Optical Sensing Approach to the Recognition of Different Types of Particulate Matters for Sustainable Indoor Environment Management
Abstract
1. Introduction
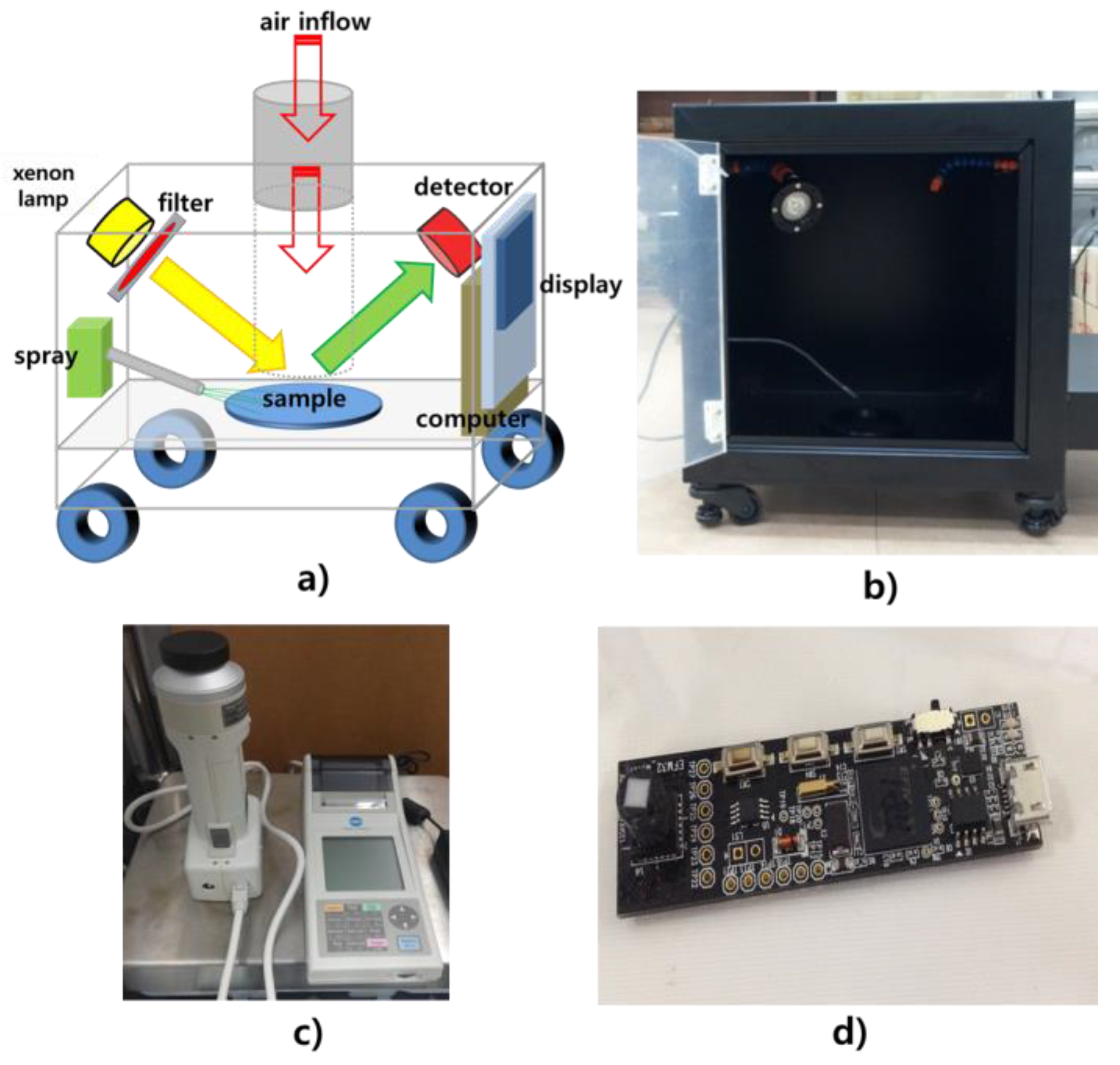
2. Materials and Methods
3. Results
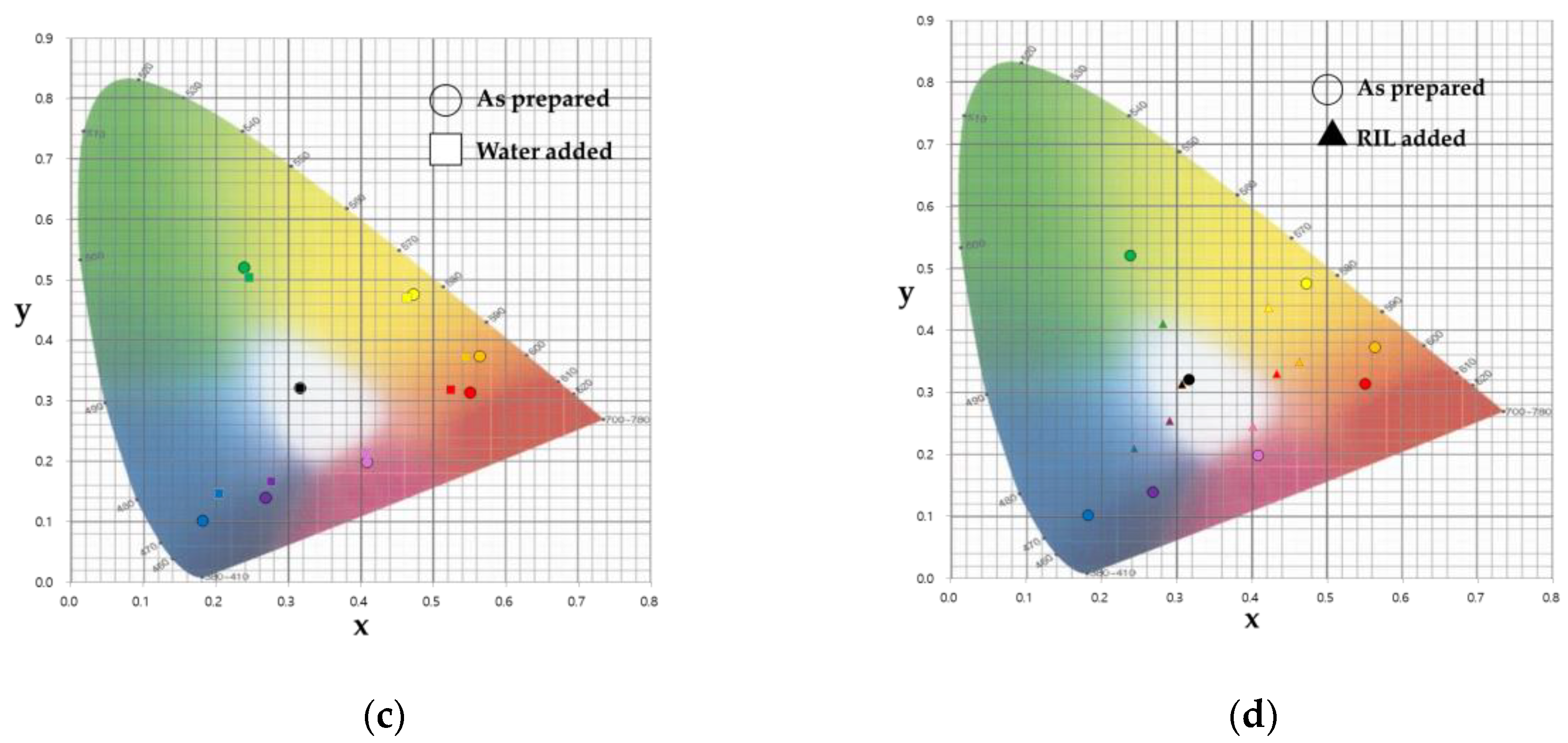
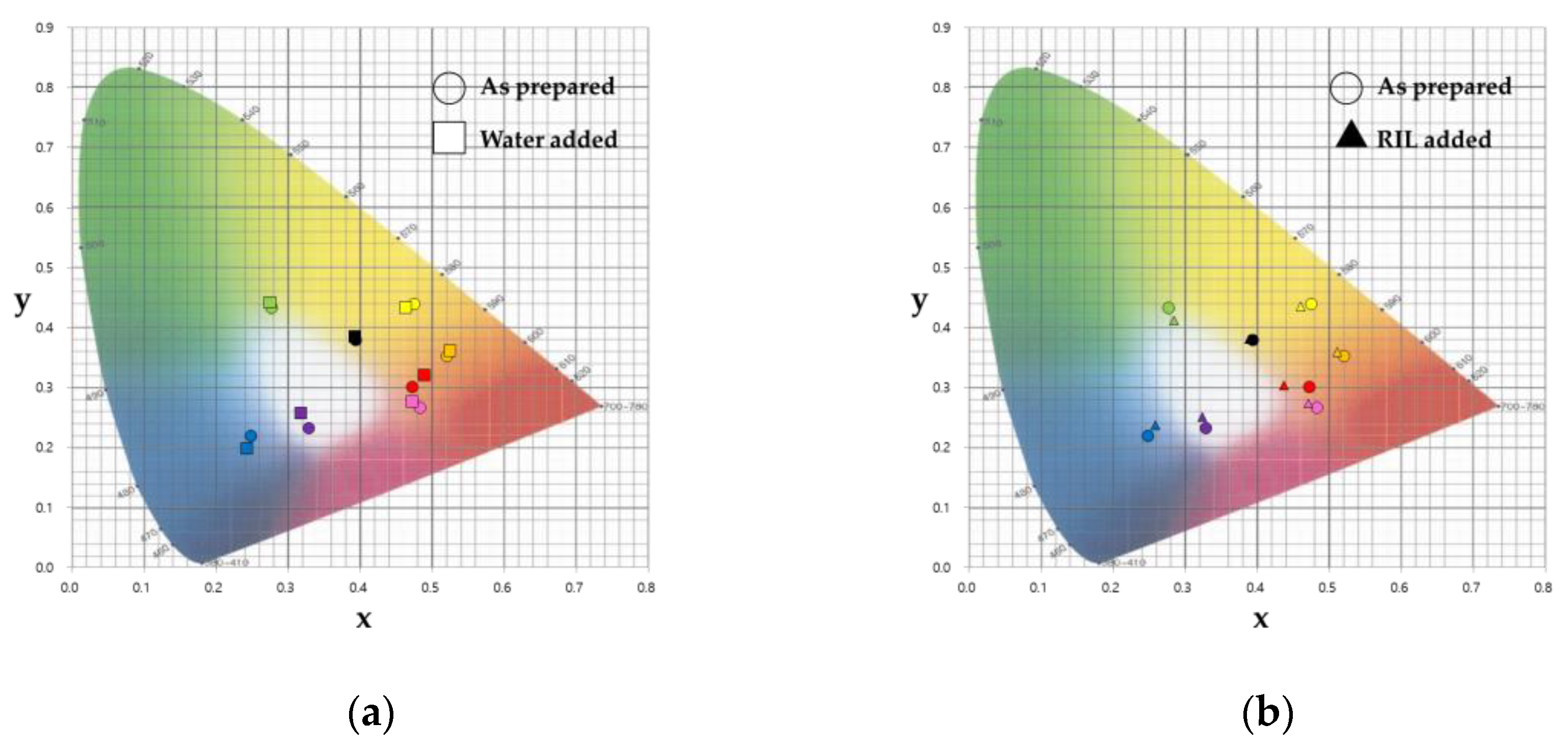
3.1. Color Detection
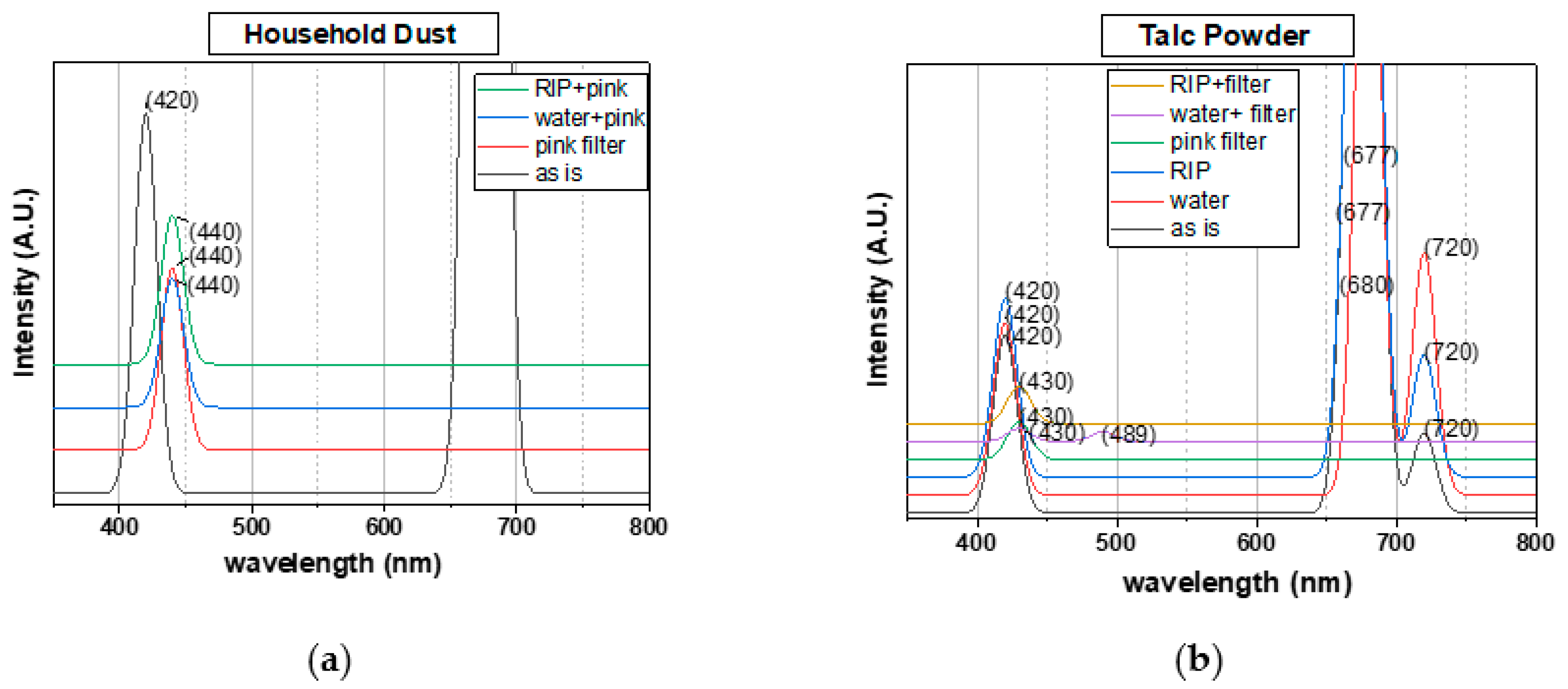
3.2. Light Spectrum Detection
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Biliaiev, M.M.; Kharytonov, M.M. Numerical Simulation of Indoor Air Pollution and Atmosphere Pollution for Regions Having Complex Topography. In Air Pollution Modeling and Its Application XXI; Springer: Berlin/Heidelberg, Germany, 2011; pp. 87–91. [Google Scholar]
- Chafe, Z.; Brauer, M.; Klimont, Z.; Dingenen, R.V.; Mehta, S.; Rao, S.; Riahi, K.; Dentener, F.; Smith, K.R. Household cooking with solid fuels contributes to ambient PM2.5 air pollution and the burden of disease. Environ. Health Perspect. 2014, 12, 1314–1320. [Google Scholar] [CrossRef]
- Afshari, A.; Matson, U.; Ekberg, L.E. Characterization of indoor sources of fine and ultrafine particles: A study conducted in a full-scale chamber. Indoor Air 2005, 15, 141–150. [Google Scholar] [CrossRef]
- Colome, S.; Kado, N.; Jaques, P.; Kleinman, M. Indoor-outdoor air pollution relations: Particulate matter less than 10 μm in aerodynamic diameter (PM10) in homes of asthmatics. Atmos. Environ. Part A Gen. Top. 1992, 26, 2173–2178. [Google Scholar] [CrossRef]
- Tasić, V.; Jovašević-Stojanović, M.; Vardoulakis, S.; Milošević, N.; Kovačević, R.; Petrović, J. Comparative assessment of a real-time particle monitor against the reference gravimetric method for PM10 and PM2.5 in indoor air. Atmos. Environ. 2012, 54, 358–364. [Google Scholar] [CrossRef]
- Kelly, K.E.; Whitaker, J.; Petty, A.; Widmer, C.; Dybwad, A.; Sleeth, D.; Martin, R.; Butterfield, A. Ambient and laboratory evaluation of a low-cost particulate matter sensor. Environ. Pollut. 2017, 211, 491–500. [Google Scholar] [CrossRef]
- Vázquez, L.; Zorzano, M.; Jimenez, S. Spectral information retrieval from integrated broadband photodiode martian ultraviolet measurements. Opt. Lett. 2007, 32, 2596–2598. [Google Scholar] [CrossRef][Green Version]
- Li, H.; Sang, X. LED array light source illuminance distribution and photoelectric detection performance analysis in dust concentration testing system. Sens. Actuators A Phys. 2018, 271, 111–117. [Google Scholar] [CrossRef]
- Vaca-Oyola, L.S.; Marín, E.; Rojas-Trigos, J.B.; Cifuentes, A.; Cabrera, H.; Alvarado, S.; Cedeño, E.; Calderón, A.; Delgado-Vasallo, O.A. Liquids refractive index spectrometer. Sens. Actuators B Chem. 2016, 229, 249–256. [Google Scholar] [CrossRef]
- Shin, S.; Jung, C.; Kim, Y. Analysis of the measurement difference for the PM10 concentrations between beta-ray absorption and gravimetric methods at gosan. Aerosol Air Qual. Res. 2011, 11, 846–853. [Google Scholar] [CrossRef]
- Sówka, I.; Chlebowska-Sty’s, A.; Pachurka, Ł.; Rogula-Kozłowska, W.; Mathews, B. Analysis of particulate matter concentration variability and origin in selected urban areas in Poland. Sustainability 2019, 11, 5735. [Google Scholar] [CrossRef]
- Jo, E.J.; Lee, W.S.; Jo, H.Y.; Kim, C.H.; Eom, J.S.; Mok, J.H.; Kim, M.H.; Lee, K.; Kim, K.U.; Lee, M.K.; et al. Effects of particulate matter on respiratory disease and the impact of meteorological factors in Busan, Korea. Respir. Med. 2017, 124, 79–87. [Google Scholar] [CrossRef]
- Zhang, K.; Shang, X.; Herrmann, H.; Meng, F.; Mo, Z.; Chen, J.; Lv, W. Approaches for identifying PM2.5 source types and source areas at a remote background site of South China in spring. Sci. Total Environ. 2019, 691, 1320–1327. [Google Scholar] [CrossRef]
- Ahn, H.; Jung, B.K.; Joo, J.C.; Park, J.R. Spectral sensing of asbestos according to concentration in various asbestos containing materials. Appl. Mech. Mater. 2014, 627, 7–11. [Google Scholar] [CrossRef]
- Mortimer, R.J.; Varley, T.S. Quantification of colour stimuli through the calculation of CIE chromaticity coordinates and luminance data for application to in situ colorimetry studies of electrochromic materials. Displays 2011, 32, 35–44. [Google Scholar] [CrossRef]
- Dang, R.Y.; Liu, Y.; Liu, G. Chromaticity changes of inorganic pigments in Chinese traditional paintings due to the illumination of frequently-used light sources in museum. Color Res. Appl. 2018, 43, 596–605. [Google Scholar] [CrossRef]
- Steuber, F.; Staudigel, J.; Stössel, M.; Simmerer, J.; Winnacker, A.; Spreitzer, H.; Weissörtel, F.; Salbeck, J. White light emission from organic LEDs utilizing spiro compounds with high-temperature stability. Adv. Mater. 2000, 12, 130–133. [Google Scholar] [CrossRef]
- Ferrer, E.; Alegría, A.; Farré, R.; Clemente, G.; Calvo, C. Fluorescence, browning index and color in infant formulas during storage. J. Agric. Food Chem. 2005, 53, 4911–4917. [Google Scholar] [CrossRef]


| Chromaticity Values | Chromaticity Values | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Cellophane | Y | x | y | Sample | Cellophane | Y | x | y | ||
| Household Dust | As prepared | - | 13.40 | 0.3229 | 0.3244 | Soil Powder | As prepared | - | 49.15 | 0.3576 | 0.3581 |
| Red | 4.87 | 0.4216 | 0.3307 | Red | 7.63 | 0.4914 | 0.3031 | ||||
| Orange | 8.01 | 0.4625 | 0.3576 | Orange | 18.21 | 0.5472 | 0.3697 | ||||
| Yellow | 13.47 | 0.4230 | 0.4305 | Yellow | 34.44 | 0.4687 | 0.4546 | ||||
| Green | 5.08 | 0.2923 | 0.3853 | Green | 8.84 | 0.2617 | 0.4733 | ||||
| Blue | 3.16 | 0.2516 | 0.2058 | Blue | 4.71 | 0.2227 | 0.1778 | ||||
| Pink | 9.91 | 0.4098 | 0.2624 | Pink | 16.94 | 0.4496 | 0.2458 | ||||
| Violet | 4.40 | 0.2970 | 0.2686 | Violet | 3.46 | 0.3027 | 0.1933 | ||||
| Water Added | - | 5.18 | 0.3190 | 0.3208 | Water Added | - | 6.25 | 0.3824 | 0.3670 | ||
| Red | 4.43 | 0.3869 | 0.3357 | Red | 4.56 | 0.3970 | 0.3341 | ||||
| Orange | 6.16 | 0.4197 | 0.3508 | Orange | 6.67 | 0.4332 | 0.3524 | ||||
| Yellow | 9.02 | 0.3955 | 0.4066 | Yellow | 9.91 | 0.4103 | 0.4069 | ||||
| Green | 4.41 | 0.3025 | 0.3586 | Green | 4.17 | 0.3002 | 0.3711 | ||||
| Blue | 3.11 | 0.2692 | 0.2320 | Blue | 3.09 | 0.2732 | 0.2384 | ||||
| Pink | 8.52 | 0.4081 | 0.2856 | Pink | 8.09 | 0.4349 | 0.2858 | ||||
| Violet | 4.35 | 0.3008 | 0.2851 | Violet | 4.32 | 0.3054 | 0.2890 | ||||
| Refractive Index Liquid Added | - | 3.48 | 0.3163 | 0.3177 | Refractive Index Liquid Added | - | 22.23 | 0.3818 | 0.3749 | ||
| Red | 4.37 | 0.3768 | 0.3369 | Red | 5.57 | 0.4647 | 0.3254 | ||||
| Orange | 5.87 | 0.3859 | 0.3305 | Orange | 11.02 | 0.5044 | 0.3627 | ||||
| Yellow | 8.17 | 0.3756 | 0.3864 | Yellow | 19.05 | 0.4484 | 0.4310 | ||||
| Green | 3.92 | 0.3021 | 0.3635 | Green | 5.67 | 0.2814 | 0.4245 | ||||
| Blue | 4.29 | 0.2800 | 0.2699 | Blue | 3.24 | 0.2475 | 0.2012 | ||||
| Pink | 7.38 | 0.4170 | 0.2834 | Pink | 12.04 | 0.4501 | 0.2742 | ||||
| Violet | 3.39 | 0.3080 | 0.2797 | Violet | 3.34 | 0.3145 | 0.2454 | ||||
| Chromaticity Values | Chromaticity Values | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Cellophane | Y | x | y | Sample | Cellophane | Y | x | y | ||
| Talc Powder | As prepared | - | 74.79 | 0.3127 | 0.3191 | Gypsum Powder | As prepared | - | 79.97 | 0.3157 | 0.3213 |
| Red | 7.53 | 0.4932 | 0.3040 | Red | 8.52 | 0.5498 | 0.3143 | ||||
| Orange | 22.17 | 0.5568 | 0.3732 | Orange | 24.57 | 0.5630 | 0.3738 | ||||
| Yellow | 50.31 | 0.4682 | 0.4770 | Yellow | 53.23 | 0.4719 | 0.4763 | ||||
| Green | 12.40 | 0.2337 | 0.5337 | Green | 12.90 | 0.2382 | 0.5213 | ||||
| Blue | 3.84 | 0.1888 | 0.1134 | Blue | 3.99 | 0.1816 | 0.1021 | ||||
| Pink | 20.73 | 0.4025 | 0.2020 | Pink | 20.89 | 0.4078 | 0.1989 | ||||
| Violet | 4.57 | 0.2687 | 0.1634 | Violet | 3.74 | 0.2681 | 0.1400 | ||||
| Water Added | - | 47.39 | 0.3136 | 0.3205 | Water Added | - | 19.09 | 0.3065 | 0.3134 | ||
| Red | 6.69 | 0.4999 | 0.3211 | Red | 5.07 | 0.4332 | 0.3300 | ||||
| Orange | 15.50 | 0.5189 | 0.3610 | Orange | 9.48 | 0.4626 | 0.3495 | ||||
| Yellow | 32.18 | 0.4565 | 0.4669 | Yellow | 16.81 | 0.4224 | 0.4363 | ||||
| Green | 8.73 | 0.2529 | 0.4859 | Green | 6.00 | 0.2818 | 0.4111 | ||||
| Blue | 3.61 | 0.2012 | 0.1323 | Blue | 4.44 | 0.2431 | 0.2094 | ||||
| Pink | 14.18 | 0.4069 | 0.2127 | Pink | 10.71 | 0.4011 | 0.2453 | ||||
| Violet | 3.57 | 0.2783 | 0.1835 | Violet | 4.35 | 0.2906 | 0.2543 | ||||
| Refractive Index Liquid Added | - | 15.30 | 0.3225 | 0.3297 | Refractive Index Liquid Added | - | 56.70 | 0.3159 | 0.3217 | ||
| Red | 4.91 | 0.4221 | 0.3310 | Red | 7.34 | 0.5239 | 0.3181 | ||||
| Orange | 8.70 | 0.4525 | 0.3457 | Orange | 18.51 | 0.5450 | 0.3724 | ||||
| Yellow | 14.58 | 0.4276 | 0.4370 | Yellow | 37.61 | 0.4629 | 0.4703 | ||||
| Green | 5.23 | 0.2828 | 0.4115 | Green | 9.90 | 0.2457 | 0.5043 | ||||
| Blue | 3.21 | 0.2443 | 0.1962 | Blue | 4.91 | 0.2043 | 0.1460 | ||||
| Pink | 7.38 | 0.4170 | 0.2834 | Pink | 17.27 | 0.4064 | 0.2135 | ||||
| Violet | 3.39 | 0.3080 | 0.2797 | Violet | 3.54 | 0.2757 | 0.1664 | ||||
| Chromaticity Values | Chromaticity Values | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | Cellophane | Y | x | y | Sample | Cellophane | Y | x | y | ||
| Pine tree Pollen | As prepared | - | 32.77 | 0.3938 | 0.3796 | Pine tree Pollen | Water Added | - | 28.76 | 0.3926 | 0.3846 |
| Red | 6.79 | 0.4714 | 0.3017 | Red | 6.28 | 0.4887 | 0.3205 | ||||
| Orange | 14.02 | 0.5200 | 0.3524 | Orange | 12.88 | 0.5247 | 0.3614 | ||||
| Yellow | 24.74 | 0.4745 | 0.4402 | Yellow | 23.08 | 0.4626 | 0.4336 | ||||
| Green | 6.84 | 0.2764 | 0.433 | Green | 6.30 | 0.2746 | 0.4422 | ||||
| Blue | 4.52 | 0.2479 | 0.2204 | Blue | 3.25 | 0.2432 | 0.1983 | ||||
| Pink | 13.51 | 0.4823 | 0.2669 | Pink | 13.54 | 0.4722 | 0.2769 | ||||
| Violet | 3.40 | 0.3286 | 0.2329 | Violet | 4.42 | 0.3174 | 0.2579 | ||||
| Refractive Index Liquid Added | - | 22.62 | 0.3892 | 0.3804 | Refractive Index Liquid Added | Green | 5.88 | 0.2844 | 0.4117 | ||
| Red | 6.06 | 0.4374 | 0.3039 | Blue | 4.39 | 0.2589 | 0.2374 | ||||
| Orange | 11.17 | 0.5106 | 0.3595 | Pink | 11.28 | 0.4708 | 0.2735 | ||||
| Yellow | 19.14 | 0.4601 | 0.4351 | Violet | 3.35 | 0.3240 | 0.2506 | ||||
| Sample Conditions | As Prepared | Water | Refractive Index Liquid | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Filter | Measurement | Materials | |||||||||
| No filter | Peak Intensity | Low | High | Other | Low | High | Other | Low | High | Other | |
| Peak Positions (nm) | Pine tree pollen | 421 | 681 | 601 | 421 | 681 | 591 | 421 | 676 | 721 | |
| Soil | 421 | 681 | 597 | 421 | 677 | 721 | 421 | 681 | - | ||
| Household Dust | 420 | 678 | - | 420 | 677 | 720 | 420 | 677 | - | ||
| Talc | 420 | 677 | 720 | 420 | 680 | 720 | 420 | 677 | 720 | ||
| Gypsum | 420 | 679 | - | 433 | 691 | - | 433 | 690 | - | ||
| Pink filter | Peak Intensity | Low | High | Other | Low | High | Other | Low | High | Other | |
| Peak Positions (nm) | Household Dust | 440 | - | - | 440 | - | - | 440 | - | - | |
| Talc | 430 | - | - | 430 | 490 | - | 430 | - | - | ||
| Gypsum | 439 | 820 | - | 453 | - | - | 453 | - | - | ||
| Sample Conditions | As Prepared | Water | Refractive Index Liquid | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Filter | Measurement | Materials | |||||||||
| No filter | Peak Intensity | Low | High | Other | Low | High | Other | Low | High | Other | |
| Peak Intensity Ratio | Pine tree pollen | 1 | 10.46 | 0.98 | 1 | 8.75 | 0.14 | 1 | 5.86 | 0.81 | |
| Soil | 1 | 8.51 | 0.14 | 1 | 4.61 | 0.41 | 1 | 8.12 | - | ||
| Household Dust | 1 | 9.31 | - | 1 | 6.31 | 0.44 | 1 | 9.56 | - | ||
| Talc | 1 | 6.32 | 0.45 | 1 | 4.51 | 1.41 | 1 | 6.21 | 0.68 | ||
| Gypsum | 1 | 9.63 | - | 1 | 9.65 | - | 1 | 9.71 | - | ||
| Pink filter | Peak in Intensity | Low | High | Other | Low | High | Other | Low | High | Other | |
| Peak Intensity Ratio | Household Dust | 0.48 | - | - | 0.30 | - | - | 0.39 | - | - | |
| Talc | 0.21 | - | - | 0.08 | 0.06 | - | 0.21 | - | - | ||
| Gypsum | 0.36 | 0.13 | - | 0.40 | - | - | 0.44 | - | - | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahn, H.; Kang, J.S.; Choi, G.-S.; Choi, H.-J. Optical Sensing Approach to the Recognition of Different Types of Particulate Matters for Sustainable Indoor Environment Management. Sustainability 2020, 12, 10568. https://doi.org/10.3390/su122410568
Ahn H, Kang JS, Choi G-S, Choi H-J. Optical Sensing Approach to the Recognition of Different Types of Particulate Matters for Sustainable Indoor Environment Management. Sustainability. 2020; 12(24):10568. https://doi.org/10.3390/su122410568
Chicago/Turabian StyleAhn, Hosang, Jae Sik Kang, Gyeong-Seok Choi, and Hyun-Jung Choi. 2020. "Optical Sensing Approach to the Recognition of Different Types of Particulate Matters for Sustainable Indoor Environment Management" Sustainability 12, no. 24: 10568. https://doi.org/10.3390/su122410568
APA StyleAhn, H., Kang, J. S., Choi, G.-S., & Choi, H.-J. (2020). Optical Sensing Approach to the Recognition of Different Types of Particulate Matters for Sustainable Indoor Environment Management. Sustainability, 12(24), 10568. https://doi.org/10.3390/su122410568





