Sneaker, Dweller and Commuter: Evaluating Fish Behavior in Net-Based Monitoring at Hydropower Plants—A Case Study on Brown Trout (Salmo trutta)
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites and Model Fish Tested
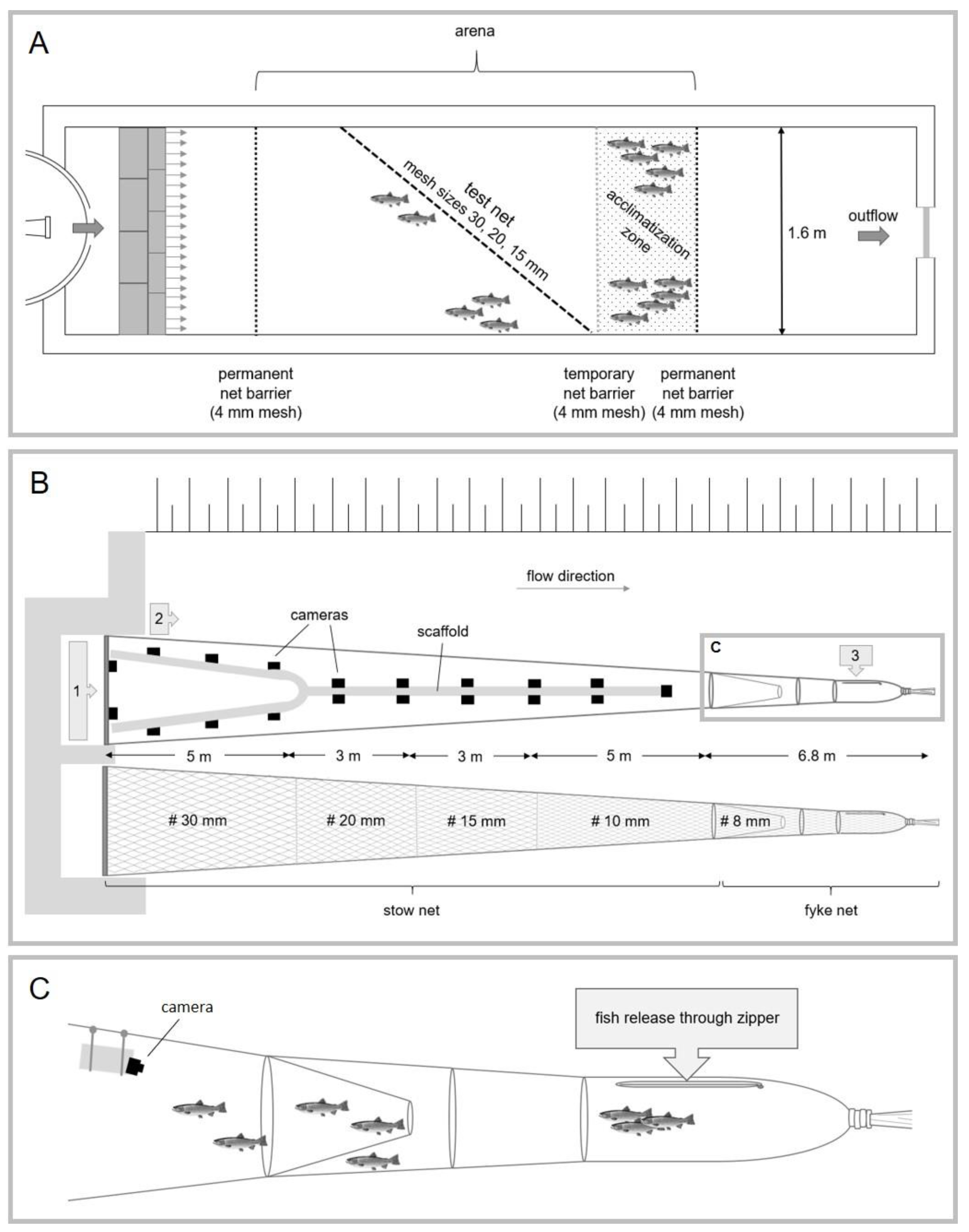
2.2. Experimental Setups
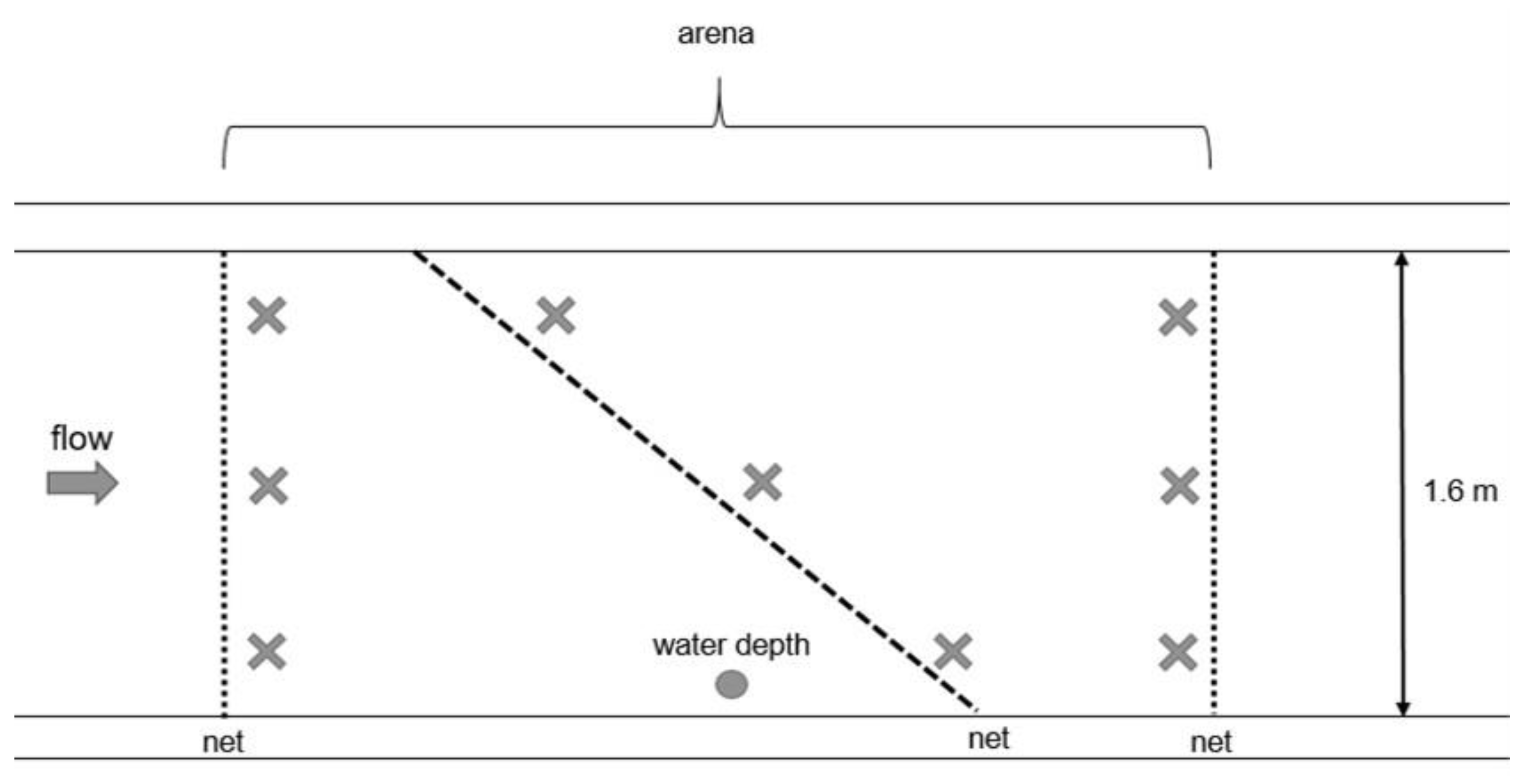
2.2.1. Fall-Through Experiment
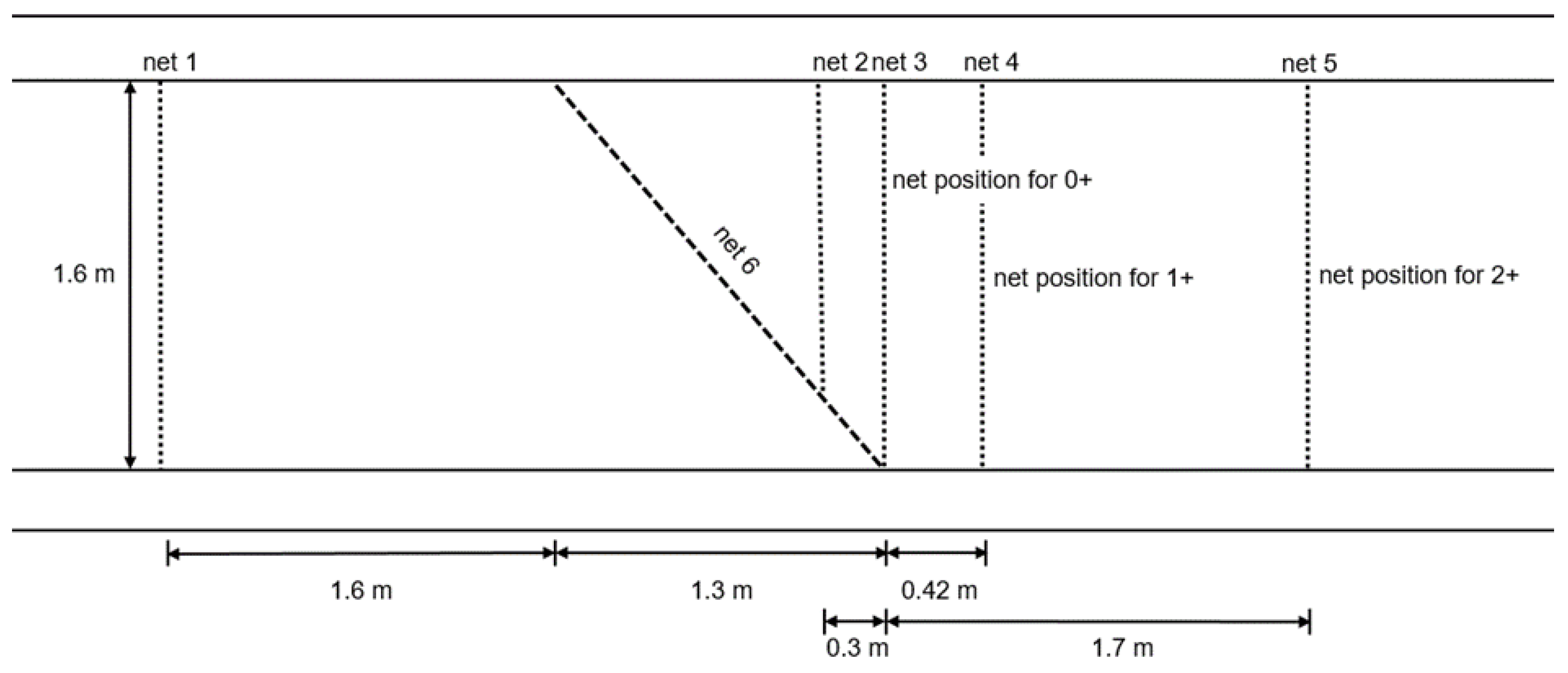
2.2.2. Net-Perception Experiment
2.2.3. Stow-Fyke-Net Experiment
2.3. Data Analysis
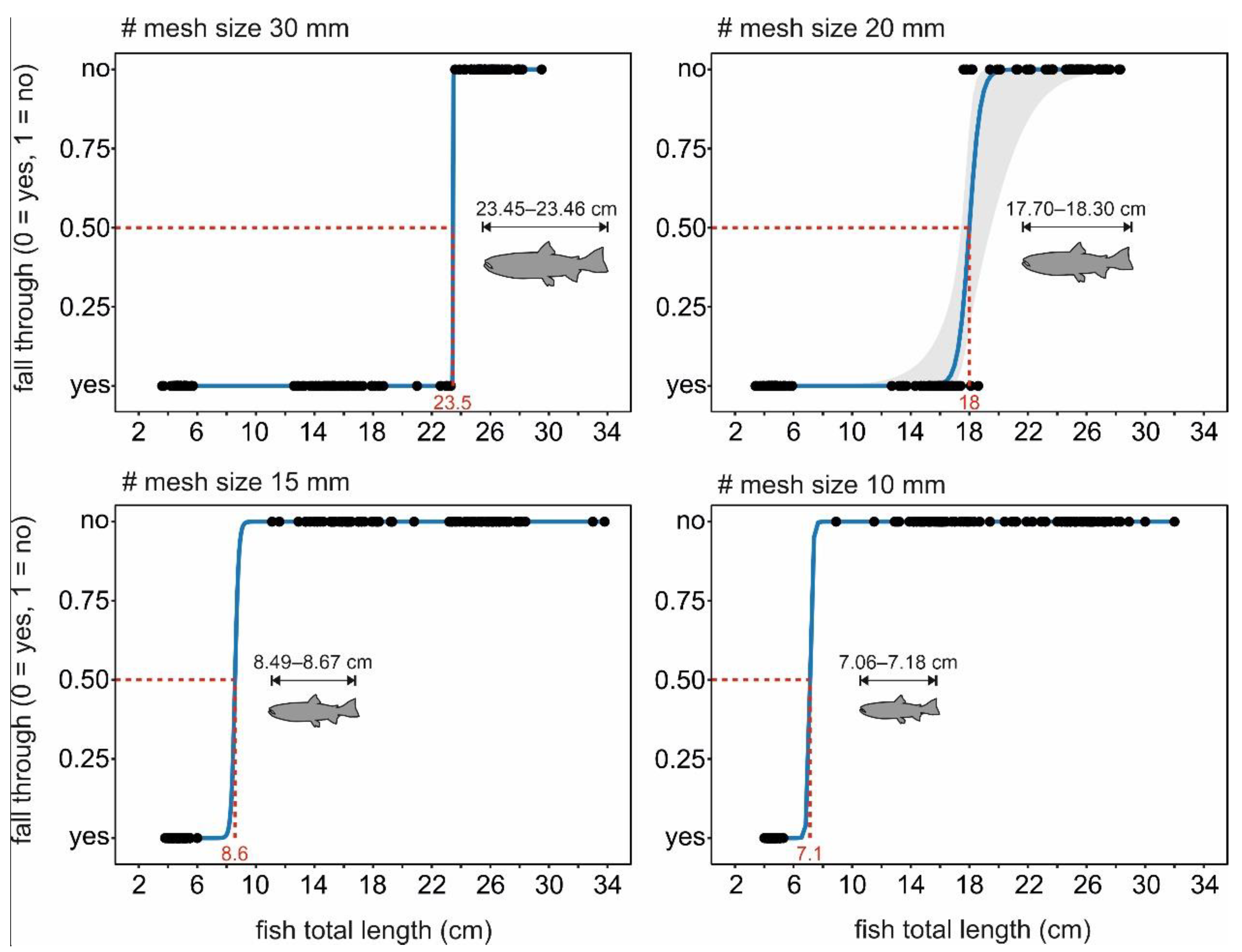
2.3.1. Fall-Through Experiment
2.3.2. Net-Perception Experiment
2.3.3. Stow-Fyke-Net Experiment
3. Results
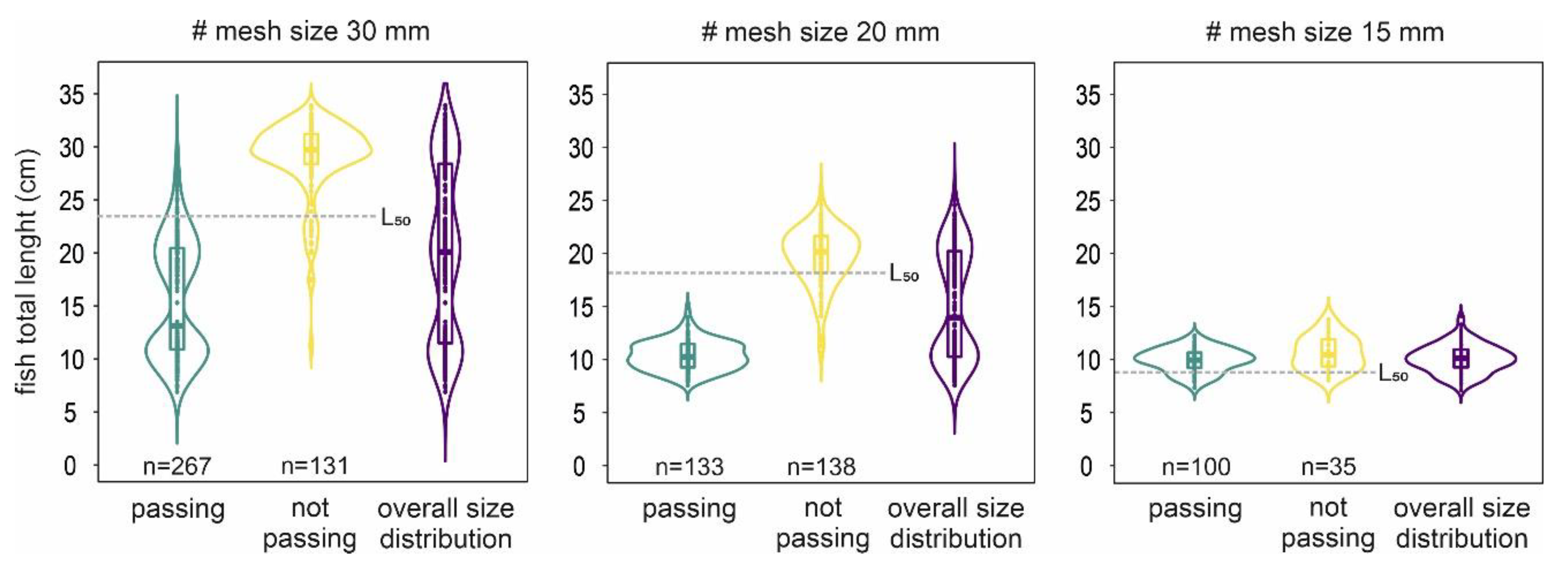
3.1. Fall-Through Experiment
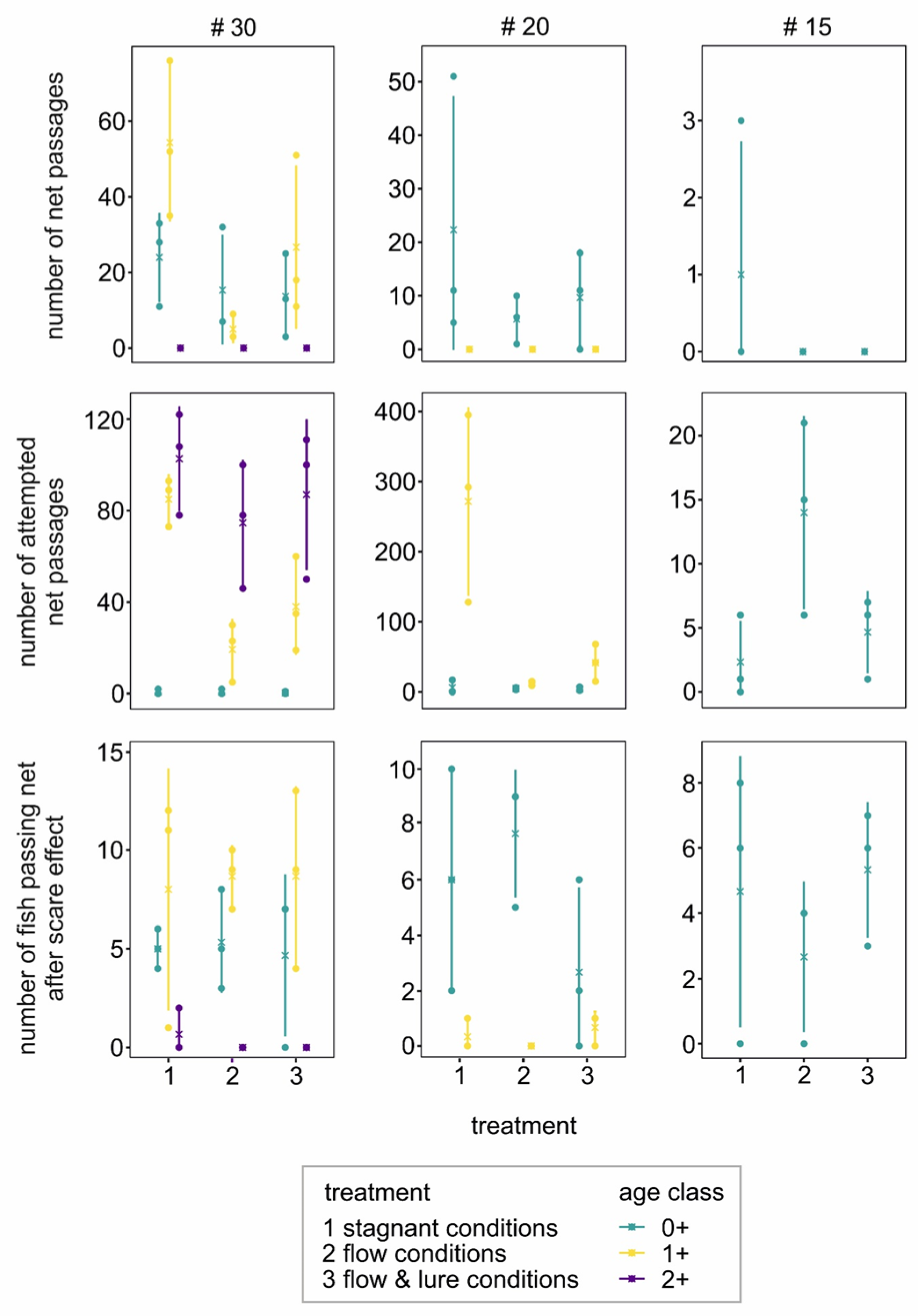
3.2. Net-Perception Experiment
3.3. Stow-Fyke-Net Experiment
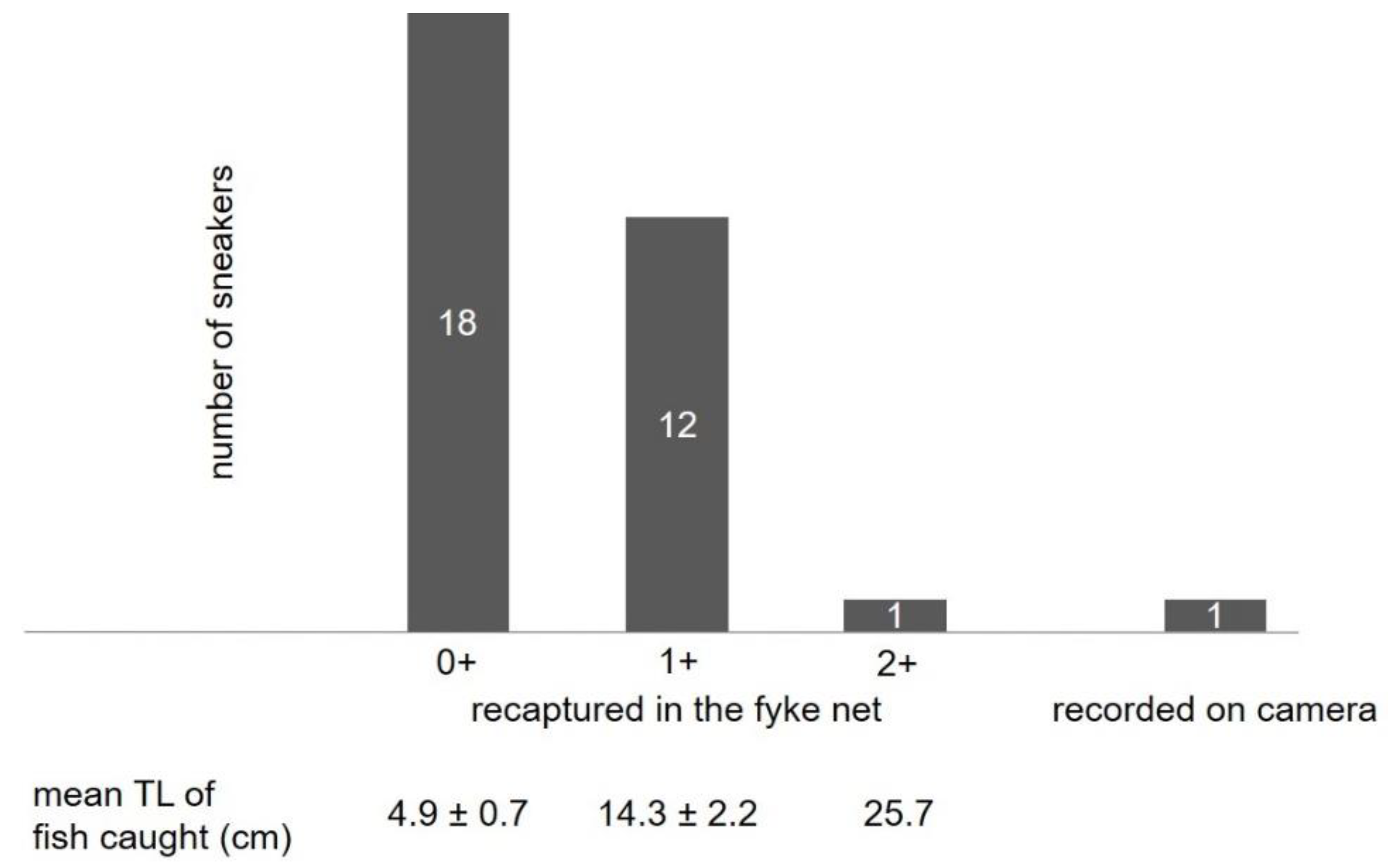
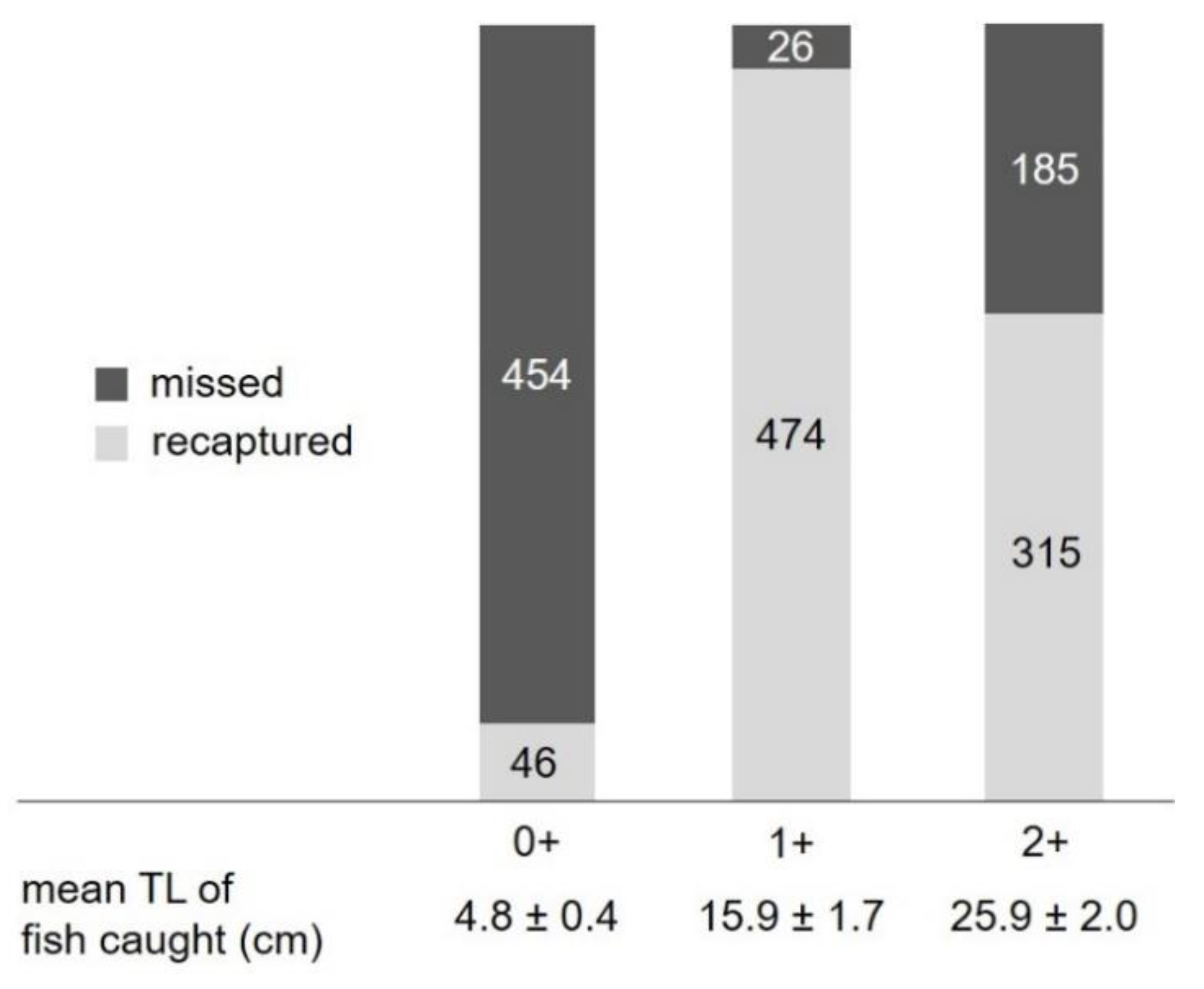
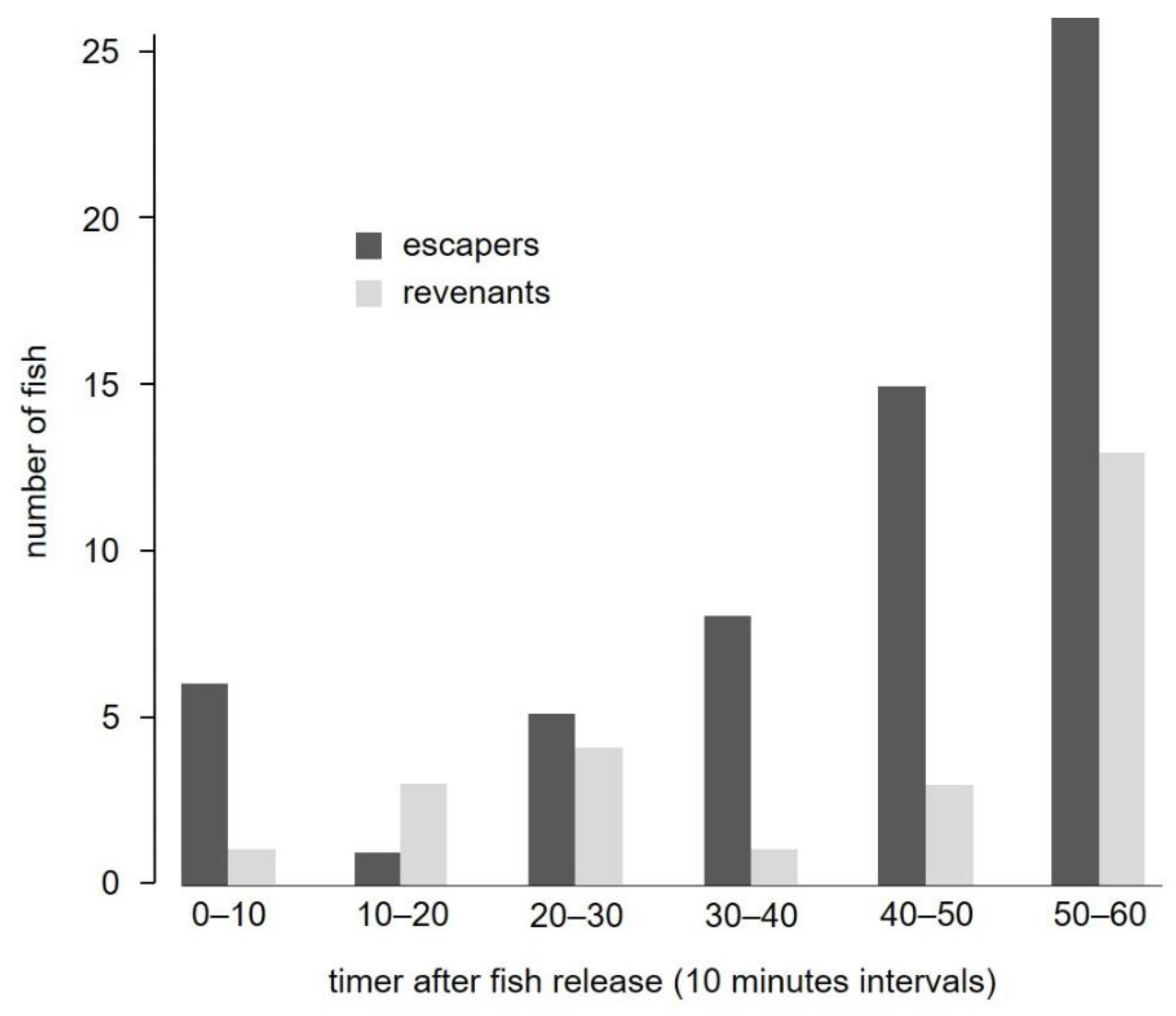
3.3.1. Fish Behavior in the Stow Net
3.3.2. Fish Behavior in the Fyke Net
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Parameter | Mean ± SD |
|---|---|
| temperature (°C) | 15.6 ± 1.0 |
| dissolved oxygen (mg·L−1) | 9.3 ± 0.7 |
| electric conductivity (μS·cm−1) | 780.2 ± 12.3 |
| pH value | 8 ± 0 |
| flow velocity (cm·s−1) treatment 1 | 1.7 ± 2.1 |
| flow velocity (cm·s−1) treatment 2 | 8.9 ± 3.7 |
| flow velocity (cm·s−1) treatment 3 | 9.0 ± 1.5 |
| Turbidity (NTU) | Oxygen (mg L−1) | Temp (°C) | pH | Conductivity (µS cm−1) | |
|---|---|---|---|---|---|
| Day 1 | 4.89 | 12.23 | 7.20 | 8.41 | 365 |
| Day 2 | 6.08 | 12.15 | 8.10 | 8.51 | 362 |
| Day 3 | 5.83 | 11.90 | 8.00 | 8.50 | 362 |
| Day 4 | 8.35 | 12.13 | 8.00 | 8.54 | 361 |
| Discharge (m3 s−1) | Flow Velocity Stow Net (m3 s−1) | Flow Velocity Fyke Net (m3 s−1) | |||
| Day 1 | 91.40 | 0.46 | 0.34 | ||
| Day 2 | 92.50 | 0.46 | 0.07 | ||
| Day 3 | 92.50 | 0.34 | 0.43 | ||
| Day 4 | 92.50 | 0.20 | 0.46 | ||
References
- Knott, J.; Mueller, M.; Pander, J.; Geist, J. Seasonal and diurnal variation of downstream fish movement at four small-scale hydropower plants. Ecol. Freshw. Fish 2020, 29, 74–88. [Google Scholar] [CrossRef]
- Knott, J.; Mueller, M.; Pander, J.; Geist, J. Fish Passage and Injury Risk at a Surface Bypass of a Small-Scale Hydropower Plant. Sustainability 2019, 11, 6037. [Google Scholar] [CrossRef]
- Pander, J.; Mueller, M.; Geist, J. Ecological functions of fish bypass channels in streams: Migration corridor and habitat for rheophilic species. River Res. Appl. 2013, 29, 441–450. [Google Scholar] [CrossRef]
- Mueller, M.; Sternecker, K.; Milz, S.; Geist, J. Assessing turbine passage effects on internal fish injury and delayed mortality using X-ray imaging. PeerJ 2020, 8, e9977. [Google Scholar] [CrossRef] [PubMed]
- Bierschenk, B.M.; Pander, J.; Mueller, M.; Geist, J. Fish injury and mortality at pumping stations: A comparison of conventional and fish-friendly pumps. Mar. Freshw. Res. 2019, 70, 449–458. [Google Scholar] [CrossRef]
- Mueller, M.; Pander, J.; Geist, J. Evaluation of external fish injury caused by hydropower plants based on a novel field-based protocol. Fish. Manag. Ecol. 2017, 24, 240–255. [Google Scholar] [CrossRef]
- Egg, L.; Pander, J.; Mueller, M.; Geist, J. Comparison of sonar-, camera- and net-based methods in detecting riverine fish-movement patterns. Mar. Freshw. Res. 2018, 69, 1905–1912. [Google Scholar] [CrossRef]
- Dedual, M. Survival of Juvenile Rainbow Trout Passing through a Francis Turbine. N. Am. J. Fish. Manag. 2007, 27, 181–186. [Google Scholar] [CrossRef]
- Dubois, R.B.; Gloss, S.P. Mortality of Juvenile American Shad and Striped Bass Passed through Ossberger Crossflow Turbines at a Small-Scale Hydroelectric Site. N. Am. J. Fish. Manag. 1993, 13, 178–185. [Google Scholar] [CrossRef]
- Cramer, F.K.; Donaldson, I.J. Evolution of Recovery Nets Used in Tests on Fish Passage through Hydraulic Turbines. Progress Fish-Cult. 1964, 26, 36–41. [Google Scholar] [CrossRef]
- Pander, J.; Mueller, M.; Knott, J.; Geist, J. Catch-related fish injury and catch efficiency of stow-net-based fish recovery installations for fish-monitoring at hydropower plants. Fish. Manag. Ecol. 2018, 25, 31–43. [Google Scholar] [CrossRef]
- Holst, R.; Madsen, N.; Moth-Poulsen, T.; Fonseca, P.; Campos, A. Manual for Gillnet Selectivity; European Commission: Brussels, Belgium, 1998; p. 43. [Google Scholar]
- Stepputtis, D.; Santos, J.; Herrmann, B.; Mieske, B. Broadening the horizon of size selectivity in trawl gears. Fish. Res. 2016, 184, 18–25. [Google Scholar] [CrossRef]
- Madsen, N. Selectivity of fishing gears used in the Baltic Sea cod fishery. Rev. Fish Biol. Fish. 2007, 17, 517–544. [Google Scholar] [CrossRef]
- Wileman, D.A.; Ferro, R.S.T.; Fonteyne, R.; Millar, R.B. Manual of Methods of Measuring the Selectivity of Towed Fishing Gears; ICES Cooperative Research Report, 215; ICES: Copenhagen, Denmark, 1996; p. 132. [Google Scholar]
- Jensen, J.W. A direct estimate of gillnet selectivity for brown trout. J. Fish Biol. 1995, 46, 857–861. [Google Scholar] [CrossRef]
- Herrmann, B.; Krag, L.A.; Frandsen, R.P.; Madsen, N.; Lundgren, B.; Stæhr, K.J. Prediction of selectivity from morphological conditions: Methodology and a case study on cod (Gadus morhua). Fish. Res. 2009, 97, 59–71. [Google Scholar] [CrossRef]
- Carol, J.; García-Berthou, E. Gillnet selectivity and its relationship with body shape for eight freshwater fish species. J. Appl. Ichthyol. 2007, 23, 654–660. [Google Scholar] [CrossRef]
- Ebel, G. Fischschutz und Fischabstieg an Wasserkraftanlagen—Handbuch Rechen—und By-Passsysteme. Ingenieurbiologische Grundlagen, Modellierung und Prognose, Bemessung und Gestaltung; Büro für Gewässerökologie und Fischereibiologie Dr. Ebel: Halle (Saale), Germany, 2013; p. 483. [Google Scholar]
- Mueller, M.; Knott, J.; Egg, L.; Bierschenk, B.; Pander, J.; Geist, J. Fischökologisches Monitoring an Innovativen Wasserkraftanlagen: Band 1 Hintergrund und Methoden; Technical Report for Bayerisches Landesamt für Umwelt; Technical University Munich: Freising, Germany, July 2020. [Google Scholar]
- Mueller, M.; Knott, J.; Pander, J.; Geist, J. Fischökologisches Monitoring an Innovativen Wasserkraftanlagen: Band 7 Eixendorf an der Schwarzach; Technical Report for Bayerisches Landesamt für Umwelt; Technical University Munich: Freising, Germany, 2020. [Google Scholar]
- Adam, B.; Schürmann, M.; Schwevers, U. Zum Umgang mit Aquatischen Organismen—Versuchstierkundliche Grundlagen; Springer Spektrum: Wiesbaden, Germany, 2013; p. 188. [Google Scholar]
- European Parliament. Directive 2010/63/EU of the European Parliament and of the Council of 22 September 2010 on the protection of animals used for scientific purposes. Off. J. Eur. Union 2010, 276, 33–77. [Google Scholar]
- Birnie-Gauvin, K.; Candee, M.M.; Baktoft, H.; Larsen, M.H.; Koed, A.; Aarestrup, K. River connectivity reestablished: Effects and implications of six weir removals on brown trout smolt migration. River Res. Appl. 2018, 34, 548–554. [Google Scholar] [CrossRef]
- Johnsson, J.I.; Näslund, J. Studying behavioural variation in salmonids from an ecological perspective: Observations questions methodological considerations. Rev. Fish. Biol. Fish. 2018, 28, 795–823. [Google Scholar] [CrossRef]
- ICES. Report of the Study Group on Turned 90° Codend Selectivity, Focusing on Baltic Cod Selectivity (SGTCOD), 4–6 May 2011; IMR: Reykjavik, Iceland, 2011; p. 40. [Google Scholar]
- Johnsson, J.I.; Brockmark, S.; Näslund, J. Environmental effects on behavioural development consequences for fitness of captive-reared fishes in the wild. J. Fish Biol. 2014, 85, 1946–1971. [Google Scholar] [CrossRef]
- Millar, R.B.; Fryer, R.J. Estimating the size-selection curves of towed gears, traps, nets and hooks. Rev. Fish Biol. Fish. 1999, 9, 89–116. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020; Available online: https://www.R-project.org/ (accessed on 1 October 2020).
- Chang, W. Extrafont: Tools for Using Fonts. R Package Version 0.17. 2014. Available online: https://CRAN.R-project.org/package=extrafont (accessed on 1 October 2020).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Garnier, S. Viridis: Default Color Maps from ‘matplotlib’. R Package Version 0.5.1. 2018. Available online: https://CRAN.R-project.org/package=viridis (accessed on 1 October 2020).
- Signorell, A.; Aho, K.; Alfons, A.; Anderegg, N.; Aragon, T.; Arachchige, C.; Arppe, A.; Baddeley, A.; Barton, K.; Bolker, B.; et al. DescTools: Tools for Descriptive Statistics. R Package Version 0.99.38. 2018. Available online: https://CRAN.R-project.org/package=DescTools (accessed on 1 October 2020).
- Canty, A.; Ripley, B. Boot: Bootstrap R (S-Plus) Functions; R Package Version 1.3-25. 2020. Available online: https://CRAN.R-project.org/package=boot (accessed on 1 October 2020).
- Davison, A.C.; Hinkley, D.V. Bootstrap Methods and Their Applications; Cambridge University Press: Cambridge, UK, 1997; ISBN 0-521-57391-2. [Google Scholar]
- Kuhn, M. Caret: Classification and Regression Training. R Package Version 6.0-86. 2020. Available online: https://CRAN.R-project.org/package=caret (accessed on 1 October 2020).
- Sachs, M.C. plotROC: A Tool for Plotting ROC Curves. J. Stat. Softw. Code Snippets 2017, 79, 1–19. [Google Scholar] [CrossRef]
- Dinno, A. dunn.test: Dunn’s Test of Multiple Comparisons Using Rank Sums. R Package Version 1.3.5. 2017. Available online: https://CRAN.R-project.org/package=dunn.test (accessed on 1 October 2020).
- Fox, J.; Weisberg, S. An {R} Companion to Applied Regression, 3rd ed.; Sage: Thousand Oaks, CA, USA, 2019; Available online: https://socialsciences.mcmaster.ca/jfox/Books/Companion/ (accessed on 1 October 2020).
- Wickham, H.; François, R.; Henry, L.; Müller, K. Dplyr: A Grammar of Data Manipulation. R Package Version 1.0.2. 2020. Available online: https://CRAN.R-project.org/package=dplyr (accessed on 1 October 2020).
- Wickham, H. The Split-Apply-Combine Strategy for Data Analysis. J. Stat. Softw. 2011, 40, 1–29. [Google Scholar] [CrossRef]
- Geist, J.; Porkka, M.; Kuehn, R. The status of host fish populations and fish species richness in European freshwater pearl mussel (Margaritifera margaritifera) streams. Aquat. Conserv. Mar. Freshw. Ecosyst. 2006, 16, 251–266. [Google Scholar] [CrossRef]
- Kottelat, M.; Freyhof, J. Handbook of European Freshwater Fishes, 1st ed.; Publications Kottelat: Cornol, Switzerland, 2007; p. 646. [Google Scholar]
- Peake, S.; McKinley, R.S.; Scruton, D.A. Swimming performance of various freshwater Newfoundland salmonids relative to habitat selection and fishway design. J. Fish Biol. 1997, 51, 710–723. [Google Scholar] [CrossRef]
- Houslay, T.M.; Vierbuchen, M.; Grimmer, A.J.; Young, A.J.; Wilson, A.J. Testing the stability of behavioural coping style across stress contexts in the Trinidadian guppy. Funct. Ecol. 2017, 1–15. [Google Scholar] [CrossRef]
- Näslund, J.; Bererhi, B.; Johnsson, J.I. Design of emergence test arenas can affect the results of boldness assays. Ethology 2015, 121, 556–565. [Google Scholar] [CrossRef]
- Shamchuk, A.L.; Tierney, K.B. Phenotyping stimulus evoked responses in larval zebrafish. Behaviour 2012, 149, 1177–1203. [Google Scholar] [CrossRef]
- Toms, C.N.; Echevarria, D.J.; Jouandot, D.J. A methodological review of personality-related studies in fish: Focus on the shy-bold axis of behavior. Int. J. Comp. Psychol. 2010, 23, 1–25. [Google Scholar]
- Sneddon, L.U. The bold and the shy: Individual differences in rainbow trout. J. Fish Biol. 2003, 62, 971–975. [Google Scholar] [CrossRef]
- Fraser, D.F.; Gilliam, J.F.; Daley, M.J.; Le, A.N.; Skalski, G.T. Explaining leptokurtic movement distributions: Intrapopulation variation in boldness and exploration. Am. Nat. 2001, 158, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Barber, I.; Mora, A.B.; Payne, E.M.; Weinersmith, K.L.; Sih, A. Parasitism, personality and cognition in fish. Behav. Process. 2017, 141, 205–219. [Google Scholar] [CrossRef] [PubMed]
- Tudorache, C.; Viaene, P.; Blust, R.; Vereecken, H.; De Boeck, G. A comparison of swimming capacity and energy use in seven European freshwater fish species. Ecol. Freshw. Fish 2007, 17, 284–291. [Google Scholar] [CrossRef]
- Bui, S.; Dempster, T.; Remen, M.; Oppedal, F. Effect of ectoparasite infestation density and life-history stages on the swimming performance of Atlantic salmon Salmo Salar. Aquac. Environ. Interact. 2016, 8, 387–395. [Google Scholar] [CrossRef]
- Taeubert, J.E.; Geist, J. Critical swimming speed of brown trout (Salmo trutta) infested with freshwater pearl mussel (Margaritifera margaritifera) glochidia and implications for artificial breeding of an endangered mussel species. Parasitol. Res. 2013, 112, 1607–1613. [Google Scholar] [CrossRef]
- Holzner, M. Untersuchungen über die Schädigung von Fischen bei der Passage des Mainkraftwerks Dettelbach. Ph.D. Thesis, Technical University Munich, Munich, Germany, September 2000. [Google Scholar]
- Heinrich, A.J. How Do Fish Interact with the Net?—First Approaches to Assess Fish-Net-Interactions in the Context of Hydropower Fish Monitoring. Master’s Thesis, Technical University Munich, Munich, Germany, December 2019. [Google Scholar]
- Schmidt, M.B.; Schletterer, M. Hydroakustik zur Analyse von Fischbeständen und Fischverhalten—Fallstudien aus Österreich. Österr. Wasser Abfallw. 2020, 72, 213–222. [Google Scholar] [CrossRef]
- Sebastian, S.M.; Caruthers, J.W. Effects of Naturally Occurring Bubbles on Multibeam Sonar Operations. In Proceedings of the Conference MTS/IEEE Oceans: An Ocean Odyssey, Honolulu, HI, USA, 5–8 November 2001; pp. 1241–1247. [Google Scholar] [CrossRef]
- Shen, C.; Lemmin, U. Ultrasonic scattering in highly turbulent clear water flow. Ultrasonics 1997, 35, 57–64. [Google Scholar] [CrossRef]
- Simon, J.; Kopp, D.; Larnaud, P.; Vacherot, J.P.; Morandeau, F.; Lavialle, G.; Morfin, M. Using automated video analysis to study fish escapement through escape panels in active fishing gears: Application to the effect of net colour. Mar. Policy 2020, 116, 103785. [Google Scholar] [CrossRef]
| Template | Mesh Size | Cord Diameter |
|---|---|---|
| template A | 30 mm | 1.3 mm |
| template B | 15 mm | 0.8 mm |
| template C | 10 mm | 0.5 mm |
| template D | 8 mm | 0.8 mm |
| Mesh Sizes (mm) | Runs | n Fish (15 per run) | Age Class | Fish Total Length (cm) | ||
|---|---|---|---|---|---|---|
| T1 | T2 | T3 | ||||
| 15, 20, 30 | 9 | 9 | 9 | 405 | 0+ | 3–10 (4.7 ± 0.5) |
| 20, 30 | 6 | 6 | 6 | 270 | 1+ | 11–20 (15.8 ± 1.83) |
| 30 | 3 | 3 | 3 | 135 | 2+ | 21–30 (25.6 ± 2) |
| total runs 54 | total fish 810 | |||||
| # 30 mm | # 20 mm | # 15 mm | # 10 mm | |
|---|---|---|---|---|
| 23.45 | 18.00 | 8.58 | 7.12 | |
| 23.45–23.46 | 17.70–18.30 | 8.49–8.67 | 7.06–7.18 | |
| pred. accuracy | 1.00 | 0.97 | 1.00 | 1.00 |
| α (intercept) | −2494.10 | −47.53 | −67.33 | −77.26 |
| β (slope) | 106.30 | 2.64 | 7.85 | 10.85 |
| explained deviance D2 | 1.00 | 0.93 | 1.00 | 1.00 |
| pseudo R2 | ||||
| McFadden | 1.00 | 0.93 | 1.00 | 1.00 |
| Nagelkerke | 1.00 | 0.97 | 1.00 | 1.00 |
| Cox & Snell | 0.70 | 0.71 | 0.72 | 0.72 |
| Run | FC | FM (%) | Esc. | Succ. Esc. | Revenants | Esc. 1 h |
|---|---|---|---|---|---|---|
| 1 | 48 | 4 | - | - | - | - |
| 2 | 44 | 12 | 13 | 12 | 8 | 4 |
| 3 | 45 | 10 | 12 | 7 | 3 | 4 |
| 4 | 48 | 4 | 6 | 5 | 2 | 3 |
| 5 | 47 | 6 | - | - | - | - |
| 6 | 49 | 2 | - | - | - | - |
| 7 | 46 | 8 | 3 | 3 | 1 | 2 |
| 8 | 50 | 0 | 9 | 5 | 1 | 4 |
| 9 | 45 | 10 | 12 | 10 | 4 | 6 |
| 10 | 50 | 0 | 3 | 3 | 4 | −1 * |
| Mean | 6 | 8 | 6 | 3 | 3 | |
| SD | 4 | 4 | 3 | 2 | 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smialek, N.; Pander, J.; Heinrich, A.; Geist, J. Sneaker, Dweller and Commuter: Evaluating Fish Behavior in Net-Based Monitoring at Hydropower Plants—A Case Study on Brown Trout (Salmo trutta). Sustainability 2021, 13, 669. https://doi.org/10.3390/su13020669
Smialek N, Pander J, Heinrich A, Geist J. Sneaker, Dweller and Commuter: Evaluating Fish Behavior in Net-Based Monitoring at Hydropower Plants—A Case Study on Brown Trout (Salmo trutta). Sustainability. 2021; 13(2):669. https://doi.org/10.3390/su13020669
Chicago/Turabian StyleSmialek, Nicole, Joachim Pander, Arne Heinrich, and Juergen Geist. 2021. "Sneaker, Dweller and Commuter: Evaluating Fish Behavior in Net-Based Monitoring at Hydropower Plants—A Case Study on Brown Trout (Salmo trutta)" Sustainability 13, no. 2: 669. https://doi.org/10.3390/su13020669
APA StyleSmialek, N., Pander, J., Heinrich, A., & Geist, J. (2021). Sneaker, Dweller and Commuter: Evaluating Fish Behavior in Net-Based Monitoring at Hydropower Plants—A Case Study on Brown Trout (Salmo trutta). Sustainability, 13(2), 669. https://doi.org/10.3390/su13020669





