Domestic Gardens Mitigate Risk of Exposure of Pollinators to Pesticides—An Urban-Rural Case Study Using a Red Mason Bee Species for Biomonitoring
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Bee Species
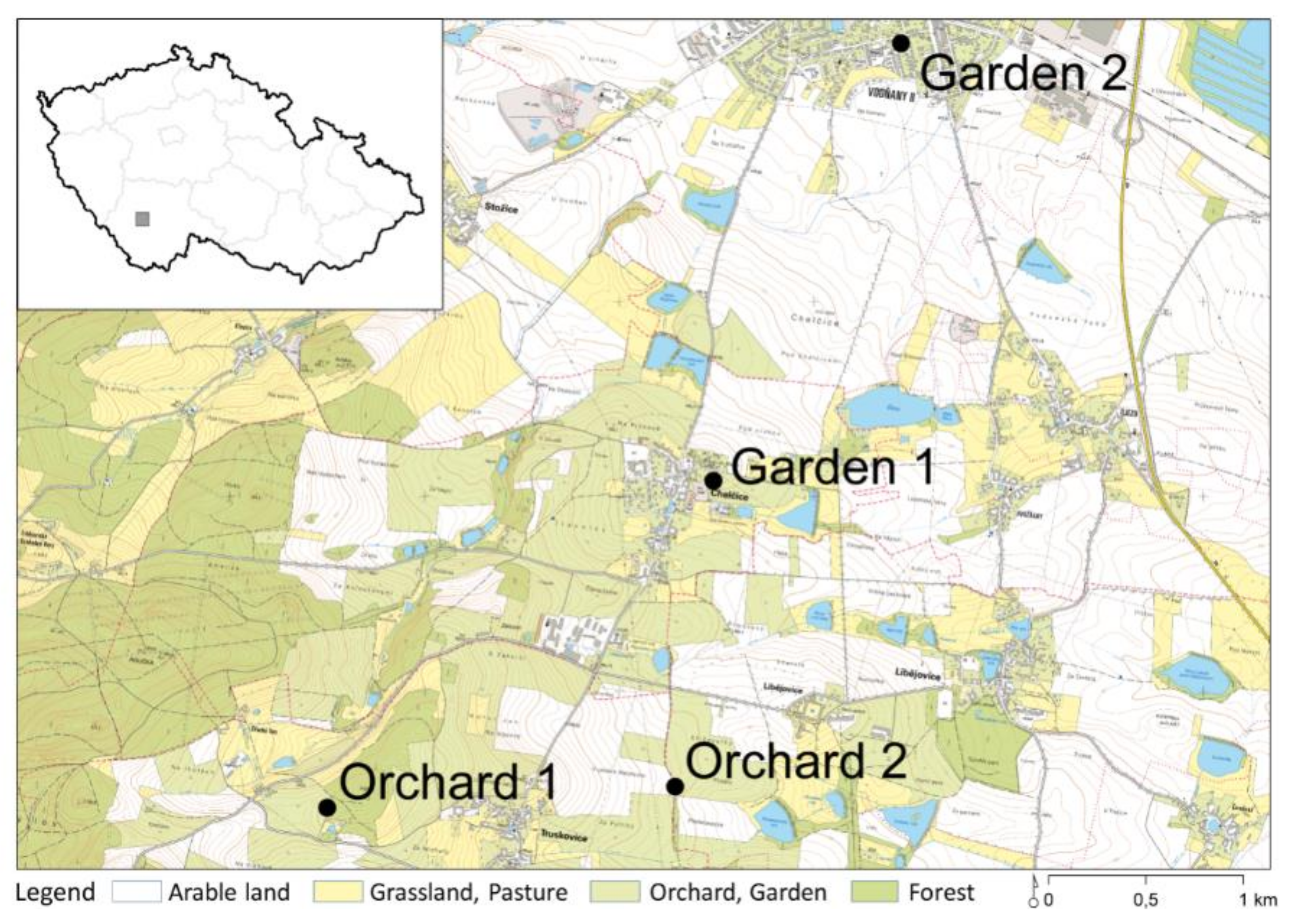
2.2. Study Sites
2.3. Collection of Pollen Samples
2.4. Palynological Analyses
2.5. Pesticide Analyses
2.6. Statistical Evaluation
3. Results
3.1. Composition of Pesticide Residues
3.2. Quantitative Comparisons of Pesticides
3.3. Plant Species Composition of Pollen
4. Discussion
4.1. Rates of Pesticide-Contamination in Gardens vs. Orchards
4.2. Source of Exposure to Pesticides
4.3. Implication for Bee Health
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Straka, J.; Bogusch, P. Anthophila. In Red List of Threatened Species of the Czech Republic. Invertebrates; Hejda, R., Farkač, J., Chobot, K., Eds.; Nature Conservation Agency of the Czech Republic: Prague, Czech Republic, 2017; Volume 36, pp. 236–249. Available online: https://www.ochranaprirody.cz/res/archive/372/058766.pdf (accessed on 26 September 2020).
- Biesmeijer, J.C.; Roberts, S.P.M.; Reemer, M.; Ohlemüller, R.; Edwards, M.; Peeters, T.; Schaffers, A.P.; Potts, S.G.; Kleukers, R.; Thomas, C.D.; et al. Parallel declines in pollinators and insect-pollinated plants in Britain and the Netherlands. Science 2006, 313, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Carvalheiro, L.G.; Kunin, W.E.; Keil, P.; Aguirre-Gutiérrez, J.; Ellis, W.N.; Fox, R.; Groom, Q.; Hennekens, S.; Van Landuyt, W.; Maes, D.; et al. Species richness declines and biotic homogenisation have slowed down for NW -European pollinators and plants. Ecol. Lett. 2013, 16, 870–878. [Google Scholar] [CrossRef] [PubMed]
- Ollerton, J.; Erenler, H.; Edwards, M.; Crockett, R. Extinctions of aculeate pollinators in Britain and the role of large-scale agricultural changes. Science 2014, 346, 1360–1362. [Google Scholar] [CrossRef]
- Goulson, D.; Nicholls, E.; Botías, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 2015, 347, 1255957. [Google Scholar] [CrossRef]
- Powney, G.D.; Carvell, C.; Edwards, M.; Morris, R.K.A.; Roy, H.E.; Woodcock, B.A.; Isaac, N.J.B. Widespread losses of pollinating insects in Britain. Nat. Commun. 2019, 10, 1–6. [Google Scholar] [CrossRef]
- Klein, A.-M.; Vaissière, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B Biol. Sci. 2007, 274, 303–313. [Google Scholar] [CrossRef]
- Garibaldi, L.A.; Steffan-Dewenter, I.; Winfree, R.; Aizen, M.A.; Bommarco, R.; Cunningham, S.A.; Kremen, C.; Carvalheiro, L.G.; Harder, L.D.; Afik, O.; et al. Wild pollinators enhance fruit set of crops regardless of honey bee abundance. Science 2013, 339, 1608–1611. [Google Scholar] [CrossRef]
- Matteson, K.C.; Ascher, J.S.; Langellotto, G.A. Bee Richness and abundance in New York City urban gardens. Ann. Entomol. Soc. Am. 2008, 101, 140–150. [Google Scholar] [CrossRef]
- Osborne, J.L.; Martin, A.P.; Shortall, C.R.; Todd, A.D.; Goulson, D.; Knight, M.E.; Hale, R.J.; Sanderson, R.A. Quantifying and comparing bumblebee nest densities in gardens and countryside habitats. J. Appl. Ecol. 2007, 45, 784–792. [Google Scholar] [CrossRef]
- Samnegård, U.; Persson, A.S.; Smith, H.G. Gardens benefit bees and enhance pollination in intensively managed farmland. Biol. Conserv. 2011, 144, 2602–2606. [Google Scholar] [CrossRef]
- Baldock, K.C.R.; Goddard, M.A.; Hicks, D.M.; Kunin, W.E.; Mitschunas, N.; Osgathorpe, L.M.; Potts, S.G.; Robertson, K.M.; Scott, A.V.; Stone, G.N.; et al. Where is the UK’s pollinator biodiversity? The importance of urban areas for flower-visiting insects. Proc. R. Soc. B Biol. Sci. 2015, 282, 20142849. [Google Scholar] [CrossRef] [PubMed]
- Langellotto, G.A.; Melathopoulos, A.P.; Messer, I.; Anderson, A.; McClintock, N.; Costner, L. Garden pollinators and the potential for ecosystem service flow to urban and peri-urban agriculture. Sustainability 2018, 10, 2047. [Google Scholar] [CrossRef]
- Ackerman, K.; Conard, M.; Culligan, P.; Plunz, R.; Sutto, M.-P.; Whittinghill, L. Sustainable food systems for future cities: The potential of urban agriculture. Econ. Soc. Rev. 2014, 45, 189–206. [Google Scholar]
- Lowenstein, D.M.; Matteson, K.C.; Minor, E.S. Diversity of wild bees supports pollination services in an urbanized landscape. Oecologia 2015, 179, 811–821. [Google Scholar] [CrossRef]
- Van Tuijl, E.; Hospers, G.-J.; Berg, L.V.D. Opportunities and challenges of urban agriculture for sustainable city development. Eur. Spat. Res. Policy 2018, 25, 5–22. [Google Scholar] [CrossRef]
- Botías, C.; David, A.; Horwood, J.; Abdulsada, A.K.; Nicholls, E.; Hill, E.M.; Goulson, D. Neonicotinoid residues in wildflowers, a potential route of chronic exposure for bees. Environ. Sci. Technol. 2015, 49, 12731–12740. [Google Scholar] [CrossRef]
- David, A.; Botías, C.; Abdul-Sada, A.; Nicholls, E.; Rotheray, E.L.; Hill, E.M.; Goulson, D. Widespread contamination of wildflower and bee-collected pollen with complex mixtures of neonicotinoids and fungicides commonly applied to crops. Environ. Int. 2016, 88, 169–178. [Google Scholar] [CrossRef]
- Long, E.Y.; Krupke, C.H. Non-cultivated plants present a season-long route of pesticide exposure for honey bees. Nat. Commun. 2016, 7, 11629. [Google Scholar] [CrossRef]
- Wood, T.J.; Goulson, D. The environmental risks of neonicotinoid pesticides: A review of the evidence post 2013. Environ. Sci. Pollut. Res. 2017, 24, 17285–17325. [Google Scholar] [CrossRef]
- Girolami, V.; Mazzon, L.; Squartini, A.; Mori, N.; Marzaro, M.; Di Bernardo, A.; Greatti, M.; Giorio, C.; Tapparo, A. Translocation of neonicotinoid insecticides from coated seeds to seedling guttation drops: A novel way of intoxication for bees. J. Econ. Entomol. 2009, 102, 1808–1815. [Google Scholar] [CrossRef] [PubMed]
- Kopit, A.M.; Pitts-Singer, T.L. Routes of pesticide exposure in solitary, cavity-nesting bees. Environ. Entomol. 2018, 47, 499–510. [Google Scholar] [CrossRef]
- Anderson, N.L.; Harmon-Threatt, A.N. Chronic contact with realistic soil concentrations of imidacloprid affects the mass, immature development speed, and adult longevity of solitary bees. Sci. Rep. 2019, 9, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Sgolastra, F.; Hinarejos, S.; Pitts-Singer, T.L.; Boyle, N.K.; Joseph, T.; Lūckmann, J.; Raine, N.E.; Singh, R.; Williams, N.M.; Bosch, J. Pesticide exposure assessment paradigm for solitary bees. Environ. Entomol. 2019, 48, 22–35. [Google Scholar] [CrossRef] [PubMed]
- Thompson, H.M. Risk assessment for honey bees and pesticides-recent developments and ‘new issues’. Pest. Manag. Sci. 2010, 66, 1157–1162. [Google Scholar] [CrossRef]
- Sánchez-Bayo, F.; Goulson, D.; Pennacchio, F.; Nazzi, F.; Goka, K.; Desneux, N. Are bee diseases linked to pesticides?–A brief review. Environ. Int. 2016, 89–90, 7–11. [Google Scholar] [CrossRef]
- Alkassab, A.T.; Kirchner, W.H. Sublethal exposure to neonicotinoids and related side effects on insect pollinators: Honeybees, bumblebees, and solitary bees. J. Plant. Dis. Prot. 2017, 124, 1–30. [Google Scholar] [CrossRef]
- Yoder, J.A.; Jajack, A.J.; Rosselot, A.E.; Smith, T.J.; Yerke, M.C.; Sammataro, D. Fungicide contamination reduces beneficial fungi in bee bread based on an area-wide field study in honey bee, Apis mellifera, colonies. J. Toxicol. Environ. Heal. Part. A 2013, 76, 587–600. [Google Scholar] [CrossRef]
- Kakumanu, M.L.; Reeves, A.M.; Anderson, T.D.; Rodrigues, R.R.; Williams, M.A. Honey bee gut microbiome is altered by in-hive pesticide exposures. Front. Microbiol. 2016, 7, 1255. [Google Scholar] [CrossRef]
- Rouzé, R.; Moné, A.; Delbac, F.; Belzunces, L.; Blot, N. The honeybee gut microbiota is altered after chronic exposure to different families of insecticides and infection by Nosema ceranae. Microbes Environ. 2019, 34, 226–233. [Google Scholar] [CrossRef]
- Dai, P.; Yan, Z.; Ma, S.; Yang, Y.; Wang, Q.; Hou, C.; Wu, Y.; Liu, Y.; Diao, Q. The herbicide glyphosate negatively affects midgut bacterial communities and survival of honey bee during larvae reared in vitro. J. Agric. Food Chem. 2018, 66, 7786–7793. [Google Scholar] [CrossRef] [PubMed]
- Motta, E.V.S.; Raymann, K.; Moran, N.A. Glyphosate perturbs the gut microbiota of honey bees. Proc. Natl. Acad. Sci. USA 2018, 115, 10305–10310. [Google Scholar] [CrossRef] [PubMed]
- Daisley, B.A.; Chmiel, J.A.; Pitek, A.P.; Thompson, G.J.; Reid, G. Missing microbes in bees: How systematic depletion of key symbionts erodes immunity. Trends Microbiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- López, J.H.; Krainer, S.; Engert, A.; Schuehly, W.; Riessberger-Gallé, U.; Crailsheim, K. Sublethal pesticide doses negatively affect survival and the cellular responses in American foulbrood-infected honeybee larvae. Sci. Rep. 2017, 7, srep40853. [Google Scholar] [CrossRef]
- DeGrandi-Hoffman, G.; Chen, Y.P.; Simonds, R. The effects of pesticides on queen rearing and virus titers in honey bees (Apis mellifera L.). Insects 2013, 4, 71–89. [Google Scholar] [CrossRef]
- Simon-Delso, N.; Martin, G.S.; Bruneau, E.; Minsart, L.-A.; Mouret, C.; Hautier, L. Honeybee colony disorder in crop areas: The role of pesticides and viruses. PLoS ONE 2014, 9, e103073. [Google Scholar] [CrossRef]
- Straub, L.; Williams, G.R.; Vidondo, B.; Khongphinitbunjong, K.; Retschnig, G.; Schneeberger, A.; Chantawannakul, P.; Dietemann, V.; Neumann, P. Neonicotinoids and ectoparasitic mites synergistically impact honeybees. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Potts, S.G.; Imperatriz-Fonseca, V.; Ngo, H.T.; Aizen, M.A.; Biesmeijer, J.C.; Breeze, S.G.P.T.D.; Dicks, L.V.; Garibaldi, L.A.; Hill, R.; Settele, J.; et al. Safeguarding pollinators and their values to human well-being. Nat. Cell Biol. 2016, 540, 220–229. [Google Scholar] [CrossRef]
- Sanchez-Bayo, F.; Goka, K. Pesticide residues and bees-A risk assessment. PLoS ONE 2014, 9, e94482. [Google Scholar] [CrossRef]
- Feltham, H.; Park, K.; Goulson, D. Field realistic doses of pesticide imidacloprid reduce bumblebee pollen foraging efficiency. Ecotoxicology 2014, 23, 317–323. [Google Scholar] [CrossRef]
- Sandrock, C.; Tanadini, L.G.; Pettis, J.S.; Biesmeijer, J.C.; Potts, S.G.; Neumann, P.G. Sublethal neonicotinoid insecticide exposure reduces solitary bee reproductive success. Agric. For. Entomol. 2013, 16, 119–128. [Google Scholar] [CrossRef]
- Rundlöf, M.; Andersson, G.K.S.; Bommarco, R.; Fries, I.; Hederström, V.; Herbertsson, L.; Jonsson, O.; Klatt, B.K.; Pedersen, T.R.; Yourstone, J.; et al. Seed coating with a neonicotinoid insecticide negatively affects wild bees. Nat. Cell Biol. 2015, 521, 77–80. [Google Scholar] [CrossRef] [PubMed]
- Stanley, D.A.; Smith, K.E.; Raine, N.E. Bumblebee learning and memory is impaired by chronic exposure to a neonicotinoid pesticide. Sci. Rep. 2015, 5, 16508. [Google Scholar] [CrossRef] [PubMed]
- Heard, M.S.; Baas, J.; Dorne, J.-L.; Lahive, E.; Robinson, A.G.; Rortais, A.; Spurgeon, D.J.; Svendsen, C.; Hesketh, H. Comparative toxicity of pesticides and environmental contaminants in bees: Are honey bees a useful proxy for wild bee species? Sci. Total. Environ. 2017, 578, 357–365. [Google Scholar] [CrossRef]
- Woodcock, B.A.; Bullock, J.M.; Shore, R.F.; Heard, M.S.; Pereira, M.G.; Redhead, J.; Ridding, L.E.; Dean, H.; Sleep, D.; Henrys, P.A.; et al. Country-specific effects of neonicotinoid pesticides on honey bees and wild bees. Science 2017, 356, 1393–1395. [Google Scholar] [CrossRef]
- Alkassab, A.T.; Kunz, N.; Bischoff, G.; Pistorius, J. Comparing response of buff-tailed bumblebees and red mason bees to application of a thiacloprid-prochloraz mixture under semi-field conditions. Ecotoxicology 2020, 29, 846–855. [Google Scholar] [CrossRef]
- Azpiazu, C.; Bosch, J.; Viñuela, E.; Medrzycki, P.; Teper, D.; Sgolastra, F. Chronic oral exposure to field-realistic pesticide combinations via pollen and nectar: Effects on feeding and thermal performance in a solitary bee. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Robinson, A.; Hesketh, H.; Lahive, E.; Horton, A.A.; Svendsen, C.; Rortais, A.; Dorne, J.L.; Baas, J.; Heard, M.S.; Spurgeon, D.J. Comparing bee species responses to chemical mixtures: Common response patterns? PLoS ONE 2017, 12, e0176289. [Google Scholar] [CrossRef] [PubMed]
- Botías, C.; David, A.; Hill, E.M.; Goulson, D. Quantifying exposure of wild bumblebees to mixtures of agrochemicals in agricultural and urban landscapes. Environ. Pollut. 2017, 222, 73–82. [Google Scholar] [CrossRef]
- Lentola, A.; David, A.; Abdul-Sada, A.; Tapparo, A.; Goulson, D.; Hill, E.M. Ornamental plants on sale to the public are a significant source of pesticide residues with implications for the health of pollinating insects. Environ. Pollut. 2017, 228, 297–304. [Google Scholar] [CrossRef]
- Cameron, R.W.F.; Blanuša, T.; Taylor, J.E.; Salisbury, A.; Halstead, A.J.; Henricot, B.; Thompson, K. The domestic garden-Its contribution to urban green infrastructure. Urban. For. Urban. Green. 2012, 11, 129–137. [Google Scholar] [CrossRef]
- Jehlička, P.; Kostelecký, T.; Smith, J. Food self-provisioning in Czechia: Beyond coping strategy of the poor: A response to Alber and Kohler’s ‘informal food production in the enlarged European Union’ (2008). Soc. Indic. Res. 2012, 111, 219–234. [Google Scholar] [CrossRef]
- Schoen, V.; Caputo, S.; Blythe, C. Valuing physical and social output: A Rapid assessment of a London community garden. Sustainability 2020, 12, 5452. [Google Scholar] [CrossRef]
- Egerer, M.; Cecala, J.M.; Cohen, H. Wild bee conservation within urban gardens and nurseries: Effects of local and landscape management. Sustainability 2019, 12, 293. [Google Scholar] [CrossRef]
- Robbins, P.; Polderman, A.; Birkenholtz, T. Lawns and toxins: An ecology of the city. Cities 2001, 18, 369–380. [Google Scholar] [CrossRef]
- Peters, B.; Gao, Z.; Zumkier, U. Large-scale monitoring of effects of clothianidin-dressed oilseed rape seeds on pollinating insects in Northern Germany: Effects on red mason bees (Osmia bicornis). Ecotoxicology 2016, 25, 1679–1690. [Google Scholar] [CrossRef]
- Rolke, D.; Persigehl, M.; Peters, B.; Sterk, G.; Blenau, W. Large-scale monitoring of effects of clothianidin-dressed oilseed rape seeds on pollinating insects in northern Germany: Residues of clothianidin in pollen, nectar and honey. Ecotoxicology 2016, 25, 1691–1701. [Google Scholar] [CrossRef]
- Ruddle, N.; Elston, C.; Klein, O.; Hamberger, A.; Thompson, H.M. Effects of exposure to winter oilseed rape grown from thiamethoxam-treated seed on the red mason beeOsmia bicornis. Environ. Toxicol. Chem. 2018, 37, 1071–1083. [Google Scholar] [CrossRef]
- Dietzsch, A.C.; Kunz, N.; Wirtz, I.P.; Stähler, M.; Heimbach, U.; Pistorius, J. Does winter oilseed rape grown from clothianidin-coated seeds affect experimental populations of mason bees and bumblebees? A semi-field and field study. J. Consum. Prot. Food Saf. 2019, 14, 223–238. [Google Scholar] [CrossRef]
- Eeraerts, M.; Pisman, M.; Vanderhaegen, R.; Meeus, I.; Smagghe, G. Recommendations for standardized oral toxicity test protocols for larvae of solitary bees, Osmia spp. Apidologie 2019, 51, 48–60. [Google Scholar] [CrossRef]
- Raw, A. The biology of the solitary bee Osmia rufa (L.) (Megachilidae). Ecol. Entomol. 2009, 124, 213–229. [Google Scholar] [CrossRef]
- Raw, A. Pollen preferences of three osmia species (Hymenoptera). Oikos 1974, 25, 54. [Google Scholar] [CrossRef]
- Radmacher, S.; Strohm, E. Factors affecting offspring body size in the solitary bee Osmia bicornis (Hymenoptera, Megachilidae). Apidologie 2009, 41, 169–177. [Google Scholar] [CrossRef]
- Jauker, F.; Peter, F.; Wolters, V.; Diekötter, T. Early reproductive benefits of mass-flowering crops to the solitary bee Osmia rufa outbalance post-flowering disadvantages. Basic Appl. Ecol. 2012, 13, 268–276. [Google Scholar] [CrossRef]
- Holzschuh, A.; Dormann, C.F.; Tscharntke, T.; Steffan-Dewenter, I. Mass-flowering crops enhance wild bee abundance. Oecologia 2013, 172, 477–484. [Google Scholar] [CrossRef]
- Persson, A.S.; Mazier, F.; Smith, H.G. When beggars are choosers-How nesting of a solitary bee is affected by temporal dynamics of pollen plants in the landscape. Ecol. Evol. 2018, 8, 5777–5791. [Google Scholar] [CrossRef]
- Gathmann, A.; Tscharntke, T. Foraging ranges of solitary bees. J. Anim. Ecol. 2002, 71, 757–764. [Google Scholar] [CrossRef]
- Hofmann, M.M.; Fleischmann, A.; Renner, S.S. Foraging distances in six species of solitary bees with body lenghts of 6 to 15 mm, inferred from individual tagging, suggest 150 m-rule-of-thumb for flower strip distances. J. Hymenopt. Res. 2020, 77, 105–117. [Google Scholar] [CrossRef]
- ČÚZK (Czech Office for Surveying, Mapping and Cadastre). Base Map of the Czech Republic 1:10,000: WMS View Service-ZM 10. Available online: https://geoportal.cuzk.cz/(S(naqemgklqsek3b3ydggftiqe))/Default.aspx?menu=3115&mode=TextMeta&side=wms.verejne&metadataID=CZ-CUZK-WMS-ZM10-P&metadataXSL=metadata.sluzba (accessed on 25 September 2020).
- Faegri, K.; Iversen, J. Textbook of Pollen Analysis, 4th ed.; John Wiley and Sons: Chichester, UK, 1989; p. 328. [Google Scholar]
- Beug, H.-J. Leitfaden der Pollenbestimmung für Mitteleuropa und angrenzende Gebiete, 1st ed.; Verlag Dr. Friedrich Pfeil: München, Germany, 2004; p. 542. [Google Scholar]
- Anastassiades, M.; Lehotay, S.J.; Štajnbaher, D.; Schenck, F.J. Fast and easy multiresidue method employing acetonitrile extraction/partitioning and “dispersive solid-phase extraction” for the determination of pesticide residues in produce. J. AOAC Int. 2003, 86, 412–431. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing. Vienna, Austria, 2019. Available online: http://www.R-project.org/ (accessed on 25 September 2020).
- Kasiotis, K.M.; Anagnostopoulos, C.; Anastasiadou, P.; Machera, K. Pesticide residues in honeybees, honey and bee pollen by LC–MS/MS screening: Reported death incidents in honeybees. Sci. Total. Environ. 2014, 633–642. [Google Scholar] [CrossRef]
- Calatayud-Vernich, P.; Calatayud, F.; Simó, E.; Picó, Y. Occurrence of pesticide residues in Spanish beeswax. Sci. Total. Environ. 2017, 605, 745–754. [Google Scholar] [CrossRef] [PubMed]
- Tosi, S.; Costa, C.; Vesco, U.; Quaglia, G.; Guido, G. A 3-year survey of Italian honey bee-collected pollen reveals widespread contamination by agricultural pesticides. Sci. Total. Environ. 2018, 615, 208–218. [Google Scholar] [CrossRef]
- Böhme, F.; Bischoff, G.; Zebitz, C.P.W.; Rosenkranz, P.; Wallner, K. Pesticide residue survey of pollen loads collected by honeybees (Apis mellifera) in daily intervals at three agricultural sites in South Germany. PLoS ONE 2018, 13, e0199995. [Google Scholar] [CrossRef] [PubMed]
- Hladik, M.L.; Vandever, M.; Smalling, K.L. Exposure of native bees foraging in an agricultural landscape to current-use pesticides. Sci. Total. Environ. 2016, 542, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Thompson, H.M.; Fryday, S.L.; Harkin, S.; Milner, S. Potential impacts of synergism in honeybees (Apis mellifera) of exposure to neonicotinoids and sprayed fungicides in crops. Apidologie 2014, 45, 545–553. [Google Scholar] [CrossRef]
- Schmuck, R.; Stadler, T.; Schmidt, H.-W. Field relevance of a synergistic effect observed in the laboratory between an EBI fungicide and a chloronicotinyl insecticide in the honeybee (Apis mellifera L., Hymenoptera). Pest. Manag. Sci. 2003, 59, 279–286. [Google Scholar] [CrossRef]
- Iwasa, T.; Motoyama, N.; Ambrose, J.T.; Roe, R.M. Mechanism for the differential toxicity of neonicotinoid insecticides in the honey bee, Apis mellifera. Crop. Prot. 2004, 23, 371–378. [Google Scholar] [CrossRef]
- Zhu, W.; Schmehl, D.R.; Mullin, C.A.; Frazier, J.L. Four common pesticides, their mixtures and a formulation solvent in the hive environment have high oral toxicity to honey bee larvae. PLoS ONE 2014, 9, e77547. [Google Scholar] [CrossRef]
- Biddinger, D.J.; Robertson, J.L.; Mullin, C.; Frazier, J.; Ashcraft, S.A.; Rajotte, E.G.; Joshi, N.K.; Vaughn, M. Comparative toxicities and synergism of apple orchard pesticides to Apis mellifera (L.) and Osmia cornifrons (Radoszkowski). PLoS ONE 2013, 8, e72587. [Google Scholar] [CrossRef]
- Sgolastra, F.; Medrzycki, P.; Bortolotti, L.; Renzi, M.T.; Tosi, S.; Bogo, G.; Teper, D.; Porrini, C.; Molowny-Horas, R.; Bosch, J. Synergistic mortality between a neonicotinoid insecticide and an ergosterol-biosynthesis-inhibiting fungicide in three bee species. Pest. Manag. Sci. 2016, 73, 1236–1243. [Google Scholar] [CrossRef]
- Sgolastra, F.; Arnan, X.; Cabbri, R.; Isani, G.; Medrzycki, P.; Teper, D.; Bosch, J. Combined exposure to sublethal concentrations of an insecticide and a fungicide affect feeding, ovary development and longevity in a solitary bee. Proc. R. Soc. B Biol. Sci. 2018, 285, 20180887. [Google Scholar] [CrossRef] [PubMed]
- Steffan-Dewenter, I.; Kuhn, A. Honeybee foraging in differentially structured landscapes. Proc. R. Soc. B Biol. Sci. 2003, 270, 569–575. [Google Scholar] [CrossRef] [PubMed]
- Wolf, S.; Moritz, R.F.A. Foraging distance in Bombus terrestris L. (Hymenoptera: Apidae). Apidologie 2008, 39, 419–427. [Google Scholar] [CrossRef]
- Redhead, J.W.; Dreier, S.; Bourke, A.F.G.; Heard, M.S.; Jordan, W.C.; Sumner, S.; Wang, J.; Carvell, C. Effects of habitat composition and landscape structure on worker foraging distances of five bumble bee species. Ecol. Appl. 2016, 26, 726–739. [Google Scholar] [CrossRef] [PubMed]
- Jehlička, P.; Daněk, P.; Vávra, J. Rethinking resilience: Home gardening, food sharing and everyday resistance. Can. J. Dev. Stud. 2018, 40, 511–527. [Google Scholar] [CrossRef]
- Jehlička, P.; Smith, J. Sustainability and the “urban peasant”: Rethinking the cultural politics of food self-provisioning in the Czech Republic. In New Perspectives on Consumer Culture Theory and Research; Zahrádka, P., Sedláková, R., Eds.; Cambridge Scholars Publishing: Newcastle upon Tyne, UK, 2012; pp. 78–96. [Google Scholar]
- Russo, L.; Park, M.; Blitzer, E.; Danforth, B. Flower handling behavior and abundance determine the relative contribution of pollinators to seed set in apple orchards. Agric. Ecosyst. Environ. 2017, 246, 102–108. [Google Scholar] [CrossRef]
- Krupke, C.H.; Hunt, G.J.; Eitzer, B.D.; Andino, G.; Given, K. Multiple routes of pesticide exposure for honey bees living near agricultural fields. PLoS ONE 2012, 7, e29268. [Google Scholar] [CrossRef]
- McArt, S.H.; Fersch, A.A.; Milano, N.J.; Truitt, L.L.; Böröczky, K. High pesticide risk to honey bees despite low focal crop pollen collection during pollination of a mass blooming crop. Sci. Rep. 2017, 7, srep46554. [Google Scholar] [CrossRef]
- Bonmatin, J.-M.; Giorio, C.; Girolami, V.; Goulson, D.; Kreutzweiser, D.P.; Krupke, C.; Liess, M.; Long, E.; Marzaro, M.; Mitchell, E.A.D.; et al. Environmental fate and exposure; neonicotinoids and fibronil. Environ. Sci. Pollut. Res. 2015, 22, 35–67. [Google Scholar] [CrossRef]
- Vijver, M.G.; Hunting, E.R.; Nederstigt, T.A.; Tamis, W.L.; Brink, P.V.D.; Van Bodegom, P.M. Postregistration monitoring of pesticides is urgently required to protect ecosystems. Environ. Toxicol. Chem. 2017, 36, 860–865. [Google Scholar] [CrossRef]
- Hvězdová, M.; Kosubová, P.; Košíková, M.; Scherr, K.E.; Šimek, Z.; Brodský, L.; Šudoma, M.; Škulcová, L.; Sáňka, M.; Svobodová, M.; et al. Currently and recently used pesticides in Central European arable soils. Sci. Total. Environ. 2018, 361–370. [Google Scholar] [CrossRef] [PubMed]
- Silva, V.; Mol, H.G.J.; Zomer, P.; Tienstra, M.; Ritsema, C.J.; Geissen, V. Pesticide residues in European agricultural soil-A hidden reality unfolded. Sci. Total Environ. 2019, 653, 1532–1545. [Google Scholar] [CrossRef] [PubMed]
- Artz, D.R.; Pitts-Singer, T.L. Effects of fungicide and adjuvant sprays on nesting behavior in two managed solitary bees, Osmia lignaria and Megachile rotundata. PLoS ONE 2015, 10, e0135688. [Google Scholar] [CrossRef] [PubMed]
- EC (European Commission). Commission Implementing Regulation (EU) 2018/1865 of 28 November 2018 concerning the non-renewal of approval of the active substance propiconazole, in accordance with Regulation (EC) No 1107/2009 of the European Parliament and of the Council concerning the placing of plant protection products on the market, and amending Commission Implementing Regulation (EU) No 540/2011. Off. J. Eur. Union 2018, 304, 6–9. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?qid=1543505797502&uri=CELEX:32018R1865 (accessed on 25 September 2020).
- Rondeaux, G.; Sánchez-Bayo, F.; Tennekes, H.A.; Decourtye, A.; Ramírez-Romero, R.; Desneux, N. Delayed and time-cumulative toxicity of imidacloprid in bees, ants and termites. Sci. Rep. 2014, 4, srep05566. [Google Scholar] [CrossRef]
- Crall, J.D.; Switzer, C.M.; Oppenheimer, R.L.; Versypt, A.N.F.; Dey, B.; Brown, A.; Eyster, M.; Guérin, C.; Pierce, N.E.; Combes, S.A.; et al. Neonicotinoid exposure disrupts bumblebee nest behavior, social networks, and thermoregulation. Science 2018, 362, 683–686. [Google Scholar] [CrossRef]
- Erban, T.; Sopko, B.; Talacko, P.; Harant, K.; Kadlikova, K.; Halesova, T.; Riddellova, K.; Pekas, A. Chronic exposure of bumblebees to neonicotinoid imidacloprid suppresses the entire mevalonate pathway and fatty acid synthesis. J. Proteom. 2019, 196, 69–80. [Google Scholar] [CrossRef]
- EC (European Commission). Final Renewal Report for the Active Substance Thiacloprid Finalised in the Standing Committee on Plants, Animals, Food and Feed at Its Meeting on 22 October 2019 in View of the Non-Renewal of the Approval of Thiacloprid as Active Substance in Accordance with Regulation (EC) No 1107/2009. Thiacloprid, SANTE/10450/2019 Rev. 1, 22 October 2019; Directorate-General for Health and Food Safety: Bruxelles, Belgium, 2019; Available online: https://ec.europa.eu/food/plant/pesticides/eu-pesticides-database/public/?event=activesubstance.ViewReview&id=1788 (accessed on 25 September 2020).
- EC (European Commission). Commission Implementing Regulation (EU) 2020/18 of 10 January 2020 concerning the non-renewal of the approval of the active substance chlorpyrifos, in accordance with Regulation (EC) No 1107/2009 of the European Parliament and of the Council concerning the placing of plant protection products on the market, and amending the Annex to Commission Implementing Regulation (EU) No 540/2011. Off. J. Eur. Union 2020, 7, 14–16. Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?qid=1578929027090&uri=CELEX:32020R0018 (accessed on 25 September 2020).
- EC (European Commission). Final Renewal Report for the Active Substance Methiocarb Finalised in the Standing Committee on Plants, Animals, Food and Feed at Its Meeting on 17 July 2019 in View of the Non-Renewal of the Approval of Methiocarb as Active Substance in Accordance with Regulation (EC) No 1107/2009. Methiocarb, SANTE/11710/2018 Rev 1, 17 July 2019; Directorate-General for Health and Food Safety: Bruxelles, Belgium, 2019; Available online: https://ec.europa.eu/food/plant/pesticides/eu-pesticides-database/public/?event=activesubstance.ViewReview&id=1730 (accessed on 25 September 2020).
- ČÚZK (Czech Office for Surveying, Mapping and Cadastre). Orthophotos of the Czech Republic Recent Data: WMS View Service-Orthophoto. Available online: https://geoportal.cuzk.cz/(S(naqemgklqsek3b3ydggftiqe))/Default.aspx?lng=EN&menu=3121&mode=TextMeta&side=wms.verejne&metadataID=CZ-CUZK-WMS-ORTOFOTO-P&metadataXSL=metadata.sluzba (accessed on 25 September 2020).


| Site | Nest | Concentration of Pesticides (ppb) | Composition of Pollen (%) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pyrimet. | Boscalid | Tebuc. | Azoxys. | Prochloraz | Terbuthyl. | Acetamip. | Thiaclop. | Methiocarb | Quercus T | PM 1 | Bra. | Other | ||
| G 1 | A | 76.2 | 99.6 | 0.4 | ||||||||||
| B | 417.0 | 13.7 | 12.2 | 65.2 | 0.2 | 34.2 | 0.4 | |||||||
| C | 51.6 | 12.7 | 99.6 | 0.4. | ||||||||||
| G 2 | A | 14.5 | 73.9 | 0.2 | 1.4 | 24.5 | ||||||||
| B | 93.5 | 6.3 | 0.2 | |||||||||||
| O 1 | A | 1677.9 | 1603.1 | 86.9 | 19.7 | 18.5 | 97.0 | 1.6 | 1.0 | 0.4 | ||||
| B | 312.0 | 106.7 | 14.6 | 99.0 | 0.2 | 0.4 | 0.4 | |||||||
| C | 437.3 | 730.8 | 15.7 | 14.8 | 92.0 | 0.2 | 7.6 | 0.2 | ||||||
| D | 254.5 | 1359.5 | 11.4 | 275.0 | 95.3 | 4.7 | ||||||||
| O 2 | A | 631.2 | 31.3 | 27.7 | 290.4 | 10.7 | 10.9 | 83.4 | 0.2 | 16.4 | ||||
| B | 846.7 | 205.6 | 52.9 | 91.0 | 1.4 | 7.6 | ||||||||
| C | 2424.6 | 37.2 | 11.1 | 18.5 | 95.3 | 4.3 | 0.4 | |||||||
| D | 2446.3 | 375.7 | 215.9 | 18.8 | 66.3 | 33.3 | 0.4 | |||||||
| E | 10.2 | 1383.9 | 407.9 | 100.0 | ||||||||||
| Mean 2 | 109.0 (G) | 0.0 (G) | 5.3 (G) | 0.0 (G) | ||||||||||
| 1004.5 (O) | 648.2 (O) | 93.8 (O) | 66.7 (O) | |||||||||||
| M-W 3 | G < O * | G < O ** | G < O * | ns | ||||||||||
| Mean 4 | 14.2 | 22.5 | 16.5 | 7.7 | ||||||||||
| Maxim. | 83.0 | 962.0 | 33.2 | 107.0 | ||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Šlachta, M.; Erban, T.; Votavová, A.; Bešta, T.; Skalský, M.; Václavíková, M.; Halešová, T.; Edwards-Jonášová, M.; Včeláková, R.; Cudlín, P. Domestic Gardens Mitigate Risk of Exposure of Pollinators to Pesticides—An Urban-Rural Case Study Using a Red Mason Bee Species for Biomonitoring. Sustainability 2020, 12, 9427. https://doi.org/10.3390/su12229427
Šlachta M, Erban T, Votavová A, Bešta T, Skalský M, Václavíková M, Halešová T, Edwards-Jonášová M, Včeláková R, Cudlín P. Domestic Gardens Mitigate Risk of Exposure of Pollinators to Pesticides—An Urban-Rural Case Study Using a Red Mason Bee Species for Biomonitoring. Sustainability. 2020; 12(22):9427. https://doi.org/10.3390/su12229427
Chicago/Turabian StyleŠlachta, Martin, Tomáš Erban, Alena Votavová, Tomáš Bešta, Michal Skalský, Marta Václavíková, Taťána Halešová, Magda Edwards-Jonášová, Renata Včeláková, and Pavel Cudlín. 2020. "Domestic Gardens Mitigate Risk of Exposure of Pollinators to Pesticides—An Urban-Rural Case Study Using a Red Mason Bee Species for Biomonitoring" Sustainability 12, no. 22: 9427. https://doi.org/10.3390/su12229427
APA StyleŠlachta, M., Erban, T., Votavová, A., Bešta, T., Skalský, M., Václavíková, M., Halešová, T., Edwards-Jonášová, M., Včeláková, R., & Cudlín, P. (2020). Domestic Gardens Mitigate Risk of Exposure of Pollinators to Pesticides—An Urban-Rural Case Study Using a Red Mason Bee Species for Biomonitoring. Sustainability, 12(22), 9427. https://doi.org/10.3390/su12229427







