 888–894. [Google Scholar]
- Barbosa, R.; de Almeida, A.; Mielke, M.; Loguercio, L.; Mangabeira, P.; Gomes, F. A physiological analysis of Genipaamericana L.: A potential phytoremediator tree for chromium polluted watersheds. Environ. Exp. Bot. 2007, 61, 264–271. [Google Scholar] [CrossRef]
- Prado, C.; Pagano, E.; Prado, F.; Rosa, M. Detoxification of Cr (VI) in Salvinia minima is related to seasonal-induced changes of thiols, phenolics and antioxidative enzymes. J. Hazard. Mater. 2012, 239, 355–361. [Google Scholar] [CrossRef] [PubMed]
| Family | Plant | Habitat | Tolerance Mechanism | References |
|---|---|---|---|---|
| Aizoaceae | Mesembryanthemum crystallinum L. | Large, mat-forming annual with sprawling stems | Phyto-extraction | [141] |
| Amaranthaceae | Gomphrena celosoides Mart. | Perennial herb | Increased proline and antioxidant enzyme activities | [137] |
| Amaryllidaceae | Allium griffithianum Boiss. | Perennial herb | Hyper-accumulation | [118] |
| Apocynaceae | Calotropis procera (Aiton) W.T. Aiton | Large shrub or small tree | Increased activities of superoxide dismutase (SOD), catalase (CAT), and glutathione reductase (GR) | [138] |
| Araceae | Colocasia esculenta (L.) Schott | Fast growing, herbaceous | High accumulation of Cr(VI) | [116] |
| Lemna minor L. | Free floating aquatic plants | Increased anti-oxidant activity, Phyto-extraction | [142] | |
| Lemna minuta Kunth | Small aquatic floating plant | Increased anti-oxidant activity | [133] | |
| Pistia stratiotes L. | Aquatic plant | Anti-oxidant activity and accumulation | [136] | |
| Spirodela polyrrhiza (L.) Schleid | Aquatic weed | Hyper-accumulation | [119] | |
| Araliaceae | Hydrocotyle umbellata L. | Creeping, aquatic herb | Hyper-accumulation | [120] |
| Asteraceae | Cirsium vulgare (Savi) Ten. | Annual or biennial, herbaceous plant | Hyper-accumulation | [130] |
| Dicoma niccolifera Wild | Terrestrial | Hyper-accumulation | [13] | |
| Gynura pseudochina (L.) DC. | Cr VI reduction | [143] | ||
| Helianthus annuus L. | Annual forb | Hyper-accumulation | [144] | |
| Parthenium hysterophorus L. | Annual, erect, herbaceous | Hyper-accumulation | [117] | |
| Vernonia cinerea (L.) Less. | Perennial herb | [127] | ||
| Brassicaceae | Brassicanapus L. | Annual or biennialherb | Gentle Remediation Options (GROs) | [139] |
| Callitraceae | Callitriche cophocarpa Sendtn. | Aquatic macrophyte | Cr VI reduction | [145] |
| Cannabaceae | Cannabis sativa L. | Annual, herbaceous, flowering | Hyper-accumulation | [117,118] |
| Cannaceae | Canna indica L. | Long-lived, perennial herb | Hyper-accumulation | [120] |
| Commeliniaceae | Tradescantia pallida (Rose) D.R. Hunt | Succulent perennial herb | Increased anti-oxidant activity | [146] |
| Convolvulaceae | Ipomoea aquatica Forssk. | Semi-aquatic, tropical plant | Hyper-accumulation | [131] |
| Euphorbiaceae | Euphorbia helioscopia L. | Desert, herbaceous spurge | Hyper-accumulation | [117] |
| Rumex dentatus L. | ||||
| Fabaceae | Arachis hypogea L. | Annual herb | Hyper-accumulation | [114] |
| Cassia tora L. | Annual under shrub | [140] | ||
| Medicago sativa L. | Perennial flowering plant | High proline and GST accumulation | [147] | |
| Medicago truncatula Gaertn. | Small annual legume | Regulating the sulphur transport and metabolism | [14] | |
| Sesbaniasesban (L.) Merr. | Fast-growing, perennial legume tree | Phyto-stabilizer | [125] | |
| Vigna unguiculata (L.) Walp. | Annual, herbaceous legume | Hyper-accumulation | [114] | |
| Lamiaceae | Origanum vulgare L. | Mediterranean, perennial herb | Hyper-accumulation | [122] |
| Salvia moorcroftiana Wall.ex Benth. | White-woolly perennial herb | Biosorptive detoxification | [135] | |
| Plantaginaceae | Callitriche cophocarpa Sendtn. | Water-submerged, macrophyte | Hyper-accumulation | [128] |
| Poaceae | Arundo donax L. | Tall perennial cane | Hyper-accumulation | [126] |
| Brachiaria mutica (Forssk.) Stapf | Evergreen, perennial grass | Phyto-stabilizer | [125] | |
| Chrysopogon zizanioides (L.) Roberty | Perennial, bunch-grass | Hyper-accumulation | [121] | |
| Diectomis fastigiata (Sw.) P. Beauv. | Tropical grass | [127] | ||
| Triploid hybrid Napier grass (diploid Pennisetum americanus L. X tetraploid elephant grass Pennisetum purpureum Schumach) | Waste lands, roadside, tropical grass | [123] | ||
| Leersia hexandra Sw. | Aquatic perennial grass | Iron-biochar nano-complex & hyperaccumulator | [134] | |
| Miscanthus sinensis Andersson (1855) | Herbaceous perennial plant | Hyper-accumulation | [97] | |
| Oryza sativa L. | Flooded, arable land | Hyper-accumulation | [18] | |
| Phragmites australis (Cav.)Trin. ex Steud. | Perennial grass | Cr III precipitation | [148] | |
| Cr VI reduction | [144] | |||
| Phragmites communis ( Trin.) | Wetland grass | Producing indole acetic acid, siderophores etc. | [15] | |
| Spartina argentinensis (Trin.) Merr. | Perennial grass | Hyper-accumulation | [66] | |
| Vetiveria zizanoides (L.) Roberty | [115] | |||
| Pontederiaceae | Eichhornia crassipes Mart. | Aquatic, flowering plant | Hyper-accumulation | [129] |
| Aquatic plant | Anti-oxidant activity and accumulation | [136] | ||
| Free floating, perennial aquatic plant | Increased anti-oxidant activity | [149] | ||
| Pteridaceae | Pteris vittata L. | Fern species | Hyper-accumulation | [150] |
| Rubiaceae | Genipa americana L. | Wood plant | Hyper-accumulation | [151] |
| Salviniaceae | Salvinia minima Baker 1886 | Aquatic macrophyte | Increased anti-oxidant activity | [152] |
| Solanaceae | Solanum viarum Dunal | Perennial shrub | Hyper-accumulation | [124] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Srivastava, D.; Tiwari, M.; Dutta, P.; Singh, P.; Chawda, K.; Kumari, M.; Chakrabarty, D. Chromium Stress in Plants: Toxicity, Tolerance and Phytoremediation. Sustainability 2021, 13, 4629. https://doi.org/10.3390/su13094629
Srivastava D, Tiwari M, Dutta P, Singh P, Chawda K, Kumari M, Chakrabarty D. Chromium Stress in Plants: Toxicity, Tolerance and Phytoremediation. Sustainability. 2021; 13(9):4629. https://doi.org/10.3390/su13094629
Chicago/Turabian StyleSrivastava, Dipali, Madhu Tiwari, Prasanna Dutta, Puja Singh, Khushboo Chawda, Monica Kumari, and Debasis Chakrabarty. 2021. "Chromium Stress in Plants: Toxicity, Tolerance and Phytoremediation" Sustainability 13, no. 9: 4629. https://doi.org/10.3390/su13094629
APA StyleSrivastava, D., Tiwari, M., Dutta, P., Singh, P., Chawda, K., Kumari, M., & Chakrabarty, D. (2021). Chromium Stress in Plants: Toxicity, Tolerance and Phytoremediation. Sustainability, 13(9), 4629. https://doi.org/10.3390/su13094629








