Productivity-Enhancing Technologies. Can Consumer Choices Affect the Environmental Footprint of Beef?
Abstract
1. Introduction
2. Productivity-Enhancing Technologies in Beef Production
2.1. Hormonal Implants
2.2. Ionophores
2.3. Beta-Adrenergic Agonists
3. The Role of PETs in Global Beef Production
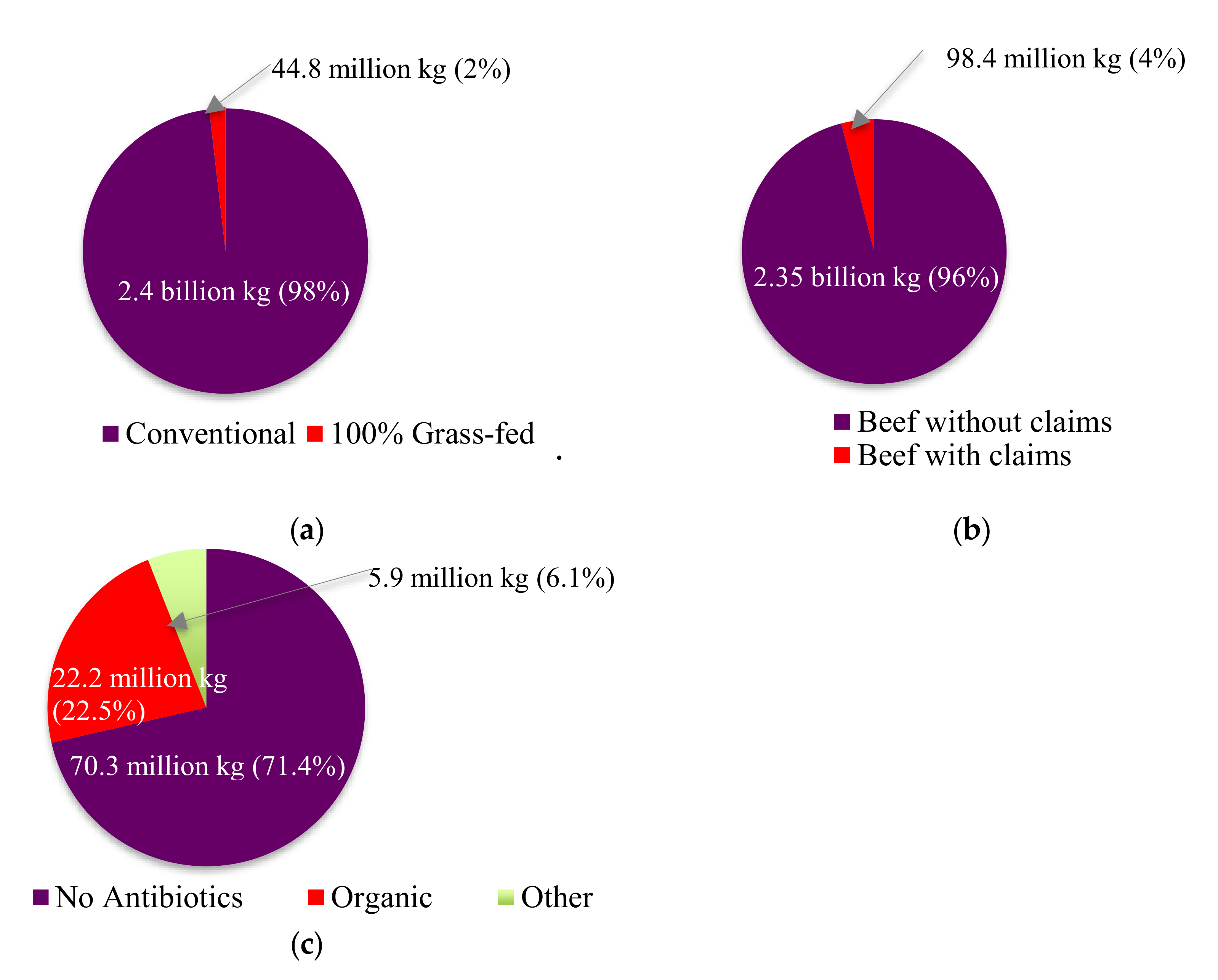
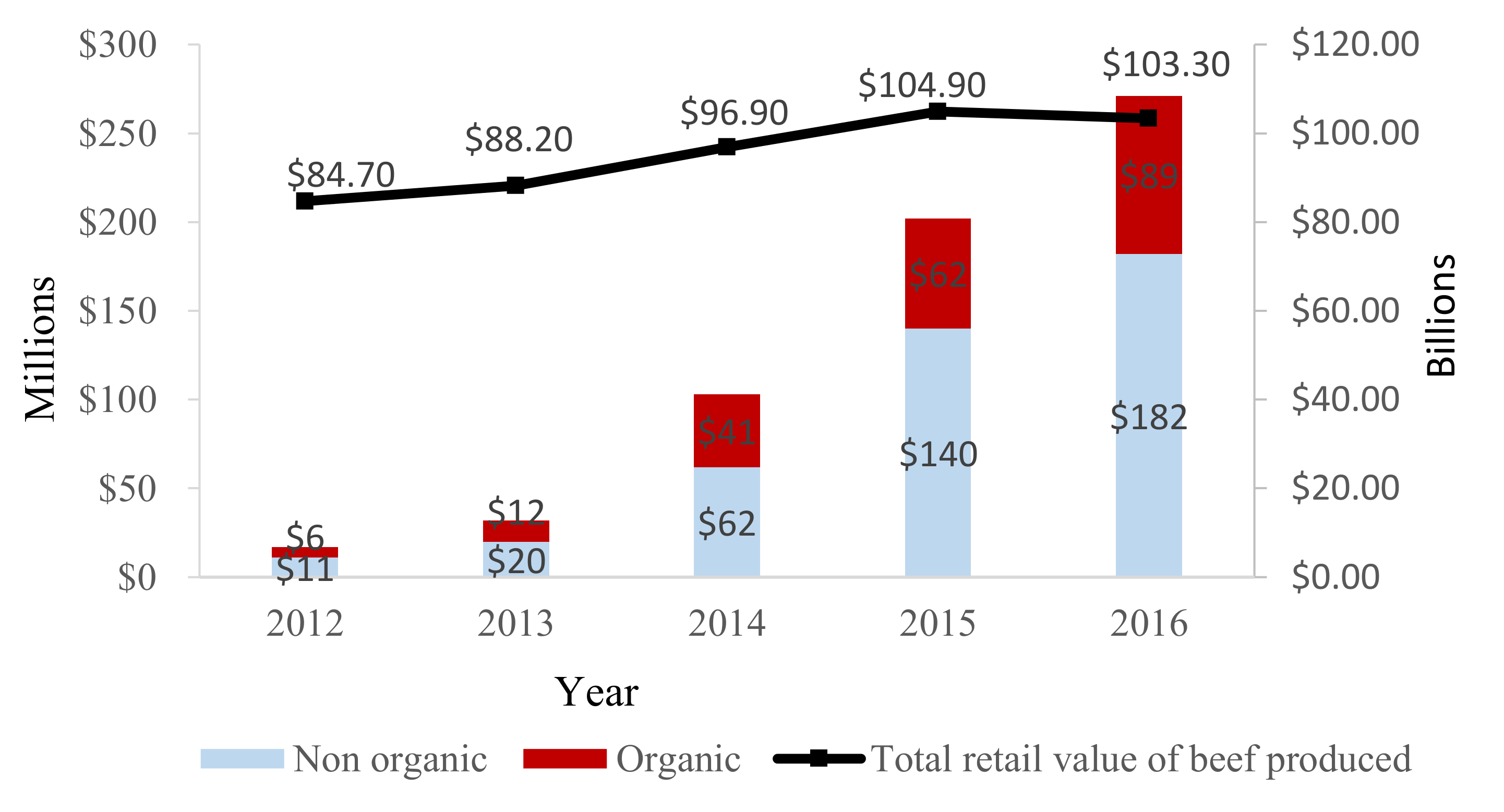
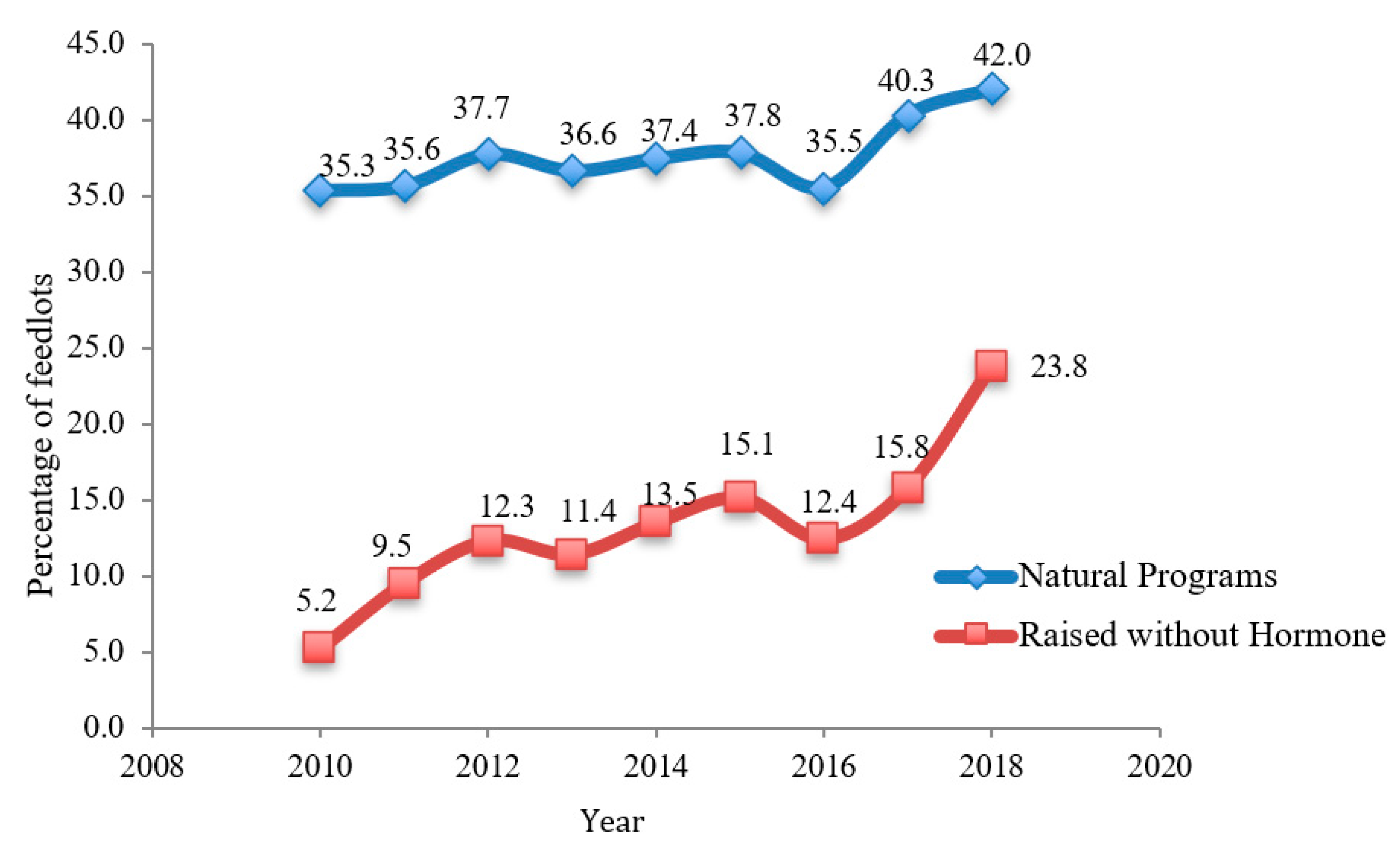
4. Impact of PET Use on Consumer Choice
5. PETs and the Environment
5.1. Greenhouse Gas Emissions and Resource Use
5.2. Environmental Contamination
6. PET, Food Safety, and Animal Welfare
7. Future Directions
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United Nations. Department of Economic and Social Affairs. Population Division. World Population Prospects. 2019. Available online: https://population.un.org/wpp/Graphs/Probabilistic/POP/TOT/900 (accessed on 10 January 2020).
- Adesogan, A.T.; Havelaar, A.H.; McKune, S.L.; Eilittä, M.; Dahl, G.E. Animal source foods: Sustainability problem or malnutrition and sustainability solution? Perspective matters. Glob. Food Sec. 2020, 25. [Google Scholar] [CrossRef]
- Lonnie, M.; Hooker, E.; Brunstrom, J.M.; Corfe, B.M.; Green, M.A.; Watson, A.W.; Williams, E.A.; Stevenson, E.J.; Penson, S.; Johnstone, A.M. Protein for life: Review of optimal protein intake, sustainable dietary sources and the effect on appetite in ageing adults. Nutrients 2018, 10, 360. [Google Scholar] [CrossRef] [PubMed]
- Van Zanten, H.H.E.; Meerburg, B.G.; Bikker, P.; Herrero, M.; De Boer, I.J.M. Opinion paper: The role of livestock in a sustainable diet: A land-use perspective. Animal 2016, 10, 547–549. [Google Scholar] [CrossRef] [PubMed]
- Food and Agriculture Organization (FAO). Dietary Protein Quality Evaluation in Human Nutrition; Report of an FAO Expert Consultation; FAO: Rome, Italy, 2013. [Google Scholar]
- Gorissen, S.H.M.; Witard, O.C. Characterising the muscle anabolic potential of dairy, meat and plant-based protein sources in older adults. Proc. Nutr. Soc. 2018, 77, 20–31. [Google Scholar] [CrossRef]
- Van Vliet, S.; Burd, N.A.; van Loon, L.J.C. The skeletal muscle anabolic response to plant- versus animal-based protein consumption. J. Nutr. 2015, 145, 1981–1991. [Google Scholar] [CrossRef]
- Obersby, D.; Chappell, D.C.; Dunnett, A.; Tsiami, A.A. Plasma total homocysteine status of vegetarians compared with omnivores: A systematic review and meta-Analysis. Br. J. Nutr. 2013, 109, 785–794. [Google Scholar] [CrossRef]
- Magkos, F.; Tetens, I.; Bügel, S.G.; Felby, C.; Schacht, S.R.; Hill, J.O.; Ravussin, E.; Astrup, A. A Perspective on the transition to plant-based diets: A diet change may attenuate climate change, but can it also attenuate obesity and chronic disease risk? Adv. Nutr. 2020, 11, 1–9. [Google Scholar] [CrossRef]
- Haider, L.M.; Schwingshackl, L.; Hoffmann, G.; Ekmekcioglu, C. The effect of vegetarian diets on iron status in adults: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2018, 58, 1359–1374. [Google Scholar] [CrossRef]
- Phillips, S.M. Nutrient-rich meat proteins in offsetting age-related muscle loss. Meat Sci. 2012, 92, 174–178. [Google Scholar] [CrossRef]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tarnopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef]
- Wilkinson, S.B.; Tarnopolsky, M.A.; Macdonald, M.J.; Macdonald, J.R.; Armstrong, D.; Phillips, S.M. Consumption of fluid skim milk promotes greater muscle protein accretion after resistance exercise than does consumption of an isonitrogenous and isoenergetic soy-protein beverage. Am. J. Clin. Nutr. 2007, 85, 1031–1040. [Google Scholar] [CrossRef]
- Herrero, M.; Havlík, P.; Valin, H.; Notenbaert, A.; Rufino, M.C.; Thornton, P.K.; Blümmel, M.; Weiss, F.; Grace, D.; Obersteiner, M. Biomass use, production, feed efficiencies, and greenhouse gas emissions from global livestock systems. Proc. Natl. Acad. Sci. USA 2013, 110, 20888–20893. [Google Scholar] [CrossRef]
- Gerber, P.J.; Steinfeld, H.; Henderson, B.; Mottet, A.; Opio, C.; Dijkman, J.; Falcucci, A.; Tempio, G. Tackling Climate Change Through Livestock: A Global Assessment of Emissions and Mitigation Opportunities; FAO: Rome, Italy, 2013. [Google Scholar]
- Poore, J.; Nemecek, T. Reducing food’s environmental impacts through producers and consumers. Science 2018, 360, 987–992. [Google Scholar] [CrossRef]
- Eshel, G.; Shepon, A.; Makov, T.; Milo, R. Land, irrigation water, greenhouse gas, and reactive nitrogen burdens of meat, eggs, and dairy production in the United States. Proc. Natl. Acad. Sci. USA 2014, 111, 11996–12001. [Google Scholar] [CrossRef]
- Lobato, J.F.P.; Freitas, A.K.; Devincenzi, T.; Cardoso, L.L.; Tarouco, J.U.; Vieira, R.M.; Dillenburg, D.R.; Castro, I. Brazilian beef produced on pastures: Sustainable and healthy. Meat Sci. 2014, 98, 336–345. [Google Scholar] [CrossRef]
- Wiedemann, S.G.; Henry, B.K.; McGahan, E.J.; Grant, T.; Murphy, C.M.; Niethe, G. Resource use and greenhouse gas intensity of Australian beef production: 1981–2010. Agric. Syst. 2015, 133, 109–118. [Google Scholar] [CrossRef]
- Capper, J.L. The environmental impact of beef production in the United States: 1977 compared with 2007. J. Anim. Sci. 2011, 89, 4249–4261. [Google Scholar] [CrossRef]
- Legesse, G.; Beauchemin, K.A.; Ominski, K.H.; McGeough, E.J.; Kroebel, R.; MacDonald, D.; Little, S.M.; McAllister, T.A. Greenhouse gas emissions of Canadian beef production in 1981 as compared with 2011. Anim. Prod. Sci. 2016, 56, 153–168. [Google Scholar] [CrossRef]
- Legesse, G.; Cordeiro, M.R.C.; Ominski, K.H.; Beauchemin, K.A.; Kroebel, R.; McGeough, E.J.; Pogue, S.; McAllister, T.A. Water use intensity of Canadian beef production in 1981 as compared to 2011. Sci. Total Environ. 2018, 619–620, 1030–1039. [Google Scholar] [CrossRef]
- Strydom, P.E. Performance-enhancing technologies of beef production. Anim. Front. 2016, 6, 22–30. [Google Scholar] [CrossRef][Green Version]
- Godfray, H.C.J.; Aveyard, P.; Garnett, T.; Hall, J.W.; Key, T.J.; Lorimer, J.; Pierrehumbert, R.T.; Scarborough, P.; Springmann, M.; Jebb, S.A. Meat consumption, health, and the environment. Science 2018, 361. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.H.; Kang, D.; Lim, M.W.; Kang, C.S.; Sung, H.J. Risk assessment of growth hormones and antimicrobial residues in meat. Toxicol. Res. 2010, 26, 301–313. [Google Scholar] [CrossRef]
- Nachman, K.E.; Smith, T.J. Hormone use in food animal production: Assessing potential dietary exposures and breast cancer risk. Curr. Environ. Health Rep. 2015, 2, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Nielsen Global Health and Ingredient-Sentiment Survey. What’s in Our Food and on Our Mind? Ingredient and Dining-Out Trends around the World. 2016. Available online: https://nutrimento.pt/activeapp/wp-content/uploads/2016/09/global-ingredient-and-out-of-home-dining-trends-aug-2016.pdf (accessed on 25 March 2020).
- Johnson, B.J.; Beckett, J. Application of Growth Enhancing Compounds in Modern Beef Production Executive Summary. Available online: https://meatscience.org/docs/default-source/publications-resources/white-papers/application-of-growth-enhancing-compounds-in-modern-beef-production-2015-final.pdf?sfvrsn=a9180b3_2 (accessed on 25 March 2020).
- Stewart, L. Implanting Beef Cattle; The University of Georgia Cooperative Extension: Athens, GA, USA, 2013; Available online: http://extension.uga.edu/publications/detail.html?number=B1302&title=Implanting%20Beef%20Cattle (accessed on 5 March 2020).
- Lone, K.P. Natural sex steroids and their xenobiotic analogs in animal production: Growth, carcass quality, pharmacokinetics, metabolism, mode of action, residues, methods, and epidemiology. Crit. Rev. Food Sci. Nutr. 1997, 37, 93–209. [Google Scholar] [CrossRef]
- McDonald, P.; Edwards, R.A.; Greenhalgh, J.F.D.; Morgan, C.A.; Sinclair, L.; Wilkinson, R.G. Animal Nutrition, 7th ed.; Pearson Education Limited: London, UK, 2011. [Google Scholar]
- Galbraith, H. Hormones in international meat production: Biological, sociological and consumer issues. Nutr. Res. Rev. 2002, 15, 293–314. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Davis, H.E.; Belk, K.E. Managing meat exports considering production technology challenges. Anim. Front. 2018, 8, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Reinhardt, C.D.; Wagner, J.J. High-dose anabolic implants are not all the same for growth and carcass traits of feedlot steers: A meta-analysis. J. Anim. Sci. 2014, 92, 4711–4718. [Google Scholar] [CrossRef][Green Version]
- Platter, W.J.; Tatum, J.D.; Belk, K.E.; Scanga, J.A.; Smith, G.C. Effects of repetitive use of hormonal implants on beef carcass quality, tenderness, and consumer ratings of beef palatability. J. Anim. Sci. 2003, 81, 984–996. [Google Scholar] [CrossRef]
- Partridge, I. Hormone Growth Promotants and Beef Production: A Best Practice Guide; Meat and Livestock Australia Limited: Sydney, Australia, 2011. [Google Scholar]
- Dunshea, F.R.; D’Souza, D.N.; Channon, H.A. Metabolic modifiers as performance-enhancing technologies for livestock production. Anim. Front. 2016, 6, 6–14. [Google Scholar] [CrossRef][Green Version]
- Zobell, D.R.; Chapman, C.K.; Heaton, K.; Birkelo, C. Beef cattle implants. In All Archived Publications; Utah State University Extension: Logan, UT, USA, 2000; Available online: https://digitalcommons.usu.edu/extension_histall/29/ (accessed on 3 February 2020).
- Gifford, C.A.; Branham, K.A.; Ellison, J.O.; Gómez, B.L.; Lemley, C.O.; Hart, C.G.; Krehbiel, C.R.; Bernhard, B.C.; Maxwell, C.L.; Goad, C.L.; et al. Effect of anabolic implants on adrenal cortisol synthesis in feedlot beef cattle implanted early or late in the finishing phase. Physiol. Behav. 2015, 138, 118–123. [Google Scholar] [CrossRef]
- Rokka, M.; Jestoi, M.; Peltonen, K. Trace level determination of polyether ionophores in feed. Biomed. Res. Int. 2013, 2013. [Google Scholar] [CrossRef]
- Ranga Niroshan Appuhamy, J.A.D.; Strathe, A.B.; Jayasundara, S.; Wagner-Riddle, C.; Dijkstra, J.; France, J.; Kebreab, E. Anti-methanogenic effects of monensin in dairy and beef cattle: A meta-analysis. J. Dairy Sci. 2013, 96, 5161–5173. [Google Scholar] [CrossRef]
- Duffield, T.F.; Merrill, J.K.; Bagg, R.N. Meta-analysis of the effects of monensin in beef cattle on feed efficiency, body weight gain, and dry matter intake. J. Anim. Sci. 2012, 90, 4583–4592. [Google Scholar] [CrossRef]
- Goodrich, R.D.; Garrett, J.E.; Gast, D.R.; Kirick, M.A.; Larson, D.A.; Meiske, J.C. Influence of monensin on the performance of cattle. J. Anim. Sci. 1984, 58, 1484–1498. [Google Scholar] [CrossRef]
- Spires, H.R.; Olmsted, A.; Berger, L.L.; Fontenot, J.P.; Gill, D.R.; Riley, J.G.; Wray, M.I.; Zinn, R.A. Efficacy of laidlomycin propionate for increasing rate and efficiency of gain by feedlot cattle. J. Anim. Sci. 1990, 68, 3382–3391. [Google Scholar] [CrossRef]
- Thompson, A.J.; Smith, Z.K.; Corbin, M.J.; Harper, L.B.; Johnson, B.J. Ionophore strategy affects growth performance and carcass characteristics in feedlot steers. J. Anim. Sci. 2016, 94, 5341–5349. [Google Scholar] [CrossRef]
- Hersom, M.; Thrift, T. Application of Ionophores in Cattle Dets. IFAS Ext.; University of Florida: Gainesville, FL, USA, 2012; Available online: https://edis.ifas.ufl.edu/pdffiles/AN/AN28500.pdf (accessed on 5 March 2020).
- Centner, T.J.; Alvey, J.C.; Stelzleni, A.M. Beta agonists in livestock feed: Status, health concerns, and international trade. J. Anim. Sci. 2014, 92, 4234–4240. [Google Scholar] [CrossRef]
- Neumeier, C.J.; Mitloehner, F.M. Cattle biotechnologies reduce environmental impact and help feed a growing planet. Anim. Front. 2013, 3, 36–41. [Google Scholar] [CrossRef]
- Lean, I.J.; Thompson, J.M.; Dunshea, F.R. A meta-analysis of zilpaterol and ractopamine effects on feedlot performance, carcass traits and shear strength of meat in cattle. PLoS ONE 2014, 9, e115904. [Google Scholar] [CrossRef]
- Smith, D.J.; Shelver, W.L.; Chakrabarty, S.; Hoffman, T.W. Detection and quantification of residues in sheep exposed to trace levels of dietary zilpaterol HCl. Food Addit Contam Part A. 2019, 36, 1289–1301. [Google Scholar] [CrossRef]
- Maxwell, C.L.; Bernhard, B.C.; O’Neill, C.F.; Wilson, B.K.; Hixon, C.G.; Haviland, C.L.; Grimes, A.N.; Calvo-Lorenzo, M.S.; VanOverbeke, D.L.; Mafi, G.G.; et al. The effects of technology use in feedlot production systems on feedlot performance and carcass characteristics. J. Anim. Sci. 2015, 93, 1340–1349. [Google Scholar] [CrossRef] [PubMed]
- Smith, Z.K.; Anderson, P.T.; Johnson, B.J. Finishing cattle in all-natural and conventional production systems. Open J. Anim. Sci. 2020, 10, 237–253. [Google Scholar] [CrossRef][Green Version]
- Kerr, W.A.; Hobbs, J.E. The North American-European union dispute over beef produced using growth hormones: A major test for the new international trade regime. World Econ. 2002, 25, 283–296. [Google Scholar] [CrossRef]
- FAO. 2020. Available online: http://www.fao.org/faostat/en/#data (accessed on 25 March 2020).
- Dilger, A. Beta-Agonists: What are They and Why Do We Use Them in Livestock Production. Available online: https://meatscience.org/docs/default-source/publications-resources/fact-sheets/beta-agonists---dilger-20158d82e7711b766618a3fcff0000a508da.pdf?sfvrsn=69f481b3_0 (accessed on 5 March 2020).
- Organization for Economic Cooperation and Development. Meat Consumption (Indicator). 2020. Available online: https://data.oecd.org/agroutput/meat-consumption.htm (accessed on 31 January 2020).
- Tait, P.; Rutherford, P.; Driver, T.; Li, X.; Saunders, C.; Dalziel, P.; Guenther, M. Consumer insights and willingness to pay for attributes: New Zealand beef products in California, USA. In Agribusiness and Economics Research Unit Research; Lincoln University New Zealand: Lincoln, New Zealand, 2018. [Google Scholar]
- Beef Checkoff. When It Comes to Beef, Consumers Have Options. 2020. Available online: https://www.beefitswhatsfordinner.com/retail/sales-data-shopper-insights/options-at-retail (accessed on 16 August 2020).
- Cheung, R.; McMahon, P.; Norell, E.; Kissel, R.; Benz, D.; Back to Grass: The Market Potential for U.S. Grass-Fed Beef. Stone Barns Center for Food and Agriculture. 2017. Available online: https://www.stonebarnscenter.org/wp-content/uploads/2017/10/Grassfed_Full_v2.pdf (accessed on 3 April 2020).
- United States Department of Agriculture. Statistics and Information; United State Department of Agriculture: Washington, DC, USA, 2021. Available online: https://www.ers.usda.gov/topics/animal-products/cattle-beef/statistics-information.aspx (accessed on 14 January 2021).
- Odde, K.G.; King, M.E.; McCabe, E.D.; Smith, M.J.; Hill, K.L.; Rogers, G.M.; Fike, K.E. Trends in “natural” value-added calf programs at superior livestock video auction. Kansas Agric. Exp. Stn. Res. Reports 2019, 5. [Google Scholar] [CrossRef]
- Olvera, I.D. Economic Implications Associated with Pharmaceutical Technology Bans in U.S. Beef Production. Ph.D. Thesis, Texas A & M University, College Station, TX, USA, 2016. [Google Scholar]
- White, R.R.; Brady, M. Can consumers’ willingness to pay incentivize adoption of environmental impact reducing technologies in meat animal production? Food Policy 2014, 49, 41–49. [Google Scholar] [CrossRef]
- Norris, A. Context Specific Factors Affecting Consumer Preferences for Antibiotic and Hormone Use during the Production of Beef in Canada. Master’s Thesis, University of Guelph, Guelph, ON, Canada, 2020. [Google Scholar]
- Lewis, K.E.; Grebitus, C.; Colson, G.; Hu, W. German and British consumer willingness to pay for beef labeled with food safety attributes. J. Agric. Econ. 2017, 68, 451–470. [Google Scholar] [CrossRef]
- Colella, F.; Ortega, D.L. Where’s the beef? Retail channel choice and beef preference in Argentina. Meat Sci. 2017, 133, 86–94. [Google Scholar] [CrossRef]
- Hirvonen, K.; Bai, Y.; Headey, D.; Masters, W.A. Affordability of the EAT–Lancet reference diet: A global analysis. Lancet Glob. Health 2020, 8, e59–e66. [Google Scholar] [CrossRef]
- Henchion, M.; Zimmermann, J. Animal food products: Policy, market and social issues and their influence on demand and supply of meat. Proc. Nutr. Soc. 2021, 1–12. [Google Scholar] [CrossRef]
- Charlebois, S.; Somogyi, S.; Music, J.; Caron, I. Planet, Ethics, Health and the New World Order in Proteins. J. Agric. Stud. 2020, 8, 171. [Google Scholar] [CrossRef]
- Stackhouse-Lawson, K.R.; Calvo, M.S.; Place, S.E.; Armitage, T.L.; Pan, Y.; Zhao, Y.; Mitloehner, F.M. Growth promoting technologies reduce greenhouse gas, alcohol, and ammonia emissions from feedlot cattle. J. Anim. Sci. 2013, 91, 5438–5447. [Google Scholar] [CrossRef]
- Cooprider, K.L.; Mitloehner, F.M.; Famula, T.R.; Kebreab, E.; Zhao, Y.; van Eenennaam, A.L. Feedlot efficiency implications on greenhouse gas sustainability. J. Anim. Sci. 2011, 89, 2643–2656. [Google Scholar] [CrossRef]
- Capper, J.L. Is the grass always greener? Comparing the environmental impact of conventional, natural and grass-fed beef production systems. Animals 2012, 2, 127–143. [Google Scholar] [CrossRef]
- Basarab, J.; Baron, V.; López-Campos, Ó.; Aalhus, J.; Haugen-Kozyra, K.; Okine, E. Greenhouse gas emissions from calf- and yearling-fed beef production systems, with and without the use of growth promotants. Animals 2012, 2, 195–220. [Google Scholar] [CrossRef]
- Capper, J.L.; Hayes, D.J. The environmental and economic impact of removing growth-enhancing technologies from U.S. beef production. J. Anim. Sci. 2012, 90, 3527–3537. [Google Scholar] [CrossRef]
- Stackhouse, K.R.; Rotz, C.A.; Oltjen, J.W.; Mitloehner, F.M. Growth-promoting technologies decrease the carbon footprint, ammonia emissions, and costs of California beef production systems. J. Anim. Sci. 2012, 90, 4656–4665. [Google Scholar] [CrossRef]
- Webb, M.J. Influence of Production System on Animal Performance, Carcass Characteristics, Meat Quality, Environmental Impacts, Production Economics, and Consumer Preference for Beef. Ph.D. Thesis, South Dakota State University, Brookings, SD, USA, 2018. [Google Scholar]
- Biswas, S.; Shapiro, C.A.; Kranz, W.L.; Mader, T.L.; Shelton, D.P.; Snow, D.D.; Bartelt-Hunt, S.L.; Tarkalson, D.D.; Van Donk, S.J.; Zhang, T.C.; et al. Current knowledge on the environmental fate, potential impact, and management of growth-promoting steroids used in the US beef cattle industry. J. Soil Water Conserv. 2013, 68, 325–336. [Google Scholar] [CrossRef]
- Zhao, S.; Zhang, P.; Melcer, M.E.; Molina, J.F. Estrogens in streams associated with a concentrated animal feeding operation in upstate New York, USA. Chemosphere 2010, 79, 420–425. [Google Scholar] [CrossRef]
- Hafner, S.C.; Harter, T.; Parikh, S.J. Evaluation of monensin transport to shallow groundwater after irrigation with dairylagoon water. J. Environ. Qual. 2016, 45, 480–487. [Google Scholar] [CrossRef]
- Blackwell, B.R.; Wooten, K.J.; Buser, M.D.; Johnson, B.J.; Cobb, G.P.; Smith, P.N. Occurrence and characterization of steroid growth promoters associated with particulate matter originating from beef cattle feedyards. Environ. Sci. Technol. 2015, 49, 8796–8803. [Google Scholar] [CrossRef]
- Wooten, K.J.; Sandoz, M.A.; Smith, P.N. Ractopamine in particulate matter emitted from beef cattle feedyards and playa wetlands in the Central Plains. Environ. Toxic. Chem. 2018, 37, 970–974. [Google Scholar] [CrossRef] [PubMed]
- Challis, J.K.; Sura, S.; Cantin, J.; Curtis, A.W.; Shade, K.M.; McAllister, T.A.; Jones, P.D.; Giesy, J.P.; Larney, F.J. Ractopamine and other growth-promoting compounds in beef cattle operations: Fate and transport in feedlot pens and adjacent environments. Environ. Sci. Technol. 2021, 55, 1730–1739. [Google Scholar] [CrossRef] [PubMed]
- Bartelt-Hunt, S.; Snow, D.D.; Damon-Powell, T.; Miesbach, D. Occurrence of steroid hormones and antibiotics in shallow groundwater impacted by livestock waste control facilities. J. Contam. Hydrol. 2011, 123, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Hanselman, T.A.; Graetz, D.A.; Wilkie, A.C. Manure-borne estrogens as potential environmental contaminants: A review. Environ. Sci. Technol. 2003, 37, 5471–5478. [Google Scholar] [CrossRef]
- Henry, P.F.P.; Akuffo, V.G.; Chen, Y.; Karouna-Renier, N.K.; Sprague, D.T.; Bakst, M.R. Effect of 17β-trenbolone on male and female reproduction in Japanese quail (Coturnix Japonica). Avian Biol. Res. 2012, 5, 61–68. [Google Scholar] [CrossRef]
- Quinn, M.J.; Lavoie, E.T.; Ottinger, M.A. Reproductive toxicity of trenbolone acetate in embryonically exposed Japanese quail. Chemosphere 2007, 66, 1191–1196. [Google Scholar] [CrossRef]
- Shappell, N.W.; Hyndman, K.M.; Bartell, S.E.; Schoenfuss, H.L. Comparative biological effects and potency of 17 alpha- and 17 beta-estradiol in fathead minnows. Aquatic. Toxicol. 2010, 100, 1–8. [Google Scholar] [CrossRef]
- Finch, B.E.; Blackwell, B.R.; Faust, D.R.; Wooten, K.J.; Maul, J.D.; Cox, S.B.; Smith, P.N. Effects of 17α-trenbolone and melengestrol acetate on Xenopus laevis growth, development, and survival. Environ. Sci. Pollut. Res. Int. 2013, 20, 1151–1160. [Google Scholar] [CrossRef]
- Jeffries, K.M.; Jackson, L.J.; Ikonomou, M.G.; Habibi, H.R. Presence of natural and anthropogenic organic contaminants and potential fish health impacts along two river gradients in Alberta, Canada. Environ. Toxicol. Chem. 2010, 29, 2379–2387. [Google Scholar] [CrossRef]
- Johns, S.M.; Denslow, N.D.; Kane, M.D.; Watanabe, K.H.; Orlando, E.F.; Sepúlveda, M.S. Effects of estrogens and antiestrogens on gene expression of fathead minnow (Pimephales promelas) early life stages. Environ Toxicol. 2011, 26, 195–206. [Google Scholar] [CrossRef]
- Schwartz, P.; Thorpe, K.L.; Bucheli, T.D.; Wettstein, F.E.; Burkhardt-Holm, P. Short-term exposure to the environmentally relevant estrogenic mycotoxin zearalenone impairs reproduction in fish. Sci. Total Environ. 2010, 409, 326–333. [Google Scholar] [CrossRef]
- Schwartz, P.; Bucheli, T.D.; Wettstein, F.E.; Burkhardt-Holm, P. Life-cycle exposure to the estrogenic mycotoxin zearalenone affects zebrafish (Danio rerio) development and reproduction. Environ. Toxicol. 2013, 28, 276–289. [Google Scholar] [CrossRef]
- Adeel, M.; Song, X.; Wang, Y.; Francis, D.; Yang, Y. Environmental impact of estrogens on human, animal and plant life: A critical review. Environ. Int. 2017, 99, 107–119. [Google Scholar] [CrossRef]
- Biswas, S.; Kranz, W.L.; Shapiro, C.A.; Snow, D.D.; Bartelt-Hunt, S.L.; Mamo, M.; Tarkalson, D.D.; Zhang, T.C.; Shelton, D.P.; van Donk, S.J.; et al. Effect of rainfall timing and tillage on the transport of steroid hormones in runoff from manure amended row crop fields. J. Hazard Mater. 2017, 324, 436–447. [Google Scholar] [CrossRef]
- Havens, S.M.; Hedman, C.J.; Hemming, J.; Mieritz, M.G.; Shafer, M.M.; Schauer, J.J. Occurrence of estrogens, androgens and progestogens and estrogenic activity in surface water runoff from beef and dairy manure amended crop fields. Sci. Total Environ. 2020, 710, 136247. [Google Scholar] [CrossRef]
- Leusch, F.D.L.; Neale, P.A.; Arnal, C.; Aneck-Hahn, N.H.; Balaguer, P.; Bruchet, A.; Escher, B.I.; Esperanza, M.; Grimaldi, M.; Leroy, G.; et al. Analysis of endocrine activity in drinking water, surface water and treated wastewater from six countries. Water Res. 2018, 139, 10–18. [Google Scholar] [CrossRef]
- Pickford, D.B.; Jones, A.; Velez-Pelez, A.; Iguchi, T.; Mitsui, N.; Tooi, O. Screening breeding sites of the common toad (Bufo bufo) in England and Wales for evidence of endocrine disrupting activity. Ecotoxicol. Environ. Saf. 2015, 117, 7–19. [Google Scholar] [CrossRef]
- Molina-Molina, J.M.; Real, M.; Jimenez-Diaz, I.; Belhassen, H.; Hedhili, A.; Torné, P.; Fernández, M.F.; Olea, N. Assessment of estrogenic and anti-androgenic activities of the mycotoxin zearalenone and its metabolites using in vitro receptor-specific bioassays. Food Chem. Toxicol. 2014, 74, 233–239. [Google Scholar] [CrossRef]
- Bartelt-Hunt, S.L.; Snow, D.D.; Kranz, W.L.; Mader, T.L.; Shapiro, C.A.; Donk, S.J.; Shelton, D.P.; Tarkalson, D.D.; Zhang, T.C. Effect of growth promotants on the occurrence of endogenous and synthetic steroid hormones on feedlot soils and in runoff from beef cattle feeding operations. Environ. Sci. Technol. 2012, 46, 1352–1360. [Google Scholar] [CrossRef]
- Amarakoon, I.D.; Zvomuya, F.; Sura, S.; Larney, F.J.; Cessna, A.J.; Xu, S.; McAllister, T.A. Dissipation of antimicrobials in feedlot manure compost after oral administration versus fortification after excretion. J. Environ. Qual. 2016, 45, 503–510. [Google Scholar] [CrossRef]
- Carter, S.D.; Kim, H.J. Technologies to reduce environmental impact of animal wastes associated with feeding for maximum productivity. Anim. Front. 2013, 3, 42–47. [Google Scholar] [CrossRef][Green Version]
- Mersmann, H.J. Overview of the effects of beta-adrenergic receptor agonists on animal growth including mechanisms of action. J. Anim. Sci. 1998, 76, 160–172. [Google Scholar] [CrossRef] [PubMed]
- Government of Canada. Responsible Use of Medically Important Antimicrobials in Animals; Government of Canada: Ottawa, AB, Canada, 2019; Available online: https://www.canada.ca/en/public-health/services/antibiotic-antimicrobial-resistance/animals/actions/responsible-use-antimicrobials.html (accessed on 19 August 2020).
- Aidara-Kane, A.; Angulo, F.J.; Conly, J.; Minato, Y.; Silbergeld, E.K.; McEwen, S.A.; Collignon, P.J.; Balkhy, H.; Collignon, P.; Friedman, C.; et al. World Health Organization (WHO) guidelines on use of medically important antimicrobials in food-producing animals. Antimicrob. Resist. Infect. Control 2018, 7, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Global Roundtable for Sustainable Beef. Sustainability Report. 2018. Available online: https://grsbeef.org/resources/Documents/WhoWeAre/GRSB_Sustainability_Report_2018.pdf (accessed on 17 August 2020).
- Wong, A. Unknown risk on the farm: Does Agricultural use of ionophores contribute to the burden of antimicrobial resistance? mSphere 2019, 4, 1–6. [Google Scholar] [CrossRef]
- Baynes, R.E.; Dedonder, K.; Kissell, L.; Mzyk, D.; Marmulak, T.; Smith, G.; Tell, L.; Gehring, R.; Davis, J.; Riviere, J.E. Health concerns and management of select veterinary drug residues. Food Chem. Toxicol. 2016, 88, 112–122. [Google Scholar] [CrossRef]
- FAO; WHO. Carryover in feed and transfer from feed to food of unavoidable and unintended residues of approved veterinary drugs. In Report of the Joint FAO/WHO Expert Meeting from 8 to 10 January 2019; Animal Production and Health; FAO: Rome, Italy, 2019. [Google Scholar]
- Sakai, N.; Sakai, M.; Mohamad Haron, D.E.; Yoneda, M.; Ali Mohd, M. Beta-agonist residues in cattle, chicken and swine livers at the wet market and the environmental impacts of wastewater from livestock farms in Selangor State, Malaysia. Chemosphere 2016, 165, 183–190. [Google Scholar] [CrossRef]
- Davis, H.E.; Badger, C.D.; Brophy, P.; Geornaras, I.; Burnett, T.J.; Scanga, J.; Belk, K.; Prenni, J. Quantification of ractopamine residues on and in beef digestive tract tissues. J. Anim. Sci. 2019, 97, 4193–4198. [Google Scholar] [CrossRef]
- Arcella, D.; Baert, K.; Binaglia, M.; Gervelmeyer, A.; Innocenti, M.L.; Ribo, O.; Steinkellner, H.; Verhagen, H. Review of proposed MRLs, safety evaluation of products obtained from animals treated with zilpaterol and evaluation of the effects of zilpaterol on animal health and welfare. EFSA J. 2016, 14, e04579. [Google Scholar] [CrossRef]
- Canadian Food Inspection Agency. Canadian Beta Agonist-Free Beef Certification Program; Government of Canada: Ottawa, AB, Canada, 2017; Available online: https://inspection.canada.ca/exporting-food-plants-or-animals/food-exports/food-specific-export-requirements/meat/annex-v/eng/1491926348345/1491926349115#b8 (accessed on 3 April 2020).
- Australian Government Department of Agriculture. National Residue Survey 2018–2019 Cattle; Australian Government: Canberra, Australia, 2019. Available online: https://www.agriculture.gov.au/ag-farm-food/food/nrs/nrs-results-publications/industry-brochures/cattle (accessed on 25 May 2020).
- USDA-Food Safety and Inspection Service. Residue Sample Results—“Red Book”; United State Department of Agriculture: Washington, DC, USA, 2019. Available online: http://www.fsis.usda.gov/wps/portal/fsis/topics/data-collection-and-reports/chemistry/red-books/red-book (accessed on 1 May 2020).
- Groot, M.J.; Schilt, R.; Ossenkoppele, J.S.; Berende, P.L.; Haasnoot, W. Combinations of growth promoters in veal calves: Consequences for screening and confirmation methods. Zentralbl. Veterinarmed. A. 1998, 45, 425–440. [Google Scholar] [CrossRef]
- Pérez-Martínez, C.; García-Iglesias, M.J.; Ferreras-Estrada, M.C.; Bravo-Moral, A.M.; Espinosa-Alvarez, J.; Escudero-Díez, A. Effects of in-utero exposure to zeranol or diethylstilboestrol on morphological development of the fetal testis in mice. J. Comp. Pathol. 1996, 114, 407–418. [Google Scholar] [CrossRef]
- Biolatti, B.; Bollo, E.; Re, G.; Appino, S.; Tartari, E.; Benatti, G.; Elliott, C.T.; McCaughey, W.J. Pathology and residues in veal calves treated experimentally with clenbuterol. Res. Vet. Sci. 1994, 57, 365–371. [Google Scholar] [CrossRef]
- Zimmerli, U.V.; Blum, J.W. Acute and long-term metabolic, endocrine, respiratory, cardiac and skeletal-muscle activity changes in response to perorally administered β-adrenoceptor agonists in calves. J. Anim. Physiol. Anim. Nutr. 1990, 63, 157–172. [Google Scholar] [CrossRef]
- Neary, J.M.; Garry, F.B.; Gould, D.H.; Holt, T.N.; Dale Brown, R. The beta-adrenergic agonist zilpaterol hydrochloride may predispose feedlot cattle to cardiac remodeling and dysfunction [version 1; peer review: 2 approved with reservations]. F1000Research 2018, 7, 1–12. [Google Scholar] [CrossRef]
- Loneragan, G.H.; Thomson, D.U.; Scott, H.M. Increased mortality in groups of cattle administered the β-adrenergic agonists ractopamine hydrochloride and zilpaterol hydrochloride. PLoS ONE 2014, 9, e91177. [Google Scholar] [CrossRef]
- Huffstutter, P.J.; Polansek, T. Lost Hooves, Dead Cattle before Merck Halted Zilmax Sales; Reuters: London, UK, 2013. [Google Scholar]
- Merck Animal Health. Animal Safety and Well-Being; Merck Animal Health: Madison, NJ, USA, 2015. [Google Scholar]
- Carll, A.P.; Haykal-Coates, N.; Winsett, D.W.; Rowan, W.H., 3rd; Hazari, M.S.; Ledbetter, A.D.; Nyska, A.; Cascio, W.E.; Watkinson, W.P.; Costa, D.L.; et al. Particulate matter inhalation exacerbates cardiopulmonary injury in a rat model of isoproterenol-induced cardiomyopathy. Inhal. Toxicol. 2010, 22, 355–368. [Google Scholar] [CrossRef]
- Chiarella, S.E.; Soberanes, S.; Urich, D.; Morales-Nebreda, L.; Nigdelioglu, R.; Green, D.; Young, J.B.; Gonzalez, A.; Rosario, C.; Misharin, A.V.; et al. β2-Adrenergic agonists augment air pollution-induced IL-6 release and thrombosis. J. Clin. Investig. 2014, 124, 2935–2946. [Google Scholar] [CrossRef]
- Neary, J.M.; Booker, C.W.; Wildman, B.K.; Morley, P.S. Right-sided congestive heart failure in North American feedlot cattle. J. Vet. Intern. Med. 2016, 30, 326–334. [Google Scholar] [CrossRef]
- Frese, D.A.; Reinhardt, C.D.; Bartle, S.J.; Rethorst, D.N.; Bawa, B.; Thomason, J.D.; Loneragan, G.H.; Thomson, D.U. Effect of ractopamine hydrochloride and zilpaterol hydrochloride on cardiac electrophysiologic and hematologic variables in finishing steers. J. Am. Vet. Med. Assoc. 2016, 249, 668–677. [Google Scholar] [CrossRef]
- Hagenmaier, J.A.; Reinhardt, C.D.; Ritter, M.J.; Calvo-Lorenzo, M.S.; Vogel, G.J.; Guthrie, C.A.; Siemens, M.G.; Lechtenberg, K.F.; Rezac, D.J.; Thomson, D.U. Effects of ractopamine hydrochloride on growth performance, carcass characteristics, and physiological response to different handling techniques. J. Anim. Sci. 2017, 95, 1977–1992. [Google Scholar] [CrossRef]
- United State Food and Drug Administration (FDA). FDA Approves Experior for Reduction of Ammonia Gas Released from Beef Cattle Waste; Center for Veterinary Medicine: Lewisville, TX, USA, 2018. [Google Scholar]
- Government of Canada. Lubabegron—Medicating Ingredient Brochure; Government of Canada: Ottawa, AB, Canada, 2021; Available online: https://inspection.canada.ca/animal-health/livestock-feeds/medicating-ingredients/lubabegron/eng/1547583000099/1547583001862 (accessed on 23 March 2021).
- Workman, D. Top Beef Exporting Countries. Available online: http://www.worldstopexports.com/top-beef-exporting-countries/ (accessed on 1 June 2020).
- Government of Canada. Text of the Comprehensive Economic and Trade Agreement—Annex 2-A: Tariff Elimination; Government of Canada: Ottawa, AB, Canada, 2017; Available online: https://www.international.gc.ca/trade-commerce/trade-agreements-accords-commerciaux/agr-acc/ceta-aecg/text-texte/02-A.aspx?lang=eng (accessed on 1 June 2020).
- Teel, G. CCA Cheers Declaration of Complete CETA Text; Canadian Cattlemen’s Association (CCA): Calgary, AB, Canada, 2014; Available online: https://www.cattle.ca/assets/Article/d34f9112f1/09-26-2014-CCA-cheers-declaration-of-complete-CETA-text2.pdf (accessed on 1 June 2020).
- Arnason, R. EU Trade Deal Falls Short of Promises for Beef Producers; The Western Producer: Saskatoon, SK, Canada, 2020. [Google Scholar]
- United States Department of Agriculture. Export Requirements for The People’s Republic of China; United State Department of Agriculture: Washington, DC, USA, 2020. Available online: https://www.fsis.usda.gov/wps/portal/fsis/topics/international-affairs/exporting-products/export-library-requirements-by-country/peoples-republic-of-china (accessed on 30 May 2020).
| Class a | Mode of Action | Substance b | Mode of Administration |
|---|---|---|---|
| Growth hormones | |||
| Endogenous/Synthetic | Increase protein deposition at the expense of fat to increase growth rate and decrease amount of feed required for the animal to gain weight. | Estradiol-17β, Testosterone, Progesterone/Zearalenone, Trenbolone acetate | Implants |
| Melengestrol acetate | In-Feed | ||
| Beta-adrenergic agonists | Redirect nutrients from digestive organs into muscle tissue, thus increasing muscle mass accretion at the expense of fat deposition. | Ractopamine chloride, Zilpaterol chloride | In-Feed |
| Antibiotics c | |||
| Ionophores | Act against Gram-positive bacteria by altering membrane permeability to promote propionate formation in the rumen, which is more energetically favorable than acetate production. | Monensin, Lasalocid, Salinonmycin | In-Feed |
| Macrolides | Has bacteriostatic effect on both Gram-positive and Gram-negative bacteria, thus reducing microbial competition for nutrients. | Tylosin, Neomycin | In-Feed, water, or parenteral |
| Aminoglycosides | |||
| Tetracyclines | Oxytetracycline, Chlortetracyclic |
| Reference | Summary of Trial Design | Environmental Indices e,f | Country | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Methodology a | Production Stage b | Treatment c | Days on Feed | CO2eq | Land | Water | Energy | NH3 /Manure Excretion | ||
| Basarab et al. [73] | LCA | Backgrounding and finishing phases | IMP or control | Backgrounding: 312 days. Finishing: 146 to 207 days. | 5.8% ↓ | 7.8% ↓ | NR | NR | NR | Canada |
| Capper [72] | LCA | Backgrounding and finishing phases | βAA + IMP + MGA + ION (“conventional”); and no additives (“grass-fed” or “natural” animals). | Backgrounding: 123 to 159 days. Finishing: 110 to 313 days. | 14.8–40.3% ↓ | 18.3–44.7% ↓ | 17.9–75.2% ↓ | 14.9–28.6% ↓ | 17.9–50.5% ↓ N and 20.7–51.4% ↓ P excretions | US |
| Capper and Hayes [74] | LCA | Backgrounding and finishing phases | βAA + IMP + ION + MGA; or control. | Backgrounding: 148 to 159 days. Finishing: 116 to 209 days. | 8.9% ↓ | 9.1% ↓ | 4.0%↓ | 7.1% ↓ | 8.9% and 9.6%, ↓ N and P excretions, respectively. | US |
| Cooprider et al. [71] | Animal trial | Finishing phase | βAA + IMP + ION; or control. | 146 to 188 days. | 31.4% ↓ non-CO2 emissions | NR | NR | NR | NR | US |
| Stackhouse et al. [75] | LCA | Backgrounding and finishing phases | IMP + ION only; βAA + IMP + ION; or control. | Backgrounding: 182 days. Finishing: 121 to 212 days. | 6.6–8.0% ↓ | NR | NR | NR | 7.7–13.5% ↓ NH3 emissions. | US |
| Stackhouse-Lawson et al. [70] | Animal trial | Finishing phase | ION only; IMP + ION only; βAA + IMP + ION; or control | 107 days. | 9.6–16.4% ↓CH4 emissions | NR | NR | NR | 30% ↓ NH3 emissions | US |
| Webb [76] | Animal trial and LCA | Cow–calf, backgrounding, and finishing phases | ION only; IMP + ION only; βAA + IMP + ION; or control. | Backgrounding: 91 days, Finishing: 152 to 183 days | 1.1–7.7% ↓ | NR | 1.0–5.8% ↓ | 1.1–5.5% ↓ | 0.7–5.1% ↓ reactive N | US |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aboagye, I.A.; Cordeiro, M.R.C.; McAllister, T.A.; Ominski, K.H. Productivity-Enhancing Technologies. Can Consumer Choices Affect the Environmental Footprint of Beef? Sustainability 2021, 13, 4283. https://doi.org/10.3390/su13084283
Aboagye IA, Cordeiro MRC, McAllister TA, Ominski KH. Productivity-Enhancing Technologies. Can Consumer Choices Affect the Environmental Footprint of Beef? Sustainability. 2021; 13(8):4283. https://doi.org/10.3390/su13084283
Chicago/Turabian StyleAboagye, Isaac A., Marcos R. C. Cordeiro, Tim A. McAllister, and Kim H. Ominski. 2021. "Productivity-Enhancing Technologies. Can Consumer Choices Affect the Environmental Footprint of Beef?" Sustainability 13, no. 8: 4283. https://doi.org/10.3390/su13084283
APA StyleAboagye, I. A., Cordeiro, M. R. C., McAllister, T. A., & Ominski, K. H. (2021). Productivity-Enhancing Technologies. Can Consumer Choices Affect the Environmental Footprint of Beef? Sustainability, 13(8), 4283. https://doi.org/10.3390/su13084283







