Wood Ashes from Grate-Fired Heat and Power Plants: Evaluation of Nutrient and Heavy Metal Contents
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
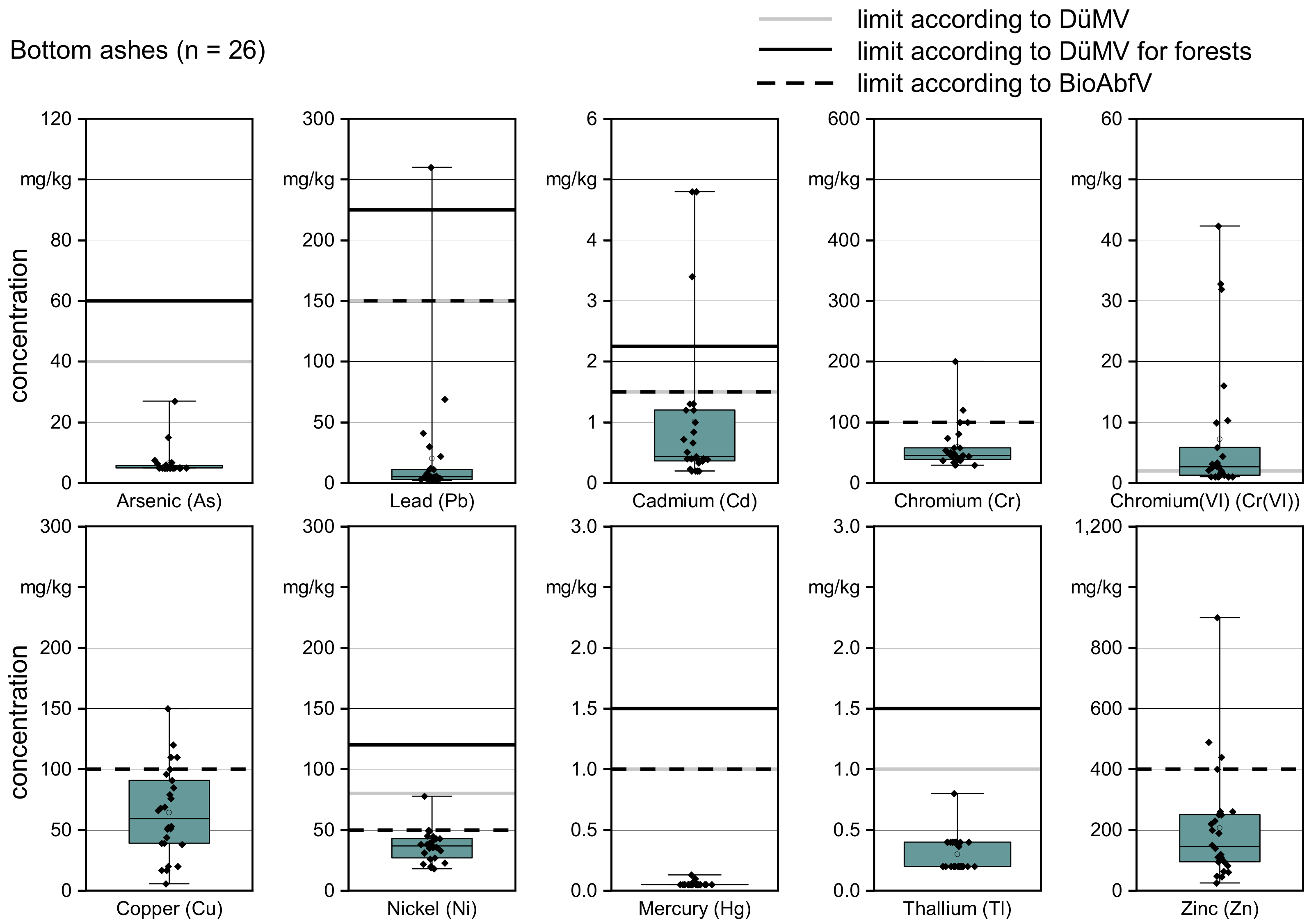
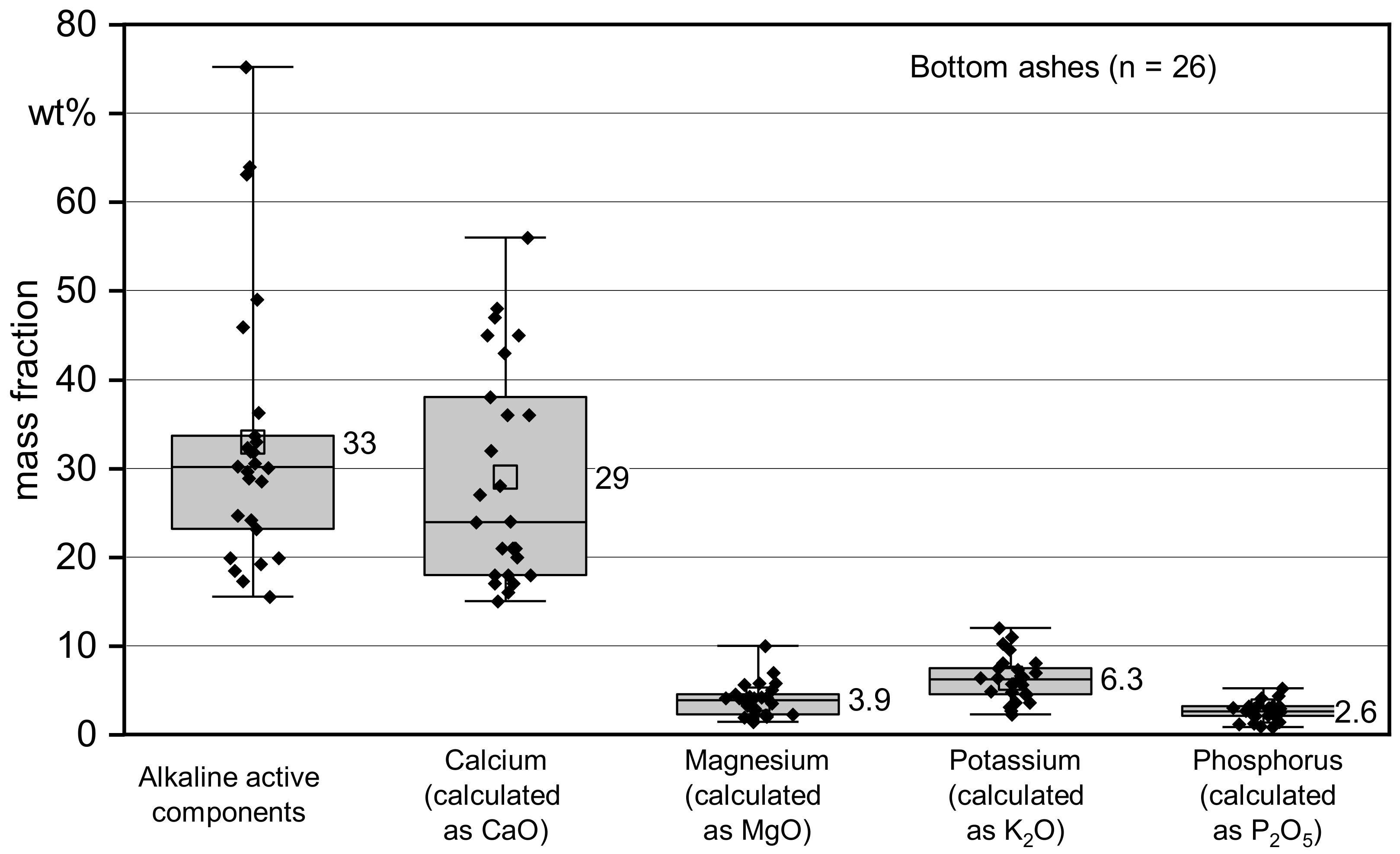
3.1. Quality of Bottom Ash
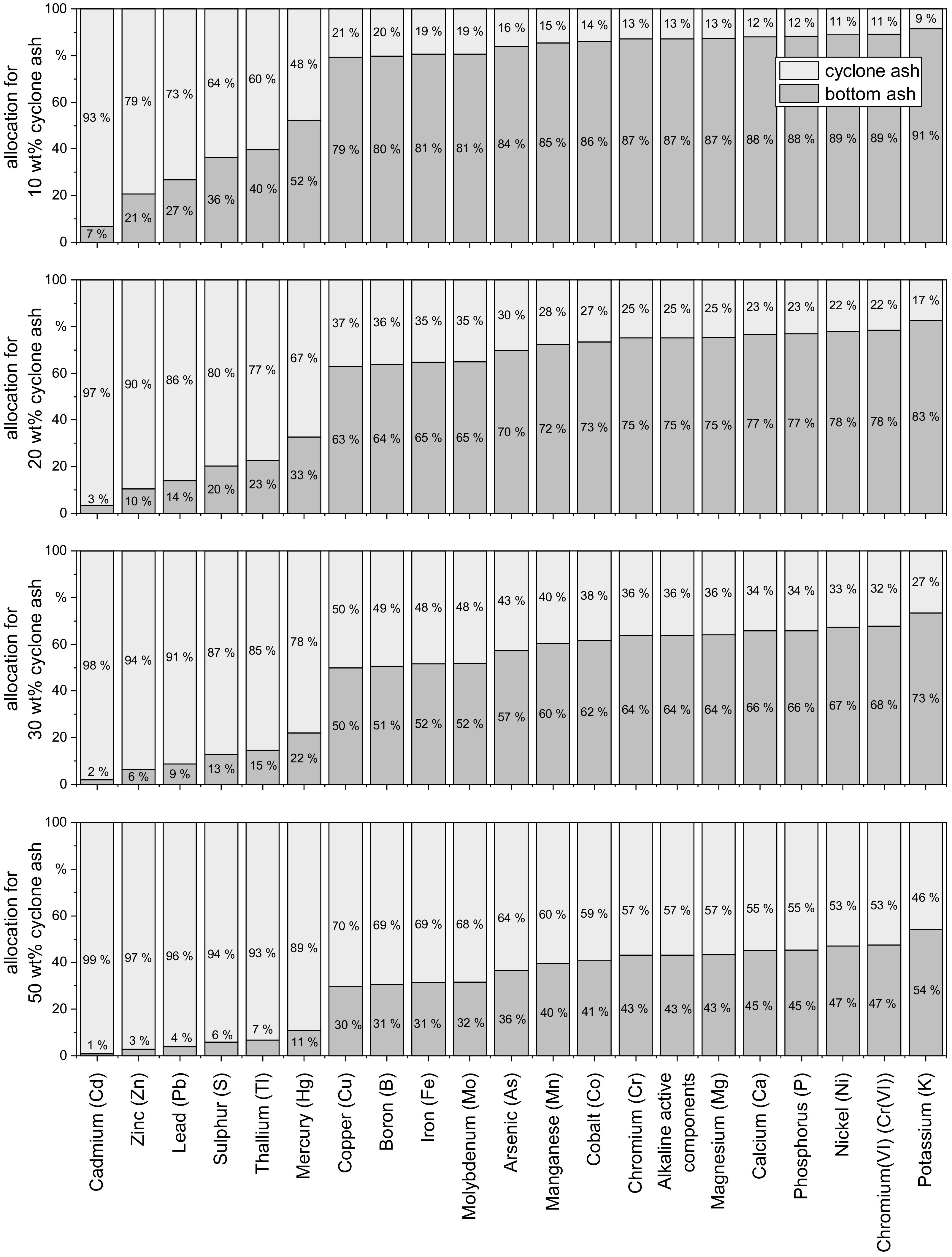
3.2. Distribution of Element Loads between Bottom Ash to Cyclone Ash (TFZ Heating Plant)
3.3. Quality of Mixtures of Bottom Ash with Cyclone Ash
4. Conclusions
- -
- As prescribed by the German Fertilizer Ordinance, only untreated wood should be used in biomass heat (and power) plants, since waste wood can contain elevated concentrations of heavy metals.
- -
- Fluctuations in fuel quality or the combustion conditions can change the heavy metal contents in bottom ash. Regular sampling and chemical analysis of the ash is therefore necessary.
- -
- Since some of the heavy metals are volatile under the usual combustion conditions in biomass heating (power) plants, care should be taken to ensure that the combustion temperatures in the boiler are constantly high enough. An average temperature of over 750 °C, for example, reliably leads to sufficiently low cadmium contents in the bottom ash.
- -
- As has been shown, mixing of bottom ash and cyclone ash leads to an increase in heavy metals. Separate collection of these ash fractions is essential.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Kaltschmitt, M.; Hartmann, H.; Hofbauer, H. Energie aus Biomasse. Grundlagen, Techniken und Verfahren; Springer: Berlin/Heidelberg, Germany, 2016; p. 1755. [Google Scholar]
- Vassilev, S.V.; Baxter, D.; Vassileva, C.G. An overview of the behaviour of biomass during combustion: Part II. Ash fusion and ash formation mechanisms of biomass types. Fuel 2014, 117, 152–183. [Google Scholar] [CrossRef]
- Gößwein, S.; Hiendlmeier, S. Energieholzmarkt Bayern 2016. Untersuchung des Energieholzmarktes in Bayern hinsichtlich Aufkommen und Verbrauch; LWF: Freising, Germany, 2018; p. 131. [Google Scholar]
- Dietz, E.; Kuptz, D.; Blum, U.; Schulmeyer, F.; Borchert, H.; Hartmann, H. Qualität von Holzhackschnitzeln in Bayern. Gehalte ausgewählter Elemente, Heizwert und Aschegehalt, Berichte aus dem TFZ, Straubing, Nr. 46; Technologie- und Förderzentrum im Kompetenzzentrum für Nachwachsende Rohstoffe (TFZ); Bayerische Landesanstalt für Wald und Forstwirtschaft (LWF): Freising-Weihenstephan, Germany, 2016; p. 141. [Google Scholar]
- Kuchler, C.; Zimmermann, D.; Kuptz, D.; Dietz, E.; Rist, E.; Riebler, M.; Schön, C.; Mack, R.; Blum, U.; Borchert, H.; et al. Contamination of wood chips with mineral soils–Fuel quality and combustion behaviour. In Proceedings of the 52nd International Symposium on Forestry Mechanization (FORMEC), Sopron, Hungary/Forchtenstein, Austria, 6–9 October 2019; University of Sopron Press: Sopron, Hungary, 2019; pp. 320–329. [Google Scholar]
- FRED Feste Regenerative Energieträger Datenbank. Straubing: Technologie- und Förderzentrum im Kompetenzzentrum für Nachwachsende Rohstoffe (TFZ). Available online: https//:www.fred.bayern.de/ (accessed on 10 June 2020).
- Werkelin, J.; Skrifvars, B.-J.; Hupa, M. Ash-forming elements in four Scandinavian wood species: Part 1: Summer harvest. Biomass Bioenergy 2005, 29, 451–466. [Google Scholar] [CrossRef]
- Kovacs, H.; Dobo, Z.; Koos, T.; Gyimesi, A.; Nagy, G. Influence of the flue gas temperature on the behavior of metals during biomass combustion. Energy Fuels 2018, 32, 7851–7856. [Google Scholar] [CrossRef]
- Pohlandt-Schwandt, K. Treatment of wood ash containing soluble chromate. Biomass Bioenergy 1999, 16, 447–462. [Google Scholar] [CrossRef]
- Schilling, S. Steuerungsmöglichkeiten der Qualität und Eignung von Holzaschen für deren Einsatz bei der Waldkalkung. Master’s Thesis, Albert-Ludwigs-Universität Freiburg, Freiburg, Germany, 2020. [Google Scholar]
- Wilpert, K.V.; Hartmann, P.; Schäffer, J. Quality control in a wood ash re-cycling concept for forests. VGB Powertech 2016, 96, 67–72. [Google Scholar]
- Steenari, B.-M.; Karlsson, L.G.; Lindqvist, O. Evaluation of the leaching characteristics of wood ash and the influence of ash agglomeration. Biomass Bioenergy 1999, 16, 119–136. [Google Scholar] [CrossRef]
- Lanzerstorfer, C. Grate-fired biomass combustion plants using forest residues as fuel. Enrichment factors for components in the fly ash. Waste Biomass Valorization 2017, 8, 235–240. [Google Scholar] [CrossRef]
- Lienemann, P.; Vock, W. Elementgehalte in Holzaschen und Validierung der Holzaschenkontrolle; ZHAW: Wädenswil, Switzerland, 2013; p. 96. [Google Scholar]
- Nurmesniemi, H.; Manskinen, K.; Pöykiö, R.; Dahl, O. Forest fertilizer properties of the bottom ash and fly ash from a large-sized (115 MW) industrial power plant incinerating wood-based biomass residues. J. Univ. Chem. Technol. Metall. 2012, 47, 43–52. [Google Scholar]
- Reichle, E.; Müller, R.; Schmoeckel, G.; Müller, C.; Wendland, M.; Geiger, H.; Stetter, U.; Zormaier, F. Verwertung und Beseitigung von Holzaschen. Merkblatt. Stand: 01.08.2009; Bayerisches Landesamt für Umwelt (LfU); Bayerische Landesanstalt für Wald und Forstwirtschaft (LWF); Bayerische Landesanstalt für Landwirtschaft (LfL): Augsburg, Germany, 2009; p. 19. [Google Scholar]
- Schrägle, R. Innovative Ansätze und Perspektiven der Verwertung von Holzaschen. In Proceedings of the 12th Internationaler BBE-Fachkongress Holzenergie, Augsburg, Germany, 27–28 September 2012; pp. 1–41. [Google Scholar]
- Stetter, U.; Zormaier, F. Verwertung und Beseitigung von Holzaschen. Neues LfU-Merkblatt Greift Altes Thema auf; LWF aktuell: Geneva, Switzerland, 2010; Volume 74, pp. 28–30. [Google Scholar]
- Hannam, K.D.; Venier, L.; Allen, D.; Deschamps, C.; Hope, E.; Jull, M.; Kwiaton, M.; McKenney, D.; Rutherford, P.M.; Hazlett, P.W. Wood ash as a soil amendment in Canadian forests: What are the barriers to utilization? Can. J. For. Res. 2018, 48, 442–450. [Google Scholar] [CrossRef]
- Lama, I.; Sain, D. A case study review of wood ash land application programs in North America, TAPPI Journal February 2021. TAPPI J. 2021, 20, 111–120. [Google Scholar]
- Bachmaier, H.; Kuptz, D.; Hartmann, H. Ash management at biomass heating plants in Southern Germany. In Setting the Course for a Biobased Economy–Papers of the 27th European Biomass Conference, Proceedings of the 27th European Biomass Conference, Lisbon, Portugal, 27–30 May 2019; Carvalho, M.D.G., Scarlat, N., Grassi, A., Helm, P., Eds.; ETA-Florence Renewable Energies: Florence, Italy, 2019; pp. 1814–1817. [Google Scholar]
- Bohrn, G.; Stampfer, K. Untreated wood ash as a structural stabilizing material in forest roads. Croat. J. For. Eng. 2014, 35, 81–89. [Google Scholar]
- Lahtinen, P. Utilization of biomass ashes in infrastructure construction in Finland. In Proceedings of the 4th Central European Biomass Conference, Graz, Austria, 15–18 January 2014; pp. 1–36. [Google Scholar]
- Ingerslev, M.; Hansen, M.; Pedersen, L.B.; Skov, S. Effects of wood chip ash fertilization on soil chemistry in a Norway spruce plantation on a nutrient-poor soil. For. Ecol. Manag. 2014, 334, 10–17. [Google Scholar] [CrossRef]
- Hansen, M.T. Options for increased use of ash from biomass combustion. In Proceedings of the Scandinavian Bio Mass Ash Workshop 2019. BMA Workshop, Copenhagen, Denmark, 25 March 2019; pp. 1–17. [Google Scholar]
- Saarsalmi, A.; Smolander, A.; Kukkola, M.; Moilanen, M.; Saramäki, J. 30-Year effects of wood ash and nitrogen fertilization on soil chemical properties, soil microbial processes and stand growth in a Scots pine stand. For. Ecol. Manag. 2012, 278, 63–70. [Google Scholar] [CrossRef]
- Brais, S.; Bélanger, N.; Guillemette, T. Wood ash and N fertilization in the Canadian boreal forest: Soil properties and response of jack pine and black spruce. For. Ecol. Manag. 2015, 348, 1–14. [Google Scholar] [CrossRef]
- Karltun, E.; Saarsalmi, A.; Ingerslev, M.; Mandre, M.; Andersson, S.; Gaitnieks, T.; Ozolincius, R.; Varnagiryte-Kabasinskiene, I. Wood ash recycling–possibilities and risks: Chapter 4. In Sustainable Use of Forest Biomass for Energy: A Synthesis with Focus on the Baltic and Nordic Region; Röser, D., Asikainen, A., Raulund-Rasmussen, K., Stupak, I., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 79–108. ISBN 978-1-4020-5054-1. [Google Scholar]
- Ernfors, M.; Sikström, U.; Nilsson, M.; Klemedtsson, L. Effects of wood ash fertilization on forest floor greenhouse gas emissions and tree growth in nutrient poor drained peatland forests. Sci. Total Environ. 2010, 408, 4580–4590. [Google Scholar] [CrossRef]
- Walter, B.; Mostbauer, P.; Karigl, B. Biomasse–Aschenströme in Österreich; Umweltbundesamt–Report, Nr. REP-0561; Umweltbundesamt GMBH: Wien, Austria, 2016; p. 56. [Google Scholar]
- Kehres, B. Zumischung von Holzasche bei der Kompostierung. H&K Aktuell 2016, 5, 1–3. [Google Scholar]
- Korpjiarvi, K. The development in the use of ashes in Finland. In Proceedings of the Scandinavian Biomass Ash Workshop 2019. BMA Workshop, Copenhagen, Denmark, 25 March 2019; pp. 1–22. [Google Scholar]
- Obernberger, I.; Supancic, K. Fact-Sheet: Einsatz von Holzasche als Bindemittel zur Bodenstabilisierung z.B. im Straßenbau; BIOS BIOENERGIESYSTEME GmbH: Wien, Austria, 2015; p. 8. [Google Scholar]
- Tejada, J.; Grammer, P.; Kappler, A.; Thorwarth, H. Trace element concentrations in firewood and corresponding stove ashes. Energy Fuels 2019, 33, 2236–2247. [Google Scholar] [CrossRef]
- Mortensen, L.H.; Rønn, R.; Vestergård, M. Bioaccumulation of cadmium in soil organisms—With focus on wood ash application. Ecotoxicol. Environ. Saf. 2018, 156, 452–462. [Google Scholar] [CrossRef]
- Saarsalmi, A.; Mälkönen, E.; Piirainen, S. Efffects of Wood Ash Fertilization on Forest Soil Chemical Properties. Filva Fenn. 2001, 35, 355–368. [Google Scholar]
- Kehres, B. Verwertung von Holzaschen auf Flächen; 08.03.2013. 2., überarb. Fassung; Bundesgütegemeinschaft Kompost e. V., Ed.; BGK Information: Köln-Gremberghoven, Gernamy, 2013; p. 14. [Google Scholar]
- Maresca, A.; Krüger, O.; Herzel, H.; Adam, C.; Kalbe, U.; Astrup, T.F. Influence of wood ash pre-treatment on leaching behaviour, liming and fertilising potential. Waste Manag. 2019, 83, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Ingerslev, M.; Skov, S.; Sevel, L.; Pedersen, L.B. Element budgets of forest biomass combustion and ash fertilization–A Danish case-study. Biomass Bioenergy 2011, 35, 2697–2704. [Google Scholar] [CrossRef]
- Okmanis, M.; Lazdiņa, D.; Lazdiņš, A. The composition and use value of tree biomass ash. Rural Sustain. Res. 2015, 34, 32–37. [Google Scholar] [CrossRef][Green Version]
- Bundesverband Mineralischer Rohstoffe e. V. (MIRO). Deutscher Nachhaltigkeitspreis 2019–Preisträger und Projekte. Die deutsche Gesteinsindustrie–modern, Effizient, Nachhaltig; Bundesverband Mineralischer Rohstoffe e. V. (MIRO): Duisburg, Germany, 2019; p. 39. [Google Scholar]
- Bundesministerium für Ernährung, Landwirtschaft und Verbraucherschutz. Verordnung über das Inverkehrbringen von Düngemitteln, Bodenhilfsstoffen, Kultursubstraten und Pflanzenhilfsmitteln. BGBI, 2017; Part I; Volume 68, pp. 1305–1349. Available online: https://www.gesetze-im-internet.de/d_mv_2012/BJNR248200012.html (accessed on 16 June 2020).
- Ettl, R.; Weis, W.; Göttlein, A. Laborversuch zur Bewertung von Organo-Asche-Presslingen und einem Kalk-Asche-Gemisch als mögliche Produkte für eine nährstoffliche Kreislaufwirtschaft in Wäldern. Forstarchiv 2010, 81, 12–20. [Google Scholar]
- Saarsalmi, A.; Smolander, A.; Moilanen, M.; Kukkola, M. Wood ash in boreal, low-productive pine stands on upland and peatland sites: Long-term effects on stand growth and soil properties. For. Ecol. Manag. 2014, 327, 86–95. [Google Scholar] [CrossRef]
- Katzensteiner, K.; Holzner, H.; Obernberger, I. Richtlinien für den sachgerechten Einsatz von Pflanzenaschen zur Verwertung auf land- und forstwirtschaftlich genutzten Flächen; Bundesministerium für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft, Eds.; Fachbeirat für Bodenfruchtbarkeit und Bodenschutz: Wien, Austria, 2011; 74p. [Google Scholar]
- Kebli, H.; Maltas, A.; Sinaj, S. Landwirtschaftlisches Potenzial von Asche aus rezykliertem Holz. Agrar. Schweiz 2017, 8, 30–37. [Google Scholar]
- Maltas, A.; Sinaj, S. Holzasche: Ein neuer Dünger für die Landwirtschaft. Agrar. Schweiz 2014, 5, 232–239. [Google Scholar]
- Bundesministerium für Umwelt, Naturschutz und Reaktorsicherheit (BMU); Bundesministerium für Ernährung, Landwirtschaft und Forsten (BMELF); Bundesministerium für Gesundheit (BMG). Verordnung über die Verwertung von Bioabfällen auf landwirtschaftlich, forstwirtschaftlich und gärtnerisch genutzten Böden). Bioabfallverordnung—BioAbfV, in der Fassung vom 1. August, Vorschriftensammlung Version 02; Stuttgart: Gewerbeaufsicht Baden-Württemberg, Germany, 2012; pp. 1–58. [Google Scholar]
- Sachverständigenrat Bioökonomie Bayern; Empfehlungen zur Förderung der Bioökonomie in Bayern: Straubing, Germany, 2017.
- Länderarbeitsgemeinschaft Abfall. LAGA PN 98—Grundregeln für die Entnahme von Proben aus festen und stichfesten Abfällen sowie abgelagerten Materialien Richtlinie für das Vorgehen bei physikalischen, chemischen und biologischen Untersuchungen im Zusammenhang mit der Verwertung/Beseitigung von Abfällen; LAGA: Potsdam, Germany, 2019; p. 69. [Google Scholar]
- Deutsches Institut für Normung (DIN). Characterization of Sludges–Determination of the Loss on Ignition of Dry Mass; German Version (EN 12879:2000); Beuth-Verlag: Berlin, Germany, 2001. [Google Scholar]
- Deutsches Institut für Normung (DIN). Soil Quality–Extraction of Trace Elements Soluble in Aqua Regia (ISO 11466:1995); Beuth-Verlag: Berlin, Germany, 1997. [Google Scholar]
- Deutsches Institut für Normung (DIN). Water Quality–Application of Inductively Coupled Plasma Mass Spectrometry (ICP-MS)–Part 2: Determination of 62 Elements (ISO 17294-2:2003), German version (EN ISO 17294-2:2004); Beuth-Verlag: Berlin, Germany, 2005. [Google Scholar]
- Deutsches Institut für Normung (DIN). Soil Quality–Determination of Chromium(VI) in Phosphate Extract (DIN 19734:1999); Beuth-Verlag: Berlin, Germany, 1999. [Google Scholar]
- Deutsches Institut für Normung (DIN). Soil quality–Determination of pH (ISO 10390:2005); Beuth-Verlag: Berlin, Germany, 2005. [Google Scholar]
- Verband deutscher landwirtschaftlicher Untersuchungs- und Forschungsanstalten (VDLUFA). Methodenbuch II. 2 Die Untersuchung von Sekundärrohstoffdüngern, Kultursubstraten und Bodenhilfsstoffen; VDLUFA: Darmstadt, Germany, 2000. [Google Scholar]
- Eberl, G. Veränderung von Chrom(VI)-Gehalten in Holzaschen durch Bewässerung. Vortrag. In Proceedings of the 21th Österreichischer Biomassetag, Kufstein, Austria, 6–7 November 2018; pp. 1–14. [Google Scholar]
- Obernberger, I. Aschen aus Biomassefeuerungen–Zusammensetzung und Verwertung. In Proceedings of the Thermische Biomassenutzung–Technik und Realisierung. Internationale Tagung, Salzburg, Austria, 23–24 April 1997; pp. 199–222. [Google Scholar]
- Ministerium für Umwelt und Verkehr Baden-Württemberg. Schadstoffströme bei der Entsorgung von Holzasche. Schadstoffströme bei der Verbrennung naturbelassener Hölzer und holzartiger Biomassen im Hinblick auf die Ascheentsorgung. Reihe Abfall 2003, 76, 79. [Google Scholar]
- Zimmermann, S.; Hässig, J.; Landolt, W. Literaturreview Holzasche–Wald. Nährstoffentzug durch Holzernte, abiotische und biotische Wirkungen; Eidgenössische Forschungsanstalt für Wald, Schnee und Landschaft (WSL): Birmensdorf, Switzerland, 2010; p. 80. [Google Scholar]

| Element | Unit | Current National Limits | ||
|---|---|---|---|---|
| DüMV | DüMV (Forest) | BioAbfV | ||
| Arsenic | mg/kg d.b. | 40 | 60 | - |
| Lead | mg/kg d.b. | 150 | 225 | 150 |
| Cadmium | mg/kg d.b. | 1.5 | 2.25 | 1.5 |
| Chromium, total | mg/kg d.b. | - | - | 100 |
| Chromium(VI) | mg/kg d.b. | 2 | - | - |
| Copper | mg/kg d.b. | (900) | (2000) | 100 |
| Nickel | mg/kg d.b. | 80 | 120 | 50 |
| Mercury | mg/kg d.b. | 1 | 1.5 | 1 |
| Thallium | mg/kg d.b. | 1 | 1.5 | 0 |
| Zinc | mg/kg d.b. | (5000) | (5000) | 400 |
| PFT | mg/kg d.b. | 0.1 | 0.15 | - |
| I-TE Dioxines and dl-PCB | ng WHO-TEQ/kg d.b. | 30 | 45 | - |
| Parameter | Unit | Plant ID | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 | 3 | 4 | ||
| Fuel | a | a | a | a | a | a | a | b | a | a | a | a | a | a | a | a | a | a | a | a | |
| Thermal power per combustion unit | kWtherm | 650 | 750 | 750 | 1050 | 1250 | 1500 | 1900 | 1950 | 2000 | 3000 | 3000 | 3500 | 3690 | 12,100 | 18,350 | 31,600 | 800 | <1 MW | 750 | 1050 |
| Total thermal power | kWtherm | 1300 | 1500 | 1500 | 2100 | 2500 | 1500 | 1900 | 1950 | 2000 | 3000 | 3000 | 3500 | 3690 | 12,100 | 27,850 | 31,600 | 800 | - | 1500 | 2100 |
| Bottom ash (n) | 12 | 1 | 1 | 2 | 2 | 2 | 1 | 1 | 1 | 2 | 2 | 1 | 2 | 2 | 2 | 2 | - | - | - | - | |
| Mixed bottom and cyclone ash (n) | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 2 | 1 | 1 | 1 | |
| Cyclone ash (n) | 8 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | |
| Composition of bottom ashes | Composition of mixed bottom and cyclone ashes | ||||||||||||||||||||
| Arsenic | mg/kg d.b. | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.5 | 7.5 | 5.0 | 27.0 | 5.0 | 6.3 | 5.0 | 5.0 | 5.0 | 5.0 | 10.7 | 10.0 | 5.0 | 5.0 | 11.0 |
| Lead | mg/kg d.b. | 4.7 | 5.0 | 2.0 | 5.4 | 3.7 | 16.0 | 3.0 | 12.0 | 2.0 | 5.4 | 11.0 | 2.3 | 4.5 | 45.5 | 3.9 | 150.5 | 38.0 | 180.0 | 12.0 | 15.0 |
| Cadmium | mg/kg d.b. | 0.4 | 0.4 | 0.2 | 0.7 | 0.3 | 2.5 | 0.7 | 1.3 | 0.3 | 1.0 | 4.1 | 1.0 | 0.4 | 0.4 | 0.3 | 1.0 | 15.4 | 15.0 | 4.7 | 7.6 |
| Chromium, total | mg/kg d.b. | 40 | 37 | 81 | 38 | 39 | 46 | 37 | 200 | 120 | 40 | 58 | 39 | 76 | 37 | 60 | 49 | 47 | 130 | 69 | 47 |
| Chromium (VI) | mg/kg d.b. | 2.0 | 1.8 | 32.8 | 2.0 | 6.3 | 3.8 | 2.1 | 42.3 | 16.0 | 2.0 | 3.7 | 1.6 | 6.7 | 1.0 | 1.8 | 1.0 | 1.8 | 21.8 | 5.5 | 4.0 |
| Copper | mg/kg d.b. | 51 | 53 | 100 | 69 | 51 | 68 | 66 | 150 | 120 | 45 | 47 | 79 | 64 | 29 | 54 | 44 | 55 | 110 | 48 | 94 |
| Nickel | mg/kg d.b. | 45 | 18 | 41 | 28 | 50 | 31 | 38 | 39 | 43 | 32 | 41 | 36 | 37 | 28 | 62 | 21 | 42 | 44 | 42 | 36 |
| Mercury | mg/kg d.b. | 0.05 | 0.05 | 0.05 | 0.06 | 0.05 | 0.09 | 0.05 | 0.05 | 0.05 | 0.05 | 0.05 | 0.10 | 0.05 | 0.05 | 0.05 | 0.06 | 0.05 | 0.06 | 0.05 | 0.09 |
| Thallium | mg/kg d.b. | 0.37 | 0.40 | 0.20 | 0.30 | 0.30 | 0.50 | 0.20 | 0.20 | 0.20 | 0.30 | 0.30 | 0.20 | 0.30 | 0.30 | 0.30 | 0.30 | 1.45 | 0.89 | 0.66 | 0.55 |
| Zinc | mg/kg d.b. | 110 | 46 | 120 | 173 | 37 | 500 | 490 | 95 | 64 | 170 | 420 | 260 | 142 | 161 | 145 | 205 | 760 | 1.500 | 320 | 990 |
| Lime (CaO) | wt% d.b. | 23.9 | 38.0 | 21.0 | 17.5 | 50.5 | 31.5 | 16.0 | 43.0 | 18.0 | 46.5 | 24.5 | 32.0 | 22.0 | 31.0 | 19.0 | 19.5 | 44.0 | 31.0 | 17.0 | 20.0 |
| Alkaline active substances (CaO) | wt% d.b. | 33.7 | 49.0 | 30.6 | 30.2 | 69.1 | 34.3 | 19.9 | 64.0 | 19.2 | 37.4 | 26.4 | 31.8 | 27.4 | 17.7 | 23.8 | 21.3 | 13.5 | 21.0 | 16.0 | 11.0 |
| Phosphate, total (P2O5) | wt% d.b. | 3.2 | 4.1 | 2.9 | 2.3 | 3.7 | 2.9 | 2.1 | 5.2 | 2.5 | 2.2 | 2.9 | 3.2 | 2.9 | 0.9 | 2.4 | 1.3 | 2.5 | 3.2 | 2.3 | 2.5 |
| Potassium oxide, total (K2O) | wt% d.b. | 10.2 | 11.0 | 8.1 | 5.5 | 5.1 | 6.4 | 4.5 | 12.0 | 4.8 | 5.5 | 6.0 | 6.5 | 8.1 | 2.5 | 7.0 | 3.4 | 5.6 | 11.0 | 6.1 | 6.1 |
| Magnesium oxide, total (MgO) | wt% d.b. | 4.2 | 5.0 | 4.1 | 2.9 | 6.3 | 4.2 | 2.2 | 5.8 | 2.3 | 7.9 | 3.1 | 3.5 | 3.8 | 1.7 | 3.6 | 2.0 | 3.2 | 4.3 | 3.3 | 3.3 |
| Total sulfur (S) | wt% d.b. | 0.11 | 0.14 | 0.04 | 0.07 | 0.04 | 0.23 | 0.15 | 0.16 | 0.09 | 0.15 | 0.34 | 0.10 | 0.11 | 0.07 | 0.05 | 0.25 | 0.54 | 1.40 | 0.29 | 0.41 |
| Boron (B) | mg/kg d.b. | 0.0 | 0.0 | 0.0 | 2.6 | 0.0 | 82.5 | 0.0 | 1.6 | 1.4 | 0.0 | 1.0 | 0.0 | 0.0 | 0.3 | 0.6 | 1.8 | 240.0 | 280.0 | 140.0 | 220.0 |
| Iron (Fe) | mg/kg d.b. | 26.083 | 9.500 | 18.000 | 14.000 | 5.750 | 15.500 | 16.000 | 5.200 | 9.600 | 13.500 | 14.000 | 12.000 | 17.000 | 11.300 | 21.000 | 14.500 | 47.000 | 94.000 | 51.000 | 51.000 |
| Cobalt (Co) | mg/kg d.b. | 22 | 4 | 13 | 11 | 12 | 10 | 11 | 28 | 14 | 8 | 17 | 13 | 15 | 10 | 19 | 9 | 14.000 | 17.000 | 23.000 | 16.000 |
| Manganese (Mn) | mg/kg d.b. | 17.567 | 2.600 | 12.000 | 8.250 | 25.000 | 11.550 | 5.500 | 40.000 | 17.000 | 6.250 | 15.500 | 13.000 | 11.500 | 1.450 | 13.000 | 1.700 | 11.350 | 23.000 | 8.700 | 13.000 |
| Molybdenum (Mo) | mg/kg d.b. | 2.6 | 7.0 | 5.0 | 3.5 | 3.9 | 4.1 | 5.0 | 5.0 | 5.0 | 4.6 | 3.5 | 5.0 | 3.5 | 3.5 | 3.5 | 3.6 | 3.5 | 3.1 | 2.0 | 5.0 |
| Sodium (Na) | mg/kg d.b. | 4.442 | 2.700 | 5.500 | 4.050 | 1.135 | 3.250 | 2.800 | 1.900 | 2.200 | 2.600 | 2.700 | 3.200 | 6.000 | 2.550 | 6.550 | 2.300 | 2.250 | 3.500 | 4.700 | 4.000 |
| Moisture content | wt% | 0.1 | 0.0 | 0.5 | 13.9 | 0.2 | 0.1 | 0.4 | 1.0 | 1.8 | 0.3 | 0.3 | 0.7 | 0.4 | 24.8 | 7.6 | 30.4 | 0.5 | 0.2 | 0.0 | 29.3 |
| Loss of ignition | wt% d.b. | 0.0 | 0.0 | 0.0 | 2.6 | 0.0 | 0.0 | 0.0 | 1.6 | 1.4 | 0.0 | 1.0 | 0.0 | 0.0 | 0.3 | 0.6 | 1.8 | 0.0 | 2.2 | 2.2 | 7.2 |
| pH | 12.7 | 13.1 | 12.8 | 12.9 | 12.8 | 12.8 | 12.7 | 13.3 | 12.4 | 12.8 | 12.9 | 12.9 | 12.8 | 12.7 | 12.6 | 12.7 | 12.8 | 13.1 | 12.8 | 12.5 | |
| Parameter | Unit | Min | 1st Quantile | Median | Mean | 3rd Quantile | Max |
|---|---|---|---|---|---|---|---|
| Arsenic (As) | mg/kg d.b. | 5 | 5 | 5 | 6.5 | 5.6 | 27 |
| Lead (Pb) | mg/kg d.b. | 2 | 2.8 | 5 | 20.2 | 11 | 260 |
| Cadmium (Cd) | mg/kg d.b. | 0.2 | 0.4 | 0.4 | 1 | 1.2 | 4.8 |
| Chromium, total (Cr) | mg/kg d.b. | 29 | 39 | 45 | 59 | 58 | 200 |
| Chromium(VI) (Cr(VI)) | mg/kg d.b. | 1 | 1.3 | 2.7 | 7.2 | 5.5 | 42.3 |
| Copper (Cu) | mg/kg d.b. | 5.8 | 39 | 60 | 64 | 90 | 150 |
| Nickel (Ni) | mg/kg d.b. | 18 | 28 | 37 | 37 | 43 | 78 |
| Mercury (Hg) | mg/kg d.b. | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 | 0.1 |
| Thallium (Th) | mg/kg d.b. | 0.2 | 0.2 | 0.2 | 0.3 | 0.4 | 0.8 |
| Zinc (Zn) | mg/kg d.b. | 26 | 95 | 145 | 205 | 250 | 900 |
| Parameter | Unit | Min | 1st Quantile | Median | Mean | 3rd Quantile | Max |
|---|---|---|---|---|---|---|---|
| Alkaline active components (CaO) | wt% d.b. | 15.5 | 23.5 | 30.1 | 32.9 | 33.5 | 75.2 |
| Calcium (calculated as CaO) | wt% d.b. | 15 | 18.5 | 24 | 28.9 | 37.5 | 56 |
| Magnesium (calculated as MgO) | wt% d.b. | 1.4 | 2.4 | 3.9 | 3.9 | 4.5 | 10 |
| Potassium (calculated as K2O) | wt% d.b. | 2.3 | 4.6 | 6.3 | 6.3 | 7.5 | 12 |
| Phosphorus (calculated as P2O5) | wt% d.b. | 0.9 | 2.1 | 2.6 | 2.6 | 3.1 | 5.2 |
| Total sulfur (S) | wt% d.b. | 0.0 | 0.1 | 0.1 | 0.1 | 0.2 | 0.4 |
| Boron (B) | mg/kg d.b. | 0 | 92 | 145 | 148 | 195 | 330 |
| Iron (Fe) | mg/kg d.b. | 5000 | 12,000 | 14,500 | 14,057 | 16,750 | 26,083 |
| Cobalt (Co) | mg/kg d.b. | 4.1 | 9.1 | 12.5 | 13.0 | 15.8 | 28.0 |
| Manganese (Mn) | mg/kg DM | 1000 | 4075 | 12,500 | 11,772 | 15,750 | 40,000 |
| Molybdenum (Mo) | mg/kg d.b. | 2.0 | 2.7 | 5.0 | 4.1 | 5.0 | 7.0 |
| Sodium (Na) | mg/kg d.b. | 970 | 2425 | 2750 | 3362 | 4181 | 8000 |
| Selenium (Se) | mg/kg d.b. | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 |
| pH value | - | 12.3 | 12.7 | 12.8 | 12.8 | 12.8 | 13.3 |
| Moisture content | wt% | 0.0 | 0.0 | 0.5 | 6.2 | 9.9 | 32.7 |
| Loss on ignition | wt% d.b. | 0.0 | 0.0 | 0.0 | 0.6 | 1.3 | 3.6 |
| Parameter | Unit | Thermal Power of Combustion Unit in MWtherm, n = Number of Units—Each Represented with One or Two Samples | ||
|---|---|---|---|---|
| <1 (n = 3) | 1–10 (n = 9) | >10 (n = 3) | ||
| Arsenic | mg/kg d.b. | 5.0 (5.0–5.0) | 7.6 (5.0–27.0) | 6.9 (5.0–10.7) |
| Lead | mg/kg d.b. | 3.9 (2.0–5.0) | 6.5 (2.0–16.0) | 66.6 (3.9–150.5) |
| Cadmium | mg/kg d.b. | 0.3 (0.2–0.4) | 1.2 (0.3–4.1) | 0.6 (0.3–1.0) |
| Chromium, total | mg/kg d.b. | 53 (37–81) | 69 (37–200) | 48 (37–60) |
| Chromium(VI) | mg/kg d.b. | 12.2 (1.8–32.8) | 8.7 (1.6–42.3) | 1.3 (1.0–1.8) |
| Copper | mg/kg d.b. | 68 (51–100) | 76 (45–150) | 42 (29–54) |
| Nickel | mg/kg d.b. | 35 (18–45) | 37 (28–50) | 37 (21–62) |
| Mercury | mg/kg d.b. | 0.05 (0.05–0.05) | 0.06 (0.05–0.10) | 0.05 (0.05–0.06) |
| Thallium | mg/kg d.b. | 0.3 (0.2–0.4) | 0.3 (0.2–0.5) | 0.3 (0.3–0.3) |
| Zinc | mg/kg d.b. | 92 (46–120) | 235 (37–500) | 170 (145–205) |
| Lime (CaO) | wt% d.b. | 28 (21–38) | 30 (16–51) | 23 (19–31) |
| Alkaline active substances (CaO) | wt% d.b. | 37.7 (30.6–49.0) | 36.0 (19.2–69.1) | 20.9 (17.7–23.8) |
| Phosphate, total (P2O5) | wt% d.b. | 3.4 (2.9–4.1) | 3.0 (2.1–5.2) | 1.5 (0.9–2.4) |
| Potassium oxide, total (K2O) | wt% d.b. | 9.8 (8.1–11.0) | 6.4 (4.5–12.0) | 4.3 (2.5–7.0) |
| Magnesium oxide, total (MgO) | wt% d.b. | 4.4 (4.1–5.0) | 4.2 (2.2–7.9) | 2.4 (1.7–3.6) |
| Total sulfur (S) | wt% d.b. | 0.10 (0.04–0.14) | 0.14 (0.04–0.34) | 0.12 (0.05–0.25) |
| Boron (B) | mg/kg d.b. | 0.02 (0.00–0.05) | 8.91 (0.00–82.50) | 0.90 (0.30–1.76) |
| Iron (Fe) | mg/kg d.b. | 17,861 (9500–26,083) | 12,255 (5200–17,000) | 15,600 (11,300–21,000) |
| Cobalt (Co) | mg/kg d.b. | 13 (4–22) | 14 (8–28) | 13 (9–19) |
| Manganese (Mn) | mg/kg d.b. | 10,722 (2600–17,567) | 15,355 (5500–40,000) | 5383 (1450–13,000) |
| Molybdenum (Mo) | mg/kg d.b. | 4.9 (2.6–7.0) | 4.3 (3.5–5.0) | 3.5 (3.5–3.6) |
| Sodium (Na) | mg/kg d.b. | 4213.9 (2700.0–5500.0) | 2983.5 (1135.0–6000.0) | 3800.0 (2300.0–6550.0) |
| Parameter | Unit | Bottom Ash | Cyclone Ash | ||
|---|---|---|---|---|---|
| Mean * | Standard Deviation | Mean * | Standard Deviation | ||
| Arsenic | mg/kg d.b. | <5.0 | 8.7 | 2.5 | |
| Lead | mg/kg d.b. | <5.0 | 123.3 | 65.6 | |
| Cadmium | mg/kg d.b. | 0.5 | 0.1 | 56.3 | 14.7 |
| Chromium, total | mg/kg d.b. | 41.1 | 7.3 | 54.4 | 10.4 |
| Chromium(VI) | mg/kg d.b. | 1.9 | 1.3 | 2.1 | 0.7 |
| Copper | mg/kg d.b. | 32.9 | 27.8 | 77.1 | 20.5 |
| Nickel | mg/kg d.b. | 46.0 | 9.1 | 51.9 | 8.7 |
| Mercury | mg/kg d.b. | <0.1 | 0.4 | 0.2 | |
| Thallium | mg/kg d.b. | <0.4 | 5.5 | 2.1 | |
| Zinc | mg/kg d.b. | 110 | 25.4 | 3787 | 788.1 |
| Lime (CaO) | wt% d.b. | 20.9 | 5.9 | 25.3 | 8.8 |
| Alkaline active substances (CaO) | wt% d.b. | 30.7 | 10.4 | 40.5 | 6.1 |
| Phosphate, total (P2O5) | wt% d.b. | 3.3 | 1.8 | 3.9 | 1.4 |
| Potassium oxide, total (K2O) | wt% d.b. | 10.1 | 3.3 | 8.5 | 3.0 |
| Magnesium oxide, total (MgO) | wt% d.b. | 4.0 | 0.9 | 5.3 | 1.1 |
| Total sulfur (S) | wt% d.b. | <0.1 | 1.8 | 0.3 | |
| Boron (B) | mg/kg d.b. | 191.3 | 40.8 | 435.0 | 72.6 |
| Iron (Fe) | mg/kg d.b. | 16,125 | 3370 | 35,125 | 11,263 |
| Cobalt (Co) | mg/kg d.b. | 20.6 | 6.5 | 29.9 | 13.4 |
| Manganese (Mn) | mg/kg d.b. | 15,850 | 7284 | 24,213 | 11,097 |
| Molybdenum (Mo) | mg/kg d.b. | 2.2 | 0.2 | 4.7 | 0.9 |
| Sodium (Na) | mg/kg d.b. | 4525 | 1253 | 2788 | 491 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bachmaier, H.; Kuptz, D.; Hartmann, H. Wood Ashes from Grate-Fired Heat and Power Plants: Evaluation of Nutrient and Heavy Metal Contents. Sustainability 2021, 13, 5482. https://doi.org/10.3390/su13105482
Bachmaier H, Kuptz D, Hartmann H. Wood Ashes from Grate-Fired Heat and Power Plants: Evaluation of Nutrient and Heavy Metal Contents. Sustainability. 2021; 13(10):5482. https://doi.org/10.3390/su13105482
Chicago/Turabian StyleBachmaier, Hans, Daniel Kuptz, and Hans Hartmann. 2021. "Wood Ashes from Grate-Fired Heat and Power Plants: Evaluation of Nutrient and Heavy Metal Contents" Sustainability 13, no. 10: 5482. https://doi.org/10.3390/su13105482
APA StyleBachmaier, H., Kuptz, D., & Hartmann, H. (2021). Wood Ashes from Grate-Fired Heat and Power Plants: Evaluation of Nutrient and Heavy Metal Contents. Sustainability, 13(10), 5482. https://doi.org/10.3390/su13105482






