3.1. Reduction Smelting for Ironmaking
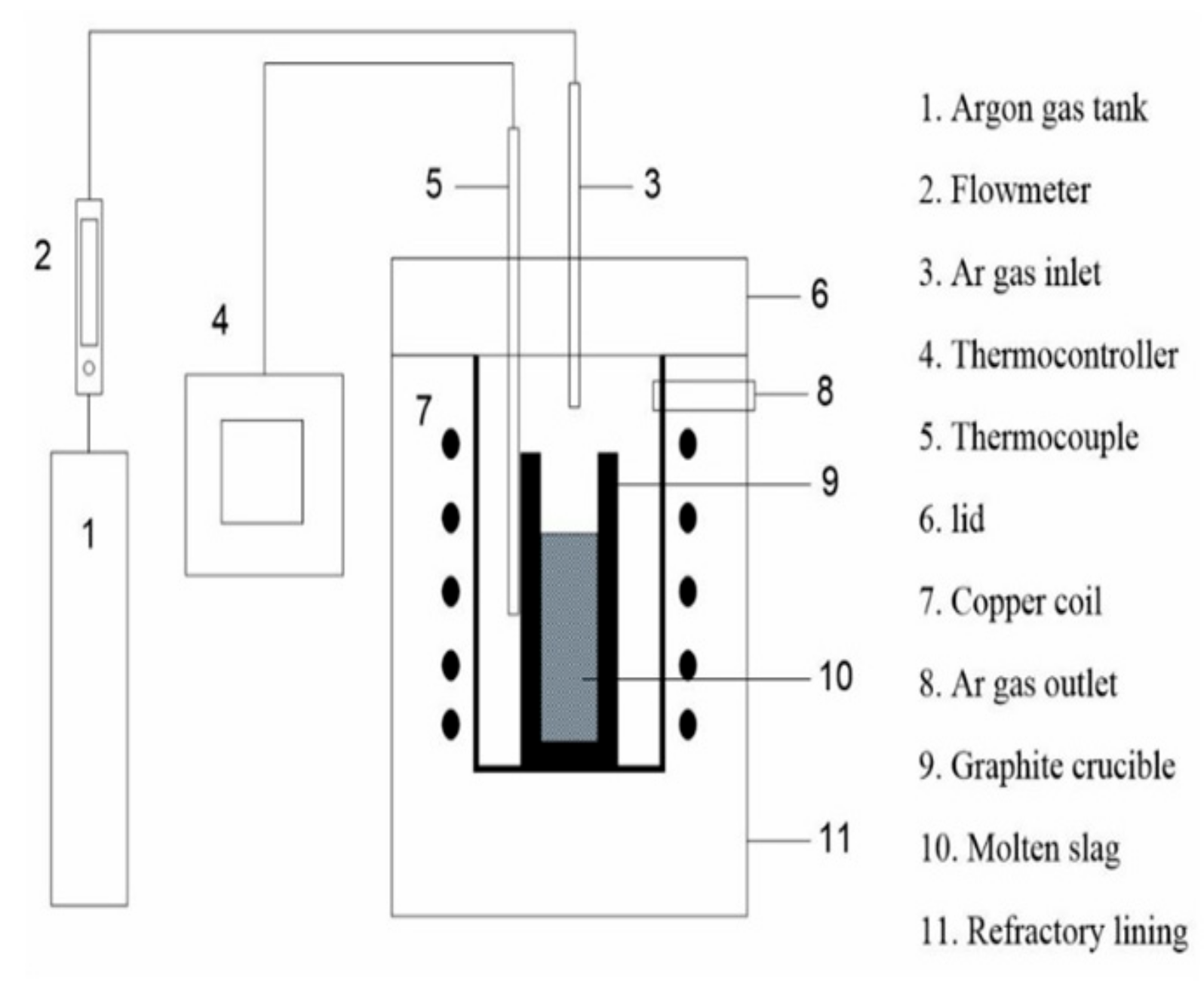
The reduction smelting experiments on the recovery of iron from copper slag were performed by reduction agent carbon at high temperatures under holding temperature and holding time factors. In the reduction smelting process of copper slag, iron oxide might be reduced from the decomposition of fayalite by direct reduction with solid carbon and indirect reduction with CO gas following this reaction:
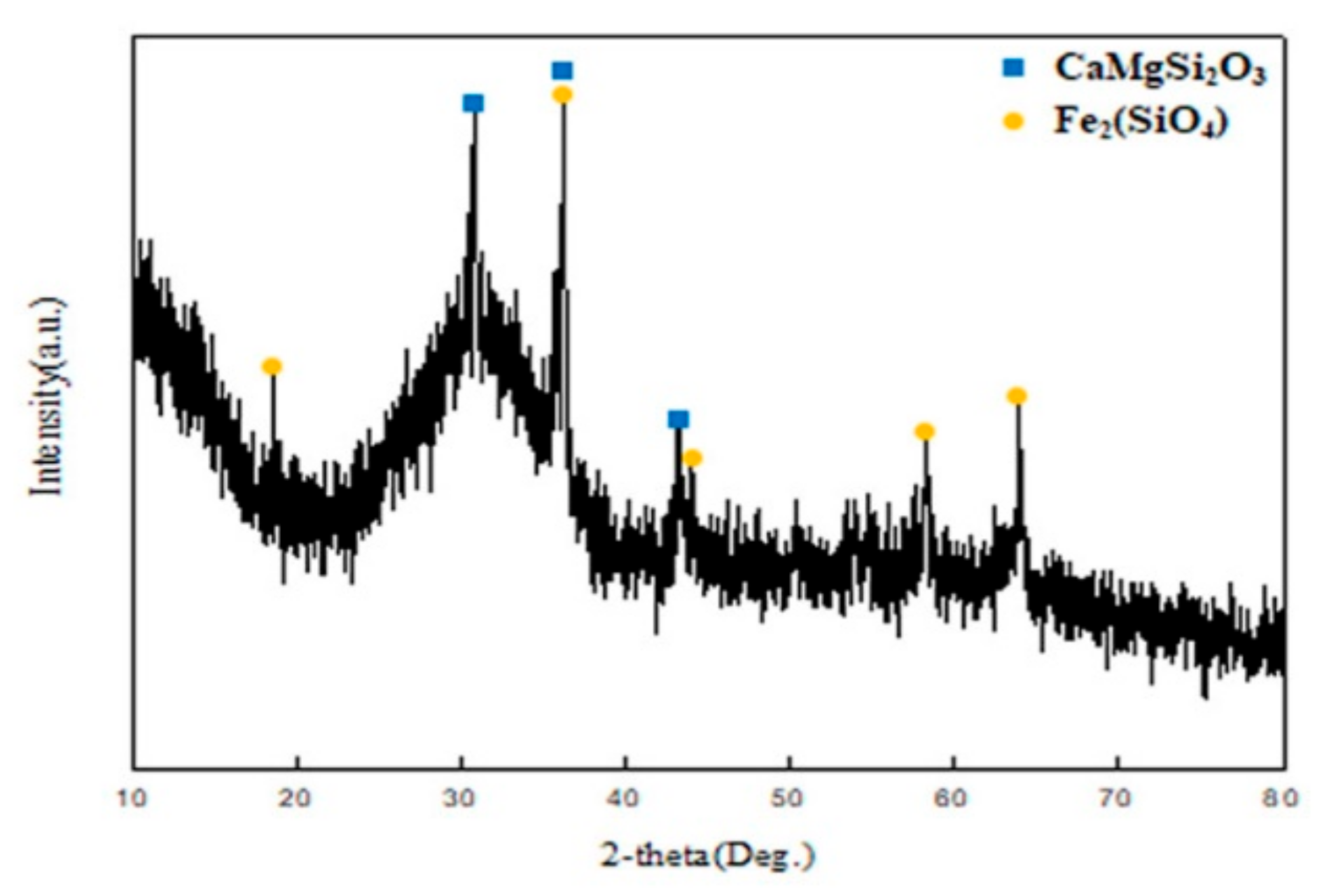
The copper slag used in this study contained 16% CaO. Therefore, it was considered that slag consists of Fe
2SiO
4 (fayalite), Ca
2SiO
4 (calcium orthosilicate), CaFeSiO
4 and Fe
2O
3 [
14,
15]. CaO affects the decomposition of fayalite and reduction of iron oxide, and changes the chemical composition of slag. Copper oxide can be easily reduced and also dissolved into a molten metal phase (as shown reaction 6), and zinc oxide is reduced to a volatile zinc, which is removed from the furnace as fumes, by carbon at a temperature of 980~1000 °C during the reduction process.
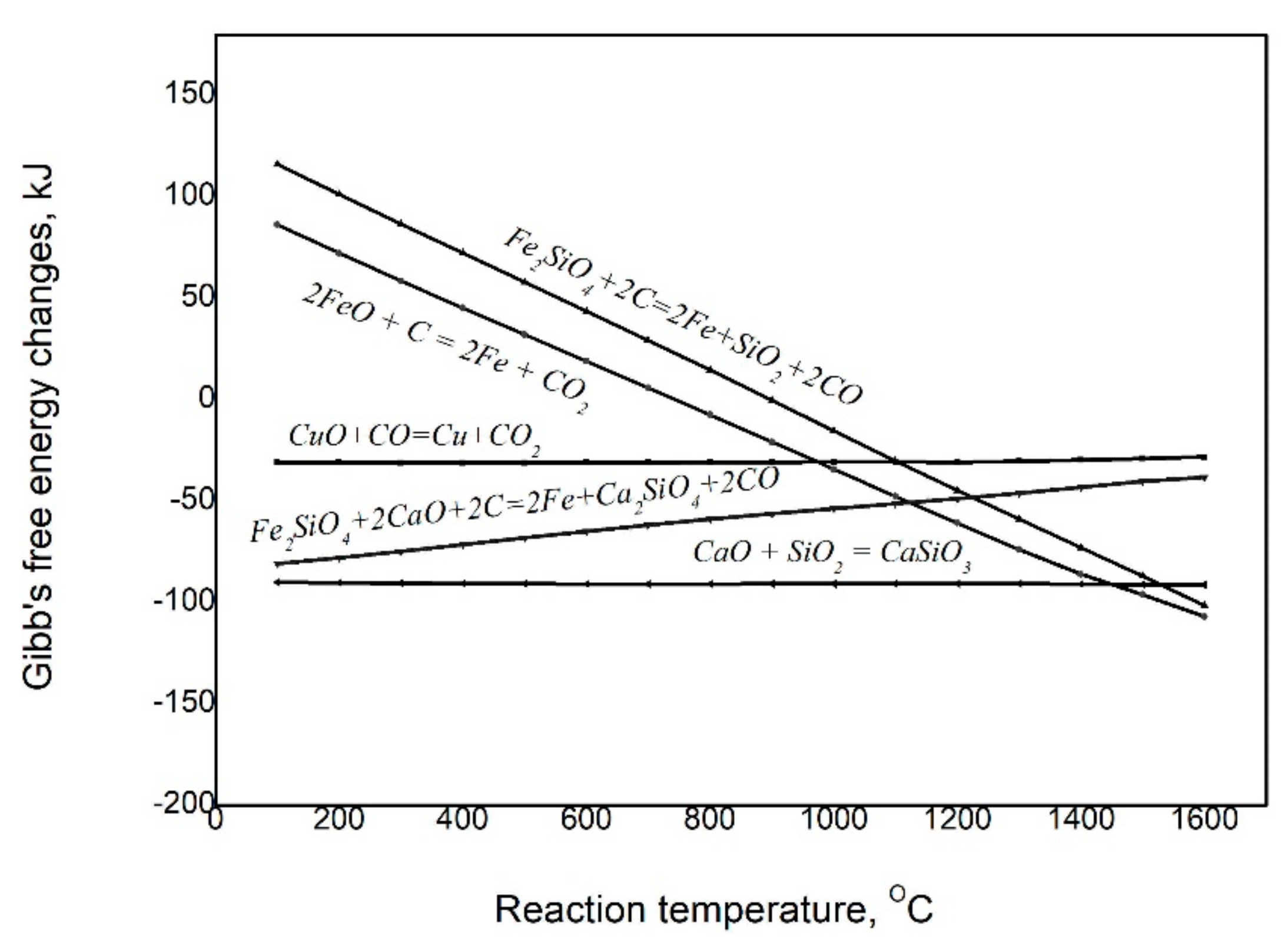
The reduction behavior of iron oxide reduced from fayalite decomposition was examined by thermodynamic analysis using HSC 5.1 chemistry software. It determined that, as in the results of the thermodynamic analysis in
Figure 3, iron can be reduced from the decomposition of fayalite in the reduction smelting process at high temperatures above 1400°C, and moreover, due to the impact of CaO, the reduction of iron in fayalite is more intensified. As illustrated, CuO can be reduced by CO gas at low temperatures, as is shown in
Figure 3.
In the reduction smelting experiment with carbon additives, Fe and Cu completely reduced from the slag and then formed a metal phase, separated by the specific gravity difference with the slag consisting of SiO
2, CaO, Al
2O
3, etc., at 1600 °C for 30 min of holding. Additionally, the reformed slag with a relatively specific gravity floated on the molten pig iron.
Table 2 shows the XRF results of the chemical composition of the reformed slag separated from copper slag by high temperature reduction according to the change of the reaction temperature condition for 30 min of holding. The main constituent elements were measured such as oxides of SiO
2, CaO and Al
2O
3. The iron oxide content in the slag decreased from 24.62% to 16.13% according to the increasing reaction temperature from 1400 °C to 1500 °C. At a reaction temperature of 1600 °C, Fe in the slag could not be measured, indicating complete separation from the slag. The recovery rate of pig iron varied greatly depending on the reaction temperature. As shown in
Table 3, it was found that the Fe
2O
3 content in the slag was decreased according to the increasing reaction time when the reaction time was 30 min, and Fe
2O
3 was not detected, whereas SiO
2, CaO and Al
2O
3 contents increased. The Cu content in the slag was not detected because it was completely dissolved into molten pig iron. Furthermore, ZnO was removed with the furnace gas while smelting of copper slag.
Table 4 shows EDS results of the chemical composition of the pig iron separated from copper smelting slag by high temperature reduction according to the change of the reaction temperature condition. It was confirmed that there was no significant difference according to the changes of the reaction temperature conditions, and it contained about 86% Fe and 3% Cu, indicating the formation of ferrous alloy contained Cu.
Cast iron is widely used in multicomponent ferrous alloy in place of steel at considerable low cost and with optimum combination properties such as castability, machinability, and excellent wear resistance, and has high hardness and high inherent damping capabilities [
19,
20,
21]. It is known that the addition of alloy elements, cooling rate and formation of graphite influence the properties of cast iron. Copper leads to the graphitizing and pearlitization of cast iron, which are phase-stabilizing, which substantially affects mechanical properties such as strength, toughness and corrosion resistance [
19,
20,
21,
22,
23,
24]. Many researchers have studied the effect of Cu addition on the mechanical properties and structure formation of cast iron. Nassef et al. [
19], Agunsove et al. [
20] and Razumakov et al. [
23] concluded that the structure and mechanical properties of cast iron are affected by the addition of 1 to 3.5% copper, which resulted in better wear resistance and the lowest wear loss, and hardness and tensile strength initially increased according to the decreased to volume fraction of graphite and stabilization of pearlite. Cast iron contains such main elements as carbon ranging from 1.8 to 4% and silicon from 1 to 3%, and other alloying elements, and has graphite flakes. In carbothermal reduction smelting, the recovered ferrous alloys appeared as cast iron according to the content-ranges of C and Si, which represented about 4.1% and 4.18%, respectively, in the ferrous alloys recovered from the copper slag. Consequently, it was considered that the recovered ferrous alloy can be used in place of cast iron ingots.
In reduction smelting by solid carbon, iron is completely reduced and forms liquid pig iron at 1600 °C for 30 min. The reformed slag contains about 46% SiO2, 26% CaO and 14% Al2O3, and is an acid slag. Therefore, the next experiment with the addition of CaO was conducted to change the chemical composition of reformed slag with the ratio B = %CaO/%SiO2 = 0.8, 0.9, 1.0, 1.1 and 1.2 at a temperature of 1600 °C for 30 min.
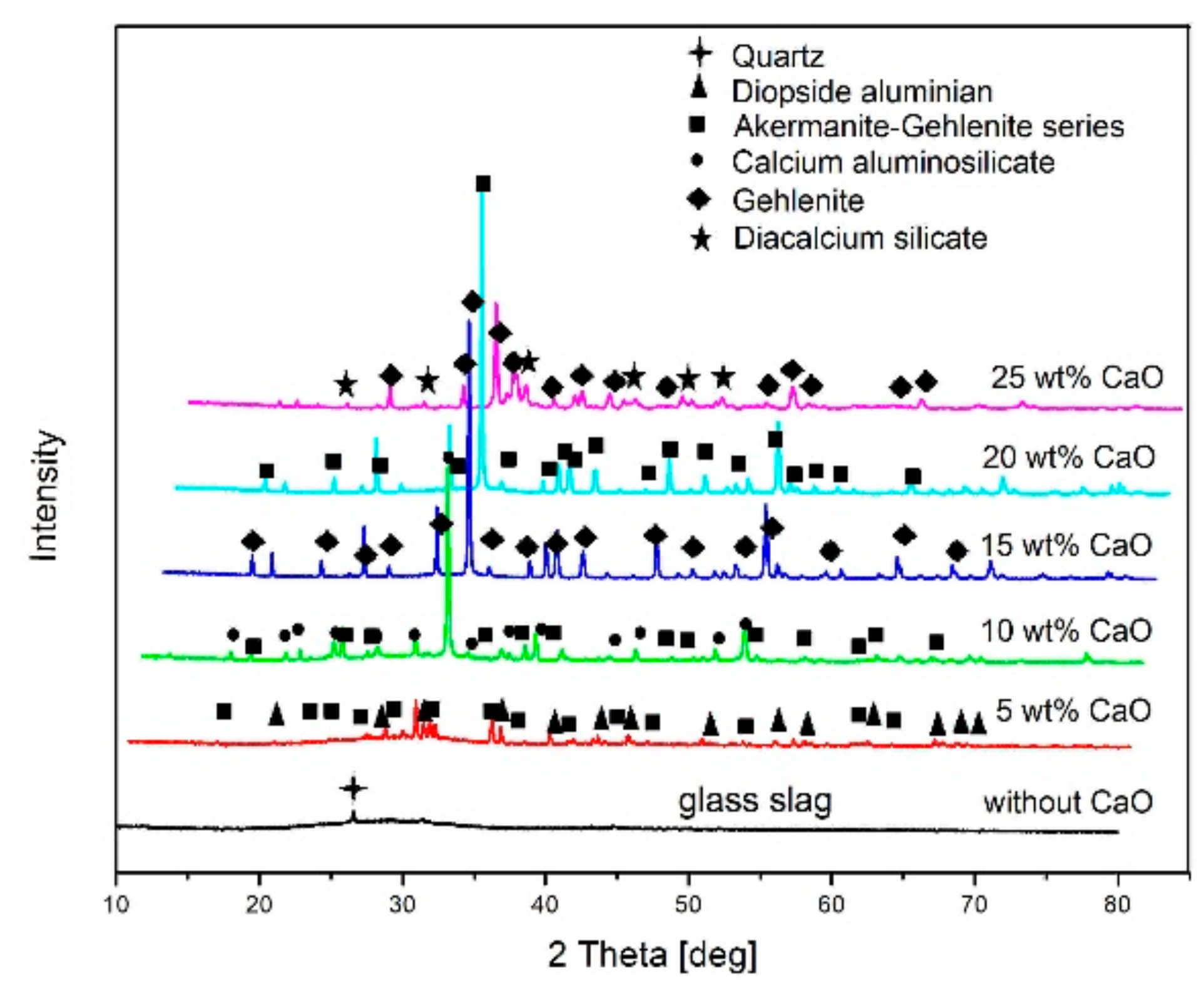
As with reduction smelting with addition of CaO, the fayalite (2FeO·SiO
2) in the copper slag is decomposed in a high temperature and reduction process, and iron oxide is reduced and separated from the liquid pig iron according to reactions (1) to (5) in order. In this case, it might be seen that the remaining oxides such as the SiO
2, CaO, Al
2O
3 and MgO interact with each other, and hence generate the reformed slag. This reformed slag might consist of typically eutectic compounds such as CaO·SiO
2, 2CaO·SiO
2, 3CaO·2SiO
2, CaO·Al
2O
3·2SiO
2, 2CaO·Al
2O
3·SiO
2, CaO·MgO·SiO
2, etc. [
25,
26]. The basic slag with a high content of CaO transforms into calcium silicates slag by the addition of CaO, and hence changing the chemical compositions of the slag may be effectively conducted in the reducing process. The chemical composition of the slag reformed from copper slag in the reduction smelting with addition of CaO was determined by X-ray fluorescence (XRF) to evaluate the difference according to the addition of CaO, as shown in
Table 5. As the XRF results show, it was found that the SiO
2 content in the slag decreased according to the increase in CaO content.
Table 6 shows EDS results of the chemical composition of the pig iron separated from copper smelting slag by high temperature reduction according to that of the addition of CaO. It was again confirmed that there was no significant difference according to the addition of CaO, and it contained about 86% of Fe and 3% of Cu, indicating the formation of ferrous alloy contained Cu.
Portland cement is manufactured with a closely controlled chemical combination of Ca, Si, Al, Fe and other ingredients, and common materials for cement include limestone, shells, and chalk or marl combined with shale, clay, blast furnace slag, silica sand, and iron ore. Blast furnace slag (BFS) is one of the massive raw materials acquired through Portland blast furnace slag cement (PBFSC) because BFS has a similar chemical composition with the chemical composition of Portland cement [
27,
28,
29,
30]. The general chemical composition of BFS is about 32 to 40% SiO
2, 32 to 50% CaO, 7 to 16% Al
2O
3 and 5 to 10% MgO, etc. For the manufacture of Portland cement, limestone as the principal raw material is burned at a high temperature causing air pollution, which creates large CO
2 emissions, therefore, BFS was discovered a partial replacement in raw material of Portland cement as PBFSC could be a kind of eco-friendly cement that emits about 40% less CO
2 than Portland cement supported by the Green Purchasing law [
29,
30]. South Korea has also used BFS as a replacement for the raw material of Portland cement and it is widely employed in civil engineering structures due to its eco-friendly processing and lower cost of PBFSC. The qualities of PBFSC are specified in KS L 5210: Blast furnace slag cement of Korean standard has three types such as 1 type is over 5, less than 30 of BFS ratio, 2 types (over 30, less than 60) and 3 type (over 60, less than 70). In 2017, the BFS has been generated about 16 million tons a year, and 92% of BFS used in cement industry [
31].
In the present study, it was observed that reformed slag from copper slag for iron recovery was similar to BFS. When the addition of CaO are up to 15%, the chemical composition of reformed slag is to that of BFS. Chemical analysis of both the produced slag and BFS showed that major oxides such as CaO, SiO
2, MgO and Al
2O
3 comprised about 95% of the total in accordance with the requirements of BFS as cement material, as shown in
Table 5.