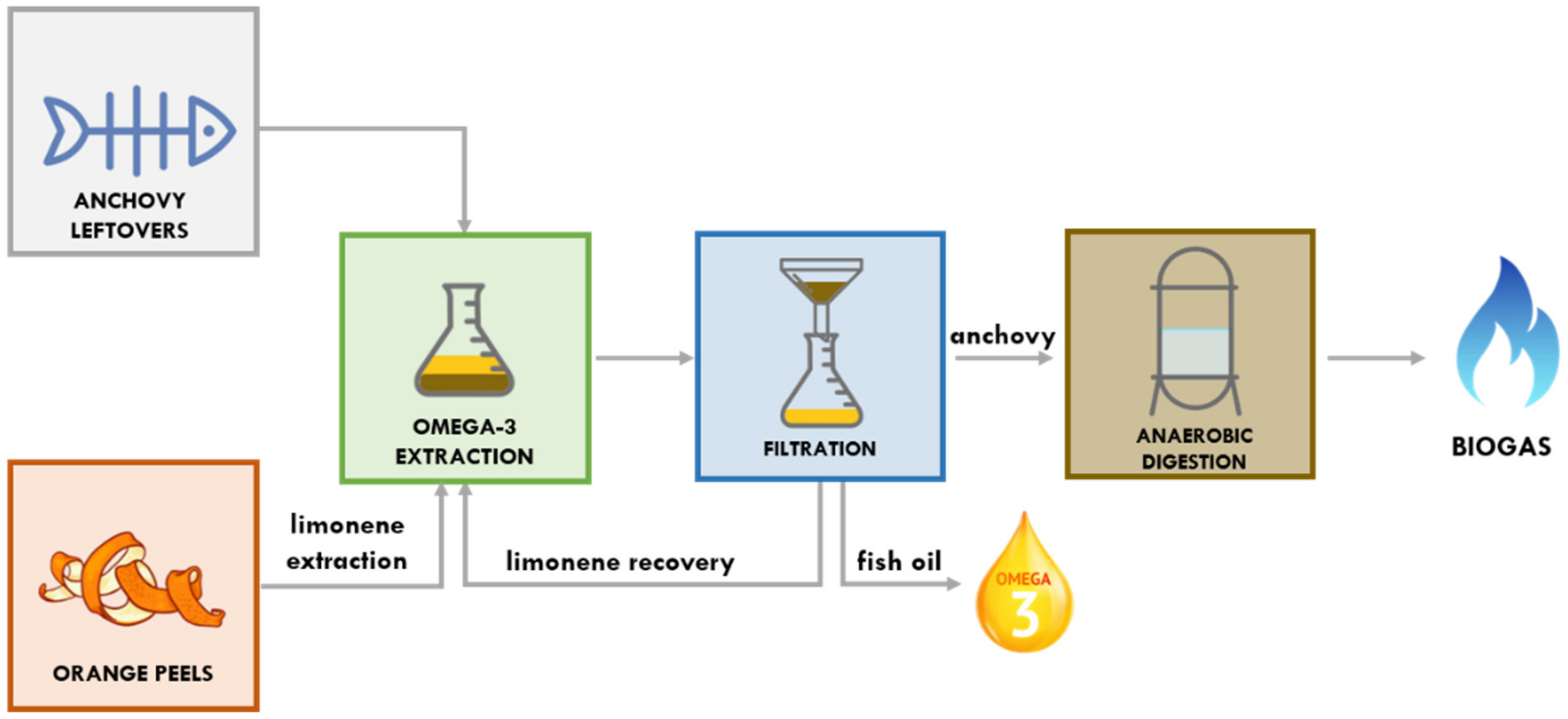
Towards the Anchovy Biorefinery: Biogas Production from Anchovy Processing Waste after Fish Oil Extraction with Biobased Limonene
Abstract
1. Introduction
2. Materials and Methods
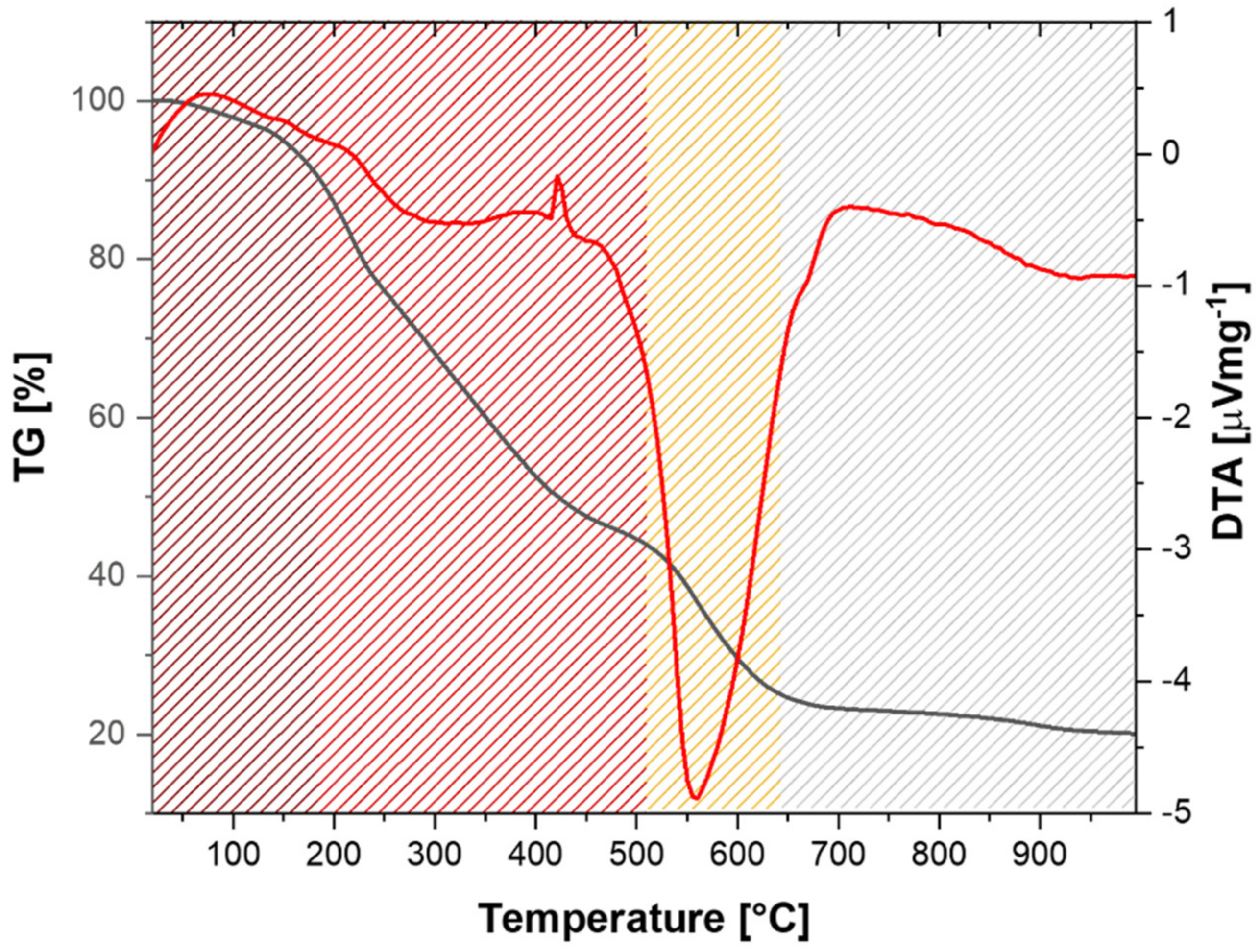
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lucarini, M.; Zuorro, A.; Di Lena, G.; Lavecchia, R.; Durazzo, A.; Benedetti, B.; Lombardi-Boccia, G. Sustainable management of secondary raw materials from the marine food-chain: A case-study perspective. Sustainability 2020, 12, 8997. [Google Scholar] [CrossRef]
- Ido, A.; Kaneta, M. Fish oil and fish meal production from urban fisheries biomass in Japan. Sustainability 2020, 12, 3345. [Google Scholar] [CrossRef]
- Goossens, Y.; Schmidt, T.G.; Kuntscher, M. Evaluation of food waste prevention measures-the use of fish products in the food service sector. Sustainability 2020, 12, 6613. [Google Scholar] [CrossRef]
- The Blue Economy: 10 Years—100 Innovations—100 Million. Available online: https://www.theblueeconomy.org (accessed on 1 February 2021).
- FAO. The State of World Fisheries and Aquaculture 2020. Sustainability in Action; Food and Agriculture Organization of the United Nations—FAO: New York, NY, USA, 2020; ISBN 9789251326923. [Google Scholar]
- Transforming Our World: The 2030 Agenda For Sustainable Development; United Nations: New York, NY, USA, 2016; pp. 12–14. [CrossRef]
- United Nations. Our ocean, our Future: Call for Action; General Assembly Resolution; United Nations: New York, NY, USA, 2017; A/RES/71/3; p. 6. [Google Scholar]
- Calabrò, P.S.; Gori, M.; Lubello, C. European trends in greenhouse gases emissions from integrated solid waste management. Environ. Technol. 2015, 36, 2125–2137. [Google Scholar] [CrossRef] [PubMed]
- Paone, E.; Tabanelli, T.; Mauriello, F. The rise of lignin biorefinery. Curr. Opin. Green Sustain. Chem. 2020, 24, 1–6. [Google Scholar] [CrossRef]
- Pagliaro, M.; Pizzone, D.M.; Scurria, A.; Lino, C.; Paone, E.; Mauriello, F.; Ciriminna, R. Sustainably Sourced Olive Polyphenols and Omega 3 Marine Lipids: A Synergy Fostering Public Health. ACS Food Sci. Technol. 2021. [Google Scholar] [CrossRef]
- Maschmeyer, T.; Luque, R.; Selva, M. Upgrading of marine (fish and crustaceans) biowaste for high added-value molecules and bio(nano)-materials. Chem. Soc. Rev. 2020, 49, 4527–4563. [Google Scholar] [CrossRef]
- Xu, C.; Nasrollahzadeh, M.; Selva, M.; Issaabadi, Z.; Luque, R. Waste-to-wealth: Biowaste valorization into valuable bio(nano)materials. Chem. Soc. Rev. 2019, 48, 4791–4822. [Google Scholar] [CrossRef]
- Nawaz, A.; Li, E.; Irshad, S.; Xiong, Z.; Xiong, H.; Shahbaz, H.M.; Siddique, F. Valorization of fisheries by-products: Challenges and technical concerns to food industry. Trends Food Sci. Technol. 2020, 99, 34–43. [Google Scholar] [CrossRef]
- Kim, S.K. Seafood Processing By-Products: Trends and Applications; Seaf. Process; Springer: Berlin/Heidelberg, Germany. [CrossRef]
- Lands, W.E.M. Fish, Omega-3 and Human Health; AOCS press: Champaign, IL, USA, 2005; ISBN 1893997812. [Google Scholar]
- Shahidi, F.; Ambigaipalan, P. Omega-3 Polyunsaturated Fatty Acids and Their Health Benefits. Annu. Rev. Food Sci. Technol. 2018, 9, 345–381. [Google Scholar] [CrossRef] [PubMed]
- Pike, I.H.; Jackson, A. Fish oil: Production and use now and in the future. Lipid Technol. 2010, 22, 59–61. [Google Scholar] [CrossRef]
- Ciriminna, R.; Scurria, A.; Avellone, G.; Pagliaro, M. A Circular Economy Approach to Fish Oil Extraction. ChemistrySelect 2019, 4, 5106–5109. [Google Scholar] [CrossRef]
- McCarty, P.L.; Smith, D.P. Anaerobic wastewater treatment. Environ. Sci. Technol. 1986, 20, 1200–1206. [Google Scholar] [CrossRef]
- Sawatdeenarunat, C.; Nguyen, D.; Surendra, K.C.; Shrestha, S.; Rajendran, K.; Oechsner, H.; Xie, L.; Khanal, S.K. Anaerobic biorefinery: Current status, challenges, and opportunities. Bioresour. Technol. 2016, 215, 304–313. [Google Scholar] [CrossRef] [PubMed]
- Gunaseelan, V.N. Anaerobic digestion of biomass for methane production: A review. Biomass Bioenergy 1997, 13, 83–114. [Google Scholar] [CrossRef]
- Steinhauser, A.; Deublein, D. Biogas from Waste and Renewables Energy; John Wiley & Sons: Hoboken, NJ, USA, 2011; ISBN 9783527327119. [Google Scholar]
- Schievano, A.; Adani, F.; Tamone, F.; D’Imporzano, G.; Scaglia, B.; Genevini, P.L. What is the Digestate? In Anaerobic Digestion: Opportunities for Agriculture and Environment; Adani, F., Schievano, A., Boccasile, G., Eds.; University of Milan: Milan, Italy, 2009; Volume 12, pp. 7–18. [Google Scholar]
- Koszel, M.; Lorencowicz, E. Agricultural Use of Biogas Digestate as a Replacement Fertilizers. Agric. Agric. Sci. Procedia 2015, 7, 119–124. [Google Scholar] [CrossRef]
- Makádi, M.; Tomócsik, A.; Orosz, V. Digestate: A New Nutrient Source—Review. In Biogas; Kumar, S., Ed.; InTech: London, UK, 2012; Available online: http://www.intechopen.com/books/biogas/digestate-a-new-nutrient-source-r (accessed on 1 February 2021).
- Eiroa, M.; Costa, J.C.; Alves, M.M.; Kennes, C.; Veiga, M.C. Evaluation of the biomethane potential of solid fish waste. Waste Manag. 2012, 32, 1347–1352. [Google Scholar] [CrossRef]
- Nges, I.A.; Mbatia, B.; Björnsson, L. Improved utilization of fish waste by anaerobic digestion following omega-3 fatty acids extraction. J. Environ. Manag. 2012, 110, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Bücker, F.; Marder, M.; Peiter, M.R.; Lehn, D.N.; Esquerdo, V.M.; Antonio de Almeida Pinto, L.; Konrad, O. Fish waste: An efficient alternative to biogas and methane production in an anaerobic mono-digestion system. Renew. Energy 2020, 147, 798–805. [Google Scholar] [CrossRef]
- Morales-Polo, C.; Cledera-Castro, M.D.M.; Hueso-Kortekaas, K.; Revuelta-Aramburu, M. Anaerobic digestion in wastewater reactors of separated organic fractions from wholesale markets waste. Compositional and batch characterization. Energy and environmental feasibility. Sci. Total Environ. 2020, 726, 138567. [Google Scholar] [CrossRef] [PubMed]
- Ivanovs, K.; Spalvins, K.; Blumberga, D. Approach for modelling anaerobic digestion processes of fish waste. Energy Procedia 2018, 147, 390–396. [Google Scholar] [CrossRef]
- Chen, Y.; Cheng, J.J.; Creamer, K.S. Inhibition of anaerobic digestion process: A review. Bioresour. Technol. 2008, 99, 4044–4064. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, B.; Flotats, X. Effect of limonene on batch anaerobic digestion of citrus peel waste. Biochem. Eng. J. 2016, 109, 9–18. [Google Scholar] [CrossRef]
- Ruiz, B.; Flotats, X. Citrus essential oils and their influence on the anaerobic digestion process: An overview. Waste Manag. 2014, 34, 2063–2079. [Google Scholar] [CrossRef]
- Calabrò, P.S.; Pontoni, L.; Porqueddu, I.; Greco, R.; Pirozzi, F.; Malpei, F. Effect of the concentration of essential oil on orange peel waste biomethanization: Preliminary batch results. Waste Manag. 2016, 48, 440–447. [Google Scholar] [CrossRef]
- Malara, A.; Paone, E.; Frontera, P.; Bonaccorsi, L.; Panzera, G.; Mauriello, F. Sustainable exploitation of coffee silverskin in water remediation. Sustainability 2018, 10, 3547. [Google Scholar] [CrossRef]
- Calabrò, P.S.; Fazzino, F.; Folino, A.; Paone, E.; Komilis, D. Semi-Continuous Anaerobic Digestion of Orange Peel Waste: Effect of Activated Carbon Addition and Alkaline Pretreatment on the Process. Sustainability 2019, 11, 3386. [Google Scholar] [CrossRef]
- Paone, E.; Beneduci, A.; Corrente, G.A.; Malara, A.; Mauriello, F. Hydrogenolysis of aromatic ethers under lignin-first conditions. Mol. Catal. 2020, 497, 111228. [Google Scholar] [CrossRef]
- Gumina, B.; Espro, C.; Galvagno, S.; Pietropaolo, R.; Mauriello, F. Bioethanol Production from Unpretreated Cellulose under Neutral Selfsustainable Hydrolysis/Hydrogenolysis Conditions Promoted by the Heterogeneous Pd/Fe3O4 Catalyst. ACS Omega 2019, 4, 352–357. [Google Scholar] [CrossRef] [PubMed]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 22nd ed.; American Public Health Association, American Water Works Association, Water Environment Federatio: Washington, DC, USA, 2012; ISBN 9780875530130. [Google Scholar]
- Calabro, P.S.; Panzera, M.F. Biomethane production tests on ensiled orange peel waste. Int. J. Heat Technol. 2017, 35, S130–S136. [Google Scholar] [CrossRef]
- Holliger, C.; Alves, M.; Andrade, D.; Angelidaki, I.; Astals, S.; Baier, U.; Bougrier, C.; Buffière, P.; Carballa, M.; De Wilde, V.; et al. Towards a standardization of biomethane potential tests. Water Sci. Technol. 2016, 74, 2515–2522. [Google Scholar] [CrossRef]
- Donoso-Bravo, A.; Pérez-Elvira, S.I.; Fdz-Polanco, F. Application of simplified models for anaerobic biodegradability tests. Evaluation of pre-treatment processes. Chem. Eng. J. 2010, 160, 607–614. [Google Scholar] [CrossRef]
- Liebetrau, J.; Pfeiffer, D.; Thrän, D. (Eds.) Collection of Measurement Methods for Biogas—Methods to Determine Parameters for Analysis Purposes and Parameters That Describe Processes in the Biogas Sector; Series of the Funding Programme “Biomass Energy Use”; Deutsches Biomasseforschungszentrum Gemeinnützige GmbH: Leipzig, Germany, 2016; Volume 7, ISSN 2364-897X. Available online: https://www.energetische-biomassenutzung.de/fileadmin/user_upload/Downloads/Ver%C3%B6_entlichungen/07_MMS_Biogas_en_web.pdf (accessed on 1 February 2021).
- Mézes, L.; Tamas, J.; Borbely, J. Novel approach of the basis of FOS/TAC method. An. Univ. Oradea Fasc. Prot. Mediu. 2011, 17, 713–718. [Google Scholar]
- Lozano-Bilbao, E.; Lozano, G.; Jiménez, S.; Jurado-Ruzafa, A.; Hardisson, A.; Rubio, C.; Weller, D.G.; Paz, S.; Gutiérrez, Á.J. Ontogenic and seasonal variations of metal content in a small pelagic fish (Trachurus picturatus) in northwestern African waters. Mar. Pollut. Bull. 2020, 156, 111251. [Google Scholar] [CrossRef] [PubMed]
- Naga, S.M.; El-Maghraby, H.F.; Mahmoud, E.M.; Talaat, M.S.; Ibrhim, A.M. Preparation and characterization of highly porous ceramic scaffolds based on thermally treated fish bone. Ceram. Int. 2015, 41, 15010–15016. [Google Scholar] [CrossRef]
- Vivekanand, V.; Mulat, D.G.; Eijsink, V.G.H.; Horn, S.J. Synergistic effects of anaerobic co-digestion of whey, manure and fish ensilage. Bioresour. Technol. 2018, 249, 35–41. [Google Scholar] [CrossRef]
- Jeung, J.H.; Chung, W.J.; Chang, S.W. Evaluation of anaerobic co-digestion to enhance the efficiency of livestock manure anaerobic digestion. Sustainability 2019, 11, 7170. [Google Scholar] [CrossRef]
- Sayara, T.; Sánchez, A. A review on anaerobic digestion of lignocellulosic wastes: Pretreatments and operational conditions. Appl. Sci. 2019, 9, 4655. [Google Scholar] [CrossRef]
- The EU Fish Market—EUMOFA. 2020. Available online: https://www.eumofa.eu (accessed on 1 February 2021).



| Substrate | Batch | Substrate [g] | TS Mix | pH Mix |
|---|---|---|---|---|
| -(blank) | 1 | - | 2.5% | 7.38 |
| 2 | 7.42 | |||
| Cellulose (control) | 3 | 2.1 | 3.1% | 7.50 |
| 4 | 7.47 | |||
| SAS | 5 | 2.7 | 3.3% | 7.39 |
| 6 | 7.30 | |||
| 7 | 7.41 |
| pH | TS [%] | VS [%TS] | COD [mgO2·gTS−1] | C/N | d-Limonene [mg·gTS−1] | |
|---|---|---|---|---|---|---|
| Inoculum | 7.50 | 5.0 ± 0.10 | 76.3 ± 0.18 | - | - | - |
| Cellulose | - | 95.6 | 100 | 1185 * | - | - |
| SAS | 6.30 | 98.0 ± 0.15 | 77.1 ± 0.27 | 918.3 | 4 | 5 |
| Substrate | Batch | Biogas [mL·gVS−1] | Average [mL·gVS−1] | BMP [mLCH4·gVS−1] | Average [mLCH4·gVS−1] | Average Methane Content |
|---|---|---|---|---|---|---|
| Cellulose (control) | 3 | 603.4 | 598.3 | 396.5 | 390.0 | 68% |
| 4 | 593.3 | 382.6 | 65% | |||
| SAS | 5 | 406.4 | 378.5 | 296.1 | 278.0 | 72% |
| 6 | 381.7 | 281.3 | 73% | |||
| 7 | 347.4 | 256.4 | 73% |
| P [mL·gVS−1] | Rm [mL·gVS−1·d−1] | λ [d] | r2 |
|---|---|---|---|
| 268.7 | 24.8 | 3.145 | 0.997 |
| Property | Blank | Cellulose | SAS |
|---|---|---|---|
| pH | 7.1 ± 0.06 | 7.0 ± 0.01 | 7.3 ± 0.00 |
| TS | 2.4 ± 0.03% | 2.5 ± 0.02% | 2.7 ± 0.01% |
| VS | 71.2 ± 0.19% | 72.1 ± 0.18% | 69.3 ± 0.57% |
| TAN [mg·L−1] | 234 ± 8.8 | 173 ± 8.0 | 697 ± 20.3 |
| Cl- [mg·L−1] | 1263 ± 17.5 | 1105 ± 55.0 | 1297 ± 107.3 |
| VFA [mgHAC·L−1] | 297.3 ± 14.11 | 290.4 ± 21.04 | 411.1 ± 28.09 |
| FOS/TAC [gHAC·gCaCO3−1] | 0.08 ± 0.001 | 0.09 ± 0.010 | 0.09 ± 0.008 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paone, E.; Fazzino, F.; Pizzone, D.M.; Scurria, A.; Pagliaro, M.; Ciriminna, R.; Calabrò, P.S. Towards the Anchovy Biorefinery: Biogas Production from Anchovy Processing Waste after Fish Oil Extraction with Biobased Limonene. Sustainability 2021, 13, 2428. https://doi.org/10.3390/su13052428
Paone E, Fazzino F, Pizzone DM, Scurria A, Pagliaro M, Ciriminna R, Calabrò PS. Towards the Anchovy Biorefinery: Biogas Production from Anchovy Processing Waste after Fish Oil Extraction with Biobased Limonene. Sustainability. 2021; 13(5):2428. https://doi.org/10.3390/su13052428
Chicago/Turabian StylePaone, Emilia, Filippo Fazzino, Daniela Maria Pizzone, Antonino Scurria, Mario Pagliaro, Rosaria Ciriminna, and Paolo Salvatore Calabrò. 2021. "Towards the Anchovy Biorefinery: Biogas Production from Anchovy Processing Waste after Fish Oil Extraction with Biobased Limonene" Sustainability 13, no. 5: 2428. https://doi.org/10.3390/su13052428
APA StylePaone, E., Fazzino, F., Pizzone, D. M., Scurria, A., Pagliaro, M., Ciriminna, R., & Calabrò, P. S. (2021). Towards the Anchovy Biorefinery: Biogas Production from Anchovy Processing Waste after Fish Oil Extraction with Biobased Limonene. Sustainability, 13(5), 2428. https://doi.org/10.3390/su13052428










