Fruit Quality Characters of Myrtle (Myrtus communis L.) Selections: Review of a Domestication Process
Abstract
:1. Myrtle Characteristics and Uses
2. The Myrtle Industry and Problems of Sustainability
- (a)
- Improve information about plant biology, particularly fruit development and ripening.
- (b)
- Evaluate the performance of myrtle propagation by seed and cutting.
- (c)
- Study spontaneous germplasm variability from morphological, phenological, and genetic points of view.
- (d)
- Identify morphological characteristics useful for cultivation and other uses.
- (e)
- Start the mass selection of valuable ecotypes for a first evaluation in situ and subsequent agamic propagation by softwood cutting of mother plants in the collection field of the University of Sassari, located at Fenosu (Central-Western Sardinia).
3. Evaluation of Spontaneous Myrtle Germplasm and Mass Selection: The First Step
3.1. Guidelines for Wild Plant Selection and Primary Characterization
3.2. Morphological and Biometric Description of the Wild Mother Plant Population
4. Cultivar Selection and Field Evaluation: The Second Step
4.1. Mother Plant Agamic Propagation, Nursery Management and Field Data Record
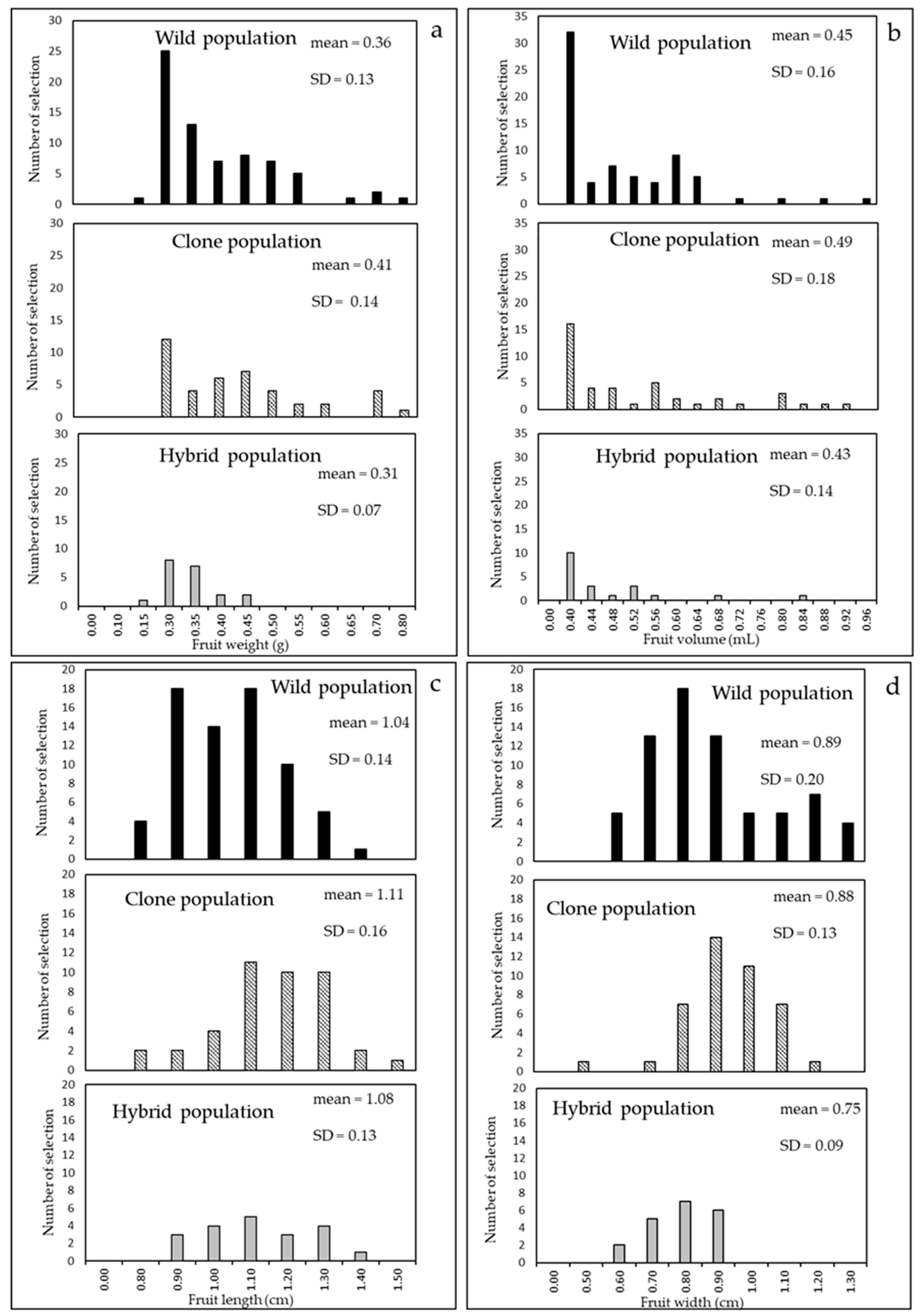
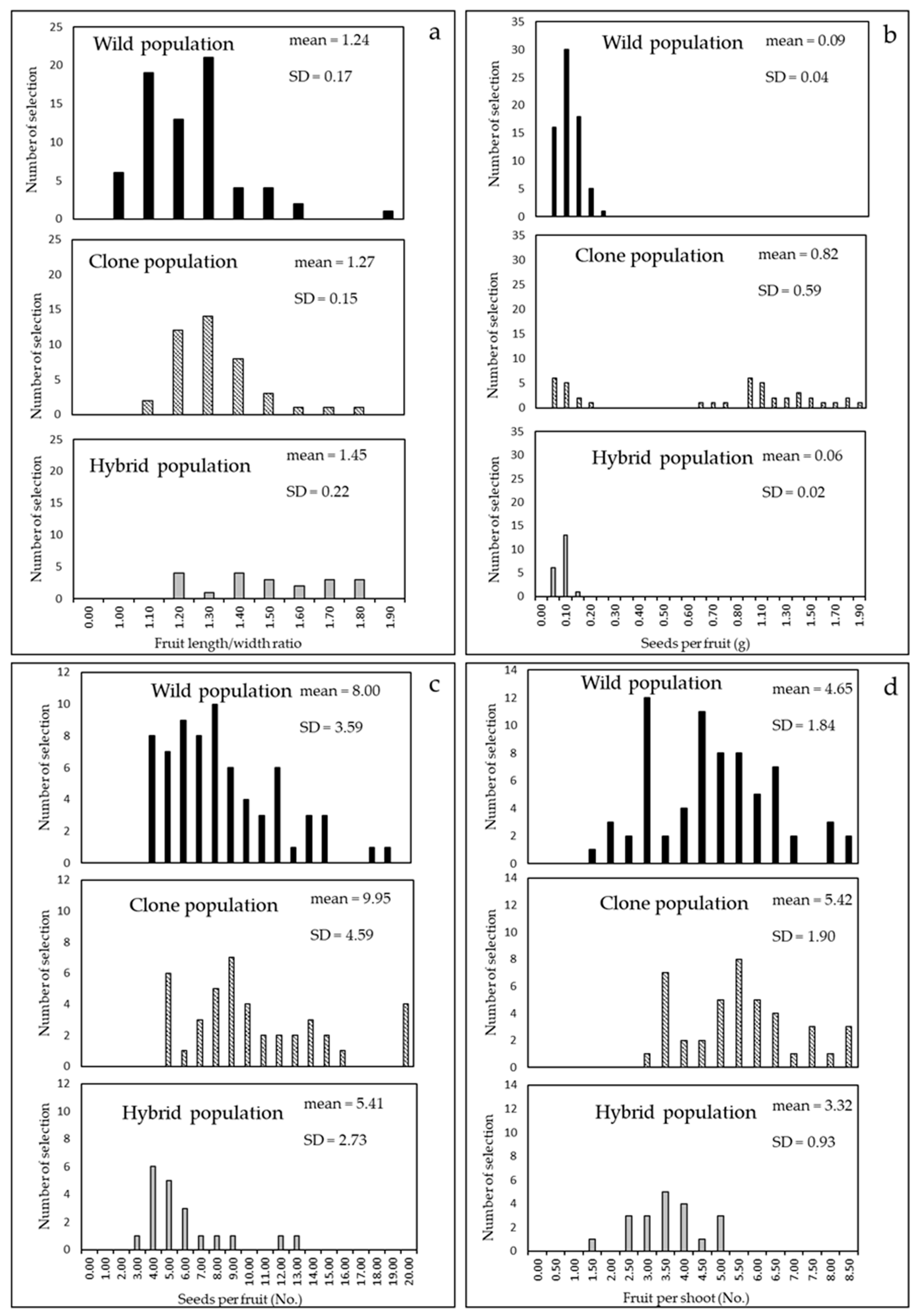
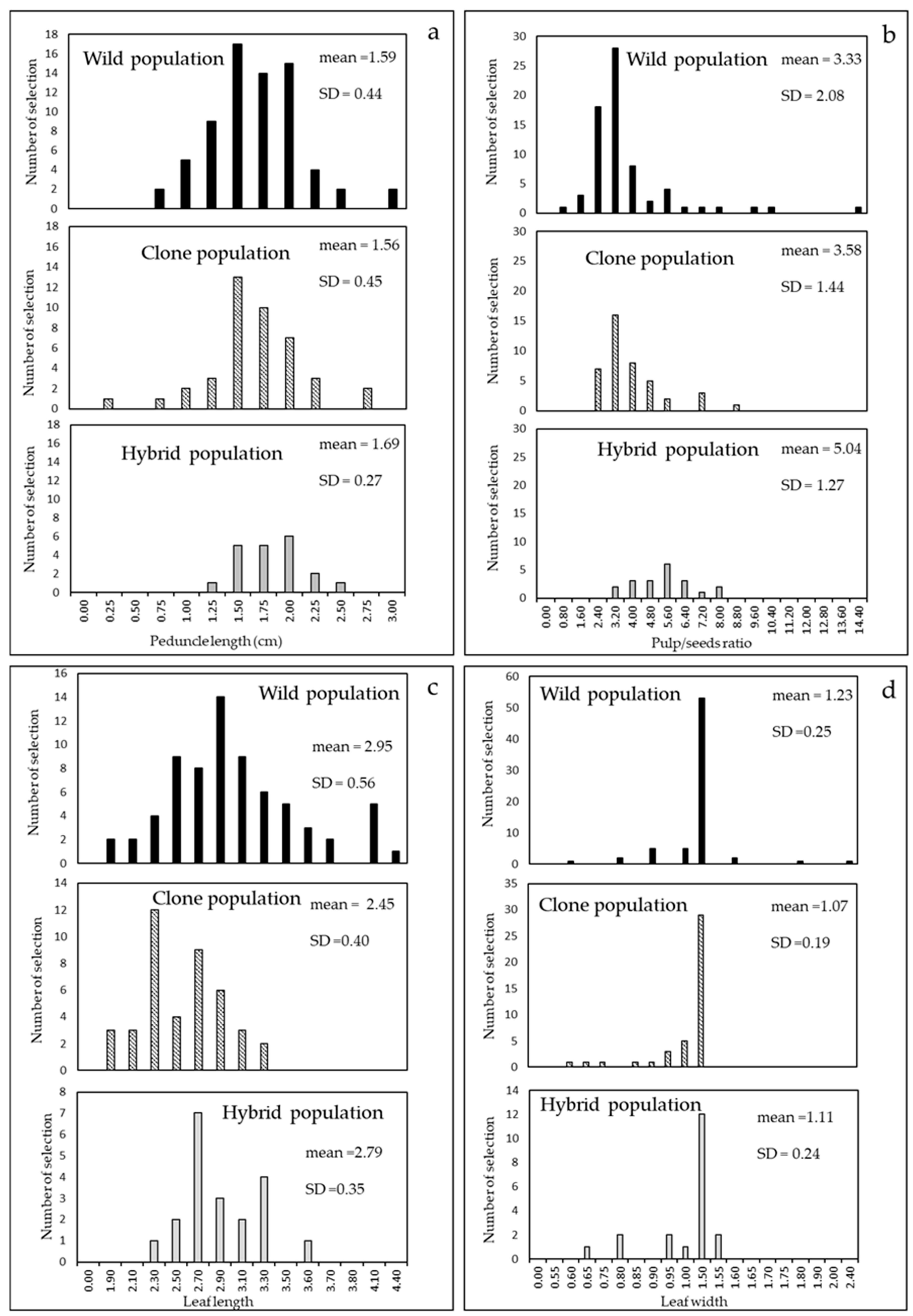
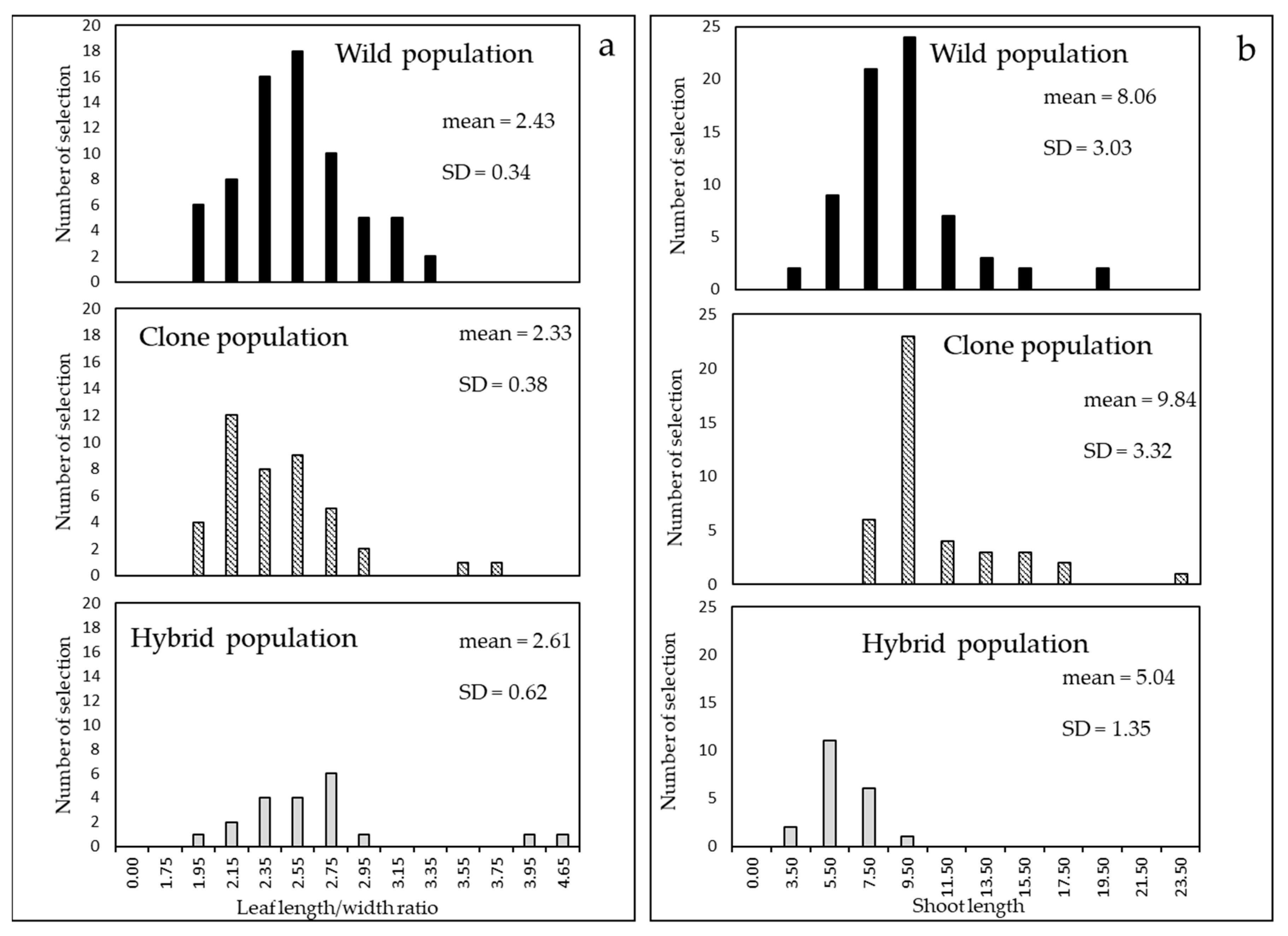
4.2. Cultivar Biometric Characters and Other Agronomic Evaluation
5. The New Group of Hybrid Selections
5.1. Open Pollination Cross, Seedling Sowing, Field Comparison, Phenotypic Selection, and Softwood Cutting Propagation
5.2. New Cultivars Originated by Hybridation: Phenotypic Characters and Biometric Data
6. Comparison of the Populations Studied in the Three Steps
7. General Evaluations and Future Perspectives of the Domestication Program
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Mulas, M.; Cani, M.R.; Brigaglia, N. Characters useful to cultivation in spontaneous populations of Myrtus communis L. Acta Hortic. 1998, 457, 271–278. [Google Scholar] [CrossRef]
- Mulas, M. The myrtle (Myrtus communis L.) case, from a wild shrub to a new fruit crop. Acta Hortic. 2012, 948, 235–242. [Google Scholar] [CrossRef]
- Mulas, M.; Fadda, A. First observations on biology and organ morphology of myrtle (Myrtus communis L.) flower. Agric. Mediterr. 2004, 134, 223–225. [Google Scholar]
- Mulas, M.; Cani, M.R. Germplasm evaluation of spontaneous myrtle (Myrtus communis L.) for cultivar selection and crop development. J. Herbs Spices Med. Plants 1999, 6, 31–49. [Google Scholar] [CrossRef]
- Usai, M.; Marchetti, M.; Culeddu, N.; Mulas, M. Chemotaxonomic Evaluation by Volatolomics Analysis of Fifty-Two Genotypes of Myrtus communis L. Plants 2020, 9, 1288. [Google Scholar] [CrossRef]
- Usai, M.; Mulas, M.; Marchetti, M. Chemical composition of essential oils of leaves and flowers from five cultivars of myrtle (Myrtus communis L.). J. Essent. Oil Res. 2015, 27, 465–476. [Google Scholar] [CrossRef]
- Mulas, M.; Melis, R.A.M. Essential oil composition of myrtle (Myrtus communis) leaves. J. Herbs Spices Med. Plants 2011, 17, 21–34. [Google Scholar] [CrossRef]
- Giampieri, F.; Cianciosi, D.; Forbes-Hernández, T.Y. Myrtle (Myrtus communis L.) berries, seeds, leaves, and essential oils: New undiscovered sources of natural compounds with promising health benefits. Food Front. 2020, 1, 276–295. [Google Scholar] [CrossRef]
- Usai, M.; Marchetti, M.; Culeddu, N.; Mulas, M. Chemical composition of myrtle (Myrtus communis L.) berries essential oils as observed in a collection of genotypes. Molecules 2018, 23, 2502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wannes, W.A.; Mhamdi, B.; Sriti, J.; Jemia, M.B.; Ouchikh, O.; Hamdaoui, G.; Marzouk, B. Antioxidant activities of the essential oils and methanol extracts from myrtle (Myrtus communis var. italica L.) leaf, stem, and flower. Food Chem. Toxicol. 2010, 48, 1362–1370. [Google Scholar] [CrossRef]
- Fadda, A.; Sarais, G.; Lai, C.; Sale, L.; Mulas, M. Control of postharvest diseases caused by Penicillium spp. with myrtle leaf phenolic extracts: In vitro and in vivo study on mandarin fruit during storage. J. Sci. Food Agric. 2021, 101, 4229–4240. [Google Scholar] [CrossRef]
- Bahadırlı, N.P.; Kahramanoğlu, İ.; Wan, C. Exposure to Volatile Essential Oils of Myrtle (Myrtus communis L.) Leaves for Improving the Postharvest Storability of Fresh Loquat Fruits. J. Food Qual. 2020, 2020, 1362–1370. [Google Scholar] [CrossRef]
- Medda, S.; Fadda, A.; Dessena, L.; Mulas, M. Quantification of Total Phenols, Tannins, Anthocyanins Content in Myrtus communis L. and Antioxidant Activity Evaluation in Function of Plant Development Stages and Altitude of Origin Site. Agronomy 2021, 11, 1059. [Google Scholar] [CrossRef]
- Sanna, D.; Mulas, M.; Molinu, M.G.; Fadda, A. Oxidative stability of plant hydroalcoholic extracts assessed by EPR spin trapping under forced ageing conditions: A myrtle case study. Food Chem. 2019, 271, 753–761. [Google Scholar] [CrossRef]
- Tuberoso, C.I.; Melis, M.; Angioni, A.; Pala, M.; Cabras, P. Myrtle hydroalcoholic extracts obtained from different selections of Myrtus communis L. Food Chem. 2007, 101, 806–811. [Google Scholar] [CrossRef]
- Montoro, P.; Tuberoso, C.I.; Piacente, S.; Perrone, A.; De Feo, V.; Cabras, P.; Pizza, C. Stability and antioxidant activity of polyphenols in extracts of Myrtus communis L. berries used for the preparation of myrtle liqueur. J. Pharm. Biomed. Anal. 2006, 41, 1614–1619. [Google Scholar] [CrossRef]
- Mulas, M.; Fadda, A.; Angioni, A. Effect of maturation and cold storage on the organic acid composition of myrtle fruits. J. Sci. Food Agric. 2013, 93, 37–44. [Google Scholar] [CrossRef]
- Fadda, A.; Palma, A.; D’Aquino, S.; Mulas, M. Effects of myrtle (Myrtus communis L.) fruit cold storage under modified atmosphere on liqueur quality. J. Food Process. Preserv. 2017, 41, e12776. [Google Scholar] [CrossRef]
- Cruciani, S.; Santaniello, S.; Fadda, A.; Sale, L.; Sarais, G.; Sanna, D.; Mulas, M.; Ginesu, G.C.; Cossu, M.L.; Serra, P.A.; et al. Extracts from myrtle liqueur processing waste modulate stem cells pluripotency under stressing conditions. BioMed Res. Int. 2019, 2019, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Cruciani, S.; Santaniello, S.; Garroni, G.; Fadda, A.; Balzano, F.; Bellu, E.; Sarais, G.; Fais, G.; Mulas, M.; Maioli, M. Myrtus polyphenols, from antioxidants to anti-inflammatory molecules: Exploring a network involving cytochromes P450 and vitamin D. Molecules 2019, 24, 1515. [Google Scholar] [CrossRef] [Green Version]
- Bellu, E.; Garroni, G.; Cruciani, S.; Balzano, F.; Serra, D.; Satta, R.; Montesu, M.A.; Fadda, A.; Mulas, M.; Sarais, G.; et al. Smart Nanofibers with Natural Extracts Prevent Senescence Patterning in a Dynamic Cell Culture Model of Human Skin. Cells 2020, 9, 2530. [Google Scholar] [CrossRef]
- Bellu, E.; Cruciani, S.; Garroni, G.; Balzano, F.; Satta, R.; Montesu, M.A.; Fadda, A.; Mulas, M.; Sarais, G.; Bandiera, P.; et al. Natural Compounds and PCL Nanofibers: A Novel Tool to Counteract Stem Cell Senescence. Cells 2021, 10, 1415. [Google Scholar] [CrossRef]
- Medda, S.; Sanchez-Ballesta, M.T.; Romero, I.; Dessena, L.; Mulas, M. Expression of Structural Flavonoid Biosynthesis Genes in Dark-Blue and White Myrtle Berries (Myrtus communis L.). Plants 2021, 10, 316. [Google Scholar] [CrossRef]
- Medda, S.; Dessena, L.; Mulas, M. Monitoring of the PAL Enzymatic Activity and Polyphenolic Compounds in Leaves and Fruits of Two Myrtle Cultivars during Maturation. Agriculture 2020, 10, 389. [Google Scholar] [CrossRef]
- Goldschmidt, E.E. The evolution of fruit tree productivity: A review. Econ. Bot. 2013, 67, 51–62. [Google Scholar] [CrossRef] [Green Version]
- Bijlsma, R.; Loeschcke, V. Genetic erosion impedes adaptive responses to stressful environments. Evol. Appl. 2012, 5, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Le Roux, J.J.; Hui, C.; Castillo, M.L.; Iriondo, J.M.; Keet, J.H.; Khapugin, A.A.; Hirsch, H. Recent anthropogenic plant extinctions differ in biodiversity hotspots and coldspots. Curr. Biol. 2019, 29, 2912–2918. [Google Scholar] [CrossRef] [PubMed]
- Mulas, M.; Cani, M.R.; Brigaglia, N.; Deidda, P. Study of myrtle (Myrtus communis L.) genetic resources to promote extensive crop as integration of spontaneous harvests. Acta Hortic. 1997, 502, 85–88. [Google Scholar] [CrossRef]
- Mulas, M.; Melis, R.A. Influence of growing area, year, season, and cultivar on the composition of myrtle leaves and infusions. HortScience 2008, 43, 549–553. [Google Scholar] [CrossRef]
- Song, G.Q.; Hancock, J.F. Vaccinium. In Wild Crop Relatives: Genomic and Breeding Resources; Springer: Berlin/Heidelberg, Germany, 2011; pp. 197–221. [Google Scholar]
- Zecca, G.A.; De Mattia, F.; Lovicu, G.; Labra, M.; Sala, F.; Grassi, F. Wild grapevine: Silvestris, hybrids or cultivars that escaped from vineyards? Molecular evidence in Sardinia. Plant Biol. 2012, 12, 558–562. [Google Scholar] [CrossRef] [PubMed]
- Zohary, D. Unconscious selection and the evolution of domesticated plants. Econ. Botan. 2004, 58, 5–10. [Google Scholar] [CrossRef]
- Mulas, M.; Francesconi, A.H.D.; Perinu, B. Myrtle (Myrtus communis L.) as a new aromatic crop: Cultivar selection. J. Herbs Spices Med. Plants 2002, 9, 127–131. [Google Scholar] [CrossRef]
- Dessena, L.; Sale, L.; Melito, S.; Mulas, M. Phenological and morphological characteristics of new selections of myrtle (Myrtus communis L.). Acta Hortic. 2015, 1172, 171–178. [Google Scholar]
- Mulas, M.; Francesconi, A.H.D.; Perinu, B.; Fadda, A. ‘Barbara’ and ‘Daniela’: Two cultivars for myrtle berries production. Acta Hortic. 2001, 576, 169–175. [Google Scholar] [CrossRef]
- FAO. 2021–23 Action Plan for the Implementation of the FAO Strategy on Mainstreaming Biodiversity across Agricultural Sectors; FAO: Rome, Italy, 2021. [Google Scholar] [CrossRef]
- Lakshman, C.D. Bio-diversity and conservation of medicinal and aromatic plants. Adv. Plants Agric. Res. 2016, 5, 561–566. [Google Scholar]
- Koerper, H.; Kolls, A.L. The silphium motif adorning ancient Libyan coinage: Marketing a medicinal plant. Econ. Bot. 1999, 53, 133–143. [Google Scholar] [CrossRef]
- Parejko, K. Pliny the Elder’s Silphium: First Recorded Species Extinction. Conserv. Biol. 2003, 17, 925–927. [Google Scholar] [CrossRef]
- Mulas, M. Traditional uses of Labiatae in the Mediterranean area. Acta Hortic. 2006, 723, 25–32. [Google Scholar] [CrossRef]
- Şekercioğlu, Ç.H.; Anderson, S.; Akçay, E.; Bilgin, R.; Can, Ö.E.; Semiz, G.; Dalfes, H.N. Turkey’s globally important biodiversity in crisis. Biol. Conserv. 2011, 144, 2752–2769. [Google Scholar] [CrossRef]
- de Medeiros, P.M.; Figueiredo, K.F.; Gonçalves, P.H.S.; de Almeida Caetano, R.; da Costa Santos, É.M.; Dos Santos, G.M.C.; Mapeli, A.M. Wild plants and the food-medicine continuum—An ethnobotanical survey in Chapada Diamantina (Northeastern Brazil). J. Ethnobiol. Ethnomed. 2021, 17, 1–10. [Google Scholar] [CrossRef]
- Pedrosa, H.C.; Clement, C.R.; Schietti, J. The domestication of the Amazon tree grape (Pourouma cecropiifolia) under an ecological lens. Front. Plant Sci. 2018, 9, 203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaughan, D.A.; Balazs, E.; Heslop-Harrison, J.S. From crop domestication to super-domestication. Ann. Bot. 2007, 100, 893–901. [Google Scholar] [CrossRef]
- Kantar, M.B.; Nashoba, A.R.; Anderson, J.E.; Blackman, B.K.; Rieseberg, L.H. The genetics and genomics of plant domestication. Bioscience 2017, 67, 971–982. [Google Scholar] [CrossRef] [Green Version]
- Mele, C.; Corona, L.; Melito, S.; Raggi, L.; Mulas, M. The genetic diversity of selections and wild populations of myrtle revealed by molecular geographic contexts. Ind. Crop. Prod. 2019, 132, 168–176. [Google Scholar] [CrossRef]
- Corona, L.; Mele, C.; Chessa, I.; Mulas, M. Analysis of Sardinian myrtle (Myrtus communis L.) germplasm selections by SSR markers. Acta Hortic. 2015, 1172, 165–170. [Google Scholar]
- Melito, S.; Dessena, L.; Sale, L.; Mulas, M. Genetic diversity and population structure of wild Sardinian myrtle (Myrtus communis L.) genotypes from different microclimatic areas. Aust. J. Crop Sci. 2017, 11, 1488–1496. [Google Scholar] [CrossRef]
- Melito, S.; Fadda, A.; Rapposelli, E.; Mulas, M. Genetic diversity and population structure of Sardinian myrtle (Myrtus communis L.) selections as obtained by AFLP markers. HortScience 2014, 49, 531–537. [Google Scholar] [CrossRef]
- Melito, S.; Chessa, I.; Erre, P.; Podani, J.; Mulas, M. The genetic diversity of Sardinian myrtle (Myrtus communis L.) populations. Electron. J. Biotechnol. 2013, 16, 7. [Google Scholar] [CrossRef]
- Aydın, C.; Özcan, M.M. Determination of nutritional and physical properties of myrtle (Myrtus communis L.) fruits growing wild in Turkey. J. Food Eng. 2007, 79, 453–458. [Google Scholar] [CrossRef]
- Şerçe, S.; Şimsek, Ö.; Gündüz, K.; Aka-Kacar, Y.; Ercisli, S. Relationships among myrtle accessions from Turkey as revealed by fruit characteristics and RAPD. Rom. Biotechnol. Lett. 2008, 13, 4054–4065. [Google Scholar]
- Ruffoni, B.; Airò, M.; Pascella, G.; Mascarello, C.; Zizzo, G.; Cervelli, C. Rooting and acclimatization of ornamental myrtle genotypes. Acta Hortic. 2002, 616, 255–258. [Google Scholar] [CrossRef]
- Gambella, F.; Paschino, F. Evaluation of feasibility of mechanical harvesting of myrtle berries (Myrtus communis L.). Int. J. Mech. Control 2010, 33–39. [Google Scholar]
- Aleksic, V.; Knezevic, P. Antimicrobial and antioxidative activity of extracts and essential oils of Myrtus communis L. Microbiol. Res. 2014, 169, 240–254. [Google Scholar] [CrossRef] [PubMed]
- Jabri, M.A.; Rtibi, K.; Sakly, M.; Marzouki, L.; Sebai, H. Role of gastrointestinal motility inhibition and antioxidant properties of myrtle berries (Myrtus communis L.) juice in diarrhea treatment. Biomed. Pharmacother. 2016, 84, 1937–1944. [Google Scholar] [CrossRef]
- Fadda, A.; Mulas, M. Chemical changes during myrtle (Myrtus communis L.) fruit development and ripening. Sci. Hortic. 2010, 125, 477–485. [Google Scholar] [CrossRef]
- Wannes, W.A.; Marzouk, B. Characterization of myrtle seed (Myrtus communis var. baetica) as a source of lipids, phenolics, and antioxidant activities. J. Food Drug Anal. 2016, 24, 316–323. [Google Scholar] [CrossRef] [Green Version]
- Franco, A.M.; Tocci, N.; Guella, G.; Dell’Agli, M.; Sangiovanni, E.; Perenzoni, D.; Vrhovsek, U.; Mattivi, F.; Manca, G. Myrtle Seeds (Myrtus communis L.) as a Rich Source of the Bioactive Ellagitannins Oenothein B and Eugeniflorin D2. ACS Omega 2019, 4, 15966–15974. [Google Scholar] [CrossRef] [Green Version]
- Shahbazian, D.; Karami, A.; Eshghi, S.; Maggi, F. Variation in the essential oil yields and compositions of Myrtle (Myrtus communis L.) populations collected from natural habitats of Southern Iran. J. Essent. Oil Res. 2018, 30, 369–378. [Google Scholar] [CrossRef]
- Bouzabata, A.; Boussaha, F.; Casanova, J.; Tomi, F. Composition and chemical variability of leaf oil of Myrtus communis from North-Eastern Algeria. Nat. Prod. Commun. 2010, 5, 1659–1662. [Google Scholar] [CrossRef] [Green Version]
- Messaoud, C.; Zaouali, Y.; Ben Salah, A.; Khoudja, M.L.; Boussaid, M. Myrtus communis in Tunisia: Variability of the essential oil composition in natural populations. Flavour Fragr. J. 2005, 20, 577–582. [Google Scholar] [CrossRef]
| Step 1—Mass Selection (1995–1997) | ||
| Target | Material | Method/Results |
| In situ selection of candidate mother plants according to a pre-designed morphotype | Wild populations of all the Sardinia region | 70 mother plants studied and agamically propagated Morphological and quality analysis according to a descriptor list |
| Step 2—Cultivar Selection (1997–2001) | ||
| Target | Material | Method/Results |
| Selections characterized by: - agamic propagation and nursery viability - agronomic performance - disease tolerance - fruit and leaf quality | 40 cultivars obtained from the in situ selected wild mother plants and field tested | Observation in ex situ collection field and other experimental plots under the same ecological and management conditions Morphological and quality analysis |
| Step 3—Open Pollination Cross and Hybrid Selection (2001–2006) | ||
| Target | Material | Method/Results |
| Explore variability of new hybrids: - seedling comparison, agamic propagation, and nursery viability - agronomic performance - disease tolerance - fruit and leaf quality | Three selections used as mother plants for open cross 387 seedlings observed and 20 selections cloned | Open pollination and seed sowing Seedling observation Agamic propagation Morphological and quality analysis according to a descriptor list |
| Character Observed | Influence on Myrtle Industry Applications |
|---|---|
| Fruit weight, volume, length, and width Fruit length/width Fruit peduncle length | Quality and concentration of phenols and of essential oils compounds in infusions Fresh/dry consumption Ornamental use Essential oils yield Manual and mechanical harvesting |
| Seeds number per fruit Seed weight per fruit Pulp/seeds ratio | Fresh consumption Quality and concentration of tannins in infusions |
| Shoot length | Yield abundance and reflowering occurrence |
| Leaf length and leaf width Leaf length/width ratio | Essential oils and other extracts yield Green biomass production Quality and concentration of white myrtle liqueur Ornamental value |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Medda, S.; Mulas, M. Fruit Quality Characters of Myrtle (Myrtus communis L.) Selections: Review of a Domestication Process. Sustainability 2021, 13, 8785. https://doi.org/10.3390/su13168785
Medda S, Mulas M. Fruit Quality Characters of Myrtle (Myrtus communis L.) Selections: Review of a Domestication Process. Sustainability. 2021; 13(16):8785. https://doi.org/10.3390/su13168785
Chicago/Turabian StyleMedda, Silvia, and Maurizio Mulas. 2021. "Fruit Quality Characters of Myrtle (Myrtus communis L.) Selections: Review of a Domestication Process" Sustainability 13, no. 16: 8785. https://doi.org/10.3390/su13168785
APA StyleMedda, S., & Mulas, M. (2021). Fruit Quality Characters of Myrtle (Myrtus communis L.) Selections: Review of a Domestication Process. Sustainability, 13(16), 8785. https://doi.org/10.3390/su13168785







