The Effects of Total Dissolved Carbon Dioxide on the Growth Rate, Biochemical Composition, and Biomass Productivity of Nonaxenic Microalgal Polyculture
Abstract
1. Introduction
2. Materials and Methods
2.1. Microalgal Polyculture and Culture Medium
2.2. Experimental System
2.3. Determination of Total Dissolved Carbon Dioxide
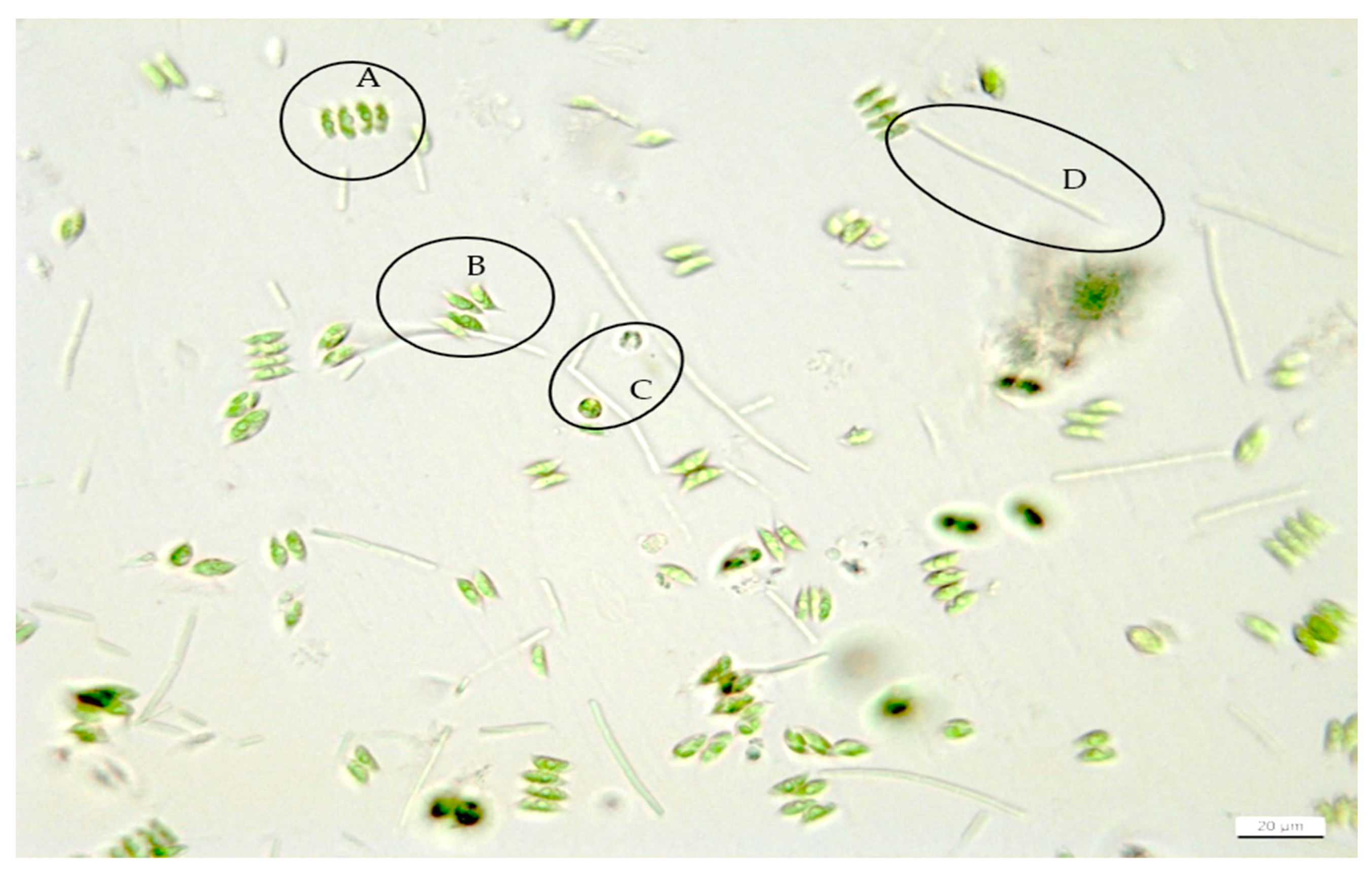
2.4. Microscopy and Morphological Analyses
2.5. Measurement of Growth Parameters
2.6. Biochemical Characterisation of Microalgal Cells
2.7. Statistical Analysis
3. Results
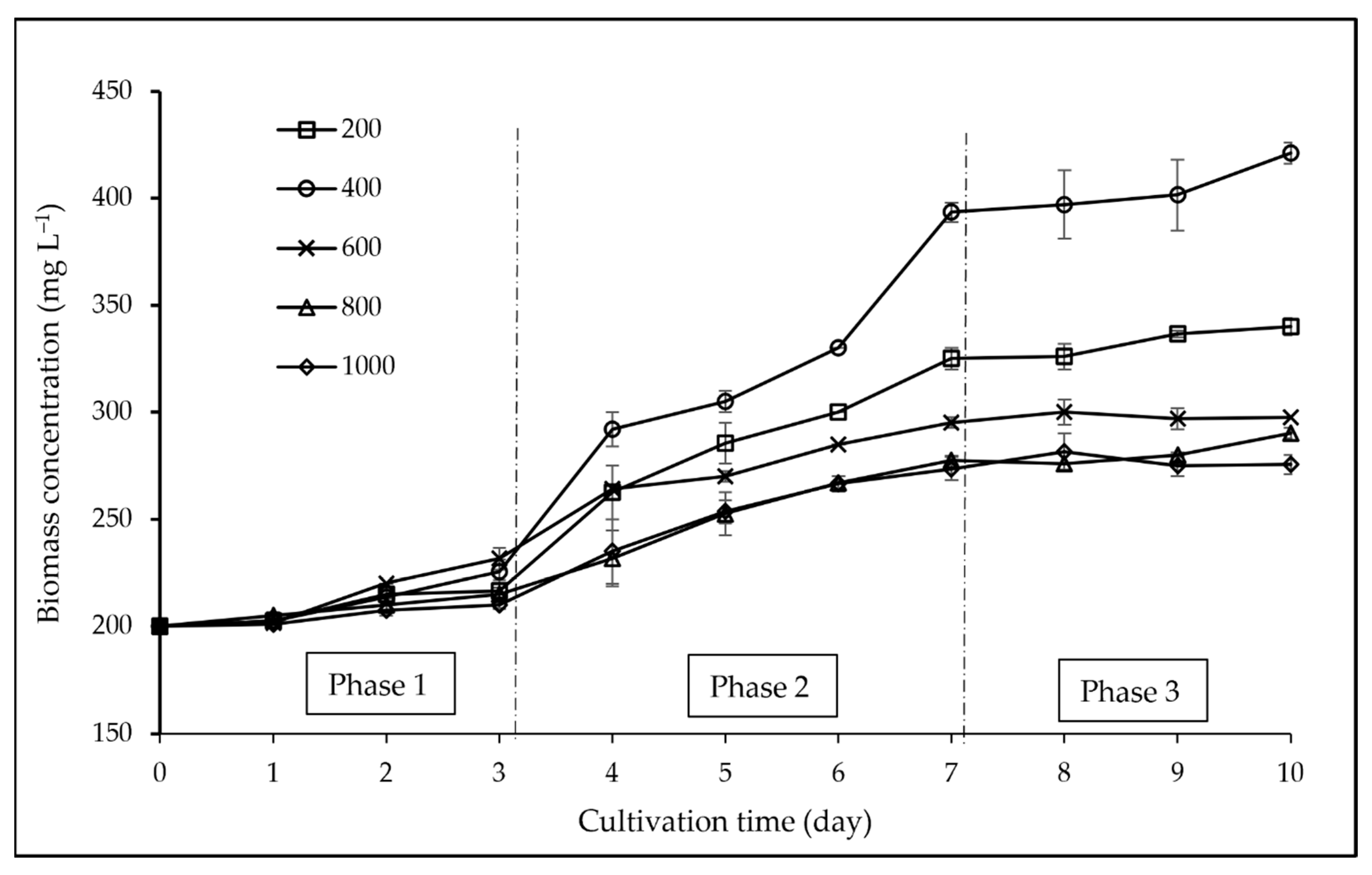
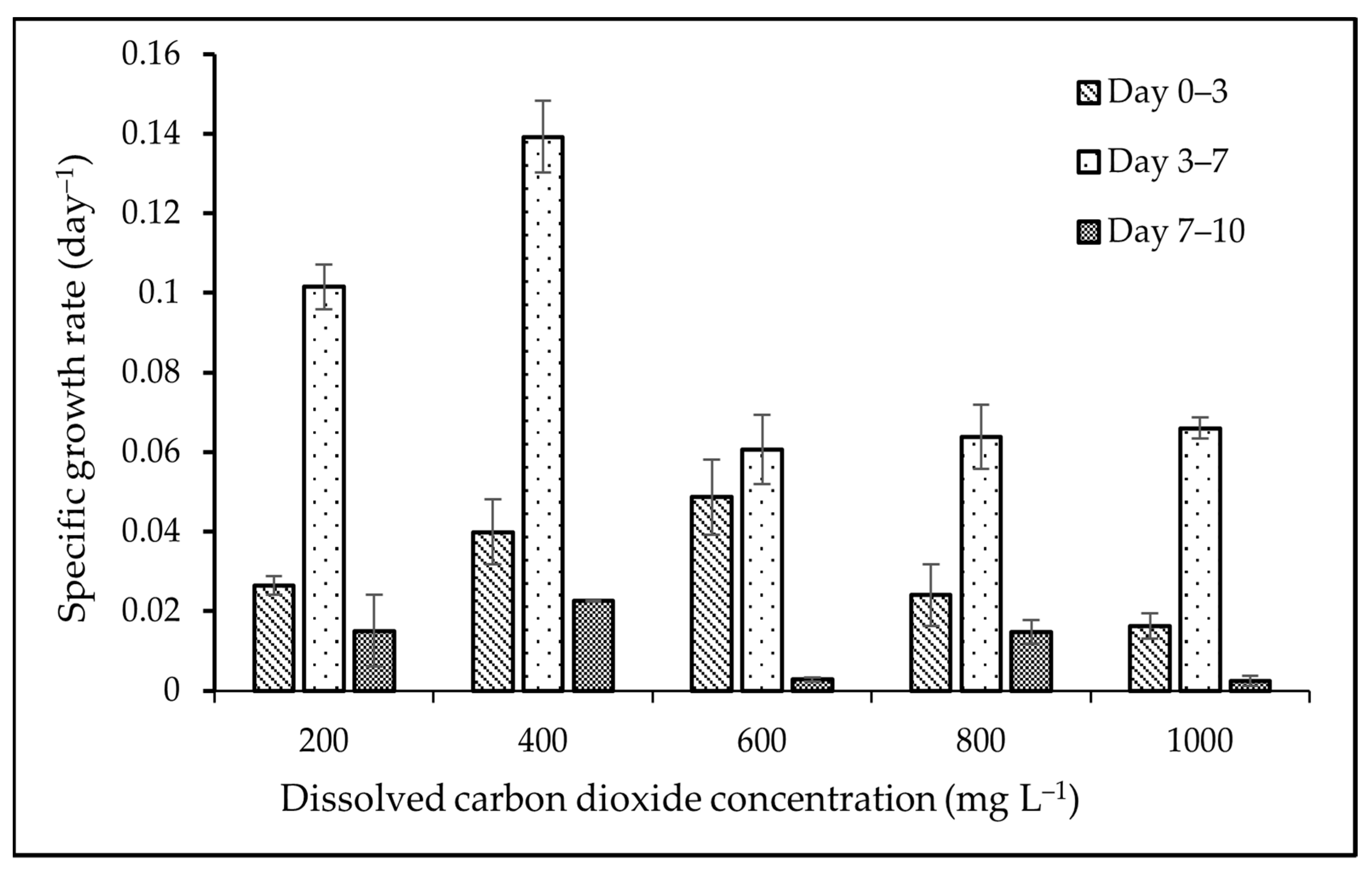
3.1. Biomass and Growth Analyses
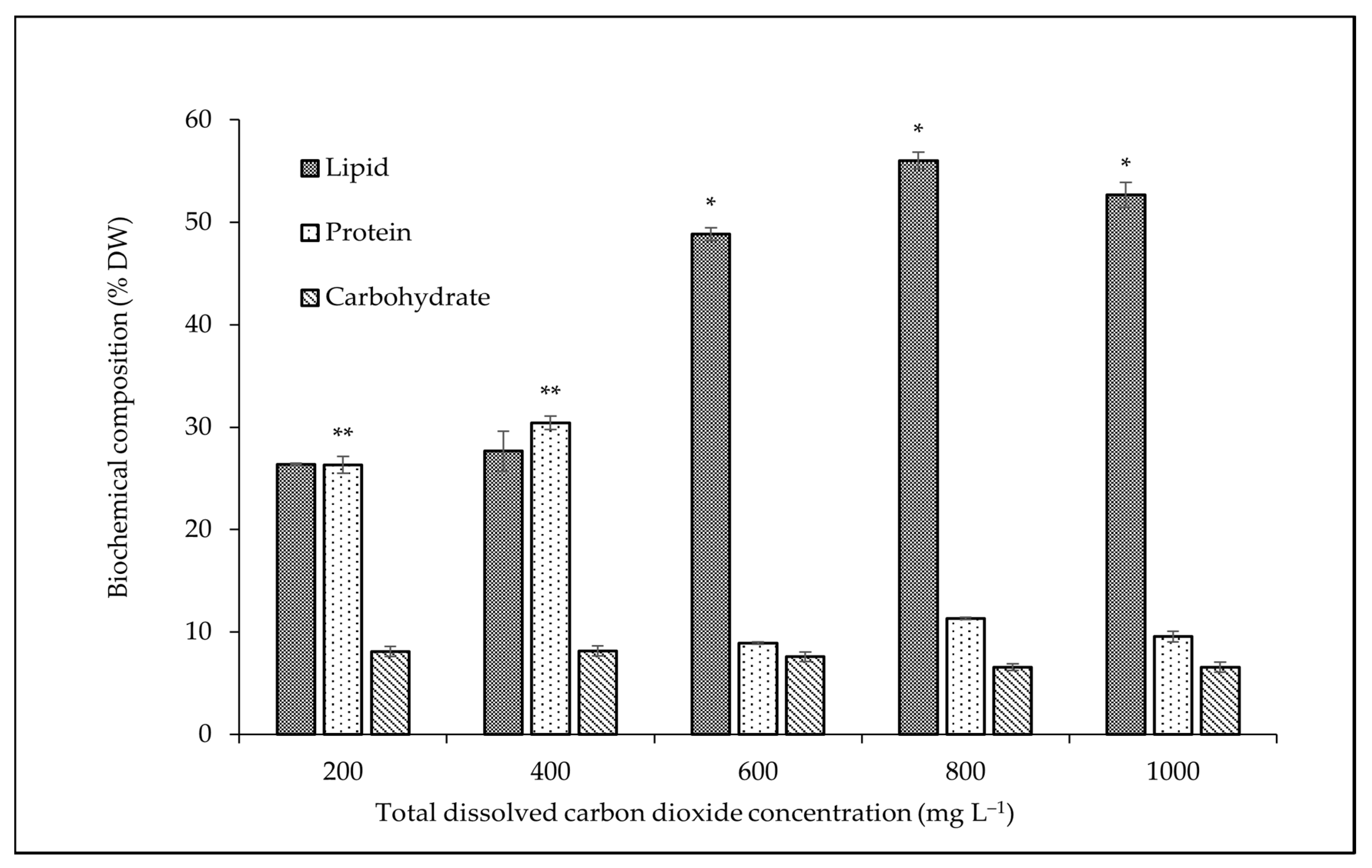
3.2. Effect of CO2 Concentration on Biochemical Composition
4. Discussion
4.1. Biomass and Growth Analyses
4.2. Effect of CO2 Concentration on Biochemical Composition
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- United States Environmental Protection Agency. Sources of Greenhouse Gas Emissions|Greenhouse Gas (GHG) Emissions. US EPA. Available online: https://www.epa.gov/ghgemissions/sources-greenhouse-gas-emissions (accessed on 27 July 2020).
- Nam, K.; Lee, S.; Jeon, H. Nonlinearity between CO2 emission and economic development: Evidence from a functional coefficient panel approach. Sustainability 2020, 12, 258. [Google Scholar] [CrossRef]
- Benemann, J.R.; Oswald, W.J. Systems and Economic Analysis of Microalgae Ponds for Conversion of Carbon Dioxide to Biomass; Final Report; U.S. Department of Energy Office of Scientific and Technical Information: Washington, DC, USA, 1996. [Google Scholar] [CrossRef]
- Hillier, J.; Hawes, C.; Squire, G.; Hilton, A.; Wale, S.; Smith, P. The carbon footprints of food crop production. Int. J. Agric. Sustain. 2009, 7, 107–118. [Google Scholar] [CrossRef]
- Kumar, K.; Mishra, S.K.; Shrivastav, A.; Park, M.S.; Yang, J.W. Recent trends in the mass cultivation of algae in raceway ponds. Renew. Sustain. Energy Rev. 2015, 51, 875–885. [Google Scholar] [CrossRef]
- Chiu, S.Y.; Kao, C.Y.; Chen, T.Y.; Chang, Y.B.; Kuo, C.M.; Lin, C.S. Cultivation of microalgal Chlorella for biomass and lipid production using wastewater as nutrient resource. Bioresour. Technol. 2015, 184, 179–189. [Google Scholar] [CrossRef]
- Wu, Y.H.; Hu, H.Y.; Yu, Y.; Zhang, T.Y.; Zhu, S.F.; Zhuang, L.L.; Zhang, X.; Lu, Y. Microalgal species for sustainable biomass/lipid production using wastewater as resource: A review. Renew. Sustain. Energy Rev. 2014, 33, 675–688. [Google Scholar] [CrossRef]
- Narwani, A.; Lashaway, A.R.; Hietala, D.C.; Savage, P.E.; Cardinale, B.J. Power of Plankton: Effects of Algal Biodiversity on Biocrude Production and Stability. Environ. Sci. Technol. 2016, 50, 13142–13150. [Google Scholar] [CrossRef] [PubMed]
- Shurin, J.B.; Abbott, R.L.; Deal, M.S.; Kwan, G.T.; Litchman, E.; Mcbride, R.C.; Mandal, S.; Smith, V.H. Industrial-strength ecology: Trade-offs and opportunities in algal biofuel production. Ecol. Lett. 2013, 16, 1393–1404. [Google Scholar] [CrossRef]
- Stockenreiter, M.; Graber, A.K.; Haupt, F.; Stibor, H. The effect of species diversity on lipid production by micro-algal communities. J. Appl. Phycol. 2012, 24, 45–54. [Google Scholar] [CrossRef]
- Wan, C.; Alam, M.A.; Zhao, X.Q.; Zhang, X.Y.; Guo, S.L.; Ho, S.H.; Chang, J.S.; Bai, F.W. Current progress and future prospect of microalgal biomass harvest using various flocculation technologies. Bioresour. Technol. 2015, 184, 251–257. [Google Scholar] [CrossRef]
- Watanabe, K.; Takihana, N.; Aoyagi, H.; Hanada, S.; Watanabe, Y.; Ohmura, N.; Saiki, H.; Tanaka, H. Symbiotic association in Chlorella culture. FEMS Microbiol. Ecol. 2005, 51, 187–196. [Google Scholar] [CrossRef] [PubMed]
- Sasongko, N.A.; Noguchi, R.; Ito, J.; Demura, M.; Ichikawa, S.; Nakajima, M.; Watanabe, M.M. Engineering study of a pilot scale process plant for microalgae-oil production utilizing municipal wastewater and flue gases: Fukushima pilot plant. Energies 2018, 11, 1693. [Google Scholar] [CrossRef]
- Padmaperuma, G.; Kapoore, R.V.; Gilmour, D.J.; Vaidyanathan, S. Microbial consortia: A critical look at microalgae co-cultures for enhanced biomanufacturing. Crit. Rev. Biotechnol. 2018, 38, 690–703. [Google Scholar] [CrossRef]
- Godwin, C.M.; Lashaway, A.R.; Hietala, D.C.; Savage, P.E.; Cardinale, B.J. Biodiversity improves the ecological design of sustainable biofuel systems. GCB Bioenergy 2018, 10, 752–765. [Google Scholar] [CrossRef]
- Demura, M.; Yoshida, M.; Yokoyama, A.; Ito, J.; Kobayashi, H.; Kayano, S.; Tamagawa, Y.; Watanobe, M.; Date, N.; Osaka, M.; et al. Biomass productivity of native algal communities in Minamisoma city, Fukushima Prefecture, Japan. Algal Res. 2018, 29, 22–35. [Google Scholar] [CrossRef]
- Riebesell, U.; Wolf-Gladrow, D.A.; Smetacek, V. Carbon dioxide limitation of marine phytoplankton growth rates. Nature 1993, 361, 249–251. [Google Scholar] [CrossRef]
- Shurin, J.B.; Mandal, S.; Abbott, R.L. Trait diversity enhances yield in algal biofuel assemblages. J. Appl. Ecol. 2014, 51, 603–611. [Google Scholar] [CrossRef]
- DuBois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric Method for Determination of Sugars and Related Substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- López, C.V.G.; del Carmen Cerón García, M.; Fernández, F.G.A.; Bustos, C.S.; Chisti, Y.; Sevilla, J.M.F. Protein measurements of microalgal and cyanobacterial biomass. Bioresour. Technol. 2010, 101, 7587–7591. [Google Scholar] [CrossRef] [PubMed]
- Folch, J.; Lees, M.; Stanley, G.H. A Simple Method for the Isolation and Purification of Total Lipides from Animal Tissues. Cancer 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Kumar, A.; Ergas, S.; Yuan, X.; Sahu, A.; Zhang, Q.; Dewulf, J.; Malcata, F.X.; van Langenhove, H. Enhanced CO2 fixation and biofuel production via microalgae: Recent developments and future directions. Trends Biotechnol. 2010, 28, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Spalding, M.H. Microalgal carbon-dioxide-concentrating mechanisms: Chlamydomonas inorganic carbon transporters. J. Exp. Bot. 2008, 59, 1463–1473. [Google Scholar] [CrossRef] [PubMed]
- Anjos, M.; Fernandes, B.D.; Vicente, A.A.; Teixeira, J.A.; Dragone, G. Optimization of CO2 bio-mitigation by Chlorella vulgaris. Bioresour. Technol. 2013, 139, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, C.; Hu, H. Screening High CO2-Tolerant Oleaginous Microalgae from Genera Desmodesmus and Scenedesmus. Appl. Biochem. Biotechnol. 2020, 192, 211–229. [Google Scholar] [CrossRef]
- Lohman, E.J.; Gardner, R.D.; Pedersen, T.; Peyton, B.M.; Cooksey, K.E.; Gerlach, R. Optimized inorganic carbon regime for enhanced growth and lipid accumulation in Chlorella vulgaris Luisa Gouveia. Biotechnol. Biofuels 2015, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Su, Y. Process effect of microalgal-carbon dioxide fixation and biomass production: A review. Renew. Sustain. Energy Rev. 2014, 31, 121–132. [Google Scholar] [CrossRef]
- Mangan, N.; Brenner, M. Systems analysis of the CO2 concentrating mechanism in cyanobacteria. Elife 2014, 3, e02043. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.-G.; Long, S.P.; Ort, D.R. Improving Photosynthetic Efficiency for Greater Yield. Annu. Rev. Plant Biol. 2010, 61, 235–261. [Google Scholar] [CrossRef] [PubMed]
- Bowes, G. Growth at elevated CO2: Photosynthetic responses mediated through Rubisco. Plant Cell Environ. 1991, 14, 795–806. [Google Scholar] [CrossRef]
- Yadav, R.R.; Krishnamurthi, K.; Mudliar, S.N.; Devi, S.S.; Naoghare, P.K.; Bafana, A.; Chakrabarti, T. Carbonic anhydrase mediated carbon dioxide sequestration: Promises, challenges and future prospects. J. Basic Microbiol. 2014, 54, 472–481. [Google Scholar] [CrossRef]
- Sültemeyer, D. Carbonic anhydrase in eukaryotic algae: Characterization, regulation, and possible function during photosynthesis. Can. J. Bot. 1998, 76, 962–972. [Google Scholar] [CrossRef]
- Lee, J.S.; Lee, J.P. Review of advances in biological CO2 mitigation technology. Biotechnol. Bioprocess Eng. 2003, 8, 354–359. [Google Scholar] [CrossRef]
- Contreras-Angulo, J.R.; Mata, T.M.; Cuellar-Bermudez, S.P.; Caetano, N.S.; Chandra, R.; Garcia-Perez, J.S.; Muylaert, K.; Parra-Saldivar, R. Symbiotic co-culture of Scenedesmus sp. and Azospirillum brasilense on N-deficient media with biomass production for biofuels. Sustainability 2019, 11, 707. [Google Scholar] [CrossRef]
- El-Katony, T.M.; El-Adl, M.F. Salt response of the freshwater microalga Scenedesmus obliquus (Turp.) Kutz is modulated by the algal growth phase. J. Oceanol. Limnol. 2020, 38, 802–815. [Google Scholar] [CrossRef]
- Ali, I.; Naqvi, S.R.; Bahadar, A. Kinetic analysis of Botryococcus braunii pyrolysis using model-free and model fitting methods. Fuel 2018, 214, 369–380. [Google Scholar] [CrossRef]
- Basu, S.; Roy, A.S.; Mohanty, K.; Ghoshal, A.K. CO2 biofixation and carbonic anhydrase activity in Scenedesmus obliquus SA1 cultivated in large scale open system. Bioresour. Technol. 2014, 164, 323–330. [Google Scholar] [CrossRef] [PubMed]
- Varshney, P.; Beardall, J.; Bhattacharya, S.; Wangikar, P.P. Effect of elevated carbon dioxide and nitric oxide on the physiological responses of two green algae, Asterarcys quadricellulare and Chlorella sorokiniana. J. Appl. Phycol. 2020, 32, 189–204. [Google Scholar] [CrossRef]
- Arora, N.; Pienkos, P.T.; Pruthi, V.; Poluri, K.M.; Guarnieri, M.T. Leveraging algal omics to reveal potential targets for augmenting TAG accumulation. Biotechnol. Adv. 2018, 36, 1274–1292. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Zhu, J.; Wakisaka, M. Effect of protocatechuic acid on Euglena gracilis growth and accumulation of metabolites. Sustainability 2020, 12, 9158. [Google Scholar] [CrossRef]
- Sake, C.L.; Metcalf, A.J.; Boyle, N.R. The challenge and potential of photosynthesis: Unique considerations for metabolic flux measurements in photosynthetic microorganisms. Biotechnol. Lett. 2019, 41, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Livne, A.; Sukenik, A. Lipid Synthesis and Abundance of Acetyl CoA Carboxylase in Isochrysis galbana (Prymnesiophyceae) Following Nitrogen Starvation. Plant Cell Physiol. 1992, 33, 1175–1181. [Google Scholar] [CrossRef]
| Tco2 (mg L−1) | Increase in Biomass (%) | |||
|---|---|---|---|---|
| Phase 1 | Phase 2 | Phase 3 | Overall | |
| 200 | 8.25 | 50.12 | 4.62 * | 70.00 |
| 400 | 12.75 | 74.50 * | 6.99 | 110.50 * |
| 600 | 15.75 * | 27.43 | 0.85 | 48.75 |
| 800 | 7.50 | 29.07 | 4.50 | 45.00 |
| 1000 | 5.00 | 30.24 | 0.73 | 37.75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kandasamy, L.C.; Neves, M.A.; Demura, M.; Nakajima, M. The Effects of Total Dissolved Carbon Dioxide on the Growth Rate, Biochemical Composition, and Biomass Productivity of Nonaxenic Microalgal Polyculture. Sustainability 2021, 13, 2267. https://doi.org/10.3390/su13042267
Kandasamy LC, Neves MA, Demura M, Nakajima M. The Effects of Total Dissolved Carbon Dioxide on the Growth Rate, Biochemical Composition, and Biomass Productivity of Nonaxenic Microalgal Polyculture. Sustainability. 2021; 13(4):2267. https://doi.org/10.3390/su13042267
Chicago/Turabian StyleKandasamy, Lenin C., Marcos A. Neves, Mikihide Demura, and Mitsutoshi Nakajima. 2021. "The Effects of Total Dissolved Carbon Dioxide on the Growth Rate, Biochemical Composition, and Biomass Productivity of Nonaxenic Microalgal Polyculture" Sustainability 13, no. 4: 2267. https://doi.org/10.3390/su13042267
APA StyleKandasamy, L. C., Neves, M. A., Demura, M., & Nakajima, M. (2021). The Effects of Total Dissolved Carbon Dioxide on the Growth Rate, Biochemical Composition, and Biomass Productivity of Nonaxenic Microalgal Polyculture. Sustainability, 13(4), 2267. https://doi.org/10.3390/su13042267







