Seaweeds as a “Palatable” Challenge between Innovation and Sustainability: A Systematic Review of Food Safety
Abstract
1. Introduction
2. Materials and Methods
- Food AND (seaweed* OR “novel food*” OR alga*) AND (microbiota OR “microb* community” OR “microb* count*” OR “microb* load” OR “microb* risk” OR “microb* hazard” OR “microb* saf*” OR “food safety”) for microbiological risk;
- Food AND (seaweed* OR “novel food*” OR alga*) AND (allergen* OR allerg*) for allergenic risk;
- Food AND (seaweed* OR “novel food*” OR alga*) AND (“physical risk*” OR “physical hazard*” OR “physical safety” OR “foreign bod*” OR “breeding substrate*”) for physical risk;
- Food AND (seaweed* OR “novel food*” OR alga*) AND (“chemical risk*” OR “chemical hazard*” OR “chemical safety” OR radionuclide* OR metal* OR arsenic OR cadmium OR copper OR zinc OR chrome OR lead OR aluminium OR mercury OR toxin*) for chemical risk.
3. Results
3.1. Microbiological Risk
3.2. Allergenic Risk
3.3. Physical Risk
3.4. Chemical Risk
4. Discussion
Limitation of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Food and Agriculture Organization of the United Nations. The State of Food Security and Nutrition in the World 2019. Safeguarding against Economic Slowdowns and Downturns; FAO: Rome, Italy, 2019. [Google Scholar]
- Who.int. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 11 February 2021).
- Ipcc.ch. Available online: https://www.ipcc.ch/srccl/download/ (accessed on 11 February 2021).
- Sustainabledevelopment.un.org. Available online: https://sustainabledevelopment.un.org/post2015/transformingourworld/publication (accessed on 11 February 2021).
- Food and Agriculture Organization of the United Nations. The Future of Food and Agriculture—Alternative Pathways to 2050; FAO: Rome, Italy, 2018; 224p. [Google Scholar]
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food Security: The Challenge of Feeding 9 Billion People. Science 2010, 327, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Lindgren, E.; Harris, F.; Dangour, A.D.; Gasparatos, A.; Hiramatsu, M.; Javadi, F.; Loken, B.; Murakami, T.; Scheelbeek, P.; Haines, A. Sustainable Food Systems—A Health Perspective. Sustain. Sci. 2018, 13, 1505–1517. [Google Scholar] [CrossRef] [PubMed]
- Eur-lex.europa.eu. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:31997R0258&from=EN (accessed on 16 February 2021).
- Clugston, G.A.; Smith, T.E. Global Nutrition Problems and Novel Foods: Global Nutrition Problems and Novel Foods. Asia Pac. J. Clin. Nutr. 2002, 11, S100–S111. [Google Scholar] [CrossRef]
- Bonaccorsi, G.; Garamella, G.; Cavallo, G.; Lorini, C. A Systematic Review of Risk Assessment Associated with Jellyfish Consumption as a Potential Novel Food. Foods 2020, 9, 935. [Google Scholar] [CrossRef]
- Calabrese, M.G.; Ferranti, P. Novel Foods: New Food Sources. In Encyclopedia of Food Security and Sustainability; Elsevier: Amsterdam, The Netherlands; Cambridge, MA, USA, 2019; pp. 271–275. [Google Scholar] [CrossRef]
- Buschmann, A.H.; Camus, C.; Infante, J.; Neori, A.; Israel, Á.; Hernández-González, M.C.; Pereda, S.V.; Gomez-Pinchetti, J.L.; Golberg, A.; Tadmor-Shalev, N.; et al. Seaweed Production: Overview of the Global State of Exploitation, Farming and Emerging Research Activity. Eur. J. Phycol. 2017, 52, 391–406. [Google Scholar] [CrossRef]
- Banach, J.L.; Hoek-van den Hil, E.F.; Fels-Klerx, H.J. Food Safety Hazards in the European Seaweed Chain. Compr. Rev. Food Sci. Food Saf. 2020, 19, 332–364. [Google Scholar] [CrossRef]
- McHugh, D.J. A Guide to the Seaweed Industry. In FAO Fisheries Technical Paper 441; FAO: Rome, Italy, 2003. [Google Scholar]
- Morrissey, J.; Kraan, S.; Guiry, M.D. A Guide to Commercially Important Seaweeds on the Irish Coast; Bord Iascaigh Mhara/Irish Sea Fisheries Board Crofton House, Crofton Road, DunLaoghaire, Co.: Dublin, Ireland, 2001; 66p.
- Mahadevan, K. Seaweeds: A Sustainable Food Source. In Seaweed Sustainability; Elsevier: London, UK; Waltham, MA, USA, 2015; pp. 347–364. [Google Scholar] [CrossRef]
- Bobin-Dubigeon, C.; Hoebler, C.; Vincent, L.; Dagorn-Scaviner, C.; Mabeau, S.; Barry, J.-L.; Lahaye, M. Chemical Composition, Physico-Chemical Properties, Enzymatic Inhibition and Fermentative Characteristics of Dietary Fibres from Edible Seaweeds. Sci. Aliment. 1997, 17, 619–639. [Google Scholar]
- Bocanegra, A.; Bastida, S.; Benedí, J.; Ródenas, S.; Sánchez-Muniz, F.J. Characteristics and Nutritional and Cardiovascular-Health Properties of Seaweeds. J. Med. Food 2009, 12, 236–258. [Google Scholar] [CrossRef]
- Rupérez, P.; Saura-Calixto, F. Dietary Fibre and Physicochemical Properties of Edible Spanish Seaweeds. Eur. Food Res. Technol. 2001, 212, 349–354. [Google Scholar] [CrossRef]
- Yoshie, Y.; Suzuki, T.; Shirai, T.; Hirano, T. Changes in the Contents of Dietary Fibers, Minerals, Free Amino Acids, and Fatty Acids during Processing of Dried Nori. Nippon Suisan Gakkaishi 1994, 60, 117–123. [Google Scholar] [CrossRef][Green Version]
- Dawczynski, C.; Schubert, R.; Jahreis, G. Amino Acids, Fatty Acids, and Dietary Fibre in Edible Seaweed Products. Food Chem. 2007, 103, 891–899. [Google Scholar] [CrossRef]
- Galland-Irmouli, A.-V.; Fleurence, J.; Lamghari, R.; Luçon, M.; Rouxel, C.; Barbaroux, O.; Bronowicki, J.-P.; Villaume, C.; Guéant, J.-L. Nutritional Value of Proteins from Edible Seaweed Palmaria Palmata (Dulse). J. Nutr. Biochem. 1999, 10, 353–359. [Google Scholar] [CrossRef]
- Peñalver, R.; Lorenzo, J.M.; Ros, G.; Amarowicz, R.; Pateiro, M.; Nieto, G. Seaweeds as a Functional Ingredient for a Healthy Diet. Mar. Drugs 2020, 18, 301. [Google Scholar] [CrossRef]
- Luo, M.; Hu, K.; Zeng, Q.; Yang, X.; Wang, Y.; Dong, L.; Huang, F.; Zhang, R.; Su, D. Comparative Analysis of the Morphological Property and Chemical Composition of Soluble and Insoluble Dietary Fiber with Bound Phenolic Compounds from Different Algae. J. Food Sci. 2020, 85, 3843–3851. [Google Scholar] [CrossRef]
- Lahaye, M. Marine Algae as Sources of Fibres: Determination of Soluble and Insoluble Dietary Fibre Contents in Some ‘Sea Vegetables’. J. Sci. Food Agric. 1991, 54, 587–594. [Google Scholar] [CrossRef]
- MacArtain, P.; Gill, C.I.R.; Brooks, M.; Campbell, R.; Rowland, I.R. Nutritional Value of Edible Seaweeds. Nutr. Rev. 2008, 65, 535–543. [Google Scholar] [CrossRef]
- Lindsey White, W.; Wilson, P. World Seaweed Utilization. In Seaweed Sustainability; Elsevier: London, UK; Waltham, MA, USA, 2015; pp. 7–25. [Google Scholar] [CrossRef]
- Burtin, P. Nutritional Value of Seaweeds. Electron. J. Environ. Agric Food Chem. 2003, 2, 17. [Google Scholar]
- Eur-lex.europa.eu. Available online: https://eur-lex.europa.eu/eli/reg/2005/2073/2014-06-01 (accessed on 16 February 2021).
- Eur-lex.europa.eu. Available online: https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=celex%3A32006R1881 (accessed on 16 February 2021).
- Eur-lex.europa.eu. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=CELEX%3A32015R0186 (accessed on 16 February 2021).
- Eur-lex.europa.eu. Available online: https://eur-lex.europa.eu/legal-content/GA/TXT/?uri=CELEX%3A32013R1275 (accessed on 16 February 2021).
- Ceva-algues.com. Available online: https://www.ceva-algues.com/en/document/edible-algae-regulatory-update/ (accessed on 16 February 2021).
- Eur-lex.europa.eu. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/HTML/?uri=CELEX:32011R1169&from=EN (accessed on 16 February 2021).
- Blikra, M.J.; Løvdal, T.; Vaka, M.R.; Roiha, I.S.; Lunestad, B.T.; Lindseth, C.; Skipnes, D. Assessment of Food Quality and Microbial Safety of Brown Macroalgae (Alaria Esculenta and Saccharina Latissima). J. Sci. Food Agric. 2019, 99, 1198–1206. [Google Scholar] [CrossRef]
- Choi, E.S.; Kim, N.H.; Kim, H.W.; Kim, S.A.; Jo, J.I.; Kim, S.H.; Lee, S.H.; Ha, S.D.; Rhee, M.S. Microbiological Quality of Seasoned Roasted Laver and Potential Hazard Control in a Real Processing Line. J. Food Prot. 2014, 77, 2069–2075. [Google Scholar] [CrossRef]
- EFSA Panel on Nutrition, Novel Foods and Food Allergens (NDA); Turck, D.; Castenmiller, J.; De Henauw, S.; Hirsch-Ernst, K.I.; Kearney, J.; Maciuk, A.; Mangelsdorf, I.; McArdle, H.J.; Naska, A.; et al. Safety of Dried Whole Cell Euglena Gracilis as a Novel Food Pursuant to Regulation (EU) 2015/2283. EFSA J. 2020, 18. [Google Scholar] [CrossRef]
- EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA); Turck, D.; Bresson, J.; Burlingame, B.; Dean, T.; Fairweather-Tait, S.; Heinonen, M.; Hirsch-Ernst, K.I.; Mangelsdorf, I.; McArdle, H.J.; et al. Safety of Ecklonia Cava Phlorotannins as a Novel Food Pursuant to Regulation (EC) No 258/97. EFSA J. 2017, 15. [Google Scholar] [CrossRef]
- Nayyar, D.; Skonberg, D.I. Contrasting Effects of Two Storage Temperatures on the Microbial, Physicochemical, and Sensory Properties of Two Fresh Red Seaweeds, Palmaria Palmata and Gracilaria Tikvahiae. J. Appl. Phycol. 2019, 31, 731–739. [Google Scholar] [CrossRef]
- Park, J.H.; Jeong, H.S.; Lee, J.S.; Lee, S.W.; Choi, Y.H.; Choi, S.J.; Joo, I.S.; Kim, Y.R.; Park, Y.K.; Youn, S.K. First Norovirus Outbreaks Associated with Consumption of Green Seaweed (Enteromorpha Spp.) in South Korea. Epidemiol. Infect. 2015, 143, 515–521. [Google Scholar] [CrossRef]
- Sakon, N.; Sadamasu, K.; Shinkai, T.; Hamajima, Y.; Yoshitomi, H.; Matsushima, Y.; Takada, R.; Terasoma, F.; Nakamura, A.; Komano, J.; et al. Foodborne Outbreaks Caused by Human Norovirus GII.P17-GII.17–Contaminated Nori, Japan, 2017. Emerg. Infect. Dis. 2018, 24, 920–923. [Google Scholar] [CrossRef]
- Kular, H.; Dean, J.; Cook, V. A case of carrageenan allergy in a pediatric patient. Ann. Allergy Asthma Immunol. 2018, 121, S119. [Google Scholar] [CrossRef]
- Lang-Yona, N.; Kunert, A.T.; Vogel, L.; Kampf, C.J.; Bellinghausen, I.; Saloga, J.; Schink, A.; Ziegler, K.; Lucas, K.; Schuppan, D.; et al. Fresh Water, Marine and Terrestrial Cyanobacteria Display Distinct Allergen Characteristics. Sci. Total Environ. 2018, 612, 767–774. [Google Scholar] [CrossRef]
- Le, T.-M.; Knulst, A.C.; Röckmann, H. Anaphylaxis to Spirulina Confirmed by Skin Prick Test with Ingredients of Spirulina Tablets. Food Chem. Toxicol. 2014, 74, 309–310. [Google Scholar] [CrossRef] [PubMed]
- Thomas, I.; Siew, L.Q.C.; Watts, T.J.; Haque, R. Seaweed Allergy. J. Allergy Clin. Immunol. Pract. 2019, 7, 714–715. [Google Scholar] [CrossRef] [PubMed]
- Uchida, M.; Kurushima, H.; Ishihara, K.; Murata, Y.; Touhata, K.; Ishida, N.; Niwa, K.; Araki, T. Characterization of Fermented Seaweed Sauce Prepared from Nori (Pyropia Yezoensis). J. Biosci. Bioeng. 2017, 123, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Uchida, M.; Kurushima, H.; Hideshima, N.; Araki, T.; Ishihara, K.; Murata, Y.; Touhata, K.; Ishida, N. Preparation and Characterization of Fermented Seaweed Sauce Manufactured from Low-Quality Nori (Dried and Fresh Fronds of Pyropia Yezoensis). Fish. Sci. 2018, 84, 589–596. [Google Scholar] [CrossRef]
- Kusaba, Y.; Miwa, T.; Ise, M.; Minoda, R. Pharyngocutaneous Fistula Caused by Dried ‘Kombu’ (Edible Seaweed) after Total Laryngectomy. BMJ Case Rep. 2019, 12, e228091. [Google Scholar] [CrossRef]
- Panebianco, F.; Giusti, A.; Giarratana, F.; Armani, A. Ethnic Seafood Products Sold on the Italian Market: Labelling Assessment and Biological, Chemical and Physical Risk Characterization. Food Control 2019, 105, 198–208. [Google Scholar] [CrossRef]
- Ali, A.Y.A.; Idris, A.M.; Eltayeb, M.A.H.; El-Zahhar, A.A.; Ashraf, I.M. Bioaccumulation and Health Risk Assessment of Toxic Metals in Red Algae in Sudanese Red Sea Coast. Toxin Rev. 2019, 1–11. [Google Scholar] [CrossRef]
- Ardiyansyah, O.; Sudarno, R. Bioaccumulation of Cadmium (Cd) Heavy Metal on Seaweed (Gracilaria Sp.) in Traditional Fishpond of Jabon Subdistrict, Sidoarjo District. IOP Conf. Ser. Earth Environ. Sci. 2019, 236, 012059. [Google Scholar] [CrossRef]
- Arulkumar, A.; Nigariga, P.; Paramasivam, S.; Rajaram, R. Metals Accumulation in Edible Marine Algae Collected from Thondi Coast of Palk Bay, Southeastern India. Chemosphere 2019, 221, 856–862. [Google Scholar] [CrossRef]
- Biancarosa, I.; Belghit, I.; Bruckner, C.G.; Liland, N.S.; Waagbø, R.; Amlund, H.; Heesch, S.; Lock, E.-J. Chemical Characterization of 21 Species of Marine Macroalgae Common in Norwegian Waters: Benefits of and Limitations to Their Potential Use in Food and Feed: Chemical Composition of Norwegian Marine Macroalgae. J. Sci. Food Agric. 2018, 98, 2035–2042. [Google Scholar] [CrossRef]
- Chen, Q.; Pan, X.-D.; Huang, B.-F.; Han, J.-L. Distribution of Metals and Metalloids in Dried Seaweeds and Health Risk to Population in Southeastern China. Sci. Rep. 2018, 8, 3578. [Google Scholar] [CrossRef]
- Filippini, M.; Baldisserotto, A.; Menotta, S.; Fedrizzi, G.; Rubini, S.; Gigliotti, D.; Valpiani, G.; Buzzi, R.; Manfredini, S.; Vertuani, S. Heavy Metals and Potential Risks in Edible Seaweed on the Market in Italy. Chemosphere 2021, 263, 127983. [Google Scholar] [CrossRef]
- Epa.gov. Available online: https://cfpub.epa.gov/ncea/risk/recordisplay.cfm?deid=190187 (accessed on 16 February 2021).
- Francisco, J.; Cardoso, C.; Bandarra, N.; Brito, P.; Horta, A.; Pedrosa, R.; Gil, M.M.; Delgado, I.M.; Castanheira, I.; Afonso, C. Bioaccessibility of Target Essential Elements and Contaminants from Fucus Spiralis. J. Food Compos. Anal. 2018, 74, 10–17. [Google Scholar] [CrossRef]
- Kim, J.K.; Kraemer, G.; Yarish, C. Evaluation of the Metal Content of Farm Grown Gracilaria Tikvahiae and Saccharina Latissima from Long Island Sound and New York Estuaries. Algal Res. 2019, 40, 101484. [Google Scholar] [CrossRef]
- Li, J.-Y.; Yang, F.; Jin, L.; Wang, Q.; Yin, J.; He, P.; Chen, Y. Safety and Quality of the Green Tide Algal Species Ulva Prolifera for Option of Human Consumption: A Nutrition and Contamination Study. Chemosphere 2018, 210, 1021–1028. [Google Scholar] [CrossRef]
- Mise, N.; Ohtsu, M.; Ikegami, A.; Mizuno, A.; Cui, X.; Kobayashi, Y.; Nakagi, Y.; Nohara, K.; Yoshida, T.; Kayama, F. Hijiki Seaweed Consumption Elevates Levels of Inorganic Arsenic Intake in Japanese Children and Pregnant Women. Food Addit. Contam. 2019, 36, 84–95. [Google Scholar] [CrossRef]
- Paz, S.; Rubio, C.; Frías, I.; Gutiérrez, A.J.; González-Weller, D.; Revert, C.; Hardisson, A. Metal Concentrations in Wild-Harvested Phaeophyta Seaweed from the Atlantic Ocean (Canary Islands, Spain). J. Food Prot. 2018, 81, 1165–1170. [Google Scholar] [CrossRef]
- Paz, S.; Rubio, C.; Frías, I.; Gutiérrez, Á.J.; González-Weller, D.; Martín, V.; Revert, C.; Hardisson, A. Toxic Metals (Al, Cd, Pb and Hg) in the Most Consumed Edible Seaweeds in Europe. Chemosphere 2019, 218, 879–884. [Google Scholar] [CrossRef]
- Sá Monteiro, M.; Sloth, J.; Holdt, S.; Hansen, M. Analysis and Risk Assessment of Seaweed. EFSA J. 2019, 17. [Google Scholar] [CrossRef]
- Santos-Silva, M.C.; Machado, E.C.; Wallner-Kersanach, M.; Camargo, M.G.; Andrade, C.; Sá, F.; Pellizzari, F. Background Levels of Trace Elements in Brown and Red Seaweeds from Trindade, a Remote Island in South Atlantic Ocean. Mar. Pollut. Bull. 2018, 135, 923–931. [Google Scholar] [CrossRef]
- Squadrone, S.; Brizio, P.; Battuello, M.; Nurra, N.; Sartor, R.M.; Riva, A.; Staiti, M.; Benedetto, A.; Pessani, D.; Abete, M.C. Trace Metal Occurrence in Mediterranean Seaweeds. Environ. Sci. Pollut. Res. 2018, 25, 9708–9721. [Google Scholar] [CrossRef]
- Stévant, P.; Marfaing, H.; Duinker, A.; Fleurence, J.; Rustad, T.; Sandbakken, I.; Chapman, A. Biomass Soaking Treatments to Reduce Potentially Undesirable Compounds in the Edible Seaweeds Sugar Kelp (Saccharina Latissima) and Winged Kelp (Alaria Esculenta) and Health Risk Estimation for Human Consumption. J. Appl. Phycol. 2018, 30, 2047–2060. [Google Scholar] [CrossRef]
- Zhang, L.; Song, H.; Guo, Y.; Fan, B.; Huang, Y.; Mao, X.; Liang, K.; Hu, Z.; Sun, X.; Fang, Y.; et al. Benefit–Risk Assessment of Dietary Selenium and Its Associated Metals Intake in China (2017–2019): Is Current Selenium-Rich Agro-Food Safe Enough? J. Hazard. Mater. 2020, 398, 123224. [Google Scholar] [CrossRef]
- Rzymski, P.; Budzulak, J.; Niedzielski, P.; Klimaszyk, P.; Proch, J.; Kozak, L.; Poniedziałek, B. Essential and Toxic Elements in Commercial Microalgal Food Supplements. J. Appl. Phycol. 2019, 31, 3567–3579. [Google Scholar] [CrossRef]
- Liu, C.; Lin, H.; Mi, N.; Liu, F.; Song, Y.; Liu, Z.; Sui, J. Bioaccessibility and Health Risk Assessment of Rare Earth Elements in Porphyra Seaweed Species. Hum. Ecol. Risk Assess. Int. J. 2018, 24, 721–730. [Google Scholar] [CrossRef]
- Squadrone, S.; Brizio, P.; Stella, C.; Mantia, M.; Battuello, M.; Nurra, N.; Sartor, R.M.; Orusa, R.; Robetto, S.; Brusa, F.; et al. Rare Earth Elements in Marine and Terrestrial Matrices of Northwestern Italy: Implications for Food Safety and Human Health. Sci. Total Environ. 2019, 660, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Uddin, S.; Bebhehani, M.; Sajid, S.; Karam, Q. Concentration of 210Po and 210Pb in Macroalgae from the Northern Gulf. Mar. Pollut. Bull. 2019, 145, 474–479. [Google Scholar] [CrossRef] [PubMed]
- Miller, T.R.; Xiong, A.; Deeds, J.R.; Stutts, W.L.; Samdal, I.A.; Løvberg, K.E.; Miles, C.O. Microcystin Toxins at Potentially Hazardous Levels in Algal Dietary Supplements Revealed by a Combination of Bioassay, Immunoassay, and Mass Spectrometric Methods. J. Agric. Food Chem. 2020, 68, 8016–8025. [Google Scholar] [CrossRef] [PubMed]
- Cofrades, S.; Serdaroğlu, M.; Jiménez-Colmenero, F. Design of Healthier Foods and Beverages Containing Whole Algae. In Functional Ingredients from Algae for Foods and Nutraceuticals; Elsevier: Oxford, UK; Cambridge, UK, 2013; pp. 609–633. [Google Scholar] [CrossRef]
- Ścieszka, S.; Klewicka, E. Algae in Food: A General Review. Crit. Rev. Food Sci. Nutr. 2019, 59, 3538–3547. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations. The Global Status of Seaweed Production, Trade and Utilization; Globefish Research Programme; FAO: Rome, Italy, 2018; Volume 124, 120p. [Google Scholar]
- Alemañ, A.E.; Robledo, D.; Hayashi, L. Development of Seaweed Cultivation in Latin America: Current Trends and Future Prospects. Phycologia 2019, 58, 462–471. [Google Scholar] [CrossRef]
- Pina-Pérez, M.C.; Rivas, A.; Martínez, A.; Rodrigo, D. Antimicrobial Potential of Macro and Microalgae against Pathogenic and Spoilage Microorganisms in Food. Food Chem. 2017, 235, 34–44. [Google Scholar] [CrossRef]
- Cho, T.J.; Rhee, M.S. Health Functionality and Quality Control of Laver (Porphyra, Pyropia): Current Issues and Future Perspectives as an Edible Seaweed. Mar. Drugs 2019, 18, 14. [Google Scholar] [CrossRef]
- Rodríguez De Marco, E.; Steffolani, M.E.; Martínez, C.S.; León, A.E. Effects of Spirulina Biomass on the Technological and Nutritional Quality of Bread Wheat Pasta. LWT Food Sci. Technol. 2014, 58, 102–108. [Google Scholar] [CrossRef]
- Fradique, M.; Batista, A.P.; Nunes, M.C.; Gouveia, L.; Bandarra, N.M.; Raymundo, A. Incorporation of Chlorella Vulgaris and Spirulina Maxima Biomass in Pasta Products. Part 1: Preparation and Evaluation: Incorporation of Microalgae Biomass in Pasta Products. J. Sci. Food Agric. 2010, 90, 1656–1664. [Google Scholar] [CrossRef]
- Prabhasankar, P.; Ganesan, P.; Bhaskar, N.; Hirose, A.; Stephen, N.; Gowda, L.R.; Hosokawa, M.; Miyashita, K. Edible Japanese Seaweed, Wakame (Undaria Pinnatifida) as an Ingredient in Pasta: Chemical, Functional and Structural Evaluation. Food Chem. 2009, 115, 501–508. [Google Scholar] [CrossRef]
- Fradinho, P.; Raymundo, A.; Sousa, I.; Domínguez, H.; Torres, M.D. Edible Brown Seaweed in Gluten-Free Pasta: Technological and Nutritional Evaluation. Foods 2019, 8, 622. [Google Scholar] [CrossRef]
- Ak, B.; Avşaroğlu, E.; Işık, O.; Özyurt, G.; Kafkas, E.; Uslu, L. Nutritional and Physicochemical Characteristics of Bread Enriched with Microalgae Spirulina Platensis. Int. J. Eng. Res. Appl. 2016, 6, 9. [Google Scholar]
- Graça, C.; Fradinho, P.; Sousa, I.; Raymundo, A. Impact of Chlorella Vulgaris on the Rheology of Wheat Flour Dough and Bread Texture. LWT 2018, 89, 466–474. [Google Scholar] [CrossRef]
- Nunes, M.C.; Graça, C.; Vlaisavljević, S.; Tenreiro, A.; Sousa, I.; Raymundo, A. Microalgal Cell Disruption: Effect on the Bioactivity and Rheology of Wheat Bread. Algal Res. 2020, 45, 101749. [Google Scholar] [CrossRef]
- García-Segovia, P.; Pagán-Moreno, M.J.; Lara, I.F.; Martínez-Monzó, J. Effect of Microalgae Incorporation on Physicochemical and Textural Properties in Wheat Bread Formulation. Food Sci. Technol. Int. 2017, 23, 437–447. [Google Scholar] [CrossRef]
- Lafarga, T.; Acién-Fernández, F.G.; Castellari, M.; Villaró, S.; Bobo, G.; Aguiló-Aguayo, I. Effect of Microalgae Incorporation on the Physicochemical, Nutritional, and Sensorial Properties of an Innovative Broccoli Soup. LWT 2019, 111, 167–174. [Google Scholar] [CrossRef]
- Wang, T.; Jónsdóttir, R.; Kristinsson, H.G.; Thorkelsson, G.; Jacobsen, C.; Hamaguchi, P.Y.; Ólafsdóttir, G. Inhibition of Haemoglobin-Mediated Lipid Oxidation in Washed Cod Muscle and Cod Protein Isolates by Fucus Vesiculosus Extract and Fractions. Food Chem. 2010, 123, 321–330. [Google Scholar] [CrossRef]
- Gupta, S.; Abu-Ghannam, N. Recent Developments in the Application of Seaweeds or Seaweed Extracts as a Means for Enhancing the Safety and Quality Attributes of Foods. Innov. Food Sci. Emerg. Technol. 2011, 12, 600–609. [Google Scholar] [CrossRef]
- Cofrades, S.; López-López, I.; Solas, M.T.; Bravo, L.; Jiménez-Colmenero, F. Influence of Different Types and Proportions of Added Edible Seaweeds on Characteristics of Low-Salt Gel/Emulsion Meat Systems. Meat Sci. 2008, 79, 767–776. [Google Scholar] [CrossRef]
- Lucas, B.F.; de Morais, M.G.; Santos, T.D.; Costa, J.A.V. Spirulina for Snack Enrichment: Nutritional, Physical and Sensory Evaluations. LWT 2018, 90, 270–276. [Google Scholar] [CrossRef]
- Dolan, L.C.; Matulka, R.A.; Burdock, G.A. Naturally Occurring Food Toxins. Toxins 2010, 2, 2289–2332. [Google Scholar] [CrossRef] [PubMed]
- Bonaccorsi, G.; Simoncini, E.; Gestri, D.; Talini, M.; Tesi, S.; Rastelli, F.; Nardone, G.; Lorini, C. The microbial safety of edible insects: A matter of processing. Ann. Ist. Super Sanita 2019, 55, 203–204. [Google Scholar] [CrossRef] [PubMed]
- Stomp, A.-M. The Duckweeds: A Valuable Plant for Biomanufacturing. Biotechnol. Annu. Rev. 2005, 11, 69–99. [Google Scholar] [CrossRef]
- Van der Spiegel, M.; Noordam, M.Y.; van der Fels-Klerx, H.J. Safety of Novel Protein Sources (Insects, Microalgae, Seaweed, Duckweed, and Rapeseed) and Legislative Aspects for Their Application in Food and Feed Production. Compr. Rev. Food Sci. Food Saf. 2013, 12, 662–678. [Google Scholar] [CrossRef]
- Ncbi.nlm.nih.gov. Available online: https://www.ncbi.nlm.nih.gov/books/NBK482131/ (accessed on 16 February 2021).
- Wang, S.-L.; Xu, X.-R.; Sun, Y.-X.; Liu, J.-L.; Li, H.-B. Heavy Metal Pollution in Coastal Areas of South China: A Review. Mar. Pollut. Bull. 2013, 76, 7–15. [Google Scholar] [CrossRef]
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy Metal Toxicity and the Environment. In Molecular, Clinical and Environmental Toxicology; Luch, A., Ed.; Experientia Supplementum; Springer: Basel, Switzerland, 2012; Volume 101, pp. 133–164. [Google Scholar] [CrossRef]
- Li, Z.; Ma, Z.; van der Kuijp, T.J.; Yuan, Z.; Huang, L. A Review of Soil Heavy Metal Pollution from Mines in China: Pollution and Health Risk Assessment. Sci. Total Environ. 2014, 468–469, 843–853. [Google Scholar] [CrossRef]
- EFSA Panel on Contaminants in the Food Chain (CONTAM). Scientific Opinion on Lead in Food. EFSA J. 2010, 8, 151. [Google Scholar] [CrossRef]
- Roleda, M.Y.; Marfaing, H.; Desnica, N.; Jónsdóttir, R.; Skjermo, J.; Rebours, C.; Nitschke, U. Variations in Polyphenol and Heavy Metal Contents of Wild-Harvested and Cultivated Seaweed Bulk Biomass: Health Risk Assessment and Implication for Food Applications. Food Control 2019, 95, 121–134. [Google Scholar] [CrossRef]
- Vasconcelos, M.T.S.D.; Leal, M.F.C. Seasonal Variability in the Kinetics of Cu, Pb, Cd and Hg Accumulation by Macroalgae. Mar. Chem. 2001, 74, 65–85. [Google Scholar] [CrossRef]
- Priyadarshini, E.; Priyadarshini, S.S.; Pradhan, N. Heavy Metal Resistance in Algae and Its Application for Metal Nanoparticle Synthesis. Appl. Microbiol. Biotechnol. 2019, 103, 3297–3316. [Google Scholar] [CrossRef]
- Circuncisão, A.R.; Catarino, M.D.; Cardoso, S.M.; Silva, A.M.S. Minerals from Macroalgae Origin: Health Benefits and Risks for Consumers. Mar. Drugs 2018, 16, 400. [Google Scholar] [CrossRef]
- Guo, J.D.; Jie, Y.; Shuo, Z.; Jin, Y.D. A Survey of 16 Rare Earth Elements in the Major Foods in China. Biomed. Environ. Sci. 2012, 25, 267–271. [Google Scholar] [CrossRef]
- Kulaksız, S.; Bau, M. Anthropogenic Gadolinium as a Microcontaminant in Tap Water Used as Drinking Water in Urban Areas and Megacities. Appl. Geochem. 2011, 26, 1877–1885. [Google Scholar] [CrossRef]
- Gómez-Aracena, J.; Riemersma, R.A.; Gutiérrez-Bedmar, M.; Bode, P.; Kark, J.D.; Garcia-Rodríguez, A.; Gorgojo, L.; van’t Veer, P.; Fernández-Crehuet, J.; Kok, F.J.; et al. Toenail Cerium Levels and Risk of a First Acute Myocardial Infarction: The EURAMIC and Heavy Metals Study. Chemosphere 2006, 64, 112–120. [Google Scholar] [CrossRef]
- Zhang, H.; Feng, J.; Zhu, W.; Liu, C.; Xu, S.; Shao, P.; Wu, D.; Yang, W.; Gu, J. Chronic Toxicity of Rare-Earth Elements on Human Beings: Implications of Blood Biochemical Indices in REE-High Regions, South Jiangxi. Biol. Trace Elem. Res. 2000, 73, 1–17. [Google Scholar] [CrossRef]
- Wu, L.; Zhou, Y.; Zhong, H. [A Case-Control Study on the Risk Factors of Leukemia in Mining Areas of Rare-Earth in South Jiangxi]. Zhonghua Liu Xing Bing Xue Za Zhi Zhonghua Liuxingbingxue Zazhi 2003, 24, 879–882. [Google Scholar]
- Zhu, W.; Xu, S.; Shao, P.; Zhang, H.; Wu, D.; Yang, W.; Feng, J.; Feng, L. Investigation on Liver Function Among Population in High Background of Rare Earth Area in South China. Biol. Trace Elem. Res. 2005, 104, 1–7. [Google Scholar] [CrossRef]
- Habibullah-Al-Mamun, M.; Ahmed, M.K.; Islam, M.S.; Tokumura, M.; Masunaga, S. Distribution of Polycyclic Aromatic Hydrocarbons (PAHs) in Commonly Consumed Seafood from Coastal Areas of Bangladesh and Associated Human Health Implications. Environ. Geochem. Health 2019, 41, 1105–1121. [Google Scholar] [CrossRef]
- Zhang, Y.; Shi, G.-L.; Guo, C.-S.; Xu, J.; Tian, Y.-Z.; Feng, Y.-C.; Wang, Y.-Q. Seasonal Variations of Concentrations, Profiles and Possible Sources of Polycyclic Aromatic Hydrocarbons in Sediments from Taihu Lake, China. J. Soils Sediments 2012, 12, 933–941. [Google Scholar] [CrossRef]
- Gu, Y.-G.; Ke, C.-L.; Liu, Q. Characterization, Sources, and Ecological Hazards of Polycyclic Aromatic Hydrocarbons in the Intertidal Sediments of Zhelin Bay, the Biggest Mariculture Area on the Eastern Guangdong Coast of China. Mar. Pollut. Bull. 2018, 130, 192–197. [Google Scholar] [CrossRef]
- Pavoni, B.; Caliceti, M.; Sperni, L.; Sfriso, A. Organic Micropollutants (PAHs, PCBs, Pesticides) in Seaweeds of the Lagoon of Venice. Oceanol. Acta 2003, 26, 585–596. [Google Scholar] [CrossRef]
- International Atomic Energy Agency. The Environmental Behaviour of Polonium; Technical Reports Series No. 484; IAEA: Vienna, Austria, 2017; 255p, ISBN 978–92–0–112116–5. [Google Scholar]
- Aarkrog, A.; Baxter, M.S.; Bettencourt, A.O.; Bojanowski, R.; Bologa, A.; Charmasson, S.; Cunha, I.; Delfanti, R.; Duran, E.; Holm, E.; et al. A Comparison of Doses from 137Cs and 210Po in Marine Food: A Major International Study. J. Environ. Radioact. 1997, 34, 69–90. [Google Scholar] [CrossRef]
- Du, P.; Walling, D.E. Using 210Pb Measurements to Estimate Sedimentation Rates on River Floodplains. J. Environ. Radioact. 2012, 103, 59–75. [Google Scholar] [CrossRef]
- Uddin, S.; Aba, A.; Bebhehani, M. Baseline Concentration of 210Po and 210Pb in Sargassum from the Northern Gulf. Mar. Pollut. Bull. 2015, 90, 330–333. [Google Scholar] [CrossRef]
- Sirelkhatim, D.A.; Sam, A.K.; Hassona, R.K. Distribution of 226Ra–210Pb–210Po in Marine Biota and Surface Sediments of the Red Sea, Sudan. J. Environ. Radioact. 2008, 99, 1825–1828. [Google Scholar] [CrossRef]
- Eur-lex.europa.eu. Available online: https://eur-lex.europa.eu/eli/reg/2005/396/oj (accessed on 16 February 2021).
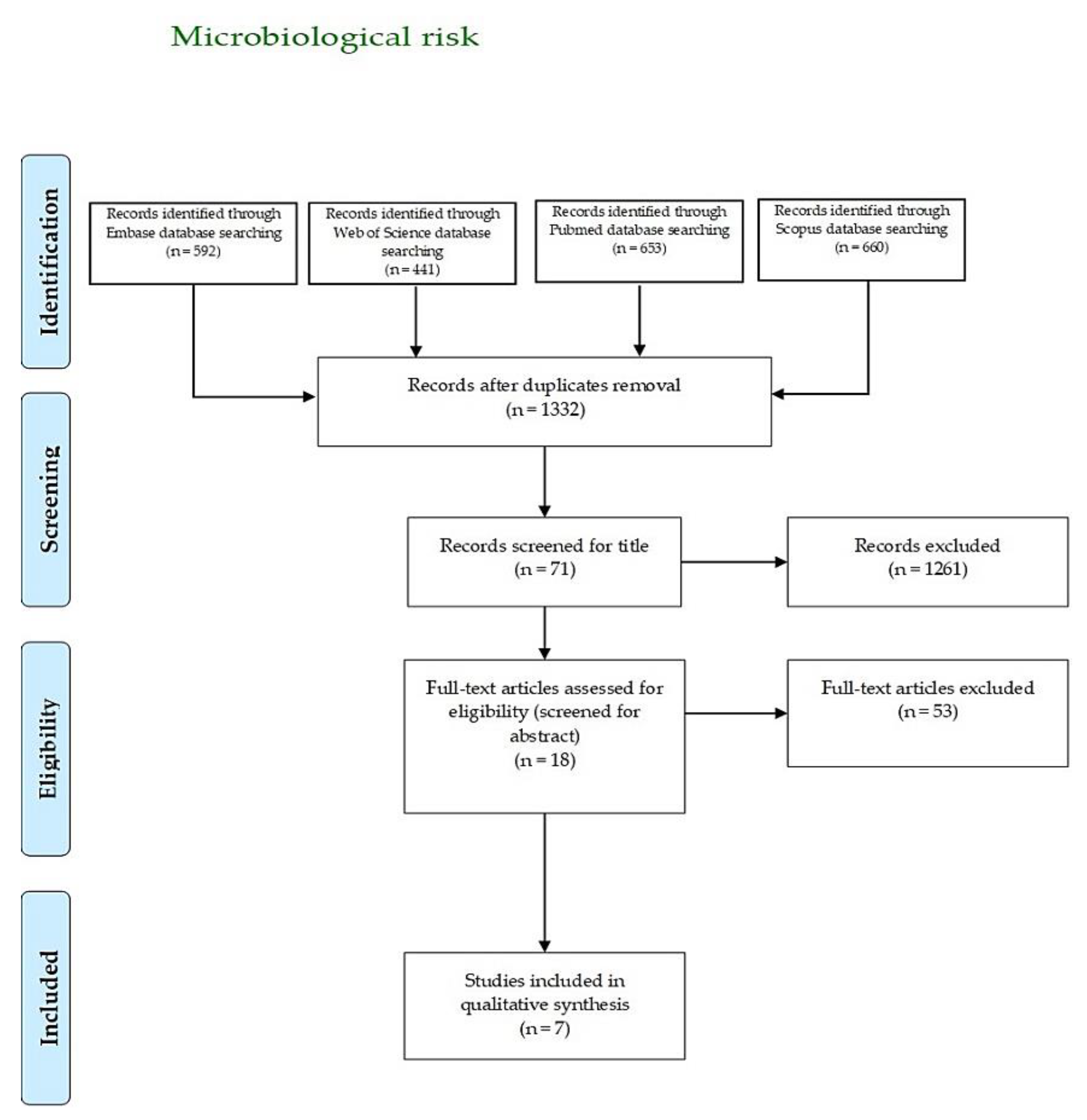

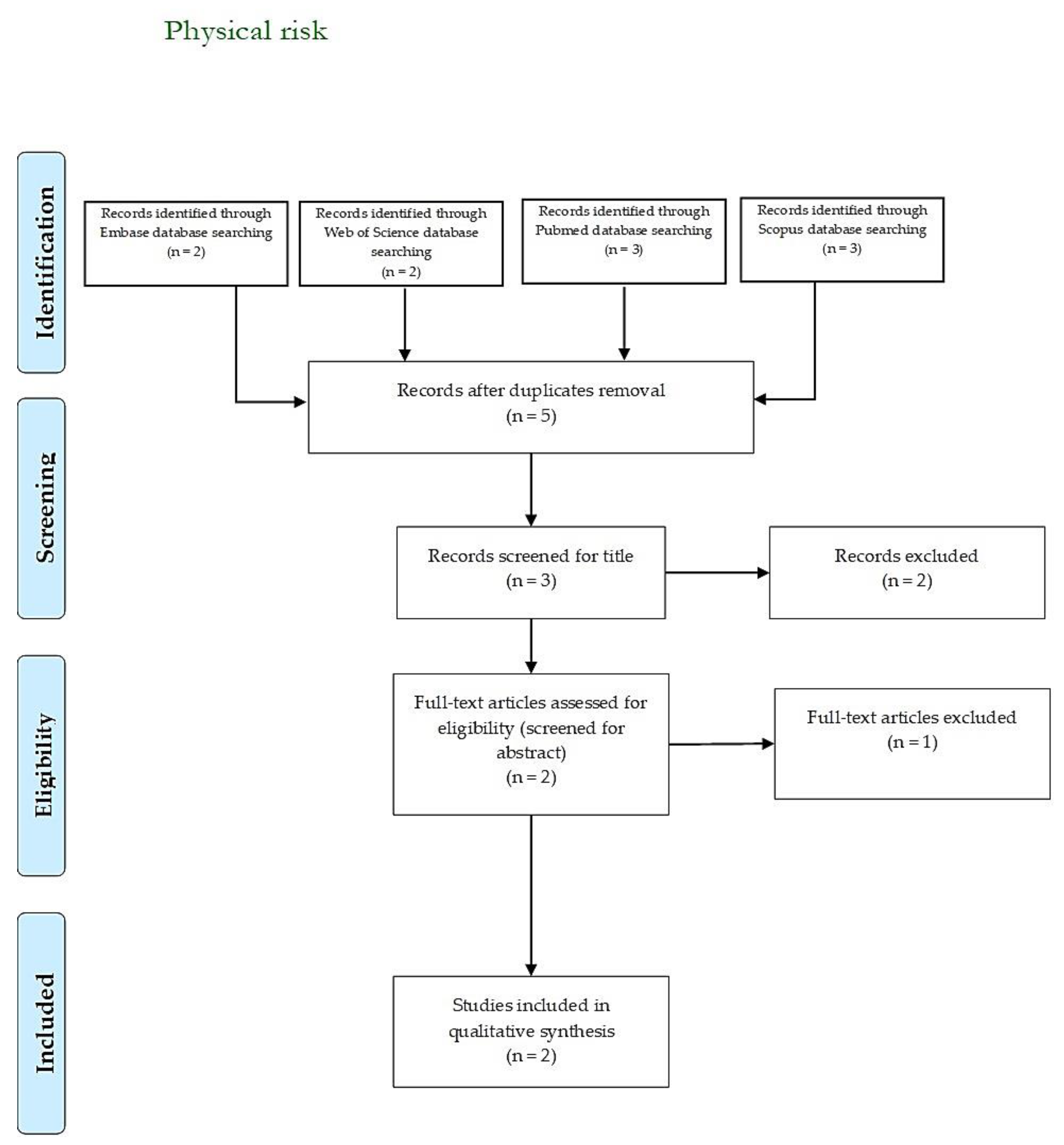
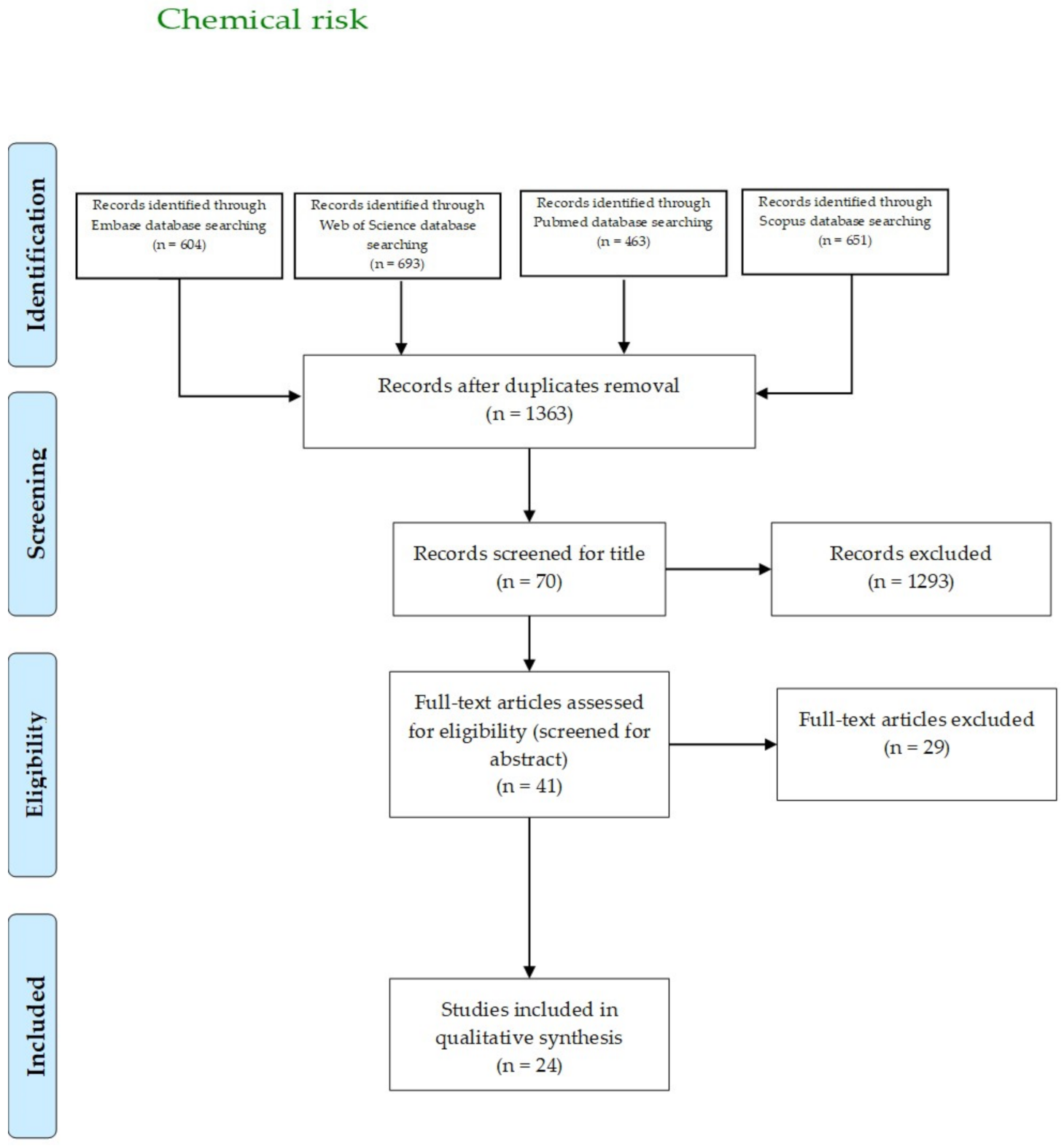
| Author, Year | Country | Aim | Sample | Main Results |
|---|---|---|---|---|
| Blikra et al., 2018 | Norway | Among other goals, this study investigated the microbiological parameters of Saccharina latissima and Alaria esculenta. | Alaria esculenta and Saccharina latissima. | The results (expressed in log CFU/g) are reported, where possible, as mean ± SD. Total microbial count: A. esculenta 2.01 ± 0.39 (raw) and 1.20 ± 0.25 (Heat-treated, HT); S. latissima 1.10 ± 0.14 (raw) and 1.13 ± 0.18 (HT). Psychotropic bacteria: A. esculenta from 1 to 1.1 (raw) and 1 ± 0.01 (HT); S. latissima from 1.01 to 1.16 (raw), and 1 ± 0.01 (HT). Spore-forming bacteria aerobic: A. esculenta from 1.70 to 2.28 (raw) and from 1 to 2.9 (HT); S. latissima 1.01 ± 0.01 (raw) and from 1 to 2.9 (HT). Spore-forming bacteria anaerobic: A. esculenta from 1.47 to 1.7 (raw) and from 1 to 2.33 (HT); S. latissima from 1.01 to 1.7 (raw), and 1.08 ± 0.20 (HT). |
| Choi et al., 2014 | South Korea | The study analyzed microbiological profile from raw materials to final seasoned roasted laver products. | Korean laver. | The dried laver: APC level of 4.4 to 7.8 log CFU/g. Supplementary Materials: APC levels from not detected to 4.5 log CFU/g. Coliforms: 2.1 log CFU/g in dried laver and 1.8 log CFU/g in the primary roasting product. Microorganism species found: the main, with 9.7%, was Moraxella followed by Clostridium (no botulinum and perfrigens) with 8.4%, Staphylococcus (not aureus) with 8.1%, Bacillus (not cereus) with 6.6%, but not B. cereus), and Neisseria with 5.3%. The microbial populations during processing decreased from 6.9 log CFU/g in the dried laver to >3.2 log CFU/g in the packaged product. Only B. cereus, among pathogenic bacteria, was detected during production process. |
| EFSA NDA Panel, 2020 | Italy | The study expressed a scientific opinion about the risk associated with the consumption of dried whole cell Euglena gracilis as a novel food (NF) pursuant to Regulation (EU) 2015/2283. | Euglena gracilis. | The Aerobic plate count (CFU/g) for each of five batches ranged from 1400 to 8000. Coliforms (most probable number, MPN/g) were <3 in all batches. Yeast and mould (CFU/g) detected varied from 20 to 70. Escherichia coli (in 10 g) was absent in all batches, as well as Staphylococcus aureus (in 10 g), Salmonella (in 25 g), and Listeria monocytogenes (in 25 g). |
| EFSA NDA Panel, 2017 | Italy | The study expressed a scientific opinion on a dietary supplement composed of Ecklonia cava phlorotannins. | Phlorotannin-rich alcohol extract of E. cava. | The viable cell count was <3000 CFU/g. Moulds and yeasts were <300 CFU/g. Ecklonia cava samples were negative for Staphylococcus aureus (CFU/g), Salmonella ssp. (CFU/25 g), and Coliforms (CFU/g). |
| Nayyar et al., 2018 | USA | The study observed, among various parameters, microbial count of two seaweeds at 2 °C and 7 °C storage temperatures. | P. palmata and G. tikvahiae. | P. palmata microbial count: at 2 °C, it started from 4 CFU/g to less than 5 log CFU/g after 11 days of storage; at 7 °C, the growth range was 3–4 CFU/g. G. tikvahiae microbial count: at 2 °C, it started from 4 to 8 CFU/g, after 12 day of storage; at 7 °C, the bacterial growth performed from 4 to 7 CFU/g at the end of storage. |
| Park et al., 2014 | South Korea | The study presented the norovirus outbreaks linked to green alga consumption. | Number of cases: ninety-one students. | The symptoms that occurred most frequently were vomit, in all cases, followed by abdominal pain and nausea. The trigger for the symptoms was the ingestion of seasoned green algae which is therefore the cause of the norovirus outbreak |
| Sakon et al., 2018 | Japan | The study reported an epidemiological investigation on norovirus outbreaks associated with nori consumption. | 2094 consumers of contaminated nori. | Gastrointestinal symptoms were reported from five schools, a commercial office, and a shop. Investigations led to the conclusion that the responsible agent was the norovirus contained in shredded nori. The possible origin of the outbreaks was identified in the different processing stages of the various manufacturing companies. |
| Author, Year | Country | Aim | Sample | Main Results |
|---|---|---|---|---|
| Kular et al., 2018 | Canada | The study described a case study, represented by an allergic reaction to the carrageenan. | A 10-month-old male with lip angioedema. | Following the consumption of a fruit cake, angioedema appeared on the lips. The skin prick test was performed which revealed the allergic reaction to carrageenan. |
| Lang-Yona et al., 2018 | Germany | The study analyzed the allergic reactivity of some cyanobacteria. | Anabaena ambigua, Cylindrospermum siamensis, Lyngbya lagerheimii, Microcystis aeruginosa, Nostoc sp., Phormidium sp., Planktothrix agardhii, and Synechocystis sp. | After laboratory analysis, C-phycocyanin (a pigment-protein complex, characteristic of the Spirulina algae) emerged as promoter of cross-reactivity with fresh marine species, compared to the other algae considered in the study. |
| Le et al., 2014 | Netherlands | The study described a case of allergic reaction after consuming Spirulina tablet. | 17-year-old male who showed symptoms associated with an allergic reaction after consuming a Spirulina food supplement. | The skin prick test confirmed the association. Phycocyanin was identified as potential allergenic element. |
| Thomas et al., 2018 | Japan | The study described a case of food allergy after consuming seaweed. | A 27-year-old man with a possible seafood allergy. | Prick to prick test for nori gave a positive result. Further investigations on red seaweeds, revealed positivity to the other species of the same algal family. Prick to prick tests for green and brown seaweeds were negative. |
| Uchida et al., 2017 | Japan | The study investigated the possible allergenic potential of nori sauce, and the cross-reactivity with other possible allergenic food. | Nori sauces set in different batches. | Allergens detected: wheat, soy, crustaceans (shrimp and crab). Results: all were negative (<0.1 mg/100 g). |
| Uchida et al., 2018 | Japan | The study investigated the allergenic potential of low-quality nori (LNs) sauce, and the cross-reactivity with other possible allergenic food. | Low-quality nori from Japan. | Allergens detected: wheat, soy, crustaceans (shrimp and crab) for low-quality nori and high-quality nori sauces. All tests were negative. |
| Author, Year | Country | Aim | Sample | Main Results |
|---|---|---|---|---|
| Kusaba et al., 2019 | Japan | The study described a case of pharyngocutaneous fistula (PCF) following Kombu ingestion. | 63-year-old person who ingested Kombu meal. | A choking episode occurred after a seaweed meal with dried Kombu. The patient underwent an emergency surgery, consisting of drainage of the abscess and the removal of a foreign body. Videoscopy revealed the presence of alga consumed in the pharyngo-esophageal tract. |
| Panebianco et al., 2019 | Italy | The study assessed chemical, microbiological, and physical hazards related to seafood commercial products. | Twenty-six differently processed seaweeds samples. | During an inspection of some seafood products, three triangular glass bodies (identified by Scanning Electron Microscopy) were found in processed nori sample. |
| Author, Year | Country | Aim | Sample | Main Results |
|---|---|---|---|---|
| Ali et al., 2019 | Saudi Arabia | The aim of this study was to detect the heavy metals levels in some red seaweeds, evaluating also the risk associated with their consumption for children and adults. | Corallma, Gracilaria, Hypnea, Jania, and Laurencia spp. | Heavy metal concentrations (μg/g) in red algae samples reported: Cr 11.7.86, Ni 6 ± 1.78, Cu 5.9 ± 2.93, Cd 0.09 ± 0.02 Pb 1.5 ± 0.41. The HI values did not represent a danger to the risk of cancer in adults and children. |
| Ardiyansyah et al., 2019 | Indonesia | This study reported data on Cd content in red alga species. | Gracilaria sp. | Cd concentration in seaweed samples: <0.0024 mg/kg. |
| Arulkumar et al., 2019 | India | The study established the concentration of Cd, Cu, Pb, and Zn in Chlorophyta, Ochrophyta, and Rhodophyta. | Thirteen edible seaweed. | Cd concentration: from 0.58 ± 0.13 to 5.24 ± 0.99 mg/kg. Pb concentration: from 14.20 ± 0.87 to 17.33 ± 0.9 mg/kg. Cu concentration: from 0.87 ± 0.07 to 8.62 ± 0.77 mg/kg. Zn concentration: from 19.59 ± 0.63 to 23.45 ± 1.03 mg/kg. |
| Biancarosa et al., 2017 | Norway | The study assessed the heavy metals and metalloids concentration in brown, green and red algae. | Twenty-one species of marine seaweeds. | Cd content: in green algae 0.12–0.18 mg/kg DW; in red algae 0.07–3.1; in brown algae 0.03–2.6 mg/kg DW. Hg concentration: from <LOQ to 0.04 mg/kg DW. Pb concentration: up to 0.58 mg/kg DW in red and brown algae; up to 3 mg/kg DW in green algae. As content: 21–120 mg/kg DW in brown algae, 6.4–24 mg/kg DW in red algae, 6.4–10 mg/kg DW in green algae. iAs concentration: <0.5 mg/kg in all samples. |
| Chen et al., 2018 | China | The study aimed to determine the metal content of algae, evaluating the risk linked to their consumption. | About three hundred Chinese seaweeds. | Concentration found in red seaweeds (mg/kg): Al 597.6 ± 594.23; As 22.05 ± 11.28; Cd 2.225 ± 1.23; Cr 2.545 ± 4.08; Cu 11.049 ± 6.277; Hg 0.01 ± 0.017; Ni 1.642 ± 1.211; Pb 0.655 ± 0.474. Concentration found in brown seaweeds (mg/kg): Al 597.65 + 655.65; As 23.01 ± 15.67; Cd 0.245 ± 0.286; Cr 2.465 ± 4.277; Cu 2.33± 5.468; Hg 0.055 ± 0.0619; Ni 1.123 ± 1.219; Pb 0.539 ± 0.63. |
| Filippini et al., 2020 | Italy | The study explored heavy metal concentration in seaweeds, and health risk for adults and children associated with their intake. | Brown, green, mixed, and red seaweeds. | Al levels found: from 0.71 mg/kg to 165.39 mg/kg. Cd concentration: from 0.02 to 1.56 mg/kg). Pb concentration: from 0.16 to 0.56 mg/kg. Iodine content: from 10.66 to 6670.8 mg/kg. Hg level: <0.03 mg/kg. |
| Francisco et al., 2018 | Portugal | One of the objectives in this work was to assess the risk linked to the heavy metal in a brown alga species. | Fucus spiralis. | As concentration: 24.36 ± 2.04 μg/g DW. Cd content: from 0.07 (unpolluted site) to up to 3.58 μg/g DW (polluted site). I levels: 190 ± 18 μg/g DW. Hg and Pb values: low. |
| Kim et al., 2019 | USA | The study measured heavy metal content in a brown alga and in a red alga. | Gracilaria tikvahiae and Saccharina latissima. | Gracilaria tikvahiae: As, Cd, and Hg high levels in western Long Island Sound; Pb high in Bronx River Estuary. Saccharina latissima: Cd and Pb high levels in Bronx River Estuary. |
| Li et al., 2018 | China | The study evaluated the contaminants content in U. prolifera and the risks for human health associated with its consumption. | Samples of U. prolifera. | Heavy metal mean concentration of two years: As 0.66–0.93, Cd 0.0068–1.6, Cr1.6–9.7, Cu 1.7–10, Pb 0.064–2.3 mg/g DW. THQ: <10−1. Potential carcinogenic PAHs: from 0.19 to 414 ng/g DW. |
| Liu et al., 2017 | China | This study investigated Porphyra REEs content and health risk related to its consumption. | Thirty-six Porphyra seaweed samples from Jiangsu province and Fujian province. | REEs content: from 2.187 to 13.452 mg/kg. Ce, La, Nd, and Y values were high in every sample. |
| Miller et al., 2020 | USA | The study analyzed the presence of anabaenopeptins, cyanopeptolins, and microginins, as well as microcystins (MC) variants in algal dietary supplements. | A total of 18 algal dietary supplement products containing cyanobacterial species Aphanizomenon flos-aquae (AFA) in pill form, whether capsule or tablet. | The analysis showed that the MAC was exceeded by forty to sixty times. Almost a quarter of the samples analysed presented an excess of MC. |
| Mise et al., 2019 | Japan | The study examined the dietary exposure to As in Japanese pregnant women and children for Hijiki seaweed consumption. | 104 pregnant women and 106 children. | Total arsenic intake: 8.46 µg/kg BW/week in pregnant women; 20.07 µg/kg BW/week in children. iAs values: 1.74 µg/kg BW/week for pregnant women and 4.81 μg/kg BW/week for children. |
| Panebianco et al., 2019 | Italy | The study assessed the presence of microbiological, chemical, and physical hazards in seaweeds. | Two seaweeds among seafood commercial products considered in the study. | For lead, the presence was 14% max in one sample and 17.8% max in the other. For As, the values were 4.1% max in the first sample and 6.7% max in the second. |
| Paz et al., 2018 | Spain | The goal was to assess the content of Al, Cd, and Pb and B, Ba, Fe, Li, Ni, and V, in several European seaweeds. | Sixty-four Phaeophyta species. | The following concentrations were expressed in mg/kg DW. Al in Halopteris scoparia 161 ± 15.6, in Padina pavonica 256 ± 179, in Sargassum fluitans 57.7 ± 15.3, in Cystoseira spp. 145 ± 147, in Haliptilum virgatum 36.3 ± 4.02. Cd in H. scoparia 0.07 ± 0.01, in P. pavonica 0.20 ± 0.21, in S. fluitans 0.16 ± 0.06, in Cystoseira spp. 0.19 ± 0.09, H. virgatum 0.19 ± 0.02. Ni in H. scoparia 1.54 ± 0.4, in P. pavonica 3.7 ± 3.02, in S. fluitans 0.9 ± 0.34, in Cystoseira spp. 1.6 ± 0.64, in H. virgatum 1.34 ± 0.17. Pb in H. scoparia 3.1 ± 0.67, in P. pavonica 3.92 ± 3.71, in S. fluitans 0.4 ± 0.22, in Cystoseira spp. 1.1 ± 1.31, H. virgatum 0.31 ± 0.06. |
| Paz et al., 2018 | Spain | The goal was to define the content of Al, Cd, Hg, and Pb in some edible seaweeds. | Seventy-three European and Asian seaweeds: Undaria pinnatifida, Himanthalia elongate, Laminaria ochroleuca, seaweed salad. | Al concentration: from a minimum of 19.1 ± 8.6 (Europe) to a maximum of 57.7 ± 35 mg/kg (Asia). Cd concentration: from 0.04 ± 0.03 to 1.11 (Europe) ± 0.3 mg/kg (Asia). Hg levels: from <LOQ (Asia) to 0.024 ± 0.001 (Europe). Pb values: from 0.23 ± 0.07 (Europe) to 0.49 ± 0.2 (Asia). |
| Rzymski et al., 2019 | Poland | The study determined Al, As, Cd, Cr, Hg, Ni, Pb, and REEs content in Spirulina and Chlorella food supplements. | Spirulina and Chlorella supplements purchased online. | Al content 2155.6 ± 1774.7 (1299.8) mg/kg in Spirulina; 1732.8 ± 1991.5 mg/kg in Chlorella. Cd concentration: 0.125 ± 0.055 mg/kg in Spirulina; 0.142 ± 0.071 mg/kg in Chlorella. Cr (VI) content: below the detection limit (0.01 mg/kg) in both species. Hg values: 0.027 ± 0.031 mg/kg in Spirulina; 0.41 ± 0.017 mg/kg in Chlorella. iAs levels: 1.7–2.2 mg/kg in Spirulina; 2.3–2.7 mg/kg in Chlorella. Ni concentration: 1.52 ± 0.72 mg/kg in Spirulina; 1.38 ± 0.63 mg/kg in Chlorella. REEs amount: 2.14 ± 1.89 mg/kg in Spirulina; 2.03 ± 11.28 mg/kg in Chlorella. Pb content: 2.6 ± 1.9 mg/kg in Spirulina; 2.6 ± 1.3 mg/kg in Chlorella. LREEs and Cu levels: high in both species. |
| Sa Monteiro et al., 2019 | Denmark | The main goal was to determine the levels of iAs, Cd, Hg, I, and Pb in edible seaweeds. | Species selected for this study: Fucus vesiculosus, Fucus serratus, Fucus spiralis, Fucus evanescens, Saccharina latissima, Ulva lactuca and Cladophora sp. | As content: from 3.2 to 116.7 μg/g fdw. Cd values: 0.017–1.97 μg/g fdw. Hg concentration: from 0.003 to 0.042 μg/g fdw. I content: from 17.2 to 4782 μg/g fdw. iAs content: not available. Pb levels: from 0.072 to 9.6 μg/g fdw. |
| Santos-Silva et al., 2018 | Brazil | This study defined, in Phaeophyta and Rhodophyta, the background levels of As, Cd, Cu, Hg, and Pb. | Species selected for this study: Dictyopteris delicatula, Canistrocarpus cervicornis, Ceratodictyon variabile and Palisada perforata. | As content: from <4.84 to 23.21 μg/g. The maximum As values, from 13.46 μg/g to 49.52 μg/g, were detected in brown seaweeds. Cu concentration: from below the detection limit to 33.55 μg/g. Cd, Hg, and Pb: below the detection limits. Hg concentration: 22.25 μg/g, maximum value. The highest mean value of Cd was found in Dictyopteris delicatula (0.18 μg/g). |
| Stévant et al., 2018 | Norway | The aim of this work was to assess toxicity due to some heavy metals in cultivated A. esculenta and S. latissima. | Samples of A. esculenta and S. latissima from the Northern coast of France. | Initial content of the Cd, I and iAs, expressed in mg/kg DW. A. esculenta Cd concentration 2.01 ± 0.09 and 1.55 ± 0.2. S. latissima Cd concentration 0.22 ± 0.03 and 0.27 ± 0.01. Limit value: 0.5. A. esculenta I concentration 213 ± 12; S. latissima I concentration 4898 ± 166 and 6568 ± 398. Limit value: 2000. S. latissima iAs content: 0.22 ± 0.04; S. latissima iAs content: 0.16 ± 0.02 and 0.23 ± 0.01. Limit value: 3. |
| Squadrone et al., 2018 | Italy | The study determined the levels of non-essential trace elements, essential trace elements, and risk for humans in several Mediterranean seaweeds. | Brown, green, and red algae species. | Main results: Brown algae: Al 9916 mg/kg, Pb 40 mg/kg. Green algae: As 37 mg/kg, Cd 0.32 mg/kg, Co 5.6 mg/kg, Cu 73 mg/kg. Brown algae contained the largest amount of total metals (20.172 mg/kg DW) the red algae the lower (8292 mg/kg DW). |
| Squadrone et al., 2019 | Italy | The study analyzed the REEs, among others, in seaweeds. | Brown, green, and red algae. | ∑REE concentration in seaweed: 12 mg/kg. |
| Uchida et al., 2016 | Japan | The present study attempted to measure the heavy metal contents of the NSs for food safety. | Dried sheets of Japanese nori alga. | Cd: 0.05 mg/100 g or lower. Total As: 0.8 mg/100 g iAs: <0.05 mg/100 g Cr: 0.01 mg/100 g or lower. Histamine: from 0.9 to 9.0 mg/100 mL. |
| Uddin et al., 2019 | Kuwait | The study registered 210Po and 210Pb and radionuclides concentrations in different algae species. | Cladophora Sargassum, and Ulva. | 210Po/210Pb ratio: from2.67 to 10.95. |
| Zhang et al., 2020 | China | The study delivered information about health risk associated with selenium-rice agro-food consumption. | Several thousands of selenium-rich agro-food purchased at the market. | Concentration of heavy metals in edible fungi and algae (μg/g wet weight): Pb content 0.3253/0.4580; As content 0.1947/0.3681; Cd content 0.2635/1.6763; Hg content 0.0086/0.1960. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cavallo, G.; Lorini, C.; Garamella, G.; Bonaccorsi, G. Seaweeds as a “Palatable” Challenge between Innovation and Sustainability: A Systematic Review of Food Safety. Sustainability 2021, 13, 7652. https://doi.org/10.3390/su13147652
Cavallo G, Lorini C, Garamella G, Bonaccorsi G. Seaweeds as a “Palatable” Challenge between Innovation and Sustainability: A Systematic Review of Food Safety. Sustainability. 2021; 13(14):7652. https://doi.org/10.3390/su13147652
Chicago/Turabian StyleCavallo, Giuseppe, Chiara Lorini, Giuseppe Garamella, and Guglielmo Bonaccorsi. 2021. "Seaweeds as a “Palatable” Challenge between Innovation and Sustainability: A Systematic Review of Food Safety" Sustainability 13, no. 14: 7652. https://doi.org/10.3390/su13147652
APA StyleCavallo, G., Lorini, C., Garamella, G., & Bonaccorsi, G. (2021). Seaweeds as a “Palatable” Challenge between Innovation and Sustainability: A Systematic Review of Food Safety. Sustainability, 13(14), 7652. https://doi.org/10.3390/su13147652









