Conservation Studies on Groundwaters’ Pollution: Challenges and Perspectives for Stygofauna Communities
Abstract
:1. Introduction
2. Pollutants Affecting Groundwater Animals
3. Relevance of Effects of Groundwater Pollution for Stygofauna in Recent Literature
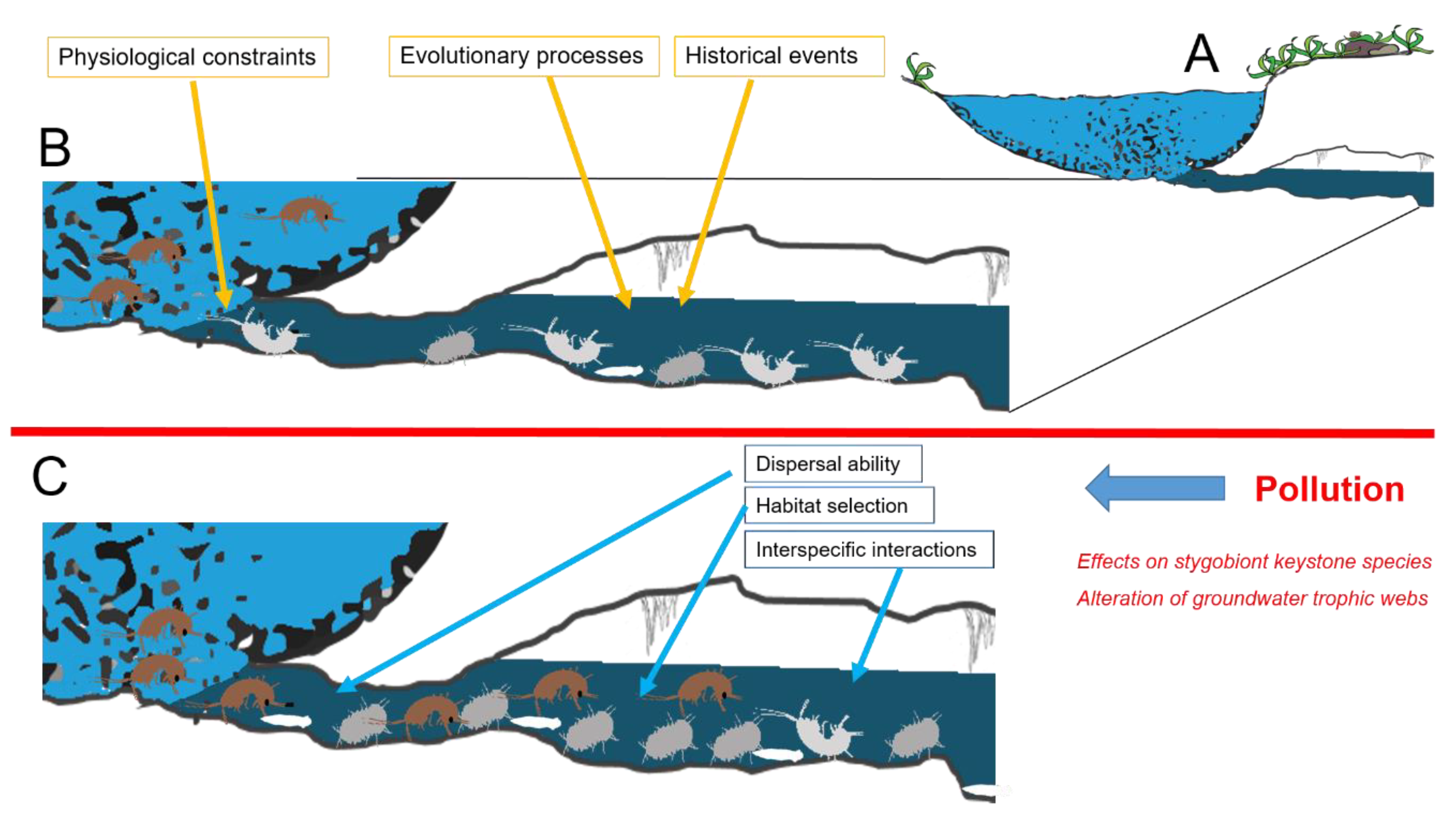
4. Effects and Research Perspectives at Community Level
4.1. Effects on Stygobiont Keystone Species
4.2. Alteration of Groundwater Trophic Webs
4.3. Effects on Interspecific Competition between Stygobionts and Epigean Animals
4.4. Effects on Ecological Niche of Epigean Mesorpedator Species
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Abell, R. Conservation biology for the biodiversity crisis: A freshwater follow-up. Conserv. Biol. 2002, 16, 1435–1437. [Google Scholar] [CrossRef] [Green Version]
- Yao, J.M.; Sanchez-Perez, J.M.; Sauvage, S.; Teissier, S.; Attard, E.; Lauga, B.; Duran, R.; Julien, F.; Bernard-Jannin, L.; Ramburn, H.; et al. Biodiversity and ecosystem purification service in an alluvial wetland. Ecol. Eng. 2017, 103, 359–371. [Google Scholar] [CrossRef]
- Richter, B.D.; Mathews, R.; Wigington, R. Ecologically sustainable water management: Managing river flows for ecological integrity. Ecol. Appl. 2003, 13, 206–224. [Google Scholar] [CrossRef]
- Clarke, A.; Mac Nally, R.; Bond, N.R.; Lake, P.S. Conserving macroinvertebrate diversity in headwater streams: The importance of knowing the relative contributions of alpha and beta diversity. Divers. Distrib. 2010, 16, 725–736. [Google Scholar] [CrossRef]
- Baldissera, M.D.; Souza, C.F.; Zanella, R.; Prestes, O.D.; Meinhart, A.D.; Da Silva, A.S.; Baldisserotto, B. Behavioral impairment and neurotoxic responses of silver catfish Rhamdia quelen exposed to organophosphate pesticide trichlorfon: Protective effects of diet containing rutin. Comp. Biochem. Phys. C 2021, 239, 108871. [Google Scholar] [CrossRef]
- Fan, B.; Li, J.; Wang, X.N.; Chen, J.; Gao, X.Y.; Li, W.W.; Ai, S.H.; Cui, L.; Gao, S.X.; Liu, Z.T. Ammonia spatiotemporal distribution and risk assessment for freshwater species in aquatic ecosystem in China. Ecotoxicol. Environ. Saf. 2021, 207, 111541. [Google Scholar] [CrossRef]
- Fulton, C.A.; Hartz, K.E.H.; Fuller, N.W.; Kent, L.N.; Anzalone, S.E.; Miller, T.M.; Connon, R.E.; Poynton, H.P.; Lydy, M.J. Fitness costs of pesticide resistance in Hyalella azteca under future climate change scenarios. Sci. Total Environ. 2021, 753, 141945. [Google Scholar] [CrossRef]
- Mao, Z.G.; Gu, X.H.; Cao, Y.; Luo, J.H.; Zeng, Q.F.; Chen, H.H.; Jeppesen, E. How does fish functional diversity respond to environmental changes in two large shallow lakes? Sci. Total Environ. 2021, 753, 142158. [Google Scholar] [CrossRef] [PubMed]
- O’Malley, Z.G.; Compson, Z.G.; Orlofske, J.M.; Baird, D.J.; Curry, R.A.; Monk, W.A. Riparian and in-channel habitat properties linked to dragonfly emergence. Sci. Rep. 2020, 10, 1–12. [Google Scholar] [CrossRef]
- Bush, A.; Nipperess, D.; Turak, E.; Hughes, L. Determining vulnerability of stream communities to climate change at the landscape scale. Freshw. Biol. 2012, 57, 1689–1701. [Google Scholar] [CrossRef]
- Howarth, F.G.; Moldovan, O.T. The ecological classification of cave animals and their adaptations. In Cave Ecology; Moldovan, O.T., Kováč, L., Halse, S., Eds.; Springer: Berlin, Germany, 2018; pp. 41–67. [Google Scholar]
- Sket, B. Distribution of Proteus (Amphibia: Urodela: Proteidae) and its possible explanation. J. Biogeogr. 1997, 24, 263–280. [Google Scholar] [CrossRef]
- Kurwadkar, S.; Kanel, S.R.; Nakarmi, A. Groundwater pollution: Occurrence, detection, and remediation of organic and inorganic pollutants. Water Environ. Res. 2020, 92, 1659–1668. [Google Scholar] [CrossRef]
- Gibert, J.; Deharveng, L. Subterranean ecosystems: A truncated functional biodiversity. Bioscience 2002, 52, 473–481. [Google Scholar] [CrossRef]
- Manenti, R.; Pennati, R.; Ficetola, G.F. Role of density and resource competition in determining aggressive behaviour in salamanders. J. Zool. 2015, 296, 270–277. [Google Scholar] [CrossRef]
- Manenti, R.; Siesa, M.E.; Ficetola, G.F. Odonata occurence in caves: Active or accidentals? A new case study. J. Cave Karst Stud. 2013, 75, 205–209. [Google Scholar] [CrossRef]
- Manenti, R. Role of cave features for aquatic troglobiont fauna occurrence: Effects on “accidentals” and troglomorphic organisms distribution. Acta Zool. Acad. Sci. Hung. 2014, 60, 257–270. [Google Scholar]
- Manenti, R.; Pezzoli, E. Think of what lies below, not only of what is visible above, or: A comprehensive zoological study of invertebrate communities of spring habitats. Eur. Zool. J. 2019, 86, 272–279. [Google Scholar] [CrossRef] [Green Version]
- Manenti, R.; Barzaghi, B. Diel activity of Niphargus amphipods in spring habitats. Crustaceana. in press.
- Manenti, R.; Piazza, B. Between darkness and light: Spring habitats provide new perspectives for modern researchers on groundwater biology. PeerJ. in press.
- Noges, P.; Argillier, C.; Borja, A.; Garmendia, J.M.; Hanganu, J.; Kodes, V.; Pletterbauer, F.; Sagouis, A.; Birk, S. Quantified biotic and abiotic responses to multiple stress in freshwater, marine and ground waters. Sci. Total Environ. 2016, 540, 43–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Culver, D.C.; Pipan, T. The Biology of Caves and Other Subterranean Habitats; Oxford University Press: New York, NY, USA, 2009. [Google Scholar]
- Culver, D.C.; Pipan, T. Shallow Subterranean Habitats: Ecology, Evolution, and Conservation; Oxford University Press: New York, NY, USA, 2014. [Google Scholar]
- Buvaneshwari, S.; Riotte, J.; Sekhar, M.; Sharma, A.K.; Helliwell, R.; Kumar, M.S.M.; Braun, J.J.; Ruiz, L. Potash fertilizer promotes incipient salinization in groundwater irrigated semi-arid agriculture. Sci. Rep. 2020, 10, 3691. [Google Scholar] [CrossRef] [PubMed]
- Fisher, I.J.; Phillips, P.J.; Bayraktar, B.N.; Chen, S.; McCarthy, B.A.; Sandstrom, M.W. Pesticides and their degradates in groundwater reflect past use and current management strategies, Long Island, New York, USA. Sci. Total Environ. 2021, 752, 141895. [Google Scholar] [CrossRef]
- Toccalino, P.L.; Gilliom, R.J.; Lindsey, B.D.; Rupert, M.G. Pesticides in groundwater of the United States: Decadal-scale changes, 1993–2011. GroundWater 2014, 52, 112–125. [Google Scholar] [CrossRef]
- Buckerfield, S.J.; Quilliam, R.S.; Bussiere, L.; Waldron, S.; Naylor, L.A.; Li, S.; Oliver, D.M. Chronic urban hotspots and agricultural drainage drive microbial pollution of karst water resources in rural developing regions. Sci. Total Environ. 2020, 744, 140898. [Google Scholar] [CrossRef] [PubMed]
- Magal, E.; Arbel, Y.; Caspi, S.; Glazman, H.; Greenbaum, N.; Yechieli, Y. Determination of pollution and recovery time of karst springs, an example from a carbonate aquifer in Israel. J. Contam. Hydrol. 2013, 145, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Graening, G.O.; Brown, A.V. Ecosystem dynamics and pollution effects in an Ozark cave stream. J. Am. Water Resour. Assoc. 2003, 39, 1497–1507. [Google Scholar] [CrossRef]
- Nikolaidis, C.; Zafiriadis, I.; Mathioudakis, V.; Constantinidis, T. Heavy metal pollution associated with an abandoned lead-zinc mine in the Kirki region, NE Greece. Bull. Environ. Contam. Toxicol. 2010, 85, 307–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoo, E.J.; Lee, J.A.; Park, J.S.; Lee, K.; Lee, W.S.; Han, J.S.; Choi, J.W. Tracing lead pollution sources in abandoned mine areas using stable Pb isotope ratios. Environ. Monit. Assess. 2014, 186, 781–789. [Google Scholar] [CrossRef]
- Castano-Sanchez, A.; Hose, G.C.; Reboleira, A. Ecotoxicological effects of anthropogenic stressors in subterranean organisms: A review. Chemosphere 2020, 244, 125422. [Google Scholar] [CrossRef]
- Hepburn, E.; Madden, C.; Szabo, D.; Coggan, T.L.; Clarke, B.; Currell, M. Contamination of groundwater with per- and polyfluoroalkyl substances (PFAS) from legacy landfills in an urban re-development precinct. Environ. Pollut. 2019, 248, 101–113. [Google Scholar] [CrossRef]
- Hoisaeter, A.; Pfaff, A.; Breedveld, G.D. Leaching and transport of PFAS from aqueous film-forming foam (AFFF) in the unsaturated soil at a firefighting training facility under cold climatic conditions. J. Contam. Hydrol. 2019, 222, 112–122. [Google Scholar] [CrossRef]
- Mammola, S.; Amorim, I.R.; Bichuette, M.E.; Borges, P.A.V.; Cheeptham, N.; Cooper, S.J.B.; Culver, D.C.; Deharveng, L.; Eme, D.; Ferreira, R.L.; et al. Fundamental research questions in subterranean biology. Biol. Rev. 2020, 95, 1855–1872. [Google Scholar] [CrossRef] [PubMed]
- Gawdzik, B.; Gawdzik, J. Impact of Pollution with Oil Derivatives on the Natural Environment and Methods of Their Removal. Ecol. Chem. Eng. S 2011, 18, 345–357. [Google Scholar]
- Mali, L.B.; Sepcic, K.; Bulog, B. Long-term starvation in cave salamander effects on liver ultrastructure and energy reserve mobilization. J. Morphol. 2013, 274, 887–900. [Google Scholar] [CrossRef]
- Mercurio, S.; Messinetti, S.; Manenti, R.; Ficetola, G.F.; Pennati, R. Embryotoxicity characterization of the flame retardant tris(1-chloro-2-propyl)phosphate (TCPP) in the invertebrate chordate Ciona intestinalis. J. Exp. Zool. Part A Ecol. Integr. Physiol. 2021, 335, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Canivet, V.; Gibert, J. Sensitivity of epigean and hypogean freshwater macroinvertebrates to complex mixtures. Part 1: Laboratory experiments. Chemosphere 2002, 46, 999–1009. [Google Scholar] [CrossRef]
- Page, M.J.; Moher, D. Evaluations of the uptake and impact of the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) Statement and extensions: A scoping review. Syst. Rev. 2017, 6, 1–14. [Google Scholar] [CrossRef]
- Bolker, B.M.; Brooks, M.E.; Clark, C.J.; Geange, S.W.; Poulsen, J.R.; Stevens, M.H.H.; White, J.S. Generalized linear mixed models: A practical guide for ecology and evolution. Trends Ecol. Evol. 2008, 24, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Asmyhr, M.G.; Hose, G.; Graham, P.; Stow, A.J. Fine-scale genetics of subterranean syncarids. Freshw. Biol. 2014, 59, 1–11. [Google Scholar] [CrossRef]
- Di Lorenzo, T.; Galassi, D.M.P. Agricultural impact on Mediterranean alluvial aquifers: Do groundwater communities respond? Fund. Appl. Limnol. 2013, 182, 271–282. [Google Scholar] [CrossRef]
- Reboleira, A.; Abrantes, N.; Oromi, P.; Goncalves, F. Acute Toxicity of Copper Sulfate and Potassium Dichromate on Stygobiont Proasellus: General Aspects of Groundwater Ecotoxicology and Future Perspectives. Water Air Soil Pollut. 2013, 224, 1–9. [Google Scholar] [CrossRef]
- Sket, B. High biodiversity in hypogean waters and its endangerment—The situation in Slovenia, the Dinaric Karst, and Europe. Crustaceana 1999, 72, 767–779. [Google Scholar] [CrossRef]
- Sket, B. The nature of biodiversity in hypogean waters and how it is endangered. Biodivers. Conserv. 1999, 8, 1319–1338. [Google Scholar] [CrossRef]
- Romero, A. Hypogean Communities as Cybernetic Systems. Diversity 2020, 12, 413. [Google Scholar] [CrossRef]
- Dole-Olivier, M.J.; Malard, F.; Ferreira, D.; Gibert, J. Groundwater biodiversity. Houille Blanche 2005, 39–44. [Google Scholar] [CrossRef]
- Mancinelli, G.; Costantini, M.L.; Rossi, L. Top-down control of reed detritus processing in a lake littoral zone: Experimental evidence of a seasonal compensation between fish and invertebrate predation. Int. Rev. Hydrobiol. 2007, 92, 117–134. [Google Scholar] [CrossRef]
- Bulog, B.; Mihajl, K.; Jeran, Z.; Toman, M.J. Trace element concentrations in the tissues of Proteus anguinus (Amphibia, Caudata) and the surrounding environment. Water Air Soil Pollut. 2002, 136, 147–163. [Google Scholar] [CrossRef]
- Bulog, B. Analyses of some microelements in the tissues of Proteus anguinus (Amphibia, Caudata) and in its habitat. Acta Carsol. 1996, 25, 309–321. [Google Scholar]
- Voituron, Y.; de Fraipont, M.; Issartel, J.; Guillaume, O.; Clobert, J. Extreme lifespan of the human fish (Proteus anguinus): A challenge for ageing mechanisms. Biol. Lett. 2011, 7, 105–107. [Google Scholar] [CrossRef] [Green Version]
- Gourbault, N. Recherches sur les Triclades Paludicoles hypogés. Mémoires Muséum Natl. d’Histoire Nat. Ser. A 1972, 73, 1–249. [Google Scholar]
- Barzaghi, B.; De Giorgi, D.; Pennati, R.; Manenti, R. Planarians, a neglected component of biodiversity in groundwaters. Diversity 2021, 13, 178. [Google Scholar] [CrossRef]
- Manenti, R.; Lunghi, E.; Barzaghi, B.; Melotto, A.; Falaschi, M.; Ficetola, G.F. Do salamanders limit the abundance of groundwater invertebrates in subterranean habitats? Diversity 2020, 12, 161. [Google Scholar] [CrossRef] [Green Version]
- Manenti, R.; Barzaghi, B.; Lana, E.; Stocchino, G.A.; Manconi, R.; Lunghi, E. The stenoendemic cave-dwelling planarians (Platyhelminthes, Tricladida) of the Italian Alps and Apennines: Conservation issues. J. Nat. Conserv. 2018, 45, 90–97. [Google Scholar] [CrossRef]
- De Beauchamp, P. Biospeleologica. Turbellariés, Hirudinées, Branchiobdellidés (Deuxième série). In Archives de Zoologie Expérimentale et Générale: Histoire Naturelle, Morphologie, Histologie, Evolution des Animaux; Centre National de la Recherche Scientifique: Paris, France, 1932; Volume 73, pp. 113–380. [Google Scholar]
- Bosnir, J.; Puntaric, D.; Skes, I.; Klaric, M.; Simic, S.; Zoric, I.; Galic, R. Toxic metals in freshwater fish from the Zagreb area as indicators of environmental pollution. Coll. Antropol. 2003, 27, 31–39. [Google Scholar] [PubMed]
- Nikanorov, A.M.; Stradomskaya, A.G. Chronic pollution of freshwater bodies: Data on accumulation of pesticides, oil products, and other toxic substances in bottom deposits. Water Resour. 2007, 34, 314–320. [Google Scholar] [CrossRef]
- Culver, D.C.; Pipan, T. Redefining the extent of the aquatic subterranean biotope-shallow subterranean habitats. Ecohydrology 2011, 4, 721–730. [Google Scholar] [CrossRef]
- Francois, C.M.; Mermillod-Blondin, F.; Malard, F.; Fourel, F.; Lecuyer, C.; Douady, C.J.; Simon, L. Trophic ecology of groundwater species reveals specialization in a low-productivity environment. Funct. Ecol. 2016, 30, 262–273. [Google Scholar] [CrossRef]
- Williamson, W.M.; Close, M.E.; Leonard, M.M.; Webber, J.B.; Lin, S. Groundwater biofilm dynamics grown in situ along a nutrient gradient. GroundWater 2012, 50, 690–703. [Google Scholar] [CrossRef]
- Wood, P.J.; Gunn, J.; Rundle, S.D. Response of benthic cave invertebrates to organic pollution events. Aquat. Conserv. 2008, 18, 909–922. [Google Scholar] [CrossRef] [Green Version]
- Ercoli, F.; Lefebvre, F.; Delangle, M.; Gode, N.; Caillon, M.; Raimond, R.; Souty-Grosset, C. Differing trophic niches of three French stygobionts and their implications for conservation of endemic stygofauna. Aquat. Conserv. 2019, 29, 2193–2203. [Google Scholar] [CrossRef]
- Holsinger, J.R. A preliminary study of the effects of organic pollution of Banners Corner Cave, Virginia. Int. J. Speleol. 1966, 2, 75–89. [Google Scholar] [CrossRef] [Green Version]
- Simon, K.S.; Buikema, A.L. Effects of organic pollution on an Appalachian cave: Changes in macroinvertebrate populations and food supplies. Am. Midl. Nat. 1997, 138, 387–401. [Google Scholar] [CrossRef]
- Raffaëlli, J. Les Urodeles du Monde; Penclen Edition: Condé-sur-Noireau, France, 2007. [Google Scholar]
- Romero, A. Cave Biology; Cambridge University Press: New York, NY, USA, 2009. [Google Scholar]
- Hervant, F.; Mathieu, J.; Durand, J.P. Metabolism and circadian rhythms of the European blind cave salamander Proteus anguinus and a facultative cave dweller, the Pyrenean newt (Euproctus asper). Can. J. Zool. 2000, 78, 1427–1432. [Google Scholar] [CrossRef]
- Wilhelm, F.M.; Taylor, S.J.; Adams, G.L. Comparison of routine metabolic rates of the stygobite, Gammarus acherondytes (Amphipoda: Gammaridae) and the stygophile, Gammarus troglophilus. Freshw. Biol. 2006, 51, 1162–1174. [Google Scholar] [CrossRef]
- Wood, P.J.; Gunn, J.; Perkins, J. The impact of pollution on aquatic invertebrates within a subterranean ecosystem—Out of sight out of mind. Arch. Hydrobiol. 2002, 155, 223–237. [Google Scholar] [CrossRef]
- Ritchie, E.G.; Johnson, C.N. Predator interactions, mesopredator release and biodiversity conservation. Ecol. Lett. 2009, 12, 982–998. [Google Scholar] [CrossRef] [PubMed]
- Ferreras-Romero, M.; Corbet, P.S. The life cycle of Cordulegaster boltonii (Donovan, 1807) (Odonata: Cordulegastridae) in the Sierra Morena Mountains (southern Spain). Hydrobiologia 1999, 405, 39–48. [Google Scholar] [CrossRef]
- Jabiol, J.; Cornut, J.; Danger, M.; Jouffroy, M.; Elger, A.; Chauvet, E. Litter identity mediates predator impacts on the functioning of an aquatic detritus-based food web. Oecologia 2014, 176, 225–235. [Google Scholar] [CrossRef] [Green Version]
- Worthen, W.B.; Horacek, H.J. The distribution of dragonfly larvae in a South Carolina stream: Relationships with sediment type, body size, and the presence of other larvae. J. Insect Sci. 2015, 15, 31. [Google Scholar] [CrossRef] [Green Version]
- Ficetola, G.F.; Barzaghi, B.; Melotto, A.; Muraro, M.; Lunghi, E.; Canedoli, C.; Lo Parrino, E.; Nanni, V.; Silva-Rocha, I.; Urso, A.; et al. N-mixture models reliably estimate the abundance of small vertebrates. Sci. Rep. 2018, 8, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Lunghi, E.; Cianferoni, F.; Ceccolini, F.; Veith, M.; Manenti, R.; Mancinelli, G.; Corti, C.; Ficetola, G.F. What shapes the trophic niche of European plethodontid salamanders? PLoS ONE 2018, 13, e0205672. [Google Scholar] [CrossRef]
- Bradley, J.G.; Eason, P.K. Predation risk and microhabitat selection by cave salamanders, Eurycea lucifuga (Rafinesque, 1822). Behaviour 2018, 155, 841–859. [Google Scholar] [CrossRef]
- Manenti, R.; Melotto, A.; Guillaume, O.; Ficetola, G.F.; Lunghi, E. Switching from mesopredator to apex predator: How do responses vary in amphibians adapted to cave living? Behav. Ecol. Sociobiol. 2020, 74, 1–13. [Google Scholar] [CrossRef]
- Lunghi, E.; Mascia, C.; Mulargia, M.; Corti, C. Is the Sardinian grass snake (Natrix natrix cetti) an active hunter in underground environments. Spixiana 2018, 41, 160. [Google Scholar]
- Elliot, J.M.; Dobson, M. Freshwater Leeches of Britain and Ireland; Freshwater Biological Association: Sudbury, ON, Canada, 2015. [Google Scholar]
- Franciscolo, M. Fauna cavernicola del Savonese (Res Ligusticae XCIV). Ann. Mus. Civ. St. Nat. Genova 1955, 67, 1–223. [Google Scholar]
- Sanfilippo, N. Le grotte della provincia di Genova e la loro Fauna. Mem. Com. Sci. Cent. CAI 1950, 2, 1–93. [Google Scholar]
- Manenti, R.; Denoel, M.; Ficetola, G.F. Foraging plasticity favours adaptation to new habitats in fire salamanders. Anim. Behav. 2013, 86, 375–382. [Google Scholar] [CrossRef]
- Manenti, R.; Ficetola, G.F.; Bianchi, B.; De Bernardi, F. Habitat features and distribution of Salamandra salamandra in underground springs. Acta Herpetol. 2009, 4, 143–151. [Google Scholar]
- Lunghi, E.; Manenti, R. Cave Communities: From the Surface Border to the Deep Darkness. Diversity 2020, 12, 167. [Google Scholar] [CrossRef]
| Variable | Estimate | χ2 | P |
|---|---|---|---|
| Ecological papers | −0.31 | 0.31 | 0.52 ta |
| Taxonomical papers | −18.05 | 17.5 | <0.01 |
| Fauna assessments | −2.32 | 14.2 | <0.01 |
| Conservation papers | −0.69 | 0.44 | 0.50 |
| Behavior | −17.8 | 0.95 | 0.32 |
| Surface species underground | 1.42 | 3.61 | 0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manenti, R.; Piazza, B.; Zhao, Y.; Padoa Schioppa, E.; Lunghi, E. Conservation Studies on Groundwaters’ Pollution: Challenges and Perspectives for Stygofauna Communities. Sustainability 2021, 13, 7030. https://doi.org/10.3390/su13137030
Manenti R, Piazza B, Zhao Y, Padoa Schioppa E, Lunghi E. Conservation Studies on Groundwaters’ Pollution: Challenges and Perspectives for Stygofauna Communities. Sustainability. 2021; 13(13):7030. https://doi.org/10.3390/su13137030
Chicago/Turabian StyleManenti, Raoul, Beatrice Piazza, Yahui Zhao, Emilio Padoa Schioppa, and Enrico Lunghi. 2021. "Conservation Studies on Groundwaters’ Pollution: Challenges and Perspectives for Stygofauna Communities" Sustainability 13, no. 13: 7030. https://doi.org/10.3390/su13137030
APA StyleManenti, R., Piazza, B., Zhao, Y., Padoa Schioppa, E., & Lunghi, E. (2021). Conservation Studies on Groundwaters’ Pollution: Challenges and Perspectives for Stygofauna Communities. Sustainability, 13(13), 7030. https://doi.org/10.3390/su13137030










