Comparative Gate-to-Gate Life Cycle Assessment for the Alkali and Acid Pre-Treatment Step in the Chemical Recycling of Waste Cotton
Abstract
1. Introduction
2. Materials and Methods
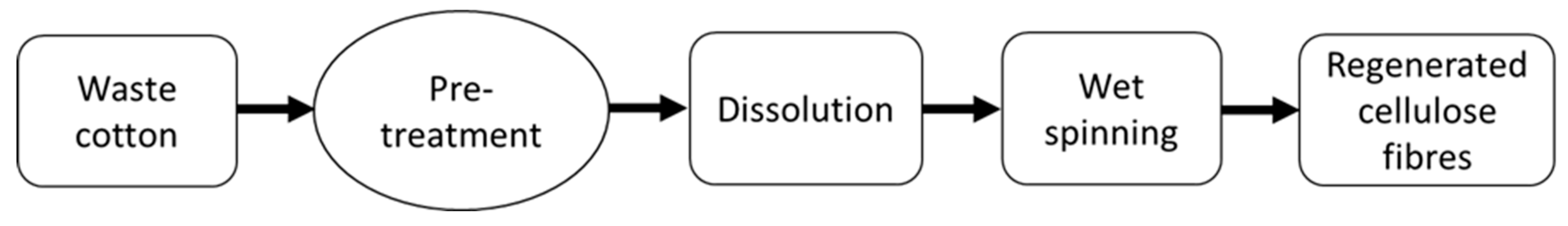
2.1. Pre-Treatment Process
2.1.1. Sodium Hydroxide Pre-Treatment
2.1.2. Sulphuric Acid Pre-Treatment
2.2. Goal Definition
2.3. Scope Definition
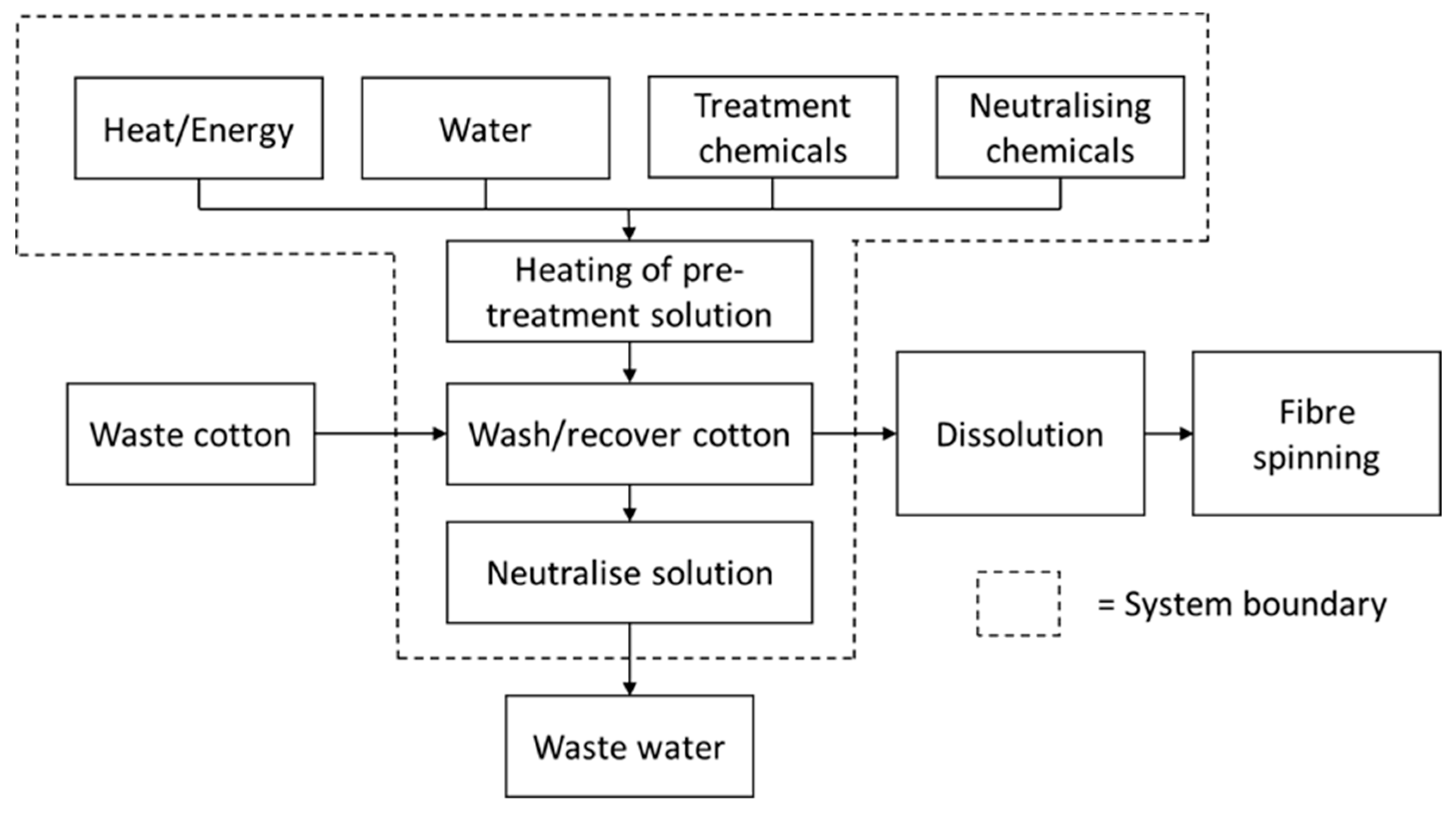
2.4. System Boundaries
2.5. Functional Unit
2.6. Life-Cycle Inventory
2.7. Impact Assessment
- Abiotic depletion of minerals (ADP)—measured in kilograms of antimony equivalents (kg Sb-eq)
- Abiotic depletion of fossil fuels (ADP-ff)—measured in megajoules (MJ)
- Global warming (GWP100a)—measured in kilograms of carbon dioxide equivalents (kg CO2-eq)
- Ozone layer depletion (ODP)—measured in kilograms of CFC-11 equivalents (kg CFC-11-eq)
- Human toxicity (HTP)—measured in kilograms of 1,4-dichlorobenzene equivalents (kg 1,4-DB-eq)
- Freshwater aquatic ecotoxicity (FAETP)—measured in kilograms of 1,4-dichlorobenzene equivalents (kg 1,4-DB-eq)
- Marine aquatic ecotoxicity (MAETP)—measured in kilograms of 1,4-dichlorobenzene equivalents (kg 1,4-DB-eq)
- Terrestrial ecotoxicity (TETP)—measured in kilograms of 1,4-dichlorobenzene equivalents (kg 1,4-DB-eq)
- Photochemical oxidation (PCOP)—measured in kilograms of ethylene equivalents (kg C2H4-eq)
- Acidification (AP)—measured in kilograms of sulfur dioxide equivalents (kg SO2-eq)
- Eutrophication (EP)—measured in kilograms of phosphate equivalents (kg PO43−-eq).
3. Results and Discussion
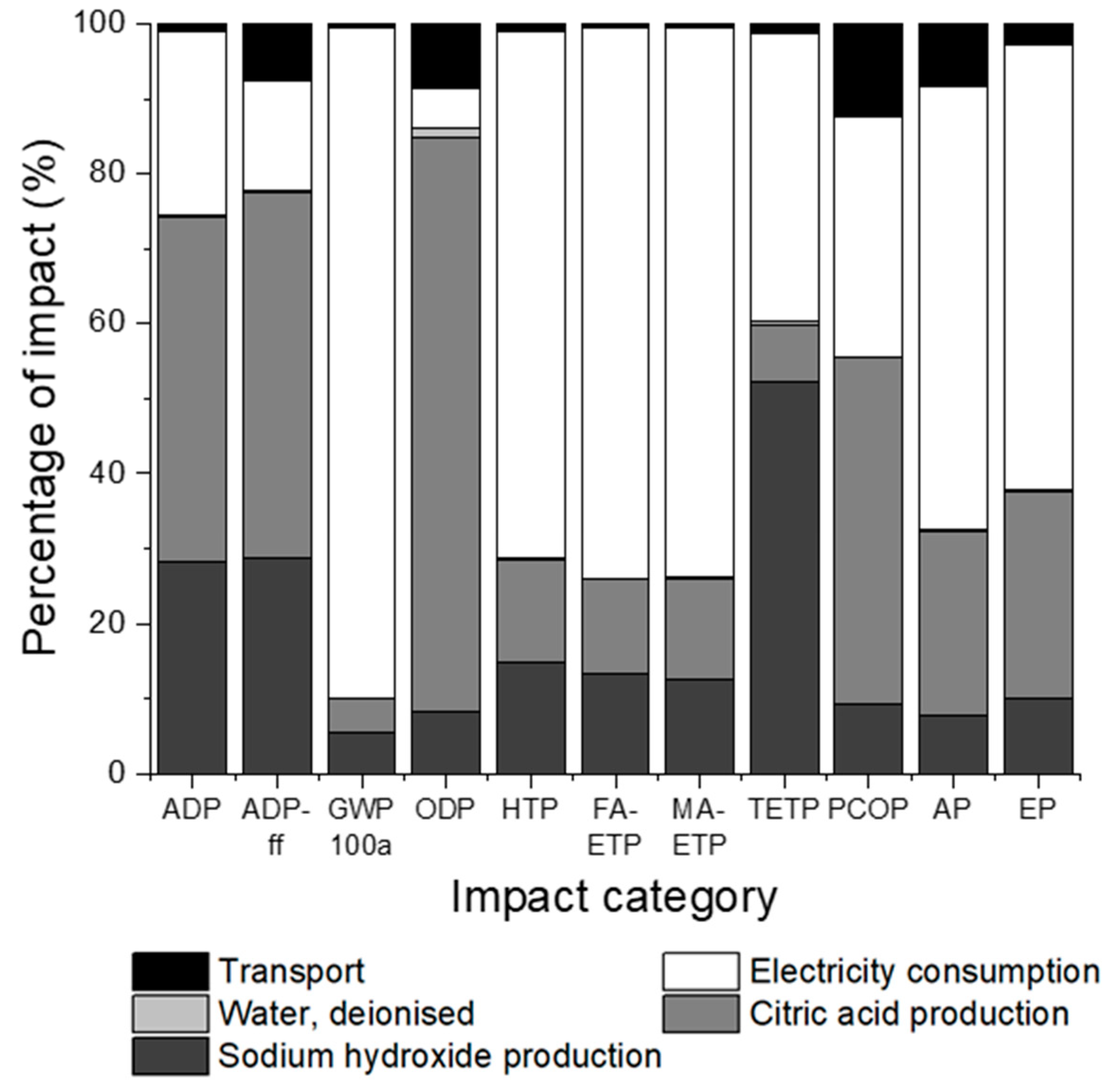
3.1. Impact Assessment for Sodium Hydroxide Pre-Treatment
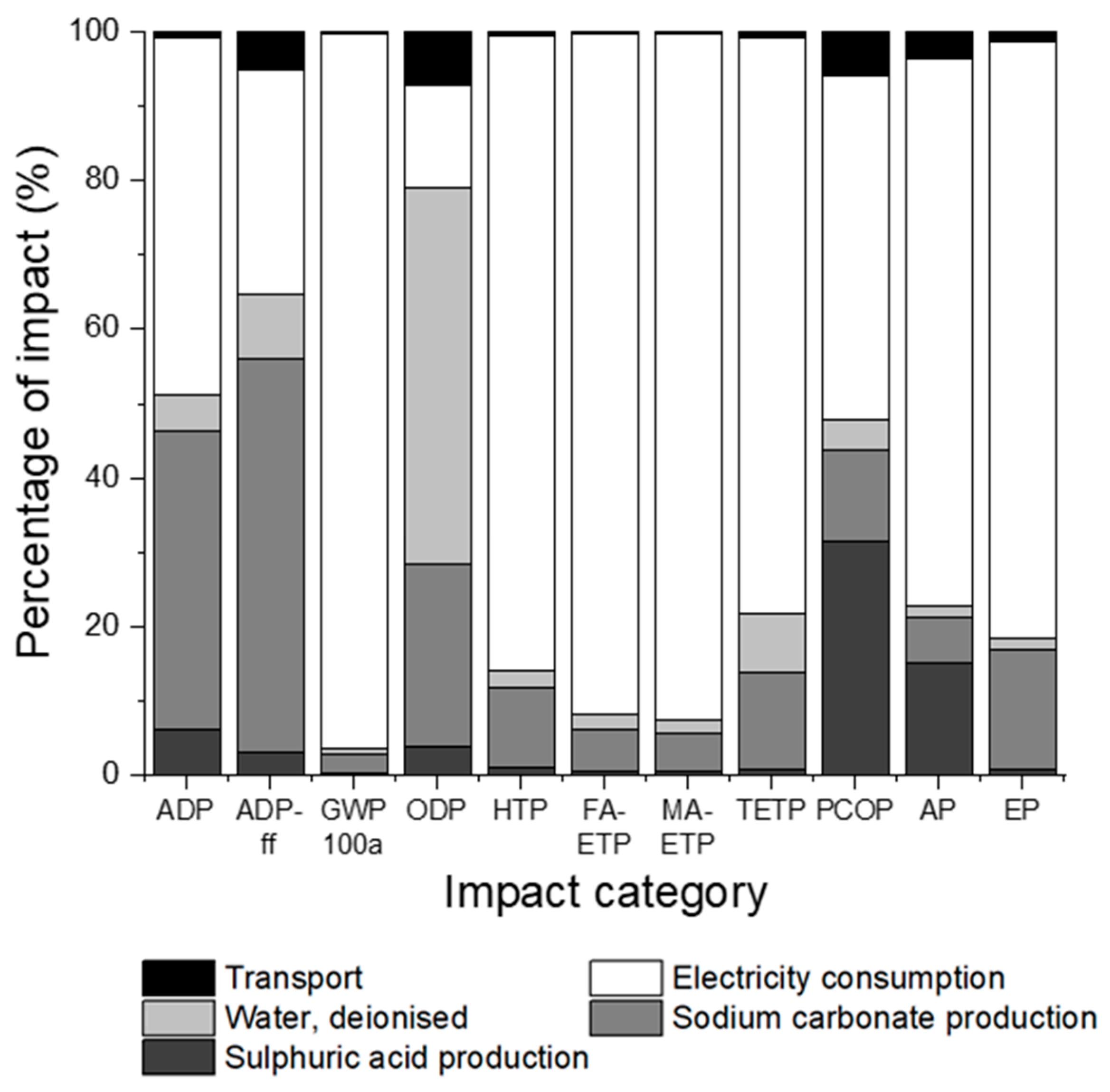
3.2. Impact Assessment for Sulphuric Acid Pre-Treatment
3.3. Comparison of Pre-Treatment Methods
3.4. Normalisation
3.5. Process Improvement and Further Implications
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- The Fiber Year. The Fiber Year 2018. 2018. Available online: www.thefiberyear.com (accessed on 14 March 2019).
- Haemmerle, F.M. The Cellulose Gap (the Future of Cellulose Fibres). In Lenzing—Nature Jeju Festival; Lenzinger Berichte: Jeju, Korea, 2011. [Google Scholar]
- United Nations. World Population Prospects 2019; United Nations: New York, NY, USA, 2019. [Google Scholar]
- Bick, R.; Halsey, E.; Ekenga, C.C. The global environmental injustice of fast fashion. Environ. Health 2018, 17, 92. [Google Scholar] [CrossRef]
- The Fiber Year Consulting. The Fiber Year 2019 World Survey on Textiles and Nonwovens; The Fiber Year GmbH: Speicher, Switzerland, 2019. [Google Scholar]
- Wanassi, B.; Azzouz, B.; Hassen, M.B. Value-added waste cotton yarn: Optimization of recycling process and spinning of reclaimed fibers. Ind. Crops Prod. 2016, 87, 27–32. [Google Scholar] [CrossRef]
- Nurminen, M.; Hernberg, S. Effects of intervention on the cardiovascular mortality of workers exposed to carbon disulphide: A 15 year follow up. Br. J. Ind. Med. 1985, 42, 32. [Google Scholar] [CrossRef] [PubMed]
- Enneking, J.C. Control of carbon disulfide emissions from viscose processes. Environ. Prog. 2002, 21, 169–174. [Google Scholar] [CrossRef]
- Ma, Y.; Hummel, M.; Kontro, I.; Sixta, H. High performance man-made cellulosic fibres from recycled newsprint. Green Chem. 2018, 20, 160–169. [Google Scholar] [CrossRef]
- White, P. Lyocell: The Production Process and Market Development. In Regenerated Cellulose Fibres; Woodings, C., Ed.; Woodhead Publishing: Cambridge, UK, 2001. [Google Scholar]
- Common Objective. Mapping the Global Fashion Industry; Common Objective Ethical Fashion Group Ltd.: London, UK, 2018. [Google Scholar]
- Ma, Y.; Hummel, M.M.; Määttänen, M.; Särkilahti, A.; Harlin, A.; Sixta, H. Upcycling of waste paper and cardboard to textiles. Green Chem. 2016, 18, 858–866. [Google Scholar] [CrossRef]
- Batalha, L.A.; Colodette, J.L.; Gomide, J.L.; Barbosa, L.C.; Maltha, C.R.; Gomes, F.J. Dissolving Pulp Production from Bamboo. BioResources 2012, 7, 640–651. [Google Scholar]
- Bicu, I.; Mustata, F. Cellulose extraction from orange peel using sulfite digestion reagents. Bioresour. Technol. 2011, 102, 10013–10019. [Google Scholar] [CrossRef]
- Gao, Q.; Shen, X.; Lu, X. Regenerated bacterial cellulose fibers prepared by the NMMO·H2O process. Carbohydr. Polym. 2011, 83, 1253–1256. [Google Scholar] [CrossRef]
- Haslinger, S.; Wang, Y.; Rissanen, M.; Lossa, M.B.; Tanttu, M.; Ilen, E.; Määttänen, M.; Harlin, A.; Hummel, M.; Sixta, H. Recycling of vat and reactive dyed textile waste to new colored man-made cellulose fibers. Green Chem. 2019, 21, 5598–5610. [Google Scholar] [CrossRef]
- Shen, L.; Worrell, E.; Patel, M.K. Environmental impact assessment of man-made cellulose fibres. Resour. Conserv. Recycl. 2010, 55, 260–274. [Google Scholar] [CrossRef]
- Ma, Y.; Rosson, L.; Wang, X.; Byrne, N. Upcycling of waste textiles into regenerated cellulose fibres: Impact of pretreatments. J. Text. Inst. 2019, 111, 630–638. [Google Scholar] [CrossRef]
- Palme, A.; Theliander, H.; Brelid, H. Acid hydrolysis of cellulosic fibres: Comparison of bleached kraft pulp, dissolving pulps and cotton textile cellulose. Carbohydr. Polym. 2016, 136, 1281–1287. [Google Scholar] [CrossRef]
- Bali, G.; Meng, X.; Deneff, J.I.; Sun, Q.; Ragauskas, A.J. The Effect of Alkaline Pretreatment Methods on Cellulose Structure and Accessibility. ChemSusChem 2015, 8, 275–279. [Google Scholar] [CrossRef]
- Trache, D.; Hussin, M.H.; Haafiz, M.M.; Thakur, V.K. Recent progress in cellulose nanocrystals: Sources and production. Nanoscale 2017, 9, 1763–1786. [Google Scholar] [CrossRef]
- Ma, Y.; Zeng, B.; Wang, X.; Byrne, N. Circular Textiles: Closed Loop Fiber to Fiber Wet Spun Process for Recycling Cotton from Denim. ACS Sustain. Chem. Eng. 2019, 7, 11937–11943. [Google Scholar] [CrossRef]
- Asaadi, S.; Hummel, M.; Hellsten, S.; Härkäsalmi, T.; Ma, Y.; Michud, A.; Sixta, H. Renewable High-Performance Fibers from the Chemical Recycling of Cotton Waste Utilizing an Ionic Liquid. ChemSusChem 2016, 9, 3250–3258. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization (ISO). Environmental Management—Life Cycle Assessment—Principles and Framework; ISO: Geneve, Switzerland, 1997. [Google Scholar]
- International Organization for Standardization (ISO). Environmental management—Life Cycle Assessment—Requirements and Guidelines; ISO: Geneve, Switzerland, 2006. [Google Scholar]
- Esteve-Turrillas, F.A.; de la Guardia, M. Environmental impact of Recover cotton in textile industry. Resour. Conserv. Recycl. 2017, 116, 107–115. [Google Scholar] [CrossRef]
- Goedkoop, M.; Oele, M.; Leijting, J.; Ponsioen, T.; Meijer, E. Introduction to LCA with SimaPro; Pré Consultants: Amersfoort, The Netherlands, 2016. [Google Scholar]
- Yuan, Z.W.; Zhu, Y.N.; Shi, J.K.; Liu, X.; Huang, L. Life-cycle assessment of continuous pad-dyeing technology for cotton fabrics. Int. J. Life Cycle Assess. 2012, 18, 659–672. [Google Scholar] [CrossRef]
- Adeniran, J.A.; Yusuf, R.O.; Adetoro, M.A. Life Cycle Engineering Case Study: Sulphuric Acid Production. J. Eng. Technol. 2017, 8, 2. [Google Scholar]
- Renouf, M.A.; Grant, T.; Sevenster, M.; Logie, J.; Ridoutt, B.; Ximenes, F.; Bengtsson, J.; Cowie, A.; Lane, J. Best Practice Guide to Life Cycle Impact Assessment in Australia. Australian Life Cycle Assessment Society, 2018. Available online: www.alcas.asn.au (accessed on 1 October 2020).
- Department of Energy and Environment. Australian Energy Statistics; Department of Energy and Environment: Canberra, Australia, 2019.
- Agrawal, K.K.; Jain, S.; Jain, A.K.; Dahiya, S. Assessment of greenhouse gas emissions from coal and natural gas thermal power plants using life cycle approach. Int. J. Environ. Sci. Technol. 2014, 11, 1157–1164. [Google Scholar] [CrossRef]
- Karbowska, B. Presence of thallium in the environment: Sources of contaminations, distribution and monitoring methods. Environ. Monit. Assess. 2016, 188, 640. [Google Scholar] [CrossRef]
- Commonwealth of Australia. National Pollutant Inventory—Beryllium and Compounds; Department of Energy and Environment; Commonwealth of Australia: Canberra, Australia, 2018.
- Tanveer, M.; Wang, L. Potential targets to reduce beryllium toxicity in plants: A review. Plant Physiol. Biochem. 2019, 139, 691–696. [Google Scholar] [CrossRef]
- Yang, C.; Xia, M.; Dong, Z.; Wang, J.; Li, X. Analysis on Abatement Cost and Its Influencing Factors on Nitrogen Oxide Emissions from Power Plants in China. In Proceedings of the 2018 International Conference on Information Technology and Management Engineering (ICITME 2018), Beijing, China, 26–27 August 2018; Atlantis Press: Paris, France, 2018. [Google Scholar]
- Rokni, E.; Panahi, A.; Ren, X.; Levendis, Y.A. Curtailing the generation of sulfur dioxide and nitrogen oxide emissions by blending and oxy-combustion of coals. Fuel 2016, 181, 772–784. [Google Scholar] [CrossRef]
- Ross, R. Facts about Cadmium. 2018. Available online: https://www.livescience.com/37044-cadmium.html (accessed on 14 December 2019).
- Tomlinson, N.; Campbell, J.J.R.; Trussell, P.C. The Influence of Zinc, Iron, Copper, And Manganese on The Production of Citric Acid by Aspergillus Niger. J. Bacteriol. 1950, 59, 217. [Google Scholar] [CrossRef] [PubMed]
- Soccol, C.R.; Vandenberghe, L.P.S.; Rodrigues, C.; Pandey, A. New Perspectives for Citric Acid Production and Application. Food Technol. Biotechnol. 2006, 44, 141–149. [Google Scholar]
- Fraser, P.J.; Dunse, B.L.; Manning, A.J.; Walsh, S.; Wang, R.H.; Krummel, P.B.; Steele, L.P.; Porter, L.W.; Allison, C.; O’Doherty, S.; et al. Australian carbon tetrachloride emissions in a global context. Environ. Chem. 2014, 11, 77–88. [Google Scholar] [CrossRef]
- Max, B.; Salgado, J.M.; Rodríguez, N.; Cortés, S.; Converti, A.; Domínguez, J.M. Biotechnological production of citric acid. Braz. J. Microbiol. 2010, 41, 862–875. [Google Scholar] [CrossRef] [PubMed]
- Crook, J.; Mousavi, A. The chlor-alkali process: A review of history and pollution. Environ. Forensics 2016, 17, 211–217. [Google Scholar] [CrossRef]
- Kim, S.; Overcash, M. Energy in chemical manufacturing processes. J. Chem. Technol. Biotechnol. 2003, 78, 995–1005. [Google Scholar] [CrossRef]
- Pyka, I.; Wierzchowski, K. Estimated mercury emissions from coal combustion in the households sector in Poland. J. Sustain. Min. 2016, 15, 66–72. [Google Scholar] [CrossRef][Green Version]
- Bullock, L.A.; Parnell, J.; Feldmann, J.; Armstrong, J.G.; Henn, A.S.; Mesko, M.F.; Mello, P.A.; Flores, E.M. Selenium and tellurium concentrations of Carboniferous British coals. Geol. J. 2019, 54, 1401–1412. [Google Scholar] [CrossRef]
- Energy Use Calculator. Electricity Usage of a Refrigerator. 2020. Available online: http://energyusecalculator.com/electricity_refrigerator.htm (accessed on 10 December 2019).
- Wu, Y.; Xie, H.; Liu, T.; Wang, Y.; Wang, F.; Gao, X.; Liang, B. Soda Ash Production with Low Energy Consumption Using Proton Cycled Membrane Electrolysis. Ind. Eng. Chem. Res. 2019, 58, 3450–3458. [Google Scholar] [CrossRef]
- Office of Air and Radiation. Technical Support Document for the Soda Ash Manufacturing Sector: Proposed Rule for Mandatory Reporting of Greenhouse Gases. U.S. Environmental Protection Agency: Washington, DC, USA, 2009. Available online: https://www.epa.gov/nscep (accessed on 14 March 2019).
- Franzoni, E.; Volpi, L.; Bonoli, A. Applicability of Life Cycle Assessment methodology to conservation works in historical building: The case of cleaning. Energy Build. 2020, 214, 109844. [Google Scholar] [CrossRef]
- Boustead, I. Eco-profiles for the European Plastics Industry: Sodium Hydroxide. PlasticsEurope 2005, 3, 13. [Google Scholar]
- Garcia-Herrero, I.; Margallo, M.; Onandía, R.; Aldaco, R.; Irabien, A. Environmental challenges of the chlor-alkali production: Seeking answers from a life cycle approach. Sci. Total Environ. 2017, 580, 147–157. [Google Scholar] [CrossRef]
- Kim, Y.; McCoy, L.T.; Lee, E.; Lee, H.; Saremi, R.; Feit, C.; Hardin, I.R.; Sharma, S.; Mani, S.; Minko, S. Environmentally sound textile dyeing technology with nanofibrillated cellulose. Green Chem. 2017, 19, 4031–4035. [Google Scholar] [CrossRef]
- Sandin, G.; Peters, G.M. Environmental impact of textile reuse and recycling—A review. J. Clean. Prod. 2018, 184, 353–365. [Google Scholar] [CrossRef]
- Nasri-Nasrabadi, B.; Wang, X.; Byrne, N. Perpetual colour: Accessing the colourfastness of regenerated cellulose fibres from coloured cotton waste. J. Text. Inst. 2020, 1–10. [Google Scholar] [CrossRef]
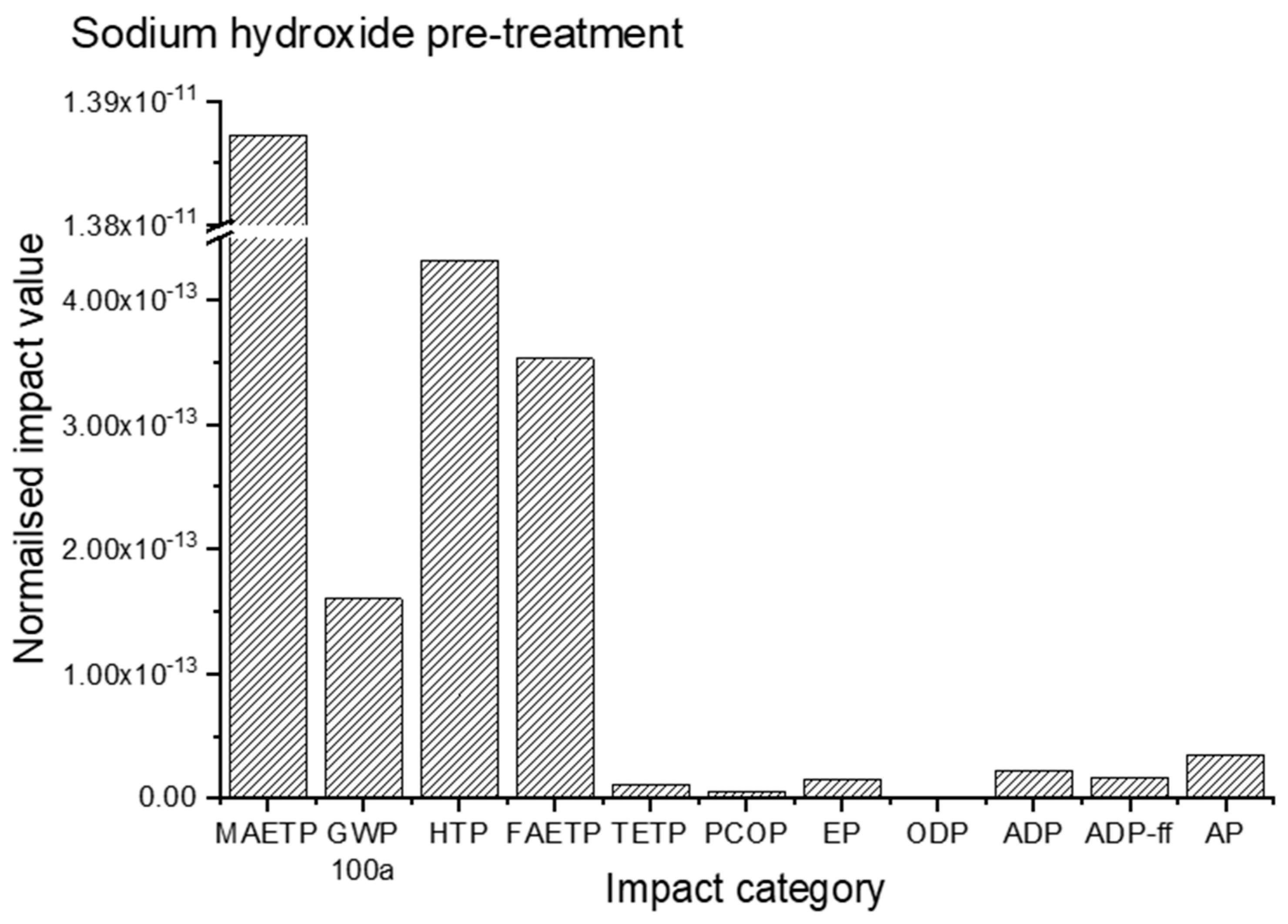
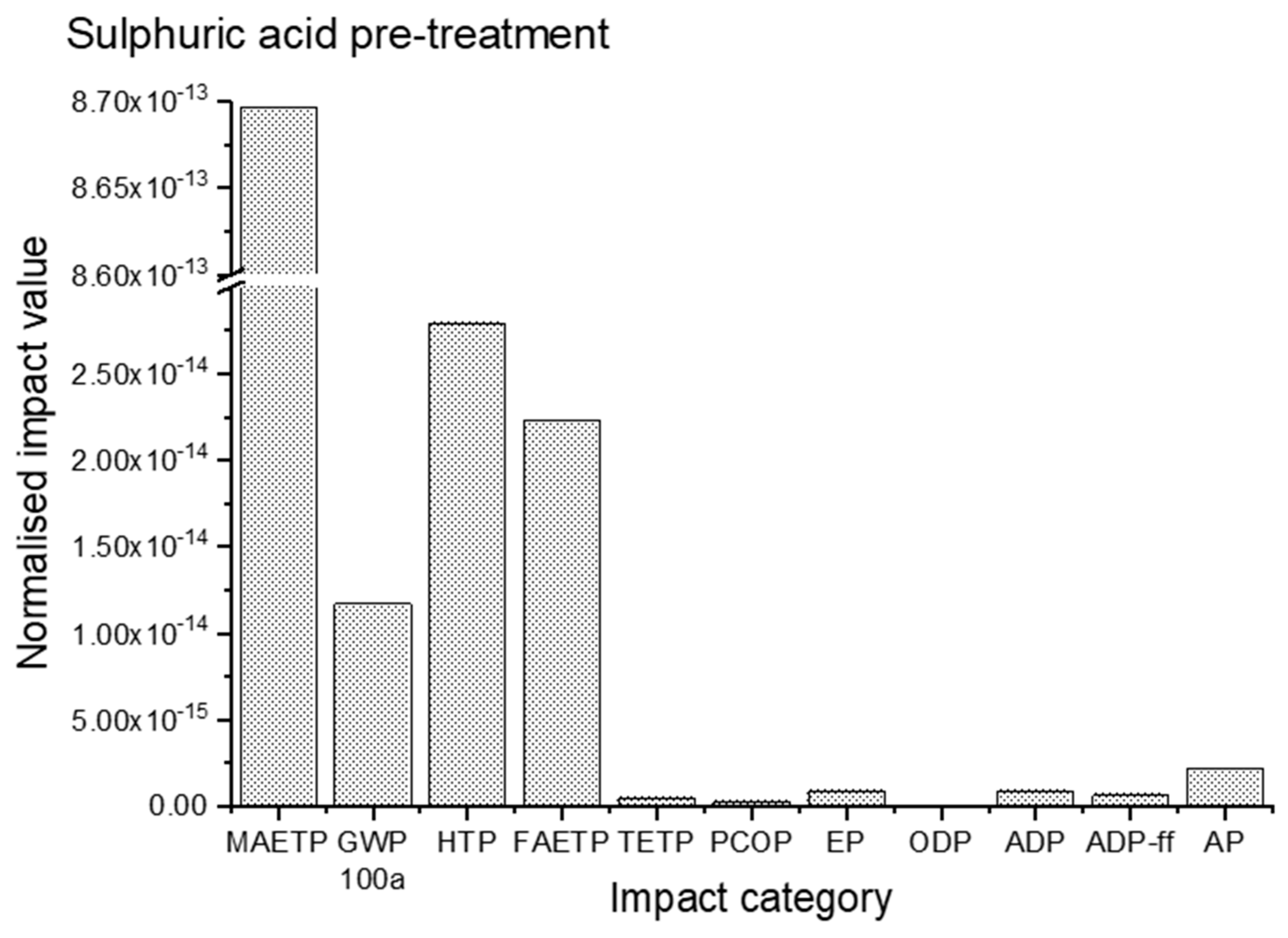
| Impact Category | Unit | Sulphuric Acid Pre-Treated Cotton Waste | Sodium Hydroxide Pre-Treated Cotton Waste |
|---|---|---|---|
| Abiotic depletion | kg Sb-eq | 1.91 × 10−7 | 4.75 × 10−6 |
| Abiotic depletion (fossil fuels) | MJ | 2.42 × 10−1 | 6.32 × 100 |
| Global warming (GWP100a) | kg CO2-eq | 4.91 × 10−1 | 6.70 × 100 |
| Ozone layer depletion (ODP) | kg CFC-11-eq | 1.41 × 10−9 | 4.67 × 10−8 |
| Human toxicity | kg 1,4-DB-eq | 7.20 × 10−2 | 1.11 × 100 |
| Fresh water aquatic ecotox. | kg 1,4-DB-eq | 5.28 × 10−2 | 8.33 × 10−1 |
| Marine aquatic ecotoxicity | kg 1,4-DB-eq | 1.69 × 102 | 2.69 × 103 |
| Terrestrial ecotoxicity | kg 1,4-DB-eq | 4.88 × 10−4 | 1.24 × 10−2 |
| Photochemical oxidation | kg C2H4-eq | 9.97 × 10−6 | 1.83 × 10−4 |
| Acidification | kg SO2-eq | 5.21 × 10−4 | 8.26 × 10−3 |
| Eutrophication | kg PO43−-eq | 1.45 × 10−4 | 2.48 × 10−3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosson, L.; Byrne, N. Comparative Gate-to-Gate Life Cycle Assessment for the Alkali and Acid Pre-Treatment Step in the Chemical Recycling of Waste Cotton. Sustainability 2020, 12, 8613. https://doi.org/10.3390/su12208613
Rosson L, Byrne N. Comparative Gate-to-Gate Life Cycle Assessment for the Alkali and Acid Pre-Treatment Step in the Chemical Recycling of Waste Cotton. Sustainability. 2020; 12(20):8613. https://doi.org/10.3390/su12208613
Chicago/Turabian StyleRosson, Lucas, and Nolene Byrne. 2020. "Comparative Gate-to-Gate Life Cycle Assessment for the Alkali and Acid Pre-Treatment Step in the Chemical Recycling of Waste Cotton" Sustainability 12, no. 20: 8613. https://doi.org/10.3390/su12208613
APA StyleRosson, L., & Byrne, N. (2020). Comparative Gate-to-Gate Life Cycle Assessment for the Alkali and Acid Pre-Treatment Step in the Chemical Recycling of Waste Cotton. Sustainability, 12(20), 8613. https://doi.org/10.3390/su12208613





