Sustainable Soil-Compacted Blocks Containing Blast Furnace Slag (BFS) Activated with Olive Stone BIOMASS Ash (OBA)
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
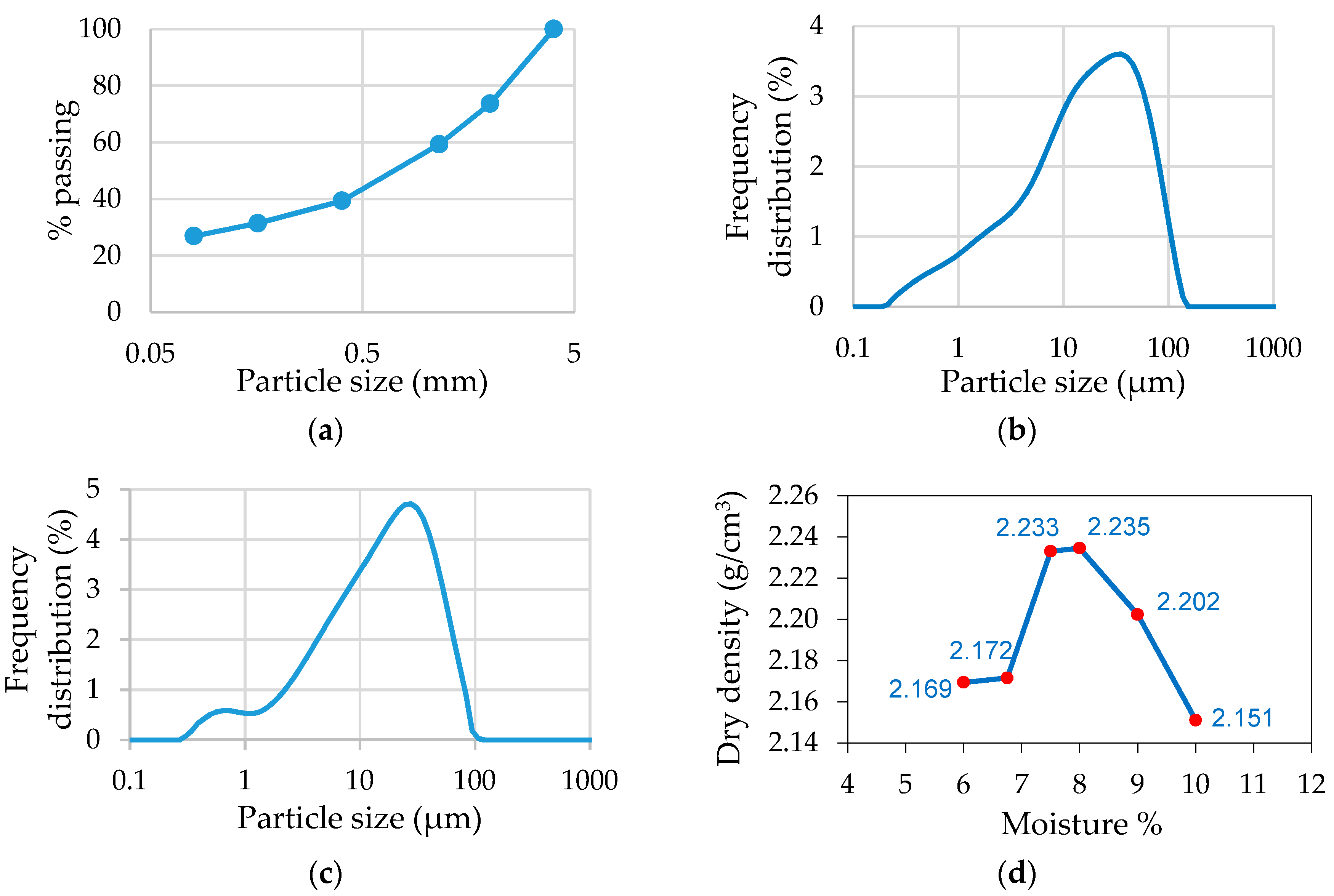
3.1. Olive Stone (OS) Biomass: Starting Raw Material
3.2. Characteristics of the Olive Stone Biomass Ash (OBA)
3.3. Mechanical Properties of Soil-Compacted Blocks
3.4. Characterization of Cementing Gel
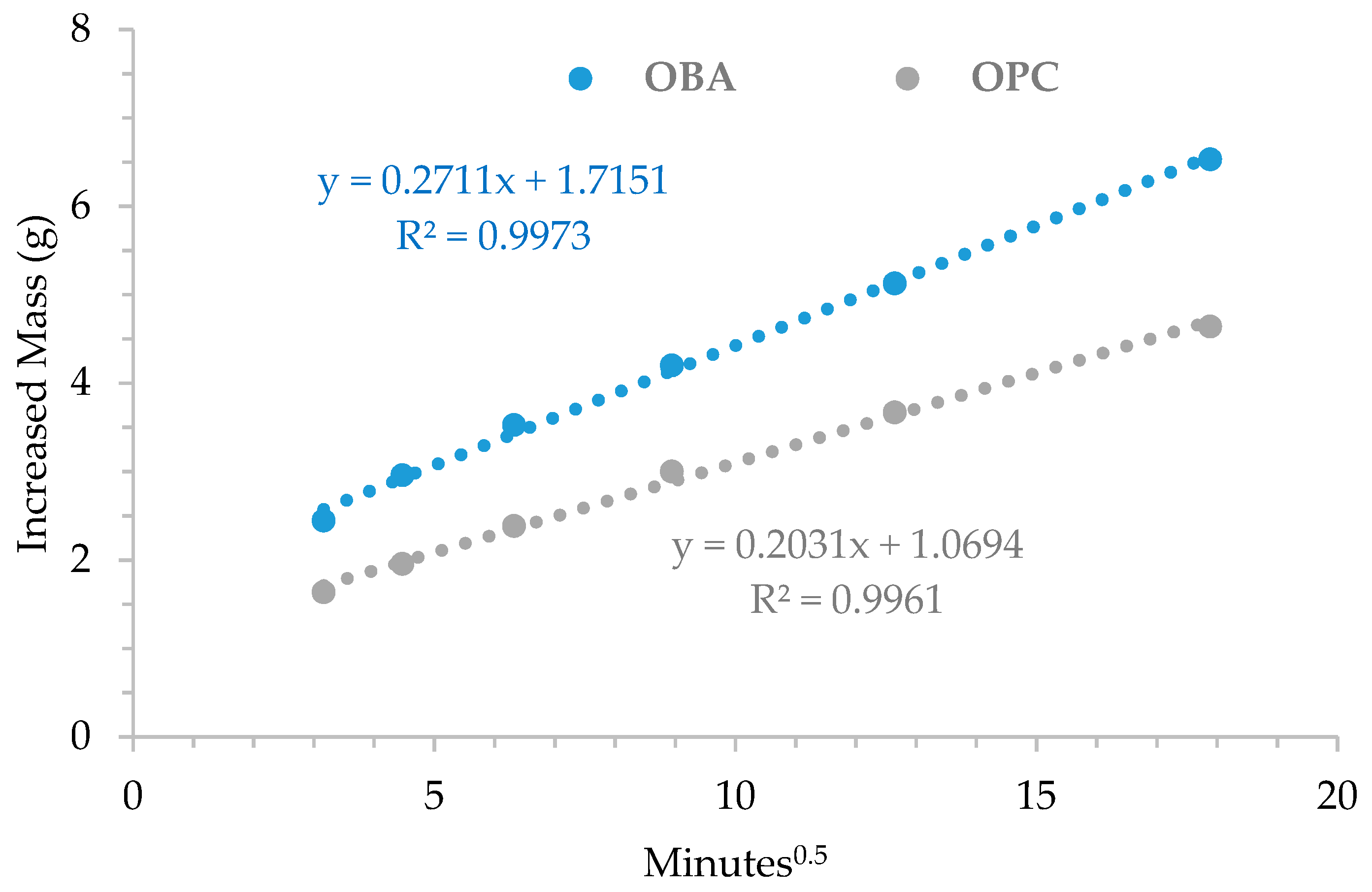
3.5. Behavior of the OBA/BFS Soil-Compacted Blocks in a Humid Environment
3.6. Sustainability and Practical Considerations
4. Conclusions
- (a)
- The energetic valorization of OS yields alkaline ash, known as OBA, which presents a sum of CaO and K2O higher than 75% per mass.
- (b)
- Dolomitic soil stabilization was conducted by adding 10% BFS, 4% OBA and 8% water with excellent compressive strength (Rc = 27.8 MPa) for the 40 mm cubic specimens obtained at 120 curing days.
- (c)
- The cementing gel formed by the reaction of OBA and BFS was characterized by thermogravimetry: the identification of the decomposition peaks in DTG curves showed the formation of C-S-H, C-A-S-H and hydrotalcite phases.
- (d)
- The FESEM study revealed the formation of the cementing gel-binding calcite and dolomite particles of soil: the chemical composition of the gel revealed the presence of magnesium and potassium and the formation of the C(K,M)-A-S-H gel.
- (e)
- The OBA/BFS soil-compacted blocks had a higher water absorption rate than the corresponding OPC ones, which is disadvantageous behavior in durability terms. However, the water strength coefficient for the OBA/BFS blocks was 0.70 for the 2-h immersion and 0.74 for the 72-h immersion, which indicates the strong adhesion of soil particles and the high insolubility of the cementing gel.
- (f)
- If we focus on the material components, the use of OBA/BFS cement reduced CO2 emissions by 96% compared to the OPC soil-compacted blocks: OBA is an excellent activator of BFS from the mechanical and environmental points of view.
- (g)
- The use of OBA as an activator allowed the preparation of one-part BFS-based cement, which is a good characteristic from the practical point of view.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Van Damme, H.; Houbenc, H. Earth concrete. Stabilization revisited. Cem. Concr. Res. 2018, 114, 90–102. [Google Scholar] [CrossRef]
- Menchaca-Ballinas, L.E.; Escalante-Garcia, J.I. Low CO2 emission cements of waste glass activated by CaO and NaOH. J. Clean. Prod. 2019, 239, 117992. [Google Scholar] [CrossRef]
- Basha, E.A.; Hashim, R.; Mahmud, H.B.; Muntohar, A.S. Stabilization of residual soil with rice husk ash and cement. Cons. Build. Mat. 2005, 19, 448–453. [Google Scholar] [CrossRef]
- Rahgozar, M.A.; Saberian, M.; Li, J. Soil stabilization with non-conventional eco-friendly agricultural waste materials: An experimental study. Trans. Geotech. 2018, 14, 52–60. [Google Scholar] [CrossRef]
- Payá, J.; Agrela, F.; Rosales, J.; Martín Morales, M.; Borrachero, M.V. Application of alkali-activated industrial waste. In New Trends in Eco-Efficient and Recycled Concrete; De Brito, J., Agrela, F., Eds.; Woodhead Publishing: Duxford, UK, 2019; pp. 357–424. [Google Scholar]
- Sisol, M.; Kudelas, D.; Marcin, M.; Holub, T.; Varga, P. Statistical Evaluation of Mechanical Properties of Slag Based Alkali-Activated Material. Sustainability 2019, 11, 5935. [Google Scholar] [CrossRef]
- Mellado, A.; Catalán, C.; Bouzón, N.; Borrachero, M.V.; Monzó, J.M.; Payá, J. Carbon footprint of geopolymeric mortar: Study of the contribution of the alkaline activating solution and assessment of an alternative route. RSC Adv. 2014, 4, 23846–23852. [Google Scholar] [CrossRef]
- Tchakouté, H.K.; Rüscher, C.H.; Kong, S.; Ranjbar, N. Synthesis of sodium waterglass from white rice husk ash as an activator to produce metakaolin-based geopolymer cements. J. Build. Eng. 2016, 6, 252–261. [Google Scholar] [CrossRef]
- Tchakouté, H.K.; Rüscher, C.H.; Hinsch, M.; Djobo, N.Y.; Kamseu, E.; Leonelli, C. Utilization of sodium waterglass from sugar cane bagasse ash as a new alternative hardener for producing metakaolin-based geopolymer cement. Chem. Erde Geochem. 2017, 77, 257–266. [Google Scholar] [CrossRef]
- Moraes, J.C.B.; Font, A.; Soriano, L.; Akasaki, J.L.; Tashima, M.M.; Monzó, J.; Borrachero, M.V.; Payá, J. New use of sugar cane straw ash in alkali-activated materials: A silica source for the preparation of the alkaline activator. Cons. Build. Mat. 2018, 171, 611–621. [Google Scholar] [CrossRef]
- Font, A.; Soriano, L.; Reig, L.; Tashima, M.M.; Borrachero, M.V.; Monzó, J.; Payá, J. Use of residual diatomaceous earth as a silica source in geopolymer production. Mat. Lett. 2018, 223, 10–13. [Google Scholar] [CrossRef]
- Samarakoon, M.H.; Ranjith, P.G.; Duan, W.H.; De Silva, V.R.S. Properties of one-part fly ash/slag-based binders activated by thermally-treated waste glass/NaOH blends: A comparative study. Cem. Concr. Comp. 2020, 112, 103679. [Google Scholar] [CrossRef]
- Zhaoa, X.; Liua, C.; Wanga, L.; Zuob, L.; Zhub, Q.; Mab, W. Physical and mechanical properties and micro characteristics of fly ash based geopolymers incorporating soda residue. Cem. Concr. Comp. 2019, 98, 125–136. [Google Scholar] [CrossRef]
- Bilginer, A.; Canbek, O.; Erdoğan, S.T. Activation of blast furnace slag with soda production waste. J. Mater. Civ. Eng. 2020, 32, 04019316. [Google Scholar] [CrossRef]
- Ban, C.; Ken, P.W.; Ramli, M. Mechanical and durability performance of novel self-activating geopolymer mortars. Proc. Eng. 2017, 171, 564–571. [Google Scholar] [CrossRef]
- Peys, A.; Rahier, H.; Pontikes, Y. Potassium-rich biomass ashes as activators in metakaolin-based inorganic polymers. Appl. Clay Sci. 2016, 119, 401–409. [Google Scholar] [CrossRef]
- Soriano, L.; Font, A.; Tashima, M.M.; Monzó, J.; Borrachero, M.V.; Payá, J. One-part blast furnace slag mortars activated with almond-shell biomass ash: A new 100% waste-based material. Mat. Lett. 2020, 272, 127882. [Google Scholar] [CrossRef]
- Abdullah, H.H.; Shahin, M.A.; Walske, M.L. Review of fly-ash-based geopolymers for soil stabilisation with special reference to clay. Geosciences 2020, 10, 249. [Google Scholar] [CrossRef]
- Cristelo, N.; Miranda, T.; Oliveira, D.V.; Rosa, I.; Soares, E.; Coelho, P.; Fernandes, L. Assessing the production of jet mix columns using alkali activated waste based on mechanical and financial performance and CO2 (eq) emissions. J. Clean. Prod. 2015, 102, 447–460. [Google Scholar] [CrossRef]
- Font, A.; Soriano, L.; Moraes, J.C.B.; Tashima, M.M.; Monzó, J.; Borrachero, M.V.; Payá, J. A 100% waste-based alkali-activated material by using olive-stone biomass ash (OBA) and blast furnace slag (BFS). Mat. Lett. 2017, 203, 46–49. [Google Scholar] [CrossRef]
- Pinheiro, S.M.M.; Font, A.; Soriano, L.; Tashima, M.M.; Monzó, J.; Borrachero, M.V.; Payá, J. Olive-stone biomass ash (OBA): An alternative alkaline source for the blast furnace slag activation. Cons. Build. Mat. 2018, 178, 327–338. [Google Scholar] [CrossRef]
- American Society for Testing and Materials. ASTM STP479. In Special Procedures for Testing Soil and Rock for Engineering Purposes; American Society for Testing and Materials: West Conshohocken, PA, USA, 1970. [Google Scholar]
- UNE 103501. In Geotechnic. Compactation Test. Modified Proctor; AENOR: Madrid, Spain, 1994. (In Spanish)
- UNE-EN 196-1. In Methods of Testing Cement—Part 1: Determination of Strength; AENOR: Madrid, Spain, 2018. (In Spanish)
- UNE 41410. In Compressed Earth Blocs for Walls and Partitions. Definitions, Specifications and Test Methods; AENOR: Madrid, Spain, 2008. (In Spanish)
- NTC 5324. In Ground Blocks Cement for Walls and Division. Definitions, Specifications. Test Methods. Conditions of Delivery; ICONTEC: Bogota, Colombia, 2004. (In Spanish)
- Alonso, M.M.; Gascó, C.; Martín Morales, M.; Suárez-Navarro, J.A.; Zamorano, M.; Puertas, F. Olive biomass ash as an alternative activator in geopolymer formation: A study of strength, radiology and leaching behaviour. Cem. Concr. Comp. 2019, 104, 103384. [Google Scholar] [CrossRef]
- Vossen, P. Olive Oil: History, Production, and Characteristics of the World’s Classic Oils. Hort. Sci. 2007, 42, 1093–1100. [Google Scholar] [CrossRef]
- Roig, A.; Cayuela, M.L.; Sánchez-Monedero, M.A. An overview on olive mill wastes and their valorisation methods. Waste Manag. 2006, 26, 960–969. [Google Scholar] [CrossRef]
- García Martín, J.F.; Cuevas, M.; Feng, C.; Álvarez-Mateos, P.; Torres-García, M.; Sánchez, S. Energetic Valorisation of Olive Biomass: Olive-Tree Pruning, Olive Stones and Pomaces. Processes 2020, 8, 511. [Google Scholar]
- Cosa, J.; Soriano, L.; Borrachero, M.V.; Payá, J.; Monzó, J. Stabilization of soil by means alternative alkali-activated cement prepared with spent FCC catalyst. Appl. Ceram. Technol. 2020, 17, 190–196. [Google Scholar] [CrossRef]
- Xing, J.; Zhao, Y.; Qiu, J.; Sun, X. Microstructural and mechanical properties of alkali activated materials from two types of blast furnace slags. Materials 2019, 12, 2089. [Google Scholar] [CrossRef] [PubMed]
- Burciaga-Díaz, O.; Escalante-García, J.I. Structure, mechanisms of reaction, and strength of an alkali-activated blast-furnace slag. J. Am. Ceram. Soc. 2013, 96, 3939–3948. [Google Scholar] [CrossRef]
- Gunasekaran, S.; Anbalagan, G. Thermal decomposition of natural dolomite. Bull. Mater. Sci. 2007, 30, 339–344. [Google Scholar] [CrossRef]
- Walkley, B.; Rackel San Nicolas, R.; Sani, M.; Rees, G.J.; Hanna, J.H.; van Deventer, J.S.J.; Provis, J.L. Phase evolution of C-(N)-A-S-H/N-A-S-H gel blends investigated via alkali-activation of synthetic calcium aluminosilicate precursors. Cem. Concr. Res. 2016, 89, 120–135. [Google Scholar] [CrossRef]
- Puertas, F.; Palacios, M.; Manzano, H.; Dolado, J.S.; Rico, A.; Rodríguez, J. A model for the C-A-S-H gel formed in alkali-activated slag cements. J. Eur. Ceram. Soc. 2011, 31, 2043–2056. [Google Scholar] [CrossRef]
- Ortega-Zavala, D.E.; Santana-Carrillo, J.L.; Burciaga-Díaz, O.; Escalante-García, J.I. An initial study on alkali activated limestone binders. Cem. Concr. Res. 2019, 120, 267–278. [Google Scholar] [CrossRef]
- Kinuthia, J.M. The durability of compressed earth-based masonry blocks, In Eco-Efficient Masonry Bricks and Blocks; Pacheco-Torgal, F., Lourenço, P.B., Labrincha, J.A., Kumar, S., Chindaprasirt, P., Eds.; Woodhead Publishing: Duxford, UK, 2015; pp. 393–421. [Google Scholar]
- Houben, H.; Verney, P.E.; Maini, S.; Webb, D.J.T. Compressed Earth Bricks. SELECTION of Production Equipment; CDI: Brussels, Belgium, 1989. [Google Scholar]
- Guettala, A.; Abibsi, A.; Houari, H. Durability study of stabilized earth concrete under both laboratory and climatic conditions exposure. Cons. Build. Mater. 2006, 20, 119–127. [Google Scholar] [CrossRef]
- Salim, R.W.; Ndambuki, J.M.; Adedokun, D.A. Improving the bearing strength of sandy loam soil compressed earth block bricks using sugarcane bagasse ash. Sustainability 2014, 6, 3686–3696. [Google Scholar] [CrossRef]
- De Brito, J.; Kurda, R. The past and future of sustainable concrete: A critical review and new strategies on cement-based materials. J. Clean. Prod. 2020; 123558, in press. [Google Scholar]
- Font, A.; Soriano, L.; Tashima, M.M.; Monzó, J.; Borrachero, M.V.; Payá, J. One-part eco-cellular concrete for the precast industry: Functional features and life cycle assessment. J. Clean. Prod. 2020, 269, 122203. [Google Scholar] [CrossRef]
- Luukkonen, T.; Abdollahnejad, Z.; Yliniemi, J.; Kinnunen, P.; Illikainen, M. One-part alkali-activated materials: A review. Cem. Concr. Res. 2018, 103, 21–34. [Google Scholar] [CrossRef]







| Oxide | % by Mass |
|---|---|
| Na2O | 0.87 |
| MgO | 7.43 |
| Al2O3 | 10.55 |
| SiO2 | 30.53 |
| P2O5 | 0.26 |
| SO3 | 1.93 |
| K2O | 0.57 |
| CaO | 40.15 |
| Fe2O3 | 1.29 |
| LOI | 5.53 |
| Component | Value |
|---|---|
| Soil | 1000 g |
| OBA (activator) | 40 g |
| BFS (precursor) | 100 g |
| Water | 91 g |
| Water/solid ratio | 0.0798 |
| Activator/precursor ratio | 0.4 |
| Oxide | % by Mass | Std. Dev |
|---|---|---|
| Na2O | 1.11 | 0.12 |
| MgO | 7.77 | 1.06 |
| Al2O3 | 0.99 | 0.25 |
| SiO2 | 5.38 | 1.97 |
| P2O5 | 3.54 | 0.59 |
| SO3 | 1.68 | 0.77 |
| Cl | 0.73 | 0.34 |
| K2O | 43.41 | 2.62 |
| CaO | 32.46 | 2.79 |
| FeO | 2.94 | 1.14 |
| Mass Loss (%) | |||
|---|---|---|---|
| Curing Time | 35–200 °C | 200–400 °C | 35–600 °C |
| 28 days | 1.09 | 2.25 | 3.97 |
| 120 days | 1.20 | 2.65 | 4.49 |
| Figure | Spot | CaO | MgO | SiO2 | Al2O3 | K2O | Fe2O3 | P2O5 | SO3 |
|---|---|---|---|---|---|---|---|---|---|
| Figure 7a | C | 82.50 | 3.43 | 6.86 | 3.97 | 2.14 | 1.10 | 0.00 | 0.00 |
| Figure 7a | D | 60.20 | 35.70 | 1.73 | 0.88 | 0.49 | 1.00 | 0.00 | 0.00 |
| Figure 7a | Ga | 38.6 | 7.8 | 28.6 | 14.6 | 6.31 | 1.97 | 0.58 | 1.14 |
| Figure 7a | Gb | 26.00 | 3.80 | 35.20 | 16.40 | 9.80 | 6.61 | 0.00 | 0.81 |
| Figure 8a | Several | 28.7–35.7 | 19.5–27.0 | 13.7–20.7 | 17.5–20.2 | 4.8–7.4 | 0.0–3.5 | 0.0–0.6 | 0.0–0.9 |
| Figure 8d | Several | 13.3–17.2 | 29.8–34.2 | 15.2–15.3 | 21.3–23.9 | 6.8–8.0 | 1.2–2.9 | 0.00 | 1.8–2.5 |
| Parameter | Energy | OPC | OBA | BFS | Soil/OPC * | Soil/OBA+BFS # |
|---|---|---|---|---|---|---|
| CO2 emission (kg/ton) | 0.907 | 0.0192 | ||||
| CO2 emission (kg/kWh) | 0.272 | |||||
| Grinding process(kWh/ton) | 35.4 | 35.4 | ||||
| CO2 emission (kg/ton) | 82.5 | 3.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Payá, J.; Monzó, J.; Roselló, J.; Borrachero, M.V.; Font, A.; Soriano, L. Sustainable Soil-Compacted Blocks Containing Blast Furnace Slag (BFS) Activated with Olive Stone BIOMASS Ash (OBA). Sustainability 2020, 12, 9824. https://doi.org/10.3390/su12239824
Payá J, Monzó J, Roselló J, Borrachero MV, Font A, Soriano L. Sustainable Soil-Compacted Blocks Containing Blast Furnace Slag (BFS) Activated with Olive Stone BIOMASS Ash (OBA). Sustainability. 2020; 12(23):9824. https://doi.org/10.3390/su12239824
Chicago/Turabian StylePayá, Jordi, José Monzó, Josefa Roselló, María Victoria Borrachero, Alba Font, and Lourdes Soriano. 2020. "Sustainable Soil-Compacted Blocks Containing Blast Furnace Slag (BFS) Activated with Olive Stone BIOMASS Ash (OBA)" Sustainability 12, no. 23: 9824. https://doi.org/10.3390/su12239824
APA StylePayá, J., Monzó, J., Roselló, J., Borrachero, M. V., Font, A., & Soriano, L. (2020). Sustainable Soil-Compacted Blocks Containing Blast Furnace Slag (BFS) Activated with Olive Stone BIOMASS Ash (OBA). Sustainability, 12(23), 9824. https://doi.org/10.3390/su12239824









