Efficacy of Hormonal Growth Promoter Implants on the Performance of Grazing Steers of Different Breeds in Southern Chile
Abstract
1. Introduction
2. Materials and Methods
2.1. Management of the Herd and Comparison Groups
2.2. Pasture and Grazing Management
2.3. Statistical Analysis
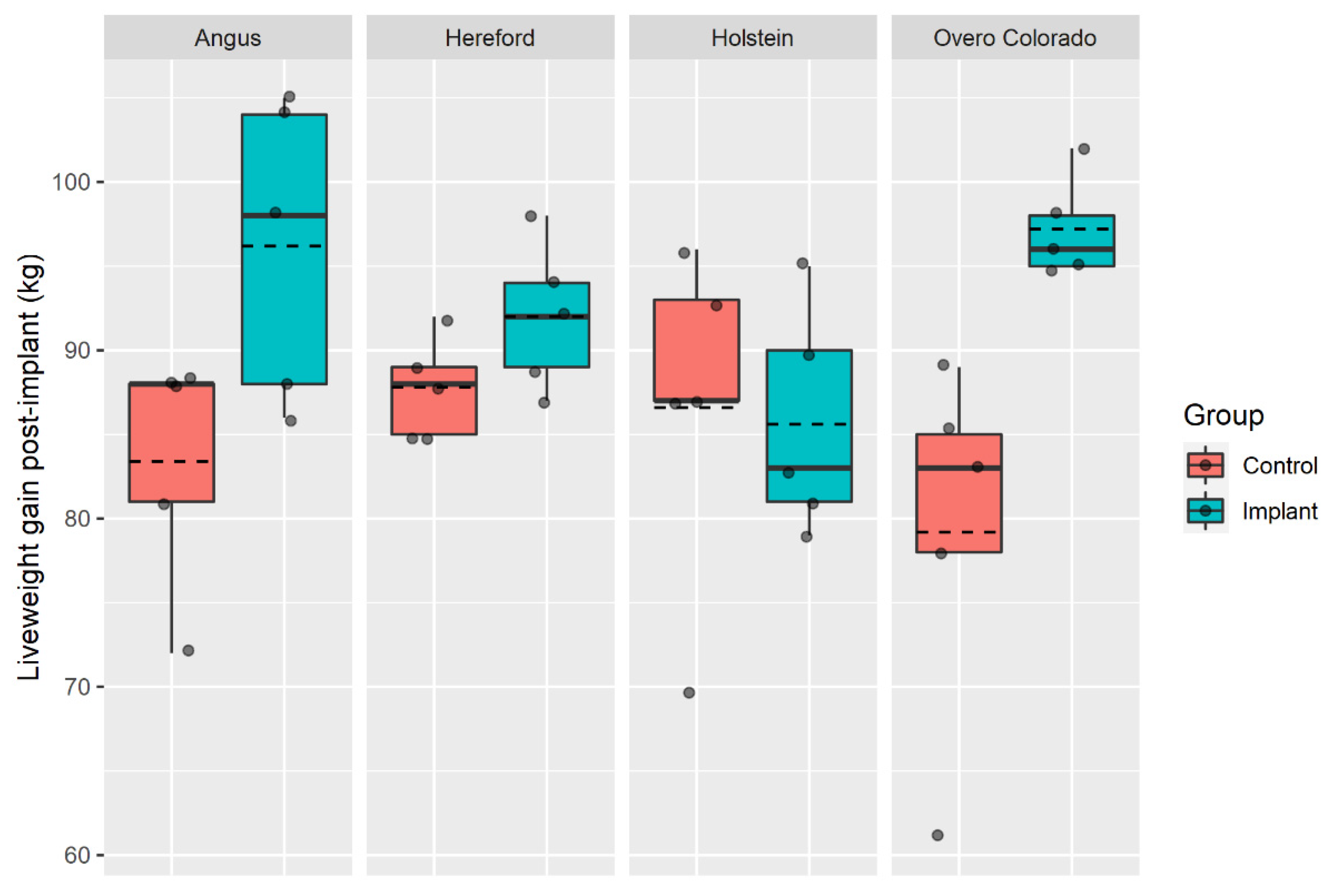
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Capper, J.L.; Bauman, D.E. The role of productivity in improving the environmental sustainability of ruminant production systems. Annu. Rev. Anim. Biosci. 2013, 1, 469–489. [Google Scholar] [CrossRef] [PubMed]
- Cederberg, C.; Persson, U.M.; Neovius, K.; Molander, S.; Clift, R. Including carbon emissions from deforestation in the carbon footprint of Brazilian beef. Environ. Sci. Technol. 2011, 45, 1773–1779. [Google Scholar] [CrossRef]
- Capper, J.L.; Hayes, D.J. The environmental and economic impact of removing growth-enhancing technologies from U.S. beef production. J. Anim. Sci. 2012, 90, 3527–3537. [Google Scholar] [CrossRef]
- Aboagye, I.A.; Cordeiro, M.R.C.; McAllister, T.A.; Ominski, K.H. Productivity-enhancing technologies. Can consumer choices affect the environmental footprint of beef? Sustainability 2021, 13, 1–19. [Google Scholar] [CrossRef]
- Thompson, J.M.; McIntyre, B.M.; Tudor, G.D.; Pethick, D.W.; Polkinghorne, R.; Watson, R. Effects of hormonal growth promotants (HGP) on growth, carcass characteristics, the palatability of different muscles in the beef carcass and their interaction with aging. Aust. J. Exp. Agric. 2008, 48, 1405–1414. [Google Scholar] [CrossRef][Green Version]
- Webb, A.S.; Rogers, R.W.; Rude, B.J. Review: Androgenic, estrogenic, and combination implants: Production and meat quality in beef. Prof. Anim. Sci. 2002, 18, 103–106. [Google Scholar] [CrossRef]
- Avery, A.; Avery, D. The Environmental Safety and Benefits of Growth Enhancing Pharmaceutical Technologies in Beef Production. Available online: https://www.thecattlesite.com/articles/1240/the-environmental-safety-and-benefits-of-growth-enhancing-pharmaceutical-technologies-in-beef-production/ (accessed on 23 July 2021).
- Jeong, S.-H.; Kang, D.; Lim, M.-W.; Kang, C.S.; Sung, H.J. Risk assessment of growth hormones and antimicrobial residues in meat. Toxicol. Res. 2010, 26, 301–313. [Google Scholar] [CrossRef]
- Stella, R.; Dervilly-Pinel, G.; Bovo, D.; Mastrorilli, E.; Royer, A.-L.; Angeletti, R.; Le Bizec, B.; Biancotto, G. Metabolomics analysis of liver reveals profile disruption in bovines upon steroid treatment. Metabolomics 2017, 13, 80. [Google Scholar] [CrossRef]
- OIE. Annual Report on Antimicrobial Agents Intended for Use in Animals; OIE: Paris, France, 2018. [Google Scholar]
- Hunter, R.A. Hormonal growth promotant use in the Australian beef industry. Anim. Prod. Sci. 2010, 50, 637–659. [Google Scholar] [CrossRef]
- Fox, L.; Butler, W.R.; Everett, R.W.; Natzke, R.P. Effect of adrenocorticotropin on milk and plasma cortisol and prolactin concentrations. J. Dairy Sci. 1981, 64, 1794–1803. [Google Scholar] [CrossRef]
- Johnson, B.J.; Ribeiro, F.R.B.; Beckett, J.L. Application of growth technologies in enhancing food security and sustainability. Anim. Front. 2013, 3, 8–13. [Google Scholar] [CrossRef]
- Smith, Z.K.; Johnson, B.J. Mechanisms of steroidal implants to improve beef cattle growth: A review. J. Appl. Anim. Res. 2020, 48, 133–141. [Google Scholar] [CrossRef]
- Song, M.K.; Choi, S.H. Growth promoters and their effects on beed production—Review. Asian Australas. J. Anim. Sci. 2001, 14, 123–135. [Google Scholar] [CrossRef]
- Johnson, B.; Beckett, J. Application of Growth Enhancing Compounds in Modern Beef Production—Executive Summary; American Meat Association: Champaign, IL, USA, 2014; pp. 1–15. [Google Scholar]
- Birkelo, C.P. Pharmaceuticals, direct-fed microbials, and enzymes for enhancing growth and feed efficiency of beef. Vet. Clin. North Am. Food Anim. Pract. 2003, 19, 599–624. [Google Scholar] [CrossRef]
- Albertí, P.; Panea, B.; Sañudo, C.; Olleta, J.L.; Ripoll, G.; Ertbjerg, P.; Christensen, M.; Gigli, S.; Failla, S.; Concetti, S.; et al. Live weight, body size and carcass characteristics of young bulls of fifteen European breeds. Livest. Sci. 2008, 114, 19–30. [Google Scholar] [CrossRef]
- Goic, L. Descripción y perspectivas de la producción de carne bovina en Chile. Informe final. In Taller Internacional: Limitaciones y Perspectivas del Sector Cárnico Bovino en Chile y el Mercosur; Catrileo, A., Ed.; FAO, Universidad Mayor: Santiago de Chile, Chile, 2001; pp. 136–148. [Google Scholar]
- Arias, R.; Santa-Cruz, C.; Velásquez, A. Effect of high potency growth implants on average daily gain of grass-fattened steers. Animals 2019, 9, 587. [Google Scholar] [CrossRef] [PubMed]
- Morales, R.; Folch, C.; Iraira, S.; Teuber, N.; Realini, C.E. Nutritional quality of beef produced in Chile from different production systems. Chil. J. Agric. Res. 2012, 72, 80–86. [Google Scholar] [CrossRef]
- Boody, G.; Vondracek, B.; Andow, D.A.; Krinke, M.; Westra, J.; Zimmerman, J.; Welle, P. Multifunctional agriculture in the United States. Bioscience 2005, 55, 27–38. [Google Scholar] [CrossRef]
- Blasi, D.A.; Kuhl, G.L. Effects of Revalor-G®, Ralgro®, and Synovex-H® on the performance of stocker heifers grazing irrigated rye pasture. In Cattlemen´s Day; Agricultural Experiment Station and Cooperative Extension Service, Kansas State University: Manhattan, KS, USA, 1998; pp. 126–128. [Google Scholar]
- Cleale, R.M.; Hilbig, D.R.; Short, T.H.; Sweiger, S.H.; Gallery, T. Effects of Synovex One Grass, Revalor-G, or Encore implants on performance of steers grazing for up to 200 days. Prof. Anim. Sci. 2018, 34, 192–201. [Google Scholar] [CrossRef]
- Berthiaume, R.; Mandell, I.; Faucitano, L.; Lafrenière, C. Comparison of alternative beef production systems based on forage finishing or grain-forage diets with or without growth promotants: 1. Feedlot performance, carcass quality, and production costs. J. Anim. Sci. 2006, 84, 2168–2177. [Google Scholar] [CrossRef] [PubMed]
- Faucitano, L.; Chouinard, P.Y.; Fortin, J.; Mandell, I.B.; Lafrenière, C.; Girard, C.L.; Berthiaume, R. Comparison of alternative beef production systems based on forage finishing or grain-forage diets with or without growth promotants: 2. Meat quality, fatty acid composition, and overall palatability. J. Anim. Sci. 2008, 86, 1678–1689. [Google Scholar] [CrossRef]
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Farney, J.K.; Corrigan, M. Short Communication: Evaluation of 2 implants for growing steers grazing tall-grass prairie when using intensive early stocking. Appl. Anim. Sci. 2019, 35, 83–87. [Google Scholar] [CrossRef]
- Guiroy, P.J.; Tedeschi, L.O.; Fox, D.G.; Hutcheson, J.P. The effects of implant strategy on finished body weight of beef cattle. J. Anim. Sci. 2002, 80, 1791–1800. [Google Scholar] [CrossRef]
- Gill, D.R.; Smith, S.C.; Nicholas, W.; Montague, M.R. Performance of stocker steers implanted with Ralgro, Synovex-S or Revalor-G. In Animal Science Research Report; Agricultural Experiment Station, Oklahoma State University: Stillwater, OK, USA, 1995; pp. 163–166. [Google Scholar]
- Merino, V.; Balocchi, O.; Pulido, R. Effect of daily herbage allowance restriction on pasture characteristics and milk production by grazing dairy cows in spring. Cienc. e Investig. Agrar. 2018, 45, 21–34. [Google Scholar] [CrossRef]
- Rivero, M.J.; Balocchi, O.A.; Moscoso, C.J.; Siebald, J.A.; Neumann, F.L.; Meyer, D.; Lee, M.R.F. Does the “high sugar” trait of perennial ryegrass cultivars express under temperate climate conditions? Grass Forage Sci. 2019. [Google Scholar] [CrossRef] [PubMed]
- Kuhl, G.L. Stocker cattle responses to implants. In Proceedings of the Symposium: Impact of Implants on Performance and Carcass Value of Beef Cattle; Oklahoma Agricultural Experiment Station: Stillwater, OK, USA, 1997; pp. 51–62. [Google Scholar]
- Goic, L.; Iraira, S. Engorda en Pastoreo. In Producción y Manejo de Carne Bovina en Chile; Catrileo, A., Ed.; Instituto de Investigaciones Agropecuarias, Ministerio de Agricultura: Temuco, Chile, 2005; pp. 275–293. [Google Scholar]
- Hayden, J.M.; Bergen, W.G.; Merkel, R.A. Skeletal muscle protein metabolism and serum growth hormone, insulin, and cortisol concentrations in growing steers implanted with estradiol-17 beta, trenbolone acetate, or estradiol-17 beta plus trenbolone acetate. J. Anim. Sci. 1992, 70, 2109–2119. [Google Scholar] [CrossRef]
- Anderson, P.T. Trenbolone acetate as a growth promotant. Compend. Contin. Educ. Pract. Vet. 1991, 13, 1179. [Google Scholar]
- Nogalski, Z.; Wielgosz-Groth, Z.; Purwin, C.; Nogalska, A.; Sobczuk-Szul, M.; Winarski, R.; Pogorzelska, P. The effect of slaughter weight and fattening intensity on changes in carcass fatness in young Holstein-Friesian bulls. Ital. J. Anim. Sci. 2014, 13, 66–72. [Google Scholar] [CrossRef]
- Catrileo, A.; Morales, R.; Rojas, C.; Cancino, D. Beef production from dairy bulls under two different production systems and its effect on the fatty acid profile and beef quality. Chil. J. Agric. Res. 2014, 74, 366–370. [Google Scholar] [CrossRef]
- Pesonen, M.; Honkavaara, M.; Huuskonen, A. Effect of breed on production, carcass traits and meat quality of aberdeen angus, limousin and aberdeen angus×limousin bulls offered a grass silage-grain-based diet. Agric. Food Sci. 2012, 21, 361–369. [Google Scholar] [CrossRef]
- Piedrafita, J.; Quintanilla, R.; Sañudo, C.; Olleta, J.L.; Campo, M.M.; Panea, B.; Renand, G.; Turin, F.; Jabet, S.; Osoro, K.; et al. Carcass quality of 10 beef cattle breeds of the Southwest of Europe in their typical production systems. Livest. Prod. Sci. 2003, 82, 1–13. [Google Scholar] [CrossRef]
- Dolezal, H.G.; Tatum, J.D.; Williams, F.L. Effects of feeder cattle frame size, muscle thickness, and age class on days fed, weight, and carcass composition. J. Anim. Sci. 1993, 71, 2975–2985. [Google Scholar] [CrossRef]
- Owens, F.N.; Gill, D.G.; Secrist, D.S.; Coleman, S.W. Review of some aspects of growth and development of feedlot cattle. J. Anim. Sci. 1995, 73, 3152–3172. [Google Scholar] [CrossRef] [PubMed]
- Elmajdoub, A.; Garbaj, A.; Abolghait, S.; El-Mahmoudy, A. Evaluation of boldenone as a growth promoter in broilers: Safety and meat quality aspects. J. Food Drug Anal. 2016, 24, 284–292. [Google Scholar] [CrossRef]
- EFSA Opinion of the Scientific Panel on contaminants in the food chain (CONTAM) related to hormone residues in bovine meat and meat products. EFSA J. 2007, 510, 1–62.
- Boerlin, P.; Wissing, A.; Aarestrup, F.M.; Frey, J.; Nicolet, J. Antimicrobial growth promoter ban and resistance to macrolides and vancomycin in enterococci from pigs. J. Clin. Microbiol. 2001, 39, 4193–4195. [Google Scholar] [CrossRef] [PubMed]
- Bengtsson, B.; Wierup, M. Antimicrobial resistance in scandinavia after ban of antimicrobial growth promoters. Anim. Biotechnol. 2006, 17, 147–156. [Google Scholar] [CrossRef] [PubMed]
- WHO. Impacts of Antimicrobial Growth Promotor Termination in Denmark; WHO: Foulum, Denmark, 2003. [Google Scholar]
- Callaway, T.R.; Edrington, T.S.; Rychlik, J.L.; Genovese, K.J.; Poole, T.L.; Jung, Y.S.; Bischoff, K.M.; Anderson, R.C.; Nisbet, D.J. Ionophores: Their use as ruminant growth promotants and impact on food safety. Curr. Issues Intest. Microbiol. 2003, 4, 43–51. [Google Scholar]
- Aroeira, C.N.; Feddern, V.; Gressler, V.; Contreras-Castillo, C.J.; Hopkins, D.L. A review on growth promoters still allowed in cattle and pig production. Livest. Sci. 2021, 247, 104464. [Google Scholar] [CrossRef]
| Component 1 | August | October |
|---|---|---|
| DM (%) | 12.2 | 16.3 |
| CP (% DM) | 26.7 | 24.6 |
| ADF (% DM) | 27.6 | 26.7 |
| NDF (% DM) | 41.6 | 40.3 |
| ME (MJ/kg DM) | 11.8 | 12.0 |
| Breed (B) | Implant Group (I) | s.e.d. | p-Value | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Variable | AA | HD | FN | OC | Yes | No | B | I | B × I | |
| Age (d) | 299 | 297 | 300 | 293 | 309 | 285 | 17.9 | 0.957 | 0.01 | 0.283 |
| Initial LW (kg) | 206 b | 208 b | 263 a | 209 b | 224 | 218 | 14.4 | <.001 | 0.41 | 0.039 |
| LWG (kg) | 87.9 b | 89.0 b | 114.8 a | 95.8 b | 100.8 | 93.0 | 11.25 | 0.07 | 0.173 | 0.635 |
| Breed (B) | Implant Group (I) | s.e.d. | p-Value | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Variable | AA | HD | FN | OC | Yes | No | B | I | B × I | Covariates | |
| LW at implant (kg) | 313 | 314 | 326 | 320 | 322 | 315 | 12.6 | 0.361 | 0.365 | 0.435 | <0.001 |
| LWG (kg) | 90.9 | 90.9 | 82.9 | 89.3 | 92.0 | 85.0 | 5.10 | 0.650 | 0.013 | 0.029 | 0.253 |
| Final LW (kg) | 404 | 405 | 409 | 410 | 413 | 400 | 30.0 | 0.730 | 0.096 | 0.116 | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivero, M.J.; Araya, L.; Oyarzo, M.; Cooke, A.S.; Morgan, S.A.; Merino, V.M. Efficacy of Hormonal Growth Promoter Implants on the Performance of Grazing Steers of Different Breeds in Southern Chile. Sustainability 2021, 13, 9135. https://doi.org/10.3390/su13169135
Rivero MJ, Araya L, Oyarzo M, Cooke AS, Morgan SA, Merino VM. Efficacy of Hormonal Growth Promoter Implants on the Performance of Grazing Steers of Different Breeds in Southern Chile. Sustainability. 2021; 13(16):9135. https://doi.org/10.3390/su13169135
Chicago/Turabian StyleRivero, M. Jordana, Luis Araya, Marcelo Oyarzo, Andrew S. Cooke, Sarah A. Morgan, and Veronica M. Merino. 2021. "Efficacy of Hormonal Growth Promoter Implants on the Performance of Grazing Steers of Different Breeds in Southern Chile" Sustainability 13, no. 16: 9135. https://doi.org/10.3390/su13169135
APA StyleRivero, M. J., Araya, L., Oyarzo, M., Cooke, A. S., Morgan, S. A., & Merino, V. M. (2021). Efficacy of Hormonal Growth Promoter Implants on the Performance of Grazing Steers of Different Breeds in Southern Chile. Sustainability, 13(16), 9135. https://doi.org/10.3390/su13169135









