Sustainability of Mussel (Mytilus Galloprovincialis) Farming in the Po River Delta, Northern Italy, Based on a Life Cycle Assessment Approach
Abstract
1. Introduction
2. Methods
2.1. General Background
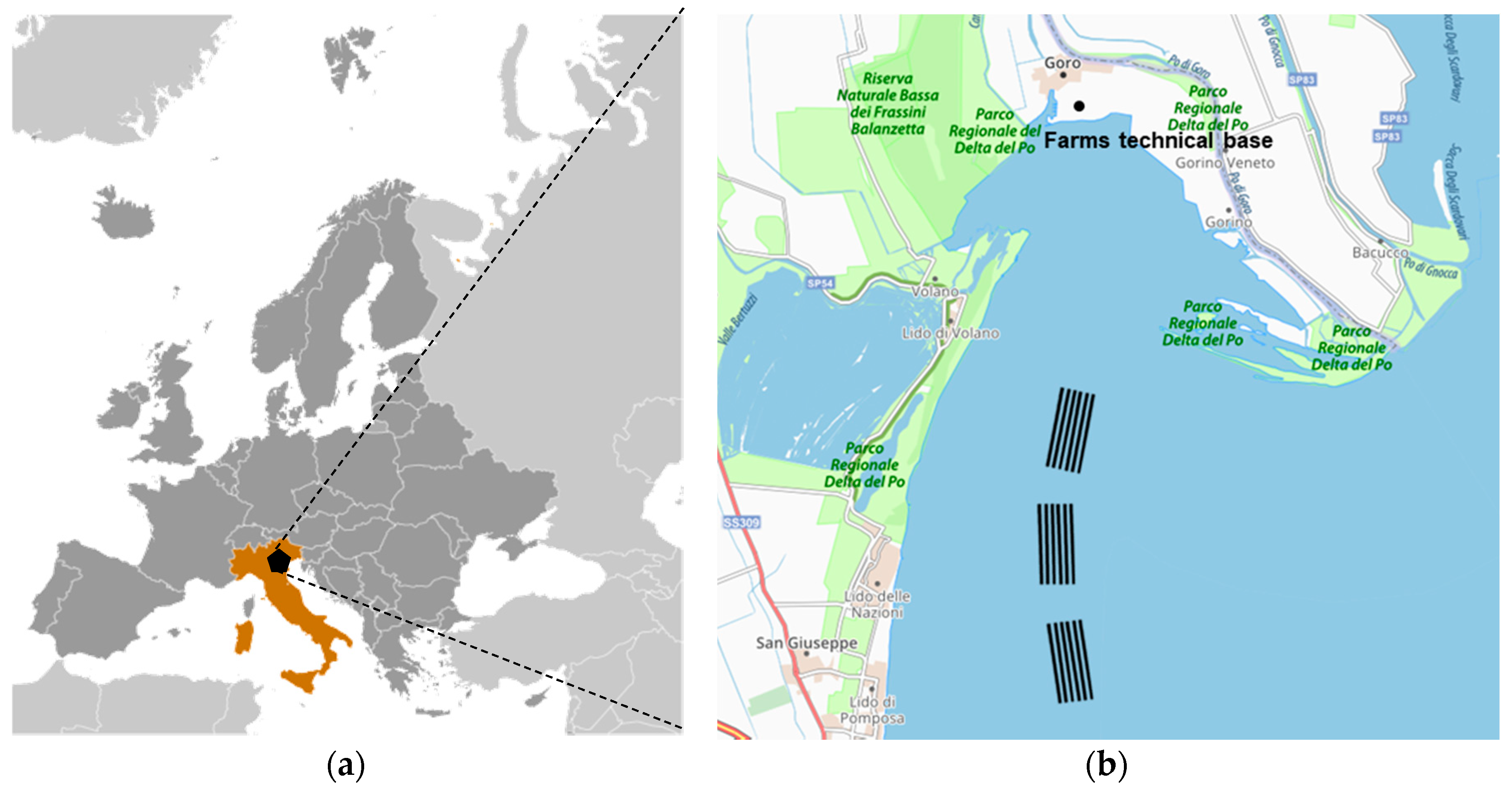
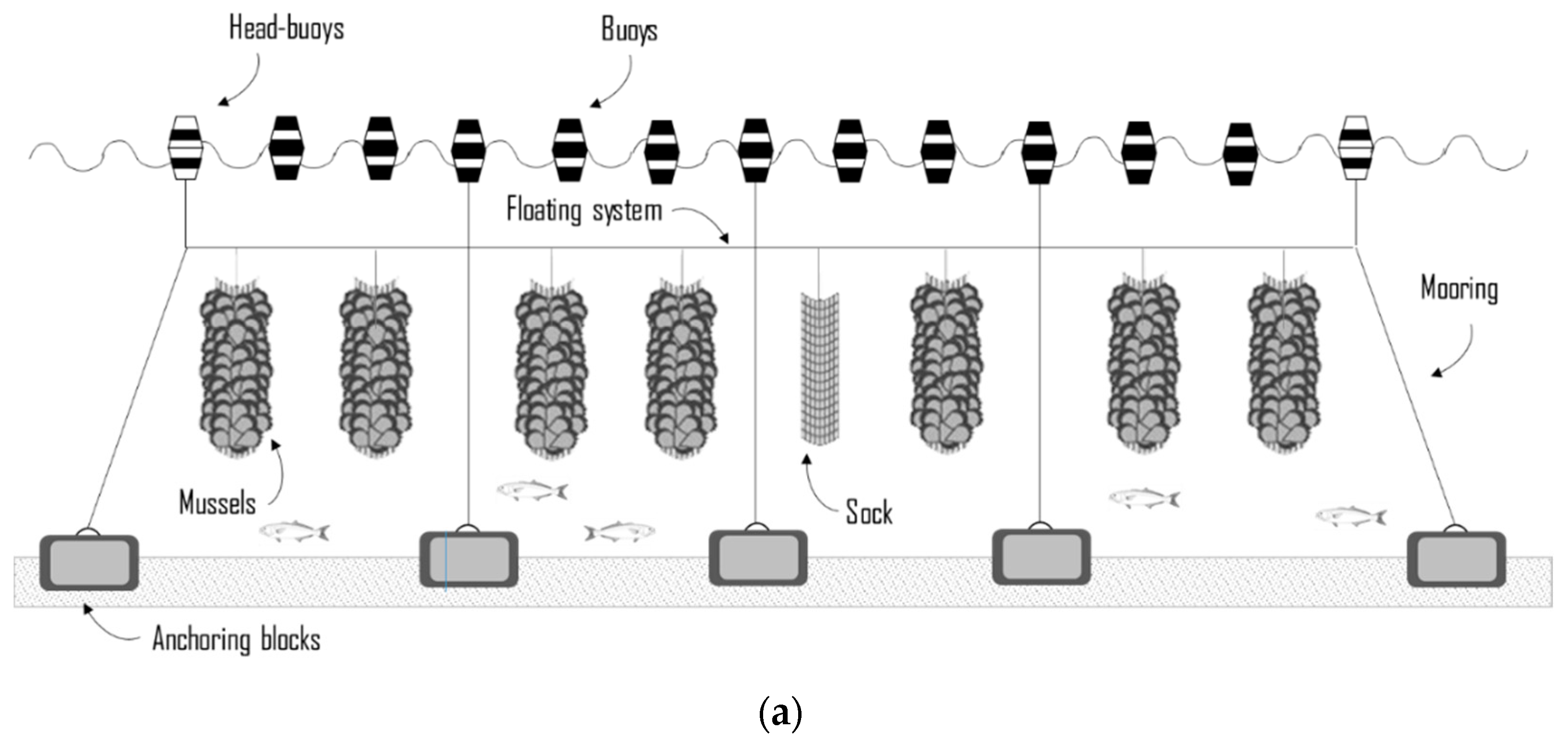
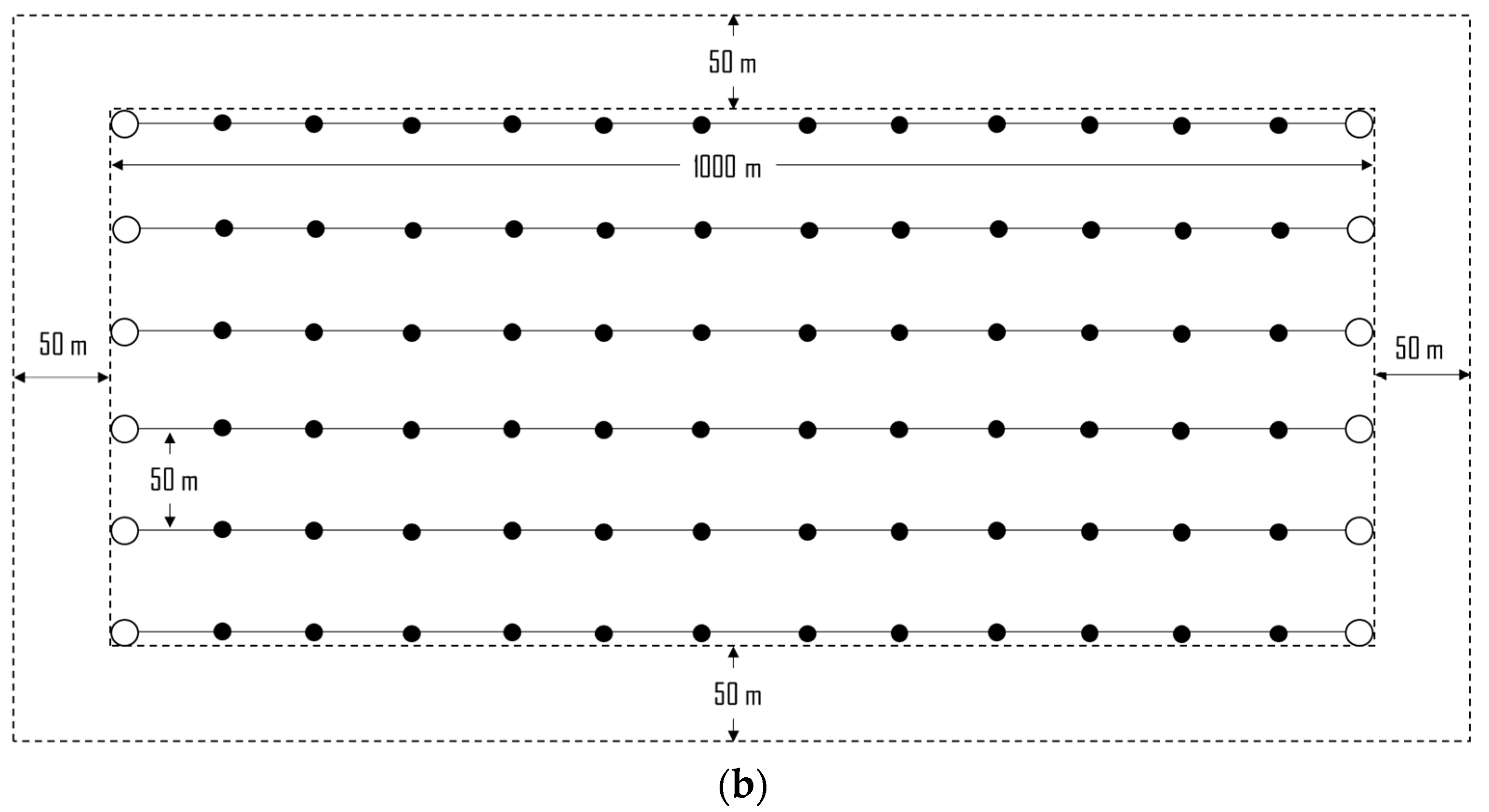
2.2. The Case Study
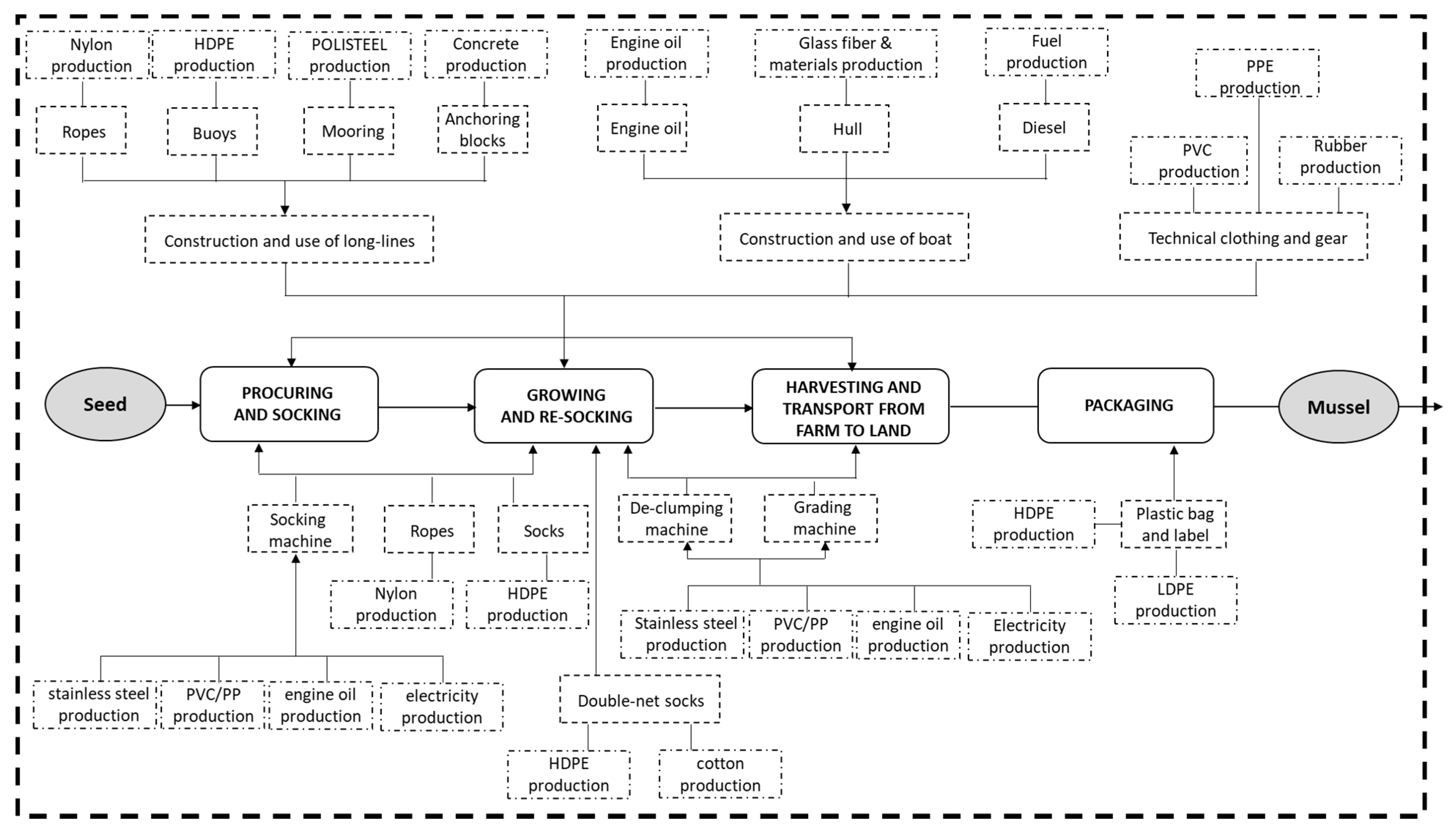
2.3. Goal, Scope Definition, Functional Unit and System Boundaries
2.4. Life Cycle Inventory
2.5. Uncertainty Analysis and Sensitivity Analysis
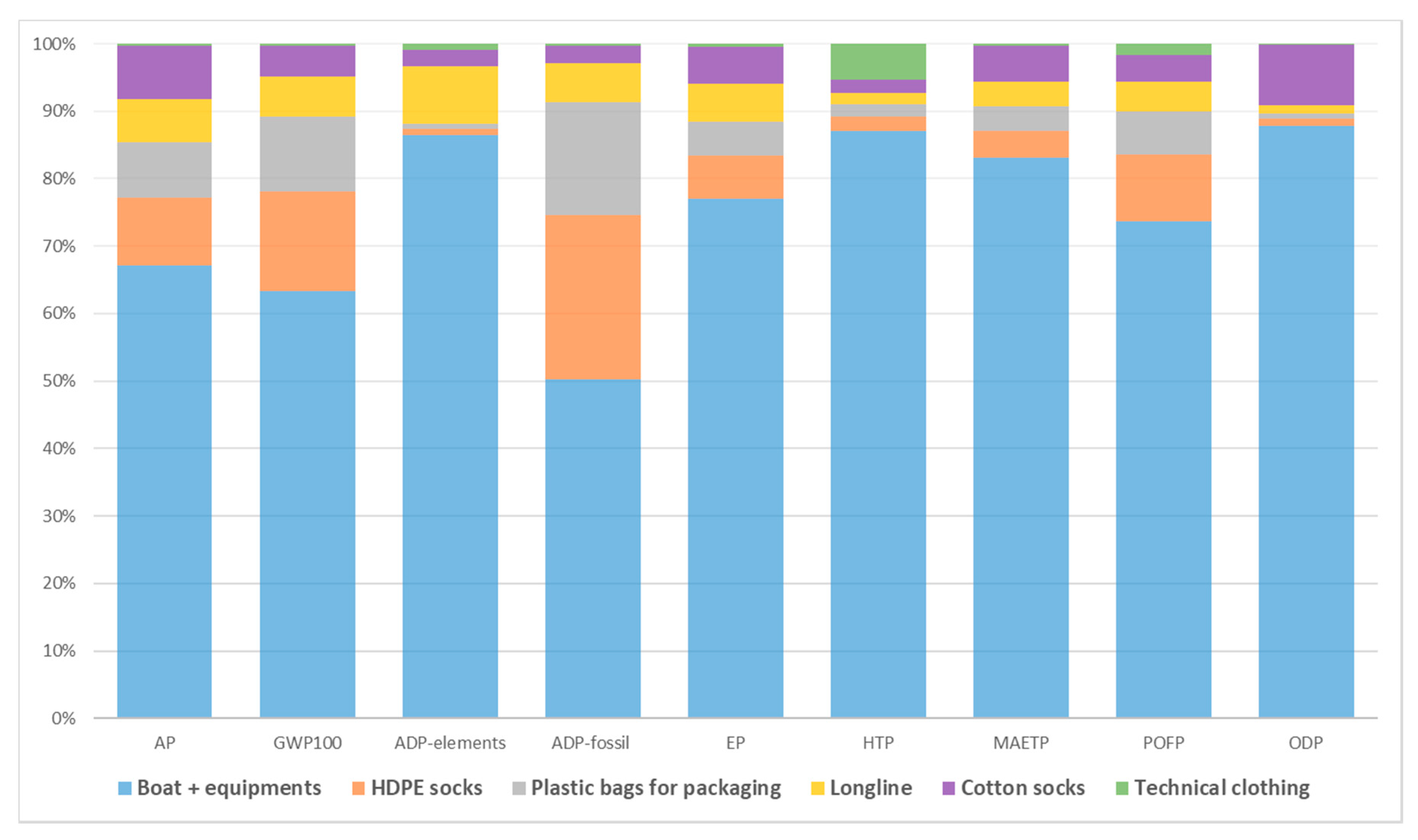
3. Results and Discussion
3.1. Environmental Performance of Mussel Farming
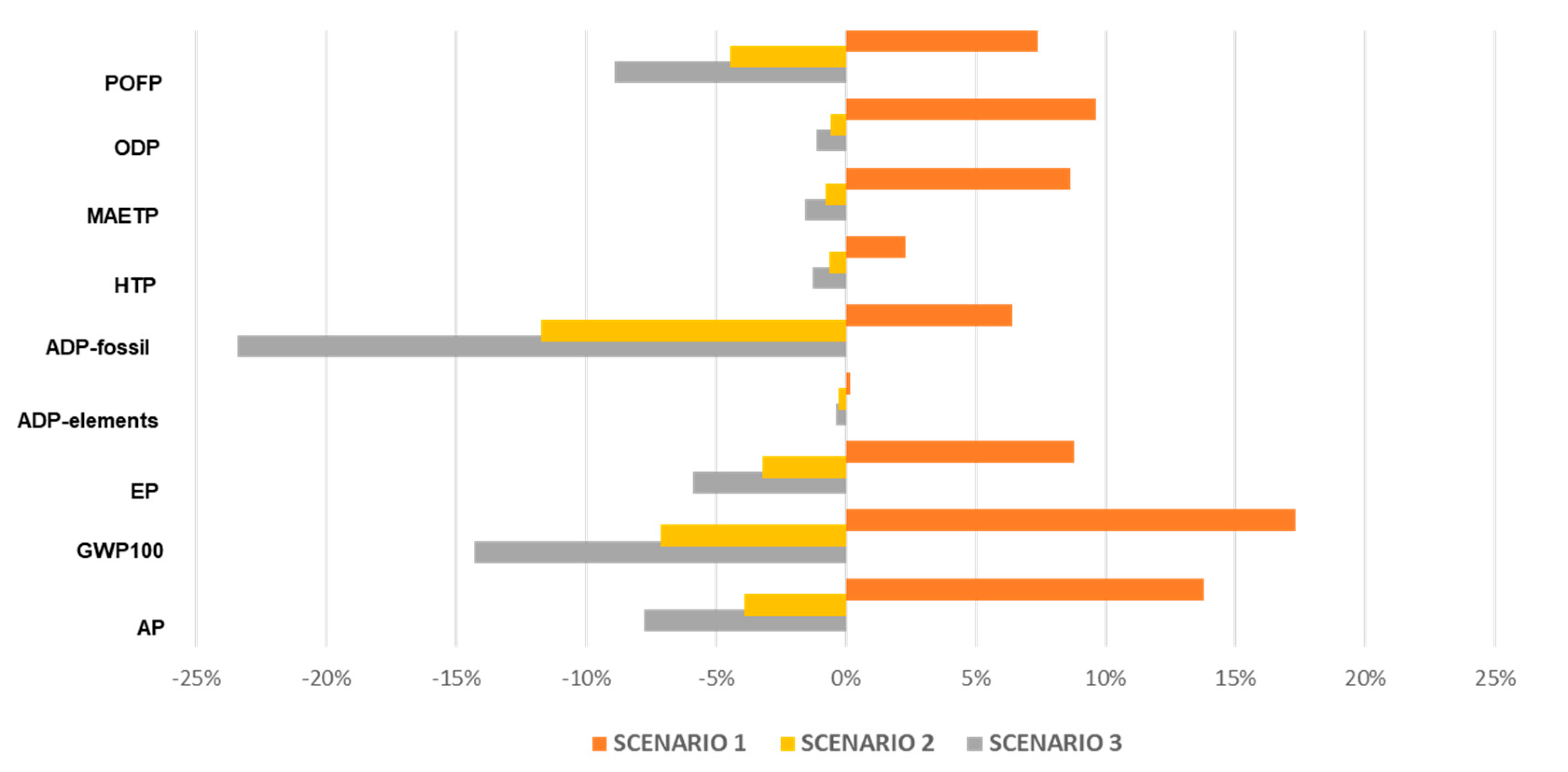
3.2. Sensitivity Analysis
3.3. Identification of Further Improvement Potential
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Anderson, J.L.; Asche, F.; Garlock, T.; Chu, J. Aquaculture: Its Role in the Future of Food. In Frontiers of Economics and Globalization; Emerald Publishing Limited: Bingley, UK, 2017; Volume 17, pp. 159–173. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture 2018 - Meeting the sustainable development goals. Rome. Licence: CC BY-NC-SA 3.0 IGO. 2018. Available online: http://www.fao.org/3/i9540en/i9540en.pdf (accessed on 13 March 2020).
- Willer, D.; Aldridge, D.C. Microencapsulated diets to improve bivalve shellfish aquaculture for global food security. Glob. Food Secur. 2019, 23, 64–73. [Google Scholar] [CrossRef]
- Jacquet, J.; Sebo, J.; Elder, M. Seafood in the future: bivalves are better. Solutions 2017, 8, 27. Available online: https://jenniferjacquet.files.wordpress.com/2018/06/jacquetseboelder_2017_solutions.pdf (accessed on 14 April 2020).
- Folke, C.; Kautsky, N. Aquaculture with its environment: Prospects for sustainability. Ocean Coast. Manag. 1992, 17, 5–24. [Google Scholar] [CrossRef]
- Diana, J.S. Aquaculture Production and Biodiversity Conservation. Biosci. 2009, 59, 27–38. [Google Scholar] [CrossRef]
- Deutsch, L.; Gräslund, S.; Folke, C.; Troell, M.; Huitric, M.; Kautsky, N.; Lebel, L. Feeding aquaculture growth through globalization: Exploitation of marine ecosystems for fishmeal. Glob. Environ. Chang. 2007, 17, 238–249. [Google Scholar] [CrossRef]
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food Security: The Challenge of Feeding 9 Billion People. Science 2010, 327, 812–818. [Google Scholar] [CrossRef]
- Dumbauld, B.R.; Ruesink, J.L.; Rumrill, S.S. The ecological role of bivalve shellfish aquaculture in the estuarine environment: A review with application to oyster and clam culture in West Coast (USA) estuaries. Aquac. 2009, 290, 196–223. [Google Scholar] [CrossRef]
- Shumway, S.E.; Davis, C.; Downey, R.; Karney, R.; Kraeuter, J.; Parsons, J.; Wikfors, G. Shellfish aquaculture–in praise of sustainable economies and environments. J. World Aquacult. Soc. 2003, 34, 8–10. [Google Scholar]
- Richter, C.; Skulas-Ray, A.C.; Champagne, C.M.; Kris-Etherton, P.M. Plant protein and animal proteins: do they differentially affect cardiovascular disease risk? Adv. Nutr. 2015, 6, 712–728. [Google Scholar] [CrossRef]
- Willer, D.; Aldridge, D.C. Microencapsulated diets to improve bivalve shellfish aquaculture. R. Soc. Open Sci. 2017, 4, 171142. [Google Scholar] [CrossRef]
- World Bank Data. CO2 Emissions. 2017. Available online: https://data.worldbank.org/indicator/EN.ATM.CO2E.PC (accessed on 3 March 2020).
- Danovaro, R.; Gambi, C.; Luna, G.; Mirto, S. Sustainable impact of mussel farming in the Adriatic Sea (Mediterranean Sea): evidence from biochemical, microbial and meiofaunal indicators. Mar. Pollut. Bull. 2004, 49, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Suplicy, F.M. A review of the multiple benefits of mussel farming. Rev. Aquac. 2018, 12, 204–223. [Google Scholar] [CrossRef]
- Venugopal, V.; Gopakumar, K. Shellfish: Nutritive Value, Health Benefits, and Consumer Safety. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1219–1242. [Google Scholar] [CrossRef]
- Prou, J.; Goulletquer, P. The French mussel industry: present status and perspectives. Bull Aquacul. Assoc. Can. 2002, 3, 17–23. [Google Scholar]
- Gangnery, A.; Bacher, C.; Buestel, D. Application of a population dynamics model to the Mediterranean mussel, Mytilus galloprovincialis, reared in Thau Lagoon (France). Aquac. 2004, 229, 289–313. [Google Scholar] [CrossRef]
- FAO. Fishery and aquaculture statistics. Global aquaculture production 1950-2015 (Fishstat). In: FAO Fisheries and Aquaculture Department [online]. Rome, Updated 2018. 2018. Available online: www.fao.org/fishery/statistics/software/fishstatj/enaccessed1.1.18 (accessed on 11 March 2020).
- European Market Observatory for Fisheries and Aquaculture Products (EUMOFA), 2019. Fresh mussel in the UE: price structure in the supply chain. Available online: https://www.eumofa.eu/documents/20178/151118/PTAT+Fresh+Mussel_EN.pdf (accessed on 11 March 2020).
- Rodríguez-Rodríguez, G.; Ramudo, R.B. Market driven management of climate change impacts in the Spanish mussel sector. Mar. Policy 2017, 83, 230–235. [Google Scholar] [CrossRef]
- Samuel-Fitwi, B.; Wuertz, S.; Schroeder, J.P.; Schulz, C. Sustainability assessment tools to support aquaculture development. J. Clean. Prod. 2012, 32, 183–192. [Google Scholar] [CrossRef]
- Finkbeiner, M.; Inaba, A.; Tan, R.; Christiansen, K.; Klüppel, H.-J. The New International Standards for Life Cycle Assessment: ISO 14040 and ISO 14044. Int. J. Life Cycle Assess. 2006, 11, 80–85. [Google Scholar] [CrossRef]
- Traverso, M.; Asdrubali, F.; Francia, A.; Finkbeiner, M. Towards life cycle sustainability assessment: an implementation to photovoltaic modules. Int. J. Life Cycle Assess. 2012, 17, 1068–1079. [Google Scholar] [CrossRef]
- Ziegler, F.; Winther, U.; Hognes, E.S.; Emanuelsson, A.; Sund, V.; Ellingsen, H. The Carbon Footprint of Norwegian Seafood Products on the Global Seafood Market. J. Ind. Ecol. 2012, 17, 103–116. [Google Scholar] [CrossRef]
- Farmery, A.K.; Gardner, C.; Green, B.S.; Jennings, S.; Watson, R.A. Domestic or imported? An assessment of carbon footprints and sustainability of seafood consumed in Australia. Environ. Sci. Policy 2015, 54, 35–43. [Google Scholar] [CrossRef]
- Cao, L.; Diana, J.S.; Keoleian, G.A. Role of life cycle assessment in sustainable aquaculture. Rev. Aquac. 2013, 5, 61–71. [Google Scholar] [CrossRef]
- Ford, J.S.; Ziegler, F.; Scholz, A.J.; Tyedmers, P.; Sonesson, U.; Kruse, S.A.; Silverman, H.; Pelletier, N.L. Proposed Local Ecological Impact Categories and Indicators for Life Cycle Assessment of Aquaculture. J. Ind. Ecol. 2012, 16, 254–265. [Google Scholar] [CrossRef]
- McGrath, K.P.; Pelletier, N.L.; Tyedmers, P. Life Cycle Assessment of a Novel Closed-Containment Salmon Aquaculture Technology. Environ. Sci. Technol. 2015, 49, 5628–5636. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Liu, Y.; Pettersen, J.B.; Brandão, M.; Ma, X.; Røberg, S.; Frostell, B. Life cycle assessment of recirculating aquaculture systems: A case of Atlantic salmon farming in China. J. Ind. Ecol. 2019, 23, 1077–1086. [Google Scholar] [CrossRef]
- Silvenius, F.; Grönroos, J.; Kankainen, M.; Kurppa, S.; Mäkinen, T.; Vielma, J. Impact of feed raw material to climate and eutrophication impacts of Finnish rainbow trout farming and comparisons on climate impact and eutrophication between farmed and wild fish. J. Clean. Prod. 2017, 164, 1467–1473. [Google Scholar] [CrossRef]
- Järviö, N.; Henriksson, P.J.G.; Guinée, J. Including GHG emissions from mangrove forests LULUC in LCA: a case study on shrimp farming in the Mekong Delta, Vietnam. Int. J. Life Cycle Assess. 2017, 23, 1078–1090. [Google Scholar] [CrossRef]
- Medeiros, M.V.; Aubin, J.; Camargo, A.F. Life cycle assessment of fish and prawn production: Comparison of monoculture and polyculture freshwater systems in Brazil. J. Clean. Prod. 2017, 156, 528–537. [Google Scholar] [CrossRef]
- Abdou, K.; Aubin, J.; Romdhane, M.S.; Le Loc’H, F.; Lasram, F.B.R. Environmental assessment of seabass (Dicentrarchus labrax) and seabream (Sparus aurata) farming from a life cycle perspective: A case study of a Tunisian aquaculture farm. Aquaculture 2017, 471, 204–212. [Google Scholar] [CrossRef]
- Lopez, P.A.; Toledo-Guedes, K.; Izquierdo-Gomez, D.; Šegvić-Bubić, T.; Sanchez-Jerez, P. Implications of Sea Bream and Sea Bass Escapes for Sustainable Aquaculture Management: A Review of Interactions, Risks and Consequences. Rev. Fish. Sci. Aquac. 2017, 26, 214–234. [Google Scholar] [CrossRef]
- Yacout, D.M.M.; Soliman, N.F.; Yacout, M.M. Comparative life cycle assessment (LCA) of Tilapia in two production systems: semi-intensive and intensive. Int. J. Life Cycle Assess. 2016, 21, 806–819. [Google Scholar] [CrossRef]
- Iribarren, D.; Moreira, M.T.; Feijoo, G. Life Cycle Assessment of fresh and canned mussel processing and consumption in Galicia (NW Spain). Resour. Conserv. Recycl. 2010, 55, 106–117. [Google Scholar] [CrossRef]
- Iribarren, D.; Hospido, A.; Moreira, M.T.; Feijoo, G. Carbon footprint of canned mussels from a business-to-consumer approach. A starting point for mussel processors and policy makers. Environ. Sci. Policy 2010, 13, 509–521. [Google Scholar] [CrossRef]
- Aubin, J.; Fontaine, C.; Callier, M.D.; D’Orbcastel, E.R. Blue mussel (Mytilus edulis) bouchot culture in Mont-St Michel Bay: potential mitigation effects on climate change and eutrophication. Int. J. Life Cycle Assess. 2017, 23, 1030–1041. [Google Scholar] [CrossRef]
- Lourguioui, H.; Brigolin, D.; Boulahdid, M.; Pastres, R. A perspective for reducing environmental impacts of mussel culture in Algeria. Int. J. Life Cycle Assess. 2017, 22, 1266–1277. [Google Scholar] [CrossRef]
- Fry, J.M. Carbon Footprint of Scottish Suspended Mussels and Intertidal Carbon Footprint of Scottish Suspended Mussels and Intertidal Oysters. SARF078, 2018, Pitlochry, UK. Available online: http://www.sarf.org.uk/cms-assets/documents/43896-326804.sarf078 (accessed on 14 April 2020).
- European Commission—Joint Research Centre—Institute for Environment and Sustainability. International Reference Life Cycle Data System (ILCD) Handbook—General Guide for Life Cycle Assessment—Detailed Guidance, 1st ed.; EUR 24708 EN; Publications Office of the European Union: Luxembourg, 2010. [Google Scholar]
- Viaroli, P.; Giordani, G.; Bartoli, M.; Naldi, M.; Azzoni, R.; Nizzoli, D.; Ferrari, I.; Comenges, J.M.Z.; Bencivelli, S.; Castaldelli, G.; et al. The Sacca di Goro Lagoon and an Arm of the Po River. In The Handbook of Environmental Chemistry; Springer Science and Business Media LLC: Berlin, Germany, 2005; Volume 5, pp. 197–232. [Google Scholar]
- Maffei, M.; Matarazzo, D.; Mietti, N.; Pasini, M.; Tontini, N.; Turolla, E.; Massaccesi, G.; Camanzi, L. Consorzio mitilicoltori dell’Emilia Romagna, Studi ed indagini rivolti al miglioramento della mitilicoltura in Emilia-Romagna. Greentime. 2011. Available online: https://agricoltura.regione.emilia-romagna.it/pesca/temi/pubblicazioni/acquacoltura/mitilicoltura-in-emilia-romagna (accessed on 16 March 2020).
- Buck, B.H.; Ebeling, M.W.; Michler-Cieluch, T. MUSSEL CULTIVATION AS A CO-USE IN OFFSHORE WIND FARMS: POTENTIAL AND ECONOMIC FEASIBILITY. Aquac. Econ. Manag. 2010, 14, 255–281. [Google Scholar] [CrossRef]
- Ecoinvent Database®. Available online: https://www.ecoinvent.org/database/database.html (accessed on 15th January 2020).
- De Bruijn, H.; Van Duin, R.; Huijbregts, M.A.J. Handbook on Life Cycle Assessment. In Ahead of the Curve; Springer Science and Business Media LLC: Berlin, Germany, 2002. [Google Scholar]
- Pelletier, N.L.; Ayer, N.W.; Tyedmers, P.H.; Kruse, S.A.; Flysjo, A.; Robillard, G.; Sonesson, U. Impact categories for life cycle assessment research of seafood production systems: review and prospectus. Int. J. Life Cycle Ass. 2007, 12, 414–421. [Google Scholar] [CrossRef]
- Muller, S.; Lesage, P.; Ciroth, A.; Mutel, C.L.; Weidema, B.P.; Samson, R. The application of the pedigree approach to the distributions foreseen in ecoinvent v3. Int. J. Life Cycle Assess. 2014, 21, 1327–1337. [Google Scholar] [CrossRef]
- Lo, S.-C.; Ma, H.-W. Quantifying and reducing uncertainty in life cycle assessment using the Bayesian Monte Carlo method. Sci. Total. Environ. 2005, 340, 23–33. [Google Scholar] [CrossRef]
- Gallardi, D.; Dörner, J.; Carbonell, P.; Pino, S.; Farías, A. Effects of Bivalve Aquaculture on the Environment and Their Possible Mitigation: A Review. Fish. Aquac. J. 2014, 5, 1–3. [Google Scholar] [CrossRef]
- Van Der Werf, H.M.G.; Turunen, L. The environmental impacts of the production of hemp and flax textile yarn. Ind. Crop. Prod. 2008, 27, 1–10. [Google Scholar] [CrossRef]
- Minami, S.; Toki, T.; Yoshikawa, N.; Hanada, T.; Ashida, M.; Kitada, S.-I.; Tsukuda, K. A Newly Developed Plug-in Hybrid Electric Boat (PHEB). J. Asian Electr. Veh. 2010, 8, 1385–1392. [Google Scholar] [CrossRef]
- Gestore dei Servizi Energetici S.p.A Rapporto Statistico 2018. Available online: http://enerweb.casaccia.enea.it/enearegioni/UserFiles/GSE_Rapporto_Statistico_FER_2018.pdf (accessed on 15 March 2020).
- Wibowo, A.C.; Mohanty, A.K.; Misra, M.; Drzal, L.T. Chopped Industrial Hemp Fiber Reinforced Cellulosic Plastic Biocomposites: Thermomechanical and Morphological Properties. Ind. Eng. Chem. Res. 2004, 43, 4883–4888. [Google Scholar] [CrossRef]
- Amaducci, S.; Amaducci, M.; Benati, R.; Venturi, G. Crop yield and quality parameters of four annual fibre crops (hemp, kenaf, maize and sorghum) in the North of Italy. Ind. Crop. Prod. 2000, 11, 179–186. [Google Scholar] [CrossRef]
- Avio, C.G.; Gorbi, S.; Regoli, F. Plastics and microplastics in the oceans: From emerging pollutants to emerged threat. Mar. Environ. Res. 2017, 128, 2–11. [Google Scholar] [CrossRef]
- Lusher, A.; Hollman, P.; Mendoza-Hill, J. Microplastics in fisheries and aquaculture. FAO Fisheries and Aquaculture Technical Paper (FAO) eng no. 615. 2017. Available online: http://www.fao.org/3/a-i7677e.pdf (accessed on 14 April 2020).
- Smith, M.; Love, D.C.; Rochman, C.M.; Neff, R.A. Microplastics in Seafood and the Implications for Human Health. Curr. Environ. Heal. Rep. 2018, 5, 375–386. [Google Scholar] [CrossRef]
- OSPAR convention, Scoping study to identify key waste items from the fishing industry and aquaculture Marine Litter Regional Action Plan Action 35. 2019. Available online: https://www.ospar.org/documents?v=41242 (accessed on 27 April 2020).
- Stamatopoulos, C. Current Status of Fisheries and Aquaculture Statistics: A Brief Review. Oceanogr. Fish. Open Access J. 2018, 7. [Google Scholar] [CrossRef]
- Sandra, M.; Devriese, L.; De Raedemaecker, F.; Lonneville, B.; Lukic, I.; Altvater, S.; Compa Ferrer, M.; Deudero, S.; Torres Hansjosten, B.; Alomar Mascaró, C.; et al. Knowledge wave on marine litter from aquaculture sources. D2.2 Aqua-Lit project. Oostende, Belgium. 2019. Available online: https://aqua-it.eu/assets/content/D2.2.%20Knowledge%20wave%20on%20marine%20litter%20from%20aquaculture%20sources_upd.pdf (accessed on 3 May 2020).
- Barile, N.B.; Scopa, M.; Nerone, E.; Mascilongo, G.; Recchi, S.; Cappabianca, S.; Antonetti, L. Study of the efficacy of a closed cycle depuration system on bivalve molluscs. Veter- Ital. 2010, 45, 555–566. [Google Scholar]
- Kinnon, A.M. Guidelines for Measuring and Managing CO2 Emission from Freight Transport Operations. The European Chemical Industry Council. 2011. Available online: https://www.ecta.com/resources/Documents/Best%20Practices%20Guidelines/guideline_for_measuring_and_managing_co2.pdf (accessed on 16 April 2020).
- Friel, S.; Schram, A.; Townsend, B. The nexus between international trade, food systems, malnutrition and climate change. Nat. Food 2020, 1, 51–58. [Google Scholar] [CrossRef]
| Parameter | Value | Standard Deviation | Unit |
|---|---|---|---|
| Hull material | Fiberglass | - | - |
| Length | 15.7 | 2.3 | m |
| Main engine power | 210 | 20 | hp |
| Boat equipment | Socking machine De-clumping machine Grading machine | - | - |
| Crew | 4.5 | 1.0 | people |
| Distance to cultivation site | 3.3 | 0.4 | miles |
| Annual consumption of diesel | 6250 | 2235 | l/years |
| Annual consumption of oil | 45 | 5 | l/years |
| Annual days of work | 205 | 5 | days |
| INPUTS | |||
|---|---|---|---|
| From the Technosphere | From the Environment | ||
| Materials and Fuels | Resources | ||
| Stainless steel AISI 316 (g) | 800.0 | Sea use (m2 year−1) | 1.54 |
| High-density polyethylene (HDPE) (g) | 27.7 | Seawater (m3) | 0.001 |
| Low-density polyethylene (LDPE) (g) | 5.0 | Freshwater (m3) | - |
| Fiberglass | 50.0 | ||
| Polypropylene (PP) (g) | 5.8 | ||
| Polisteel (g) | 19.7 | ||
| Polyvinyl chloride (PVC) (g) | 0.8 | ||
| Rubber (g) | < 0.1 | ||
| Polyphenil ether (PPE) (g) | < 0.1 | ||
| Cotton (g) | 0.2 | ||
| Nylon (g) | 9.3 | ||
| Concrete (g) | 1730.0 | ||
| Diesel for boat (g) | 20 | ||
| Engine oil (l) | 0.2 | ||
| Vehicles | |||
| Boat (no. of items) | 1 | ||
| Emissions to air | |||
| Carbon dioxide (kg) | 0.037 | ||
| Nitrous oxide (kg) | 1.1 × 10−4 | ||
| Sulfur oxide (kg) | 4.2 × 10−5 | ||
| Methane (kg) | 6.8 × 10−4 | ||
| Non-methane volatile organic carbon (NMVOC) | 1.5 × 10−4 | ||
| Particulates < 2.5 μ (kg) | 1.6 × 10−5 | ||
| Particulates < 10 μ (kg) | 4.1 × 10−6 | ||
| Particulates > 2.5 μ and < 10 μ (kg) | 5.2 × 10−6 | ||
| Emissions to Water | |||
| Absorbable organic halogen as Cl (AOX) (kg) | 1.3 × 10−9 | ||
| Biochemical oxygen demand (BOD) (kg) | 5.4 × 10−6 | ||
| Heat, waste (MJ) | 1.3 × 10−4 | ||
| Nitrate (kg) | 3.7 × 10−7 | ||
| Indicator | Score | Modes | Uncertainty Factor |
|---|---|---|---|
| Reliability | 1 | Verified data based on measurement | 1.00 |
| Completeness | 1 | Representative data from all sited relevant for the process considered | 1.00 |
| Temporal correlation | 1 | Less than three years of difference to the time period of the dataset | 1.00 |
| Geographical correlation | 1 | Data from the area under study | 1.00 |
| Further technological correlation | 2 | Data from processes and materials under study (i.e. identical technology) but from different enterprises | 1.18 |
| Impact Category | Value | Unit |
|---|---|---|
| Climate change—GWP100 * | 0.137 | kg CO2 eq. |
| Acidification potential (AP) | 7.1 × 10−4 | kg SO2 eq. |
| Eutrophication potential (EP) | 1.7 × 10−4 | kg PO4 eq. |
| Depletion of abiotic resources (ADP)—elements, ultimate reserves | 4.4 × 10−7 | kg Sb eq. |
| Depletion of abiotic resources (ADP)—fossil fuel | 2.55 | MJ |
| Ozone layer depletion potential (ODP) | 8.67 × 10−9 | kg CFC-11 eq. |
| Photochemical oxidant formation potential (POFP) | 5.12 × 10−5 | kg ethylene eq. |
| Human toxicity potential (HTP) | 0.13 | kg 1,4-DCB eq. ** |
| Marine water aquatic ecotoxicity potential (MAETP) | 130.34 | kg 1,4-DCB eq. |
| Impact Category | Mean | SD | CV% | Min | Max | Median | 5% | 95% |
|---|---|---|---|---|---|---|---|---|
| Climate change – GWP100 | 1.00 × 10−3 | 4.70 × 10−4 | 47% | 4.63 × 10−4 | 5.68 × 10−3 | 8.71 × 10−4 | 5.97 × 10−4 | 1.00 × 10−3 |
| Acidification potential (AP) | 1.58 × 10−1 | 3.09 × 10−2 | 20% | 9.95 × 10−2 | 3.78 × 10−1 | 1.53 × 10−1 | 1.20 × 10−1 | 1.58 × 10−1 |
| Eutrophication potential (EP) | 4.86 × 10−7 | 5.59 × 10−8 | 12% | 3.22 × 10−7 | 7.57 × 10−7 | 4,83 × 10−7 | 4.04 × 10−7 | 4.86 × 10−7 |
| Depletion of abiotic resources (ADP) - elements, ultimate reserves | 2.97 | 7.12 | 24% | 1.90 | 9.33 | 2.85 | 2.19 | 2.97 |
| Depletion of abiotic resources (ADP) – fossil fuel | 3.36 × 10−4 | 1.33 × 10−4 | 40% | 1.56 × 10−4 | 1.6610−3 | 3.14 × 10−4 | 1.95 × 10−4 | 3.36 × 10−4 |
| Human toxicity potential (HTP) | 2.20 × 10−1 | 1.33 × 10−1 | 60% | 1.20 × 10−1 | 2.05 | 1.95 × 10−1 | 1.43 × 10−1 | 2.20 × 10−1 |
| Marine water aquatic ecotoxicity potential (MAETP) | 249.82 | 184.23 | 74% | 111.31 | 2688.85 | 214.13 | 147.86 | 416.31 |
| Ozone layer depletion potential (ODP) | 1.10 × 10−8 | 3.65 × 10−8 | 33% | 5.16 × 10−9 | 3.04 × 10−8 | 1.03 × 10−8 | 6.64 × 10−9 | 1.10 × 10 −8 |
| Photochemical oxidant formation potential (POFP) | 7.15 × 10−5 | 2.82 × 10−5 | 39% | 3.04 × 10−5 | 3.08 × 10−4 | 6.45 × 10−5 | 4.2810−5 | 7.15 × 10−5 |
| Transport Mode | gCO2eq./kg·km * | Average km | Extra Emissions as kgCO2eq./kg of Mussel Product |
|---|---|---|---|
| From Spain | ~2000 | ||
| Intermodal road/barge | 0.034 | 0.068 | |
| Intermodal road/rail | 0.026 | 0.052 | |
| Road | 0.062 | 0.124 | |
| From Chile | ~12,000 | ||
| Deep sea/container | 0.008 | 0.096 | |
| Air freight | 0.602 | 7.224 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tamburini, E.; Turolla, E.; Fano, E.A.; Castaldelli, G. Sustainability of Mussel (Mytilus Galloprovincialis) Farming in the Po River Delta, Northern Italy, Based on a Life Cycle Assessment Approach. Sustainability 2020, 12, 3814. https://doi.org/10.3390/su12093814
Tamburini E, Turolla E, Fano EA, Castaldelli G. Sustainability of Mussel (Mytilus Galloprovincialis) Farming in the Po River Delta, Northern Italy, Based on a Life Cycle Assessment Approach. Sustainability. 2020; 12(9):3814. https://doi.org/10.3390/su12093814
Chicago/Turabian StyleTamburini, Elena, Edoardo Turolla, Elisa Anna Fano, and Giuseppe Castaldelli. 2020. "Sustainability of Mussel (Mytilus Galloprovincialis) Farming in the Po River Delta, Northern Italy, Based on a Life Cycle Assessment Approach" Sustainability 12, no. 9: 3814. https://doi.org/10.3390/su12093814
APA StyleTamburini, E., Turolla, E., Fano, E. A., & Castaldelli, G. (2020). Sustainability of Mussel (Mytilus Galloprovincialis) Farming in the Po River Delta, Northern Italy, Based on a Life Cycle Assessment Approach. Sustainability, 12(9), 3814. https://doi.org/10.3390/su12093814








