Eco-Friendly Yield and Greenhouse Gas Emissions as Affected by Fertilization Type in a Tropical Smallholder Rice System, Ghana
Abstract
1. Introduction
2. Materials and Methods
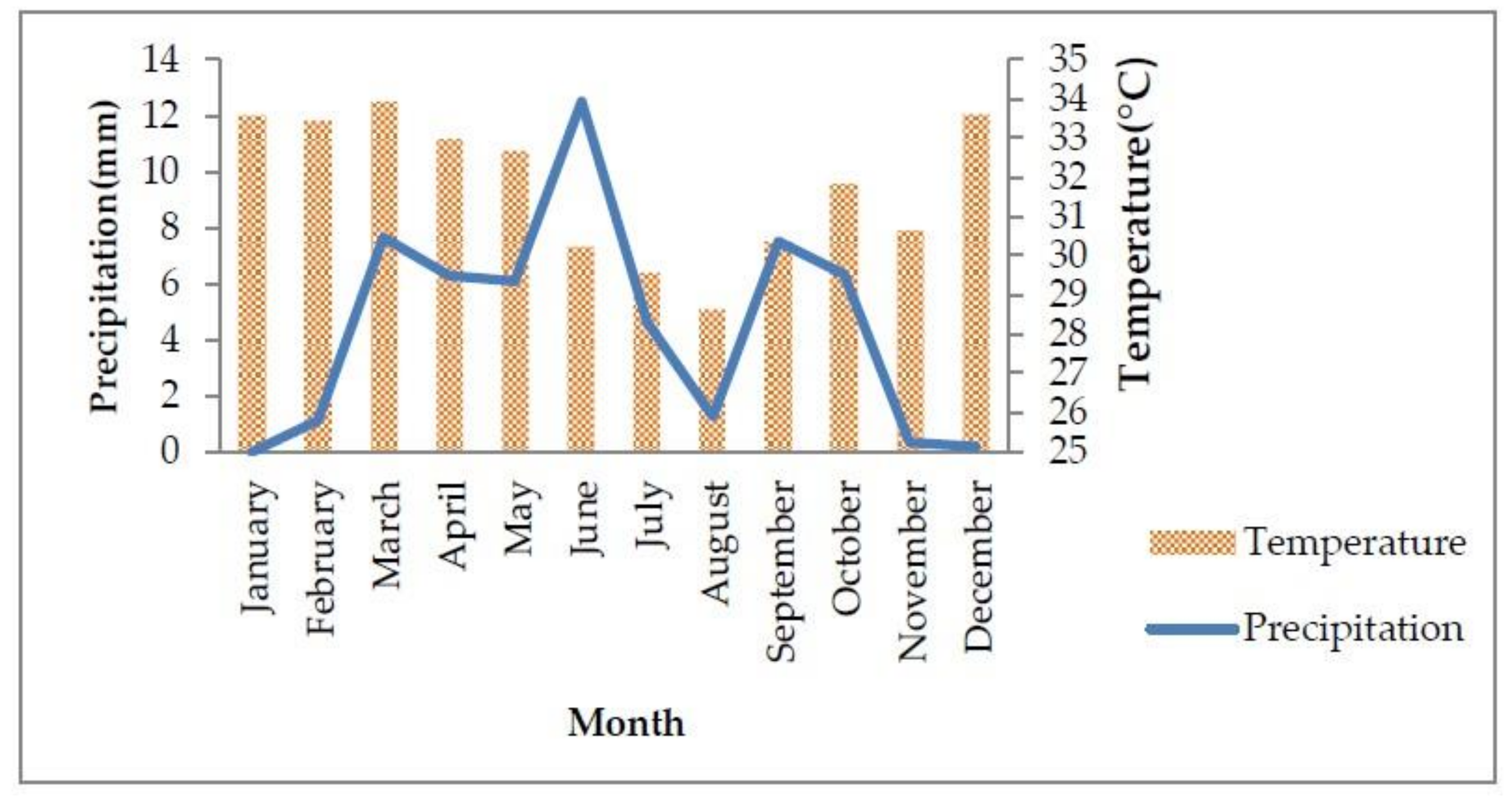
2.1. Study Area and Experimental Design
2.2. Fertilisation Types
- Farmer Practice (BAU): This treatment represented the conventional means by which smallholder farmers cultivated rice in the study area. It was designated as Business as Usual (BAU) and the fertilizer application rate under this treatment was 300 kgha−1 of NPK as base and 50 kgha−1 urea (46%N) top-up.
- Biochar (Bio) + Farmer Practice (BAU + Bio): the treatment represented Biochar and BAU soil amendment. A locally made ESLA stove made from a used lube barrel was used to char the rice straw under anoxic conditions. Biochar was applied at 28,000 kg (28t) per hectare. In rice production, 28 tha−1 and 48 tha−1 represents 50% and 100% of rice straw produced in a single season, respectively. These two rates represent low and high amendment rates. An assumption was therefore made that, should a rice farmer convert at least 50% of his straw into Biochar and incorporate it on his farm, what will the effect be on yield and GHG emissions.
- Poultry Manure (M) + Farmer Practice (BAU + M): For poultry manure, 1 tha−1, 2 tha−1, and 3 tha−1 represents low, medium, and high application rates, respectively. The high application rate of 3 tha−1 was selected for the current work.
- Biochar + Poultry Manure + Farmer Practice (BAU + Bio + M): The fourth treatment was a combination of farmer practice, poultry manure and Biochar at the same application level as the single treatments.
- Control treatment (CT): The control treatment did not have any soil amendments.
2.3. Greenhouse Gas (GHG) Sample Collection
2.4. GHG Sample Analysis
2.5. Flux Estimation and Statistical Analysis
- Model 1
- < −lmer (Parameter ~ Treatments + (1|Reps), data = Flux, REML = FALSE)
- Model 2
- < −aov (Parameter ~ Treatments, data = Flux) key: Parameter = N2O or CH4.
2.6. Global Warming Potential (GWP) and Greenhouse Gas Intensity (GHGI)
3. Results
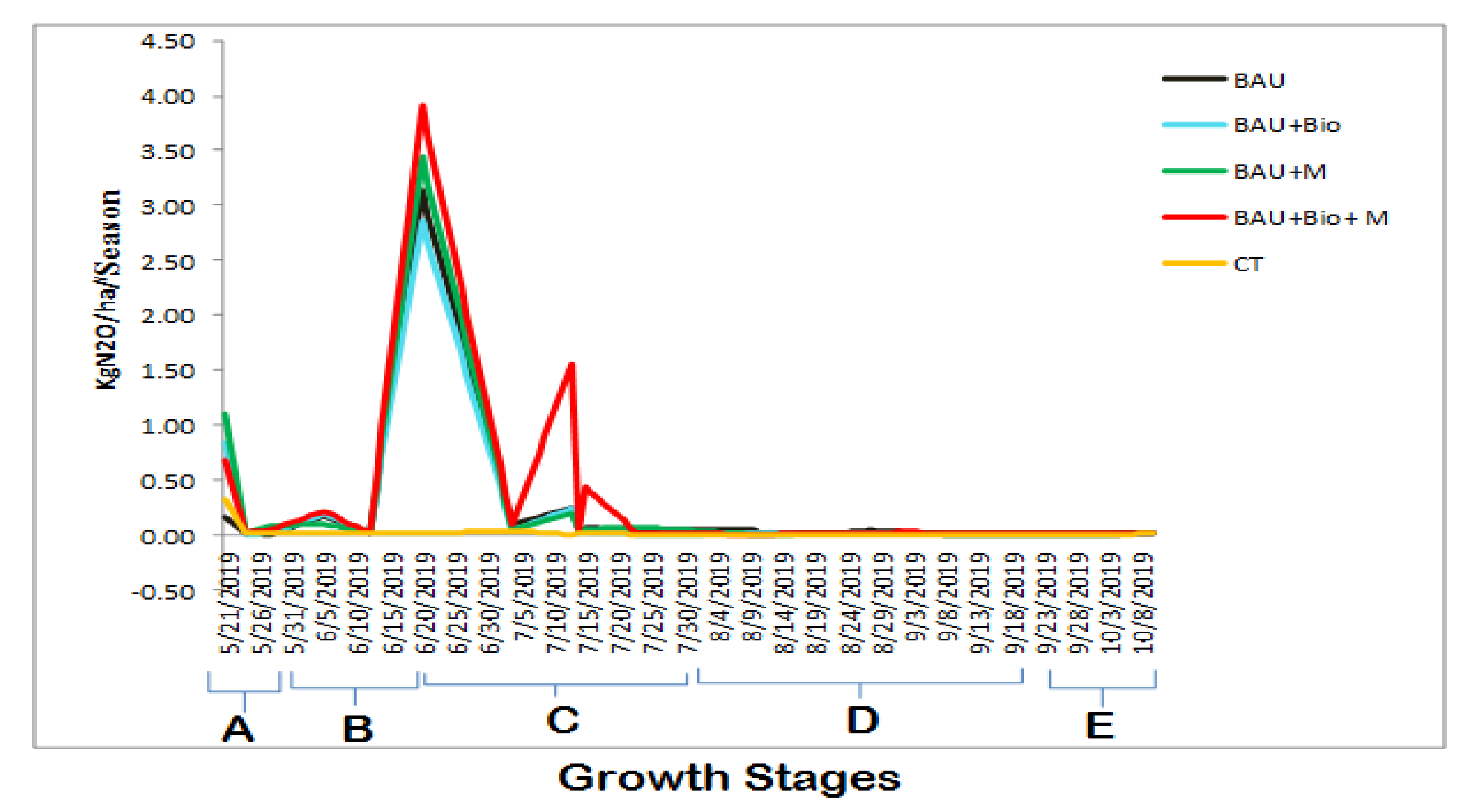
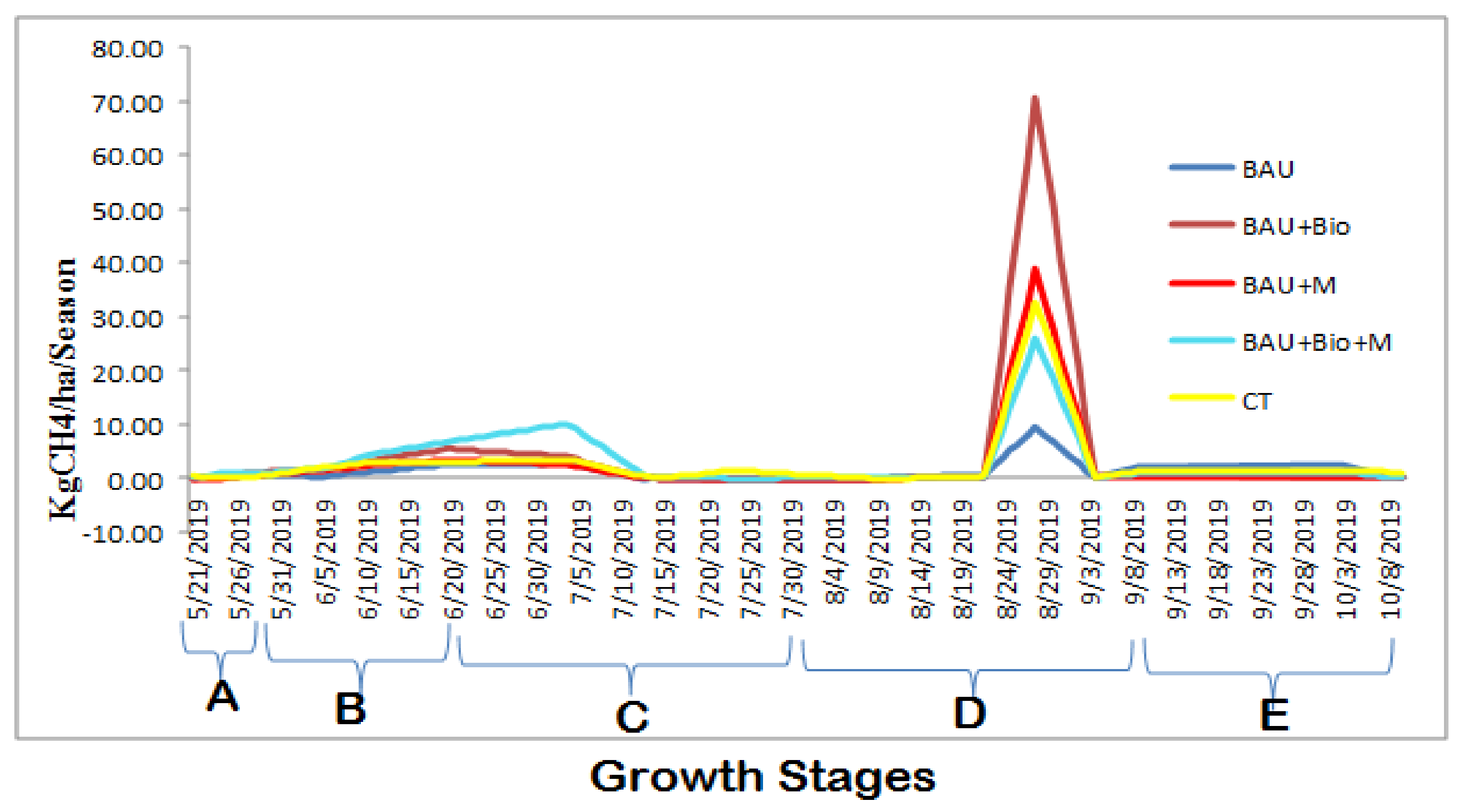
3.1. Average Daily Methane (CH4) and Nitrous Oxide (N2O) Emissions
3.2. Seasonal Cumulative Methane (CH4) and Nitrous Oxide (N2O) Emissions
3.3. Global Warming Potential, Yield and Greenhouse Gas Intensity
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Labels | pH 1:2.5 | % | % Total | % Organic | Exchangeable Cations cmol/kg | cmol/kg | cmol/kg | cmol/kg | % | AVi.BRAYS | Mechanical Analysis | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| O.C | NITROGEN | MATTER | Ca | Mg | K | Na | T.E.B | EX. ACIDITY | ECEC | Base Sat. | ppmP | % Sand | % Silt | % Clay | ||
| Soil Parameters Before Study | 5.442 | 1.37 | 0.12 | 2.36 | 2.96 | 1.32 | 0.99 | 0.05 | 5.32 | 0.74 | 6.05 | 86.84 | 1.40 | 48.00 | 32.40 | 19.60 |
| Soil Parameters After Harvesting | ||||||||||||||||
| BAU(T1) | 6.84 | 0.88 | 0.0532 | 1.51 | 8 | 1.4 | 0.495 | 0.0782 | 46.96 | 31.28 | 21.76 | |||||
| BAU+Bio(T2) | 6.8 | 0.88 | 0.0504 | 1.51 | 7 | 3.4 | 2.027 | 0.15 | 58.38 | 19.50 | 22.12 | |||||
| BAU+M(T3) | 6.72 | 0.84 | 0.0504 | 1.44 | 5.6 | 3.2 | 0.561 | 0.12 | 49.32 | 28.92 | 21.76 | |||||
| BAU+M+Bio(T4) | 6.77 | 1.08 | 0.049 | 1.86 | 7.4 | 1.2 | 2.093 | 0.15 | 50.96 | 27.64 | 21.40 | |||||
| Control(T5) | 6.77 | 0.6 | 0.0532 | 1.03 | 5.4 | 2.6 | 0.561 | 0.15 | 76.40 | 9.28 | 14.32 |
References
- Khosa, M.K.; Sidhu, B.S.; Benbi, D.K. Effect of organic materials and rice cultivars on methane emission from rice field. J. Environ. Biol. 2010, 31, 281–285. [Google Scholar] [PubMed]
- Sander, B.O.; Samson, M.; Buresh, R.J. Methane and nitrous oxide emissions from flooded rice fields as affected by water and straw management between rice crops. Geoderma 2014, 235, 355–362. [Google Scholar] [CrossRef]
- Tariq, A.; Vu, Q.D.; Jensen, L.S.; de Tourdonnet, S.; Sander, B.O.; Wassmann, R.; van Mai, T.; de Neergaard, A. Mitigating CH4 and N2O emissions from intensive rice production systems in northern Vietnam: Efficiency of drainage patterns in combination with rice residue incorporation. Agric. Ecosyst. Environ. 2017, 249, 101–111. [Google Scholar] [CrossRef]
- Hogarth, J.R.; Haywood, C.; Whitley, S. Low-Carbon Development in Sub-Saharan Africa: 20 Cross-Sector Transitions; Overseas Development Institute: London, UK, 2015. [Google Scholar]
- Intergovernmental Panel on Climate Change. Agriculture, Forestry and Other Land Use (AFOLU). In Climate Change 2014: Mitigation of Climate Change; IPCC Working Group III Contribution to AR5; Cambridge University Press: Cambridge, UK, 2015; pp. 811–922. [Google Scholar] [CrossRef]
- Smith, P.; Bustamante, M.; Ahammad, H.; Clark, H.; Dong, H.; Elsiddig, E.A.; Haberl, H.; Harper, R.; House, J.; Jafari, M.; et al. Agriculture, Forestry and Other Land Use (AFOLU). In Climate Change. Mitigation of Climate Change; Contribution of Working Group III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change; Edenhofer, O., Pichs-Madruga, R., Sokona, Y., Farahani, E., Kadner, S., Seyboth, K., Adler, A., Baum, I., Brunner, S., Eickemeier, P., et al., Eds.; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2014. [Google Scholar]
- Chen, X.; Cui, Z.; Fan, M.; Vitousek, P.; Zhao, M.; Ma, W.; Wang, Z. Producing more grain with lower environmental costs. Nature 2014, 514, 486–489. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, H.; Tsuruta, H. Nitrous oxide, nitric oxide, and nitrogen dioxide fluxes from soils after manure and urea application. J. Environ. Qual. 2003, 32, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Dong, D.; Feng, Q.; Mcgrouther, K.; Yang, M.; Wang, H.; Wu, W. Effects of biochar amendment on rice growth and nitrogen retention in a waterlogged paddy field. J. Soils Sediments 2015, 15, 153–162. [Google Scholar] [CrossRef]
- Novais, S.V.; Zenero, M.D.O.; Junior, E.F.F.; de Lima, R.P.; Cerri, C.E.P. Mitigation of greenhouse gas emissions from tropical soils amended with poultry manure and sugar cane straw biochars. Agric. Sci. 2017, 8, 887–903. [Google Scholar]
- Chen, D.; Wang, C.; Shen, J.; Li, Y.; Wu, J. Response of CH4 emissions to straw and biochar applications in double-rice cropping systems: Insights from observations and modeling. Environ. Pollut. 2018, 235, 95–103. [Google Scholar] [CrossRef]
- Huang, M.; Fan, L.; Chen, J.; Jiang, L.; Zou, Y. Continuous applications of biochar to rice: Effects on nitrogen uptake and utilization. Sci. Rep. 2018, 8, 11461. [Google Scholar] [CrossRef]
- Trong Hung, D.; Hughes, H.J.; Keck, M.; Sauer, D. Rice-Residue Management Practices of Smallholder Farms in Vietnam and Their Effects on Nutrient Fluxes in the Soil-Plant System. Sustainability 2019, 11, 1641. [Google Scholar] [CrossRef]
- Jiang, Z.; Lian, F.; Wang, Z.; Xing, B. The role of biochars in sustainable crop production and soil resiliency. J. Exp. Bot. 2020, 71, 520–542. [Google Scholar] [CrossRef] [PubMed]
- Sander, B.O.; Quilty, J.; Balingbing, C.; Castalone, A.G.; Romasanta, R.; Alberto, M.C.R.; Sandro, J.M.; Jamieson, C.; Gummert, M. An assessment of irrigated rice production energy efficiency and environmental footprint with in-field and off-field rice straw management practices. Sci. Rep. 2019, 9, 16887. [Google Scholar]
- Duy Pham, D.; Cai, K.; Phung, L.D.; Kaku, N.; Sasaki, A.; Sasaki, Y.; Horiguchi, K.; Pham, D.V.; Watanabe, T. Rice cultivation without synthetic fertilizers and performance of Microbial Fuel Cells (MFCs) under continuous irrigation with treated wastewater. Water 2019, 11, 1516. [Google Scholar] [CrossRef]
- Arunrat, N.; Pumijumnong, N. Practices for reducing greenhouse gas emissions from rice production in Northeast Thailand. Agriculture 2017, 7, 4. [Google Scholar] [CrossRef]
- Hussain, S.; Peng, S.; Fahad, S.; Khaliq, A.; Huang, J.; Cui, K.; Nie, L. Rice management interventions to mitigate greenhouse gas emissions: A review. Environ. Sci. Pollut. Res. 2015, 22, 3342–3360. [Google Scholar] [CrossRef]
- Wang, C.; Liu, J.; Shen, J.; Chen, D.; Li, Y.; Jiang, B.; Wu, J. Effects of biochar amendment on net greenhouse gas emissions and soil fertility in a double rice cropping system: A 4-year field experiment. Agric. Ecosyst. Environ. 2018, 262, 83–96. [Google Scholar] [CrossRef]
- Aulakh, M.S.; Wassmann, R.; Bueno, C.; Kreuzwieser, J.; Rennenberg, H. Characterization of root exudates at different growth stages of ten rice (Oryza sativa L.) cultivars. Plant Biol. 2001, 3, 139–148. [Google Scholar] [CrossRef]
- Wassmann, R.; Buendia, L.V.; Lantin, R.S.; Bueno, C.S.; Lubigan, L.A.; Umali, A.; Nocon, N.N.; Javellana, A.M.; Neue, H.U. Mechanisms of crop management impact on methane emissions from rice fields in Los Baños, Philippines. Nutr. Cycl. Agroecosyst. 2000, 58, 107–119. [Google Scholar] [CrossRef]
- Yagi, K.; Tsuruta, H.; Minami, K. Possible options for mitigating methane emission from rice cultivation. Nutr. Cycl. Agroecosyst. 1997, 49, 213–220. [Google Scholar] [CrossRef]
- Linquist, B.A.; Adviento-Borbe, M.A.; Pittelkow, C.M.; van Kessel, C.; jan van Groenigen, K. Fertilizer management practices and greenhouse gas emissions from rice systems: A quantitative review and analysis. Field Crops Res. 2012, 135, 10–21. [Google Scholar] [CrossRef]
- Sampanpanish, P.; Alam, L.; Mohamed, C.A.; Mokhtar, M.B.; Han, E.C.; Huang, Y.C.; Lin, J.M.; Lin, H.J.; Wu, J.Y.; Lee, C.C.; et al. Use of organic fertilizer on paddy fields to reduce greenhouse gases. Sci. Asia 2012, 38, 323–330. [Google Scholar] [CrossRef]
- Ghana Statistical Service. 2010 Population and Housing Census Report; Ghana Statistical Service: Ghana, West Africa, 2014. [Google Scholar]
- Minamikawa, K.; Takeshi, T.; Shigeto, S.; Agnes, P.; Kazuyuki, Y. Guidelines for Measuring CH4 and N2O Emissions from Rice Paddies by a Manually Operated Closed Chamber Method; National Institute for Agro-Environmental Sciences: Tsukuba, Japan, 2015; p. 76. [Google Scholar]
- Pedersen, A.R.; Petersen, S.O.; Schelde, K. A comprehensive approach to soil-atmosphere trace-gas flux estimation with static chambers. Eur. J. Soil Sci. 2010, 61, 888–902. [Google Scholar] [CrossRef]
- Hutchinson, G.L.; Mosier, A.R. Improved soil cover method for field measurement of nitrous oxide fluxes. Soil Sci. Soc. Am. J. 1981, 45, 311–316. [Google Scholar] [CrossRef]
- Das, S.; Ghosh, A.; Adhya, T.K. Nitrous oxide and methane emission from a flooded rice field as influenced by separate and combined application of herbicides bensulfuron methyl and pretilachlor. Chemosphere 2011, 84, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Wu, X.; Chen, A.; Xie, X.; Wang, Y.; Yin, C. Mitigating effects of ex situ application of rice straw on CH4 and N2O emissions from paddy-upland coexisting system. Sci. Rep. 2016, 6, 37402. [Google Scholar] [CrossRef]
- Das, S.; Adhya, T.K. Effect of combine application of organic manure and inorganic fertilizer on methane and nitrous oxide emissions from a tropical flooded soil planted to rice. Geoderma 2014, 213, 185–192. [Google Scholar] [CrossRef]
- Butterbach-Bahl, K.; Papen, H.; Rennenberg, H. Impact of gas transport through rice cultivars on methane emission from rice paddy fields. Plant Cell Environ. 1997, 20, 1175–1183. [Google Scholar] [CrossRef]
- Nouchi, I.; Mariko, S.; Aoki, K. Mechanism of methane transport from the rhizosphere to the atmosphere through rice plants. Plant Physiol. 1990, 94, 59–66. [Google Scholar] [CrossRef]
- Wang, B.; Neue, H.U.; Samonte, H.P. Role of rice in mediating methane emission. Plant Soil 1997, 189, 107–115. [Google Scholar] [CrossRef]
- Schütz, H.; Seiler, W.; Conrad, R. Processes involved in formation and emission of methane in rice paddies. Biogeochemistry 1989, 7, 33–53. [Google Scholar] [CrossRef]
- Wassmann, R.; Neue, H.U.; Alberto, M.C.R.; Lantin, R.S.; Bueno, C.; Llenaresas, D.; Arah, J.R.M.; Papen, H.; Seiler, W.; Rennenberg, H. Fluxes and pools of methane in wetland rice soils with varying organic inputs. Environ. Monit. Assess. 1996, 42, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Cicerone, R.J.; Shetter, J.D. Sources of atmospheric methane: Measurements in rice paddies and a discussion. J. Geophys. Res. Oceans 1981, 86, 7203–7209. [Google Scholar] [CrossRef]
- Den Van der Gon, H.A.; Van Breemen, N. Diffusion-controlled transport of methane from soil to atmosphere as mediated by rice plants. Biogeochemistry 1993, 21, 177–190. [Google Scholar] [CrossRef]
- Holzapfel-Pschorn, A.; Conrad, R.; Seiler, W. Effects of vegetation on the emission of methane from submerged paddy soil. Plant Soil 1986, 92, 223–233. [Google Scholar] [CrossRef]
- Gaihre, Y.K.; Tirol-Padre, A.; Wassmann, R.; Aquino, E.; Pangga, G.V.; Sta-Cruz, P.C. Spatial and temporal variations in methane fluxes from irrigated lowland rice fields. Philipp. Agric. Sci. 2011, 94, 335–342. [Google Scholar]
- Davidson, E.A. Fluxes of nitrous oxide and nitric oxide from terrestrial ecosystems. In Microbial Production and Consumption of Greenhouse Gases: Methane, Nitrous Oxide, and Halomethanes; American Society for Microbiology: Washington, DC, USA, 1991; pp. 219–235. [Google Scholar]
- Granli, T.; Bøckman, O.C. Nitrous oxide from agriculture. Nor. J. Agric. Sci. 1994, 12, 1–25. [Google Scholar]
- Adviento-Borbe, M.A.; Pittelkow, C.M.; Anders, M.; van Kessel, C.; Hill, J.E.; McClung, A.M.; Six, J.; Linquist, B.A. Optimal fertilizer nitrogen rates and yield-scaled global warming potential in drill seeded rice. J. Environ. Qual. 2013, 42, 1623–1634. [Google Scholar] [CrossRef]
- Myint, A.K.; Yamakawa, T.; Zenmyo, T.; Thao, H.T.B.; Sarr, P.S. Effects of organic-manure application on growth, grain yield, and nitrogen, phosphorus, and potassium recoveries of rice variety Manawthuka in paddy soils of differing fertility. Commun. Soil Sci. Plant Anal. 2011, 42, 457–474. [Google Scholar] [CrossRef]
- Rahman, F.; Rahman, M.M.; Rahman, G.K.M.M.; Saleque, M.A.; Hossain, A.T.M.S.; Miah, M.G. Effect of organic and inorganic fertilizers and rice straw on carbon sequestration and soil fertility under a rice–rice cropping pattern. Carbon Manag. 2016, 7, 41–53. [Google Scholar] [CrossRef]
- Van Groenigen, J.W.; Oenema, O.; jan van Groenigen, K.; Velthof, G.; van Kessel, C. Best nitrogen management practices to decrease greenhouse gas emissions. Better Crop. 2011, 95, 16–17. [Google Scholar]
- Wang, C.; Lai, D.Y.F.; Sardans, J.; Wang, W.; Zeng, C.; Peñuelas, J. Factors related with CH4 and N2O emissions from a paddy field: Clues for management implications. PLoS ONE 2017, 12, e0169254. [Google Scholar] [CrossRef] [PubMed]
- Peco, B.; Navarro, E.; Carmona, C.P.; Medina, N.G.; Marques, M.J. Effects of grazing abandonment on soil multifunctionality: The role of plant functional traits. Agric. Ecosyst. Environ. 2017, 249, 215–225. [Google Scholar] [CrossRef]
- Nyamadzawo, G.; Wuta, M.; Chirinda, N.; Mujuru, L.; Smith, J.L. Greenhouse Gas Emissions from Intermittently Flooded (Dambo) Rice under Different Tillage Practices in Chiota Smallholder Farming Area of Zimbabwe. Atmos. Clim. Sci. 2013, 3, 13–20. [Google Scholar] [CrossRef]
- Liu, J.; Shen, J.; Li, Y.; Su, Y.; Ge, T.; Jones, D.L.; Wu, J. Effects of biochar amendment on the net greenhouse gas emission and greenhouse gas intensity in a Chinese double rice cropping system. Eur. J. Soil Biol. 2014, 65, 30–39. [Google Scholar] [CrossRef]
- Kim, D.-G.; Thomas, A.D.; Pelster, D.; Rosenstock, T.S.; Sanz-Cobena, A. Reviews and syntheses: Greenhouse gas emissions in natural and agricultural lands in sub-Saharan Africa: Synthesis of available data and suggestions for further studies. Biogeosci. Discuss. 2015, 12, 16479–16526. [Google Scholar] [CrossRef]
- Shen, J.; Tang, H.; Liu, J.; Wang, C.; Li, Y.; Ge, T.; Jones, D.L.; Wu, J. Contrasting effects of straw and straw-derived biochar amendments on greenhouse gas emissions within double rice cropping systems. Agric. Ecosyst. Environ. 2014, 188, 264–274. [Google Scholar] [CrossRef]
- Cayuela, M.L.; van Zwieten, L.; Singh, B.P.; Jeffery, S.; Roig, A.; Sánchez-Monedero, M.A. Biochar’s role in mitigating soil nitrous oxide emissions: A review and meta-analysis. Agric. Ecosyst. Environ. 2014, 191, 5–16. [Google Scholar] [CrossRef]
- Kindaichi, T.; Ito, T.; Okabe, S. Ecophysiological interaction between nitrifying bacteria and heterotrophic bacteria in autotrophic nitrifying biofilms as determined by microautoradiography-fluorescence in situ hybridization. Appl. Environ. Microbiol. 2004, 70, 1641–1650. [Google Scholar] [CrossRef]
- Signor, D.; Cerri, C.E.P. Nitrous oxide emissions in agricultural soils: A review. Pesqui. Agropecuária Trop. 2013, 43, 322–338. [Google Scholar] [CrossRef]
- Rogovska, N.; Laird, D.; Cruse, R.; Fleming, P.; Parkin, T.; Meek, D. Impact of biochar on manure carbon stabilization and greenhouse gas emissions. Soil Sci. Soc. Am. J. 2011, 75, 871–879. [Google Scholar] [CrossRef]
- Yanai, Y.; Toyota, K.; Okazaki, M. Effects of charcoal addition on N2O emissions from soil resulting from rewetting air-dried soil in short-term laboratory experiments. Soil Sci. Plant Nutr. 2007, 53, 181–188. [Google Scholar] [CrossRef]
- Gell, K.; van Groenigen, J.; Cayuela, M.L. Residues of bioenergy production chains as soil amendments: Immediate and temporal phytotoxicity. J. Hazard. Mater. 2011, 186, 2017–2025. [Google Scholar] [CrossRef] [PubMed]
- Dempster, D.N.; Gleeson, D.B.; Solaiman, Z.M.; Jones, D.L.; Murphy, D.V. Decreased soil microbial biomass and nitrogen mineralisation with Eucalyptus biochar addition to a coarse textured soil. Plant Soil 2012, 354, 311–324. [Google Scholar] [CrossRef]
- Hale, S.E.; Lehmann, J.; Rutherford, D.; Zimmerman, A.R.; Bachmann, R.T.; Shitumbanuma, V.; O’Toole, A.; Sundqvist, K.L.; Arp, H.P.H.; Cornelissen, G. Quantifying the total and bioavailable polycyclic aromatic hydrocarbons and dioxins in biochars. Environ. Sci. Technol. 2012, 46, 2830–2838. [Google Scholar] [CrossRef]
- Hilber, I.; Blum, F.; Leifeld, J.; Schmidt, H.P.; Bucheli, T.D. Quantitative determination of PAHs in biochar: A prerequisite to ensure its quality and safe application. J. Agric. Food Chem. 2012, 60, 3042–3050. [Google Scholar] [CrossRef]
- Sverdrup, L.E.; Ekelund, F.; Krogh, P.H.; Nielsen, T.; Johnsen, K. Soil microbial toxicity of eight polycyclic aromatic compounds: Effects on nitrification, the genetic diversity of bacteria, and the total number of protozoans. Environ. Toxicol. Chem. Int. J. 2002, 21, 1644–1650. [Google Scholar] [CrossRef]
- Maliszewska-Kordybach, B.; Klimkowicz-Pawlas, A.; Smreczak, B.; Janusauskaite, D. Ecotoxic effect of phenanthrene on nitrifying bacteria in soils of different properties. J. Environ. Qual. 2007, 36, 1635–1645. [Google Scholar] [CrossRef]
- Guo, G.X.; Deng, H.; Qiao, M.; Yao, H.Y.; Zhu, Y.G. Effect of long-term wastewater irrigation on potential denitrification and denitrifying communities in soils at the watershed scale. Environ. Sci. Technol. 2013, 47, 3105–3113. [Google Scholar] [CrossRef]
- Spokas, K.A.; Baker, J.M.; Reicosky, D.C. Ethylene: Potential key for biochar amendment impacts. Plant Soil 2010, 333, 443–452. [Google Scholar] [CrossRef]
- Wang, C.; Lu, H.; Da, D.; Deng, H.; Strong, P.J.; Wang, H.; Wu, W. Insight into the effects of biochar on manure composting: Evidence supporting the relationship between N2O emission and denitrifying community. Environ. Sci. Technol. 2013, 47, 7341–7349. [Google Scholar] [CrossRef]
- Mahmood, F.; Khan, I.; Ashraf, U.; Shahzad, T.; Hussain, S.; Shahid, M.; Abid, M.; Ullah, S. Effects of organic and inorganic manures on maize and their residual impact on soil physico-chemical properties. J. Soil Sci. Plant Nutr. 2017, 17, 22–32. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, S.; Ju, M.; Liu, L. Preparation and modification of biochar materials and their application in soil remediation. Appl. Sci. 2019, 9, 1365. [Google Scholar] [CrossRef]
- Feng, Y.; Xu, Y.; Yu, Y.; Xie, Z.; Lin, X. Mechanisms of biochar decreasing methane emission from Chinese paddy soils. Soil Biol. Biochem. 2012, 46, 80–88. [Google Scholar] [CrossRef]
- Karhu, K.; Mattila, T.; Bergström, I.; Regina, K. Biochar addition to agricultural soil increased CH4 uptake and water holding capacity–Results from a short-term pilot field study. Agric. Ecosyst. Environ. 2011, 140, 309–313. [Google Scholar] [CrossRef]
- Spokas, K.A.; Reicosky, D.C. Impacts of sixteen different biochars on soil greenhouse gas production. Ann. Environ. Sci. 2009, 3, 179–193. [Google Scholar]
- Zhang, A.; Cui, L.; Pan, G.; Li, L.; Hussain, Q.; Zhang, X.; Zheng, J.; Crowley, D. Effect of biochar amendment on yield and methane and nitrous oxide emissions from a rice paddy from Tai Lake plain, China. Agric. Ecosyst. Environ. 2010, 139, 469–475. [Google Scholar] [CrossRef]
- Kammann, C.; Ratering, S.; Eckhard, C.; Müller, C. Biochar and hydrochar effects on greenhouse gas (carbon dioxide, nitrous oxide, and methane) fluxes from soils. J. Environ. Qual. 2012, 41, 1052–1066. [Google Scholar] [CrossRef]
- Yamato, M.; Okimori, Y.; Wibowo, I.F.; Anshori, S.; Ogawa, M. Effects of the application of charred bark of Acacia mangium on the yield of maize, cowpea and peanut, and soil chemical properties in South Sumatra, Indonesia. Soil Sci. Plant Nutr. 2006, 52, 489–495. [Google Scholar] [CrossRef]
- Jeffery, S.; Verheijen, F.G.A.; van der Velde, M.; Bastos, A.C. A quantitative review of the effects of biochar application to soils on crop productivity using meta-analysis. Agric. Ecosyst. Environ. 2011, 144, 175–187. [Google Scholar] [CrossRef]
- Semrau, J.D.; DiSpirito, A.A.; Yoon, S. Methanotrophs and copper. FEMS Microbiol. Rev. 2010, 34, 496–531. [Google Scholar] [CrossRef]
- Watanabe, A.; Takeda, T.; Kimura, M. Evaluation of origins of CH4 carbon emitted from rice paddies. J. Geophys. Res. Atmos. 1999, 104, 23623–23629. [Google Scholar] [CrossRef]
- Ma, J.; Xu, H.; Yagi, K.; Cai, Z. Methane emission from paddy soils as affected by wheat straw returning mode. Plant Soil 2008, 313, 167–174. [Google Scholar] [CrossRef]
- Cui, Y.F.; Jun, M.E.; Wang, Q.X.; Zhang, W.M.; Cheng, X.Y.; Chen, W.F. Effects of straw and biochar addition on soil nitrogen, carbon, and super rice yield in cold waterlogged paddy soils of North China. J. Integr. Agric. 2017, 16, 1064–1074. [Google Scholar] [CrossRef]
- Spokas, K.A.; Cantrell, K.B.; Novak, J.M.; Archer, D.W.; Ippolito, J.A.; Collins, H.P.; Boateng, A.A.; Lima, I.M.; Lamb, M.C.; McAloon, A.J.; et al. Biochar: A synthesis of its agronomic impact beyond carbon sequestration. J. Environ. Qual. 2012, 41, 973–989. [Google Scholar] [CrossRef]
- Sun, D.; Meng, J.; Liang, H.; Yang, E.; Huang, Y.; Chen, W.; Jiang, L.; Lan, Y.; Zhang, W.; Gao, J. Effect of volatile organic compounds absorbed to fresh biochar on survival of Bacillus mucilaginosus and structure of soil microbial communities. J. Soils Sediments 2015, 15, 271–281. [Google Scholar] [CrossRef]
- Walsh, E.; McDonnell, K.P. The influence of added organic matter on soil physical, chemical, and biological properties: A small-scale and short-time experiment using straw. Arch. Agron. Soil Sci. 2012, 58, S201–S205. [Google Scholar] [CrossRef]
- Lehmann, J.; Joseph, S. (Eds.) Biochar for Environmental Management: Science, Technology and Implementation; Routledge: London, UK, 2015. [Google Scholar]
- Liu, Y.; Lu, H.; Yang, S.; Wang, Y. Impacts of biochar addition on rice yield and soil properties in a cold waterlogged paddy for two crop seasons. Field Crop. Res. 2016, 191, 161–167. [Google Scholar] [CrossRef]
- Noguera, D.; Barot, S.; Laossi, K.R.; Cardoso, J.; Lavelle, P.; de Carvalho, M.C. Biochar but not earthworms enhances rice growth through increased protein turnover. Soil Biol. Biochem. 2012, 52, 13–20. [Google Scholar] [CrossRef]
- Zhao, L.; Cao, X.; Mašek, O.; Zimmerman, A. Heterogeneity of biochar properties as a function of feedstock sources and production temperatures. J. Hazard. Mater. 2013, 256, 1–9. [Google Scholar] [CrossRef]
- Wang, W.; Lai, D.Y.F.; Sardans, J.; Wang, C.; Datta, A.; Pan, T.; Zeng, C.; Bartrons, M.; Penuelas, J. Rice straw incorporation affects global warming potential differently in early vs. late cropping seasons in Southeastern China. Field Crop. Res. 2015, 181, 42–51. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, Z.; Liu, Y.; Xu, X.; Wang, J.; Zhang, H.; Xiong, Z. Net global warming potential and greenhouse gas intensity in rice agriculture driven by high yields and nitrogen use efficiency: A 5-year field study. Biogeosci. Discuss. 2015, 12, 18883–18911. [Google Scholar] [CrossRef]
- Wassmann, R.; Butterbach-Bahl, K.; Dobermann, A. Irrigated rice production systems and greenhouse gas emissions: Crop and residue management trends, climate change impacts and mitigation strategies. CAB Rev. Perspect. Agric. Vet. Sci. Nutr. Nat. Resour. 2007. [CrossRef]
- Zhuang, Q.; Melack, J.M.; Zimov, S.; Walter, K.M.; Butenhoff, C.L.; Khalil, M.A. Global methan emissions from wetlands, rice paddies, and lakes. Eos Trans. Am. Geophys. Union 2009, 90, 37–38. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Naylor, R.; Crews, T.; David, M.B.; Drinkwater, L.E.; Holland, E.; Johnes, P.J. Nutrient imbalances in agricultural development. Science 2009, 324, 1519–1520. [Google Scholar] [CrossRef] [PubMed]
- Case, S.D.C. Biochar Amendment and Greenhouse Gas Emissions from Agricultural Soils. Ph.D. Thesis, University of Edinburgh, Edinburgh, UK, 2013; pp. 117–122. Available online: https://ethos.bl.uk/OrderDetails.do?uin=uk.bl.ethos.586427 (accessed on 1 December 2020).
- Zheng, H.; Huang, H.; Yao, L.; Liu, J.; He, H.; Tang, J. Impacts of rice varieties and management on yield-scaled greenhouse gas emissions from rice fields in China: A meta-analysis. Biogeosciences 2014, 11, 3685. [Google Scholar] [CrossRef]
- Xia, L.; Xia, Y.; Ma, S.; Wang, J.; Wang, S.; Zhou, W.; Yan, X. Greenhouse gas emissions and reactive nitrogen releases from rice production with simultaneous incorporation of wheat straw and nitrogen fertilizer. Biogeosciences 2016, 13, 4569–4579. [Google Scholar] [CrossRef]
- Richards, M.; Metzel, R.; Chirinda, N.; Ly, P.; Nyamadzawo, G.; Vu, Q.D.; De Neergaard, A.; Oelofse, M.; Wollenberg, E.; Keller, E.; et al. Limits of agricultural greenhouse gas calculators to predict soil N2O and CH4 fluxes in tropical agriculture. Sci. Rep. 2016, 6, 26279. [Google Scholar] [CrossRef]
- Kritee, K.; Nair, D.; Zavala-Araiza, D.; Proville, J.; Rudek, J.; Adhya, T.K.; Loecke, T.; Esteves, T.; Balireddygari, S.; Dava, O.; et al. High nitrous oxide fluxes from rice indicate the need to manage water for both long-and short-term climate impacts. Proc. Natl. Acad. Sci. USA 2018, 115, 9720–9725. [Google Scholar] [CrossRef]
- Sanchis, E.; Ferrer, M.; Torres, A.G.; Cambra-López, M.; Calvet, S. Effect of water and straw management practices on methane emissions from rice fields: A review through a meta-analysis. Environ. Eng. Sci. 2012, 29, 1053–1062. [Google Scholar] [CrossRef]
- Yao, Z.; Du, Y.; Tao, Y.; Zheng, X.; Liu, C.; Lin, S.; Butterbach-Bahl, K. Water-saving ground cover rice production system reduces net greenhouse gas fluxes in an annual rice-based cropping system. Biogeosciences 2014, 11, 6221–6236. [Google Scholar] [CrossRef]
- Xu, Y.; Ge, J.; Tian, S.; Li, S.; Nguy-Robertson, A.L.; Zhan, M.; Cao, C. Effects of water-saving irrigation practices and drought resistant rice variety on greenhouse gas emissions from a no-till paddy in the central lowlands of China. Sci. Total Environ. 2015, 505, 1043–1052. [Google Scholar] [CrossRef] [PubMed]
- Vo, T.B.T.; Wassmann, R.; Tirol-Padre, A.; Cao, V.; MacDonald, B.; Espaldon, M.V.O.; Sander, B.O. Methane emission from rice cultivation in different agro-ecological zones of the Mekong river delta: Seasonal patterns and emission factors for baseline water management. Soil Sci. Plant Nutr. 2018, 64, 47–58. [Google Scholar] [CrossRef]
- Hussain, M.; Farooq, M.; Nawaz, A.; Al-Sadi, A.M.; Solaiman, Z.M.; Alghamdi, S.S.; Ammara, U.; Ok, Y.S.; Siddique, K.H.M. Biochar for crop production: Potential benefits and risks. J. Soils Sediments 2017, 17, 685–716. [Google Scholar] [CrossRef]
- Major, J.; Rondon, M.; Molina, D.; Riha, S.J.; Lehmann, J. Maize yield and nutrition during 4 years after biochar application to a Colombian savanna oxisol. Plant Soil 2010, 333, 117–128. [Google Scholar] [CrossRef]

| Block 1 | Block 2 | Block 3 | Block 4 |
|---|---|---|---|
| T3 | T1 | T2 | T1 |
| CT | T2 | T1 | CT |
| T4 | T3 | T3 | T2 |
| T2 | T4 | CT | T4 |
| T1 | CT | T4 | T3 |
| Treatment | Soil Amendment | N2O (kgNha−1day−1) | SD | CH4 (kgCH4ha−1day−1) | SD |
|---|---|---|---|---|---|
| BAU | NPK (300 kgha−1 + 50 kgha−1Urea (46%N)) | 0.22 ab | ±0.09 | 1.10 a | ±1.53 |
| BAU + BIO | BAU + 28tha−1Biochar | 0.20 b | ±0.12 | 4.76 a | ±4.87 |
| BAU+M | Manure (3tha−1) + BAU | 0.27 ab | ±0.08 | 1.20 a | ±1.77 |
| BAU + BIO + M | BAU + Biochar + Manure | 0.38 a | ±0.18 | 1.57 a | ±2.20 |
| CONTROL | No amendment | 0.02 c | ±0.03 | 2.71 a | ±2.77 |
| Treatment | N2O | SD | CH4 | SD |
|---|---|---|---|---|
| BAU | 39.93 b | ±15.76 | 153 a | ±206.35 |
| BAU + BIO | 26.84 bc | ±29.04 | 626.83 a | ±589.35 |
| BAU + M | 44.75 b | ±10.28 | 169.18 a | ±241.72 |
| BAU + BIO + M | 58.08 a | ±19.69 | 230.94 a | ±302.75 |
| CONTROL | 1.61 c | ±1.63 | 389.53 a | ±284.24 |
| Net Seasonal GWP (kgCO2eqha−1 Season−1) | Gross Seasonal GWP (kgCO2eqha−1 Season−1) | Yield (Kgha−1) | Gross Greenhouse Gas Intensity (GGHGI) kgCO2eq kg−1 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Treatment | N2O | SD | CH4 | SD | GGWP | SD | YIELD | SD | GGHGI | SD |
| BAU | 18,698.64 ab | ±7383.78 | 5099.16 a | ±6878.43 | 23,797.82 a | ±7411.43 | 5290.83 b | ±222.36 | 4.49 b | ±1.40 |
| BAU + BIO | 12,571.13 bc | ±13602.96 | 20,894.58 a | ±19,645.04 | 33,465.63 a | ±21,002.97 | 5318.33 b | ±148.49 | 8.19 a | ±1.25 |
| BAU + M | 20,959.88 ab | ±4814.37 | 5639.33 a | ±8057.59 | 26,599.22 a | ±11,562.23 | 6070.00 a | ±323.19 | 4.38 b | ±1.90 |
| BAU + BIO + M | 27,201.54 a | ±9223.51 | 7698.25 a | ±10,091.92 | 34,899.80 a | ±18,620.33 | 5935.83 a | ±332.68 | 6.35 ab | ±0.94 |
| CONTROL | 756.28 c | ±763.44 | 12,984.58 a | ±9474.70 | 13,740.86 a | ±9609.36 | 4395.00 c | ±301.28 | 3.13 b | ±2.19 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boateng, K.K.; Obeng, G.Y.; Mensah, E. Eco-Friendly Yield and Greenhouse Gas Emissions as Affected by Fertilization Type in a Tropical Smallholder Rice System, Ghana. Sustainability 2020, 12, 10239. https://doi.org/10.3390/su122410239
Boateng KK, Obeng GY, Mensah E. Eco-Friendly Yield and Greenhouse Gas Emissions as Affected by Fertilization Type in a Tropical Smallholder Rice System, Ghana. Sustainability. 2020; 12(24):10239. https://doi.org/10.3390/su122410239
Chicago/Turabian StyleBoateng, Kofi Konadu, George Yaw Obeng, and Ebenezer Mensah. 2020. "Eco-Friendly Yield and Greenhouse Gas Emissions as Affected by Fertilization Type in a Tropical Smallholder Rice System, Ghana" Sustainability 12, no. 24: 10239. https://doi.org/10.3390/su122410239
APA StyleBoateng, K. K., Obeng, G. Y., & Mensah, E. (2020). Eco-Friendly Yield and Greenhouse Gas Emissions as Affected by Fertilization Type in a Tropical Smallholder Rice System, Ghana. Sustainability, 12(24), 10239. https://doi.org/10.3390/su122410239




