From Disposal to Technological Potential: Reuse of Polypropylene Waste from Industrial Containers as a Polystyrene Impact Modifier
Abstract
1. Introduction
2. Experimental
2.1. Materials
2.2. Methods
2.2.1. Preparation of Recycled Polypropylene (PPr)
2.2.2. Blends Preparation
2.2.3. Injection Molding
- Characterizations
- Statistical Analysis
3. Results and Discussion
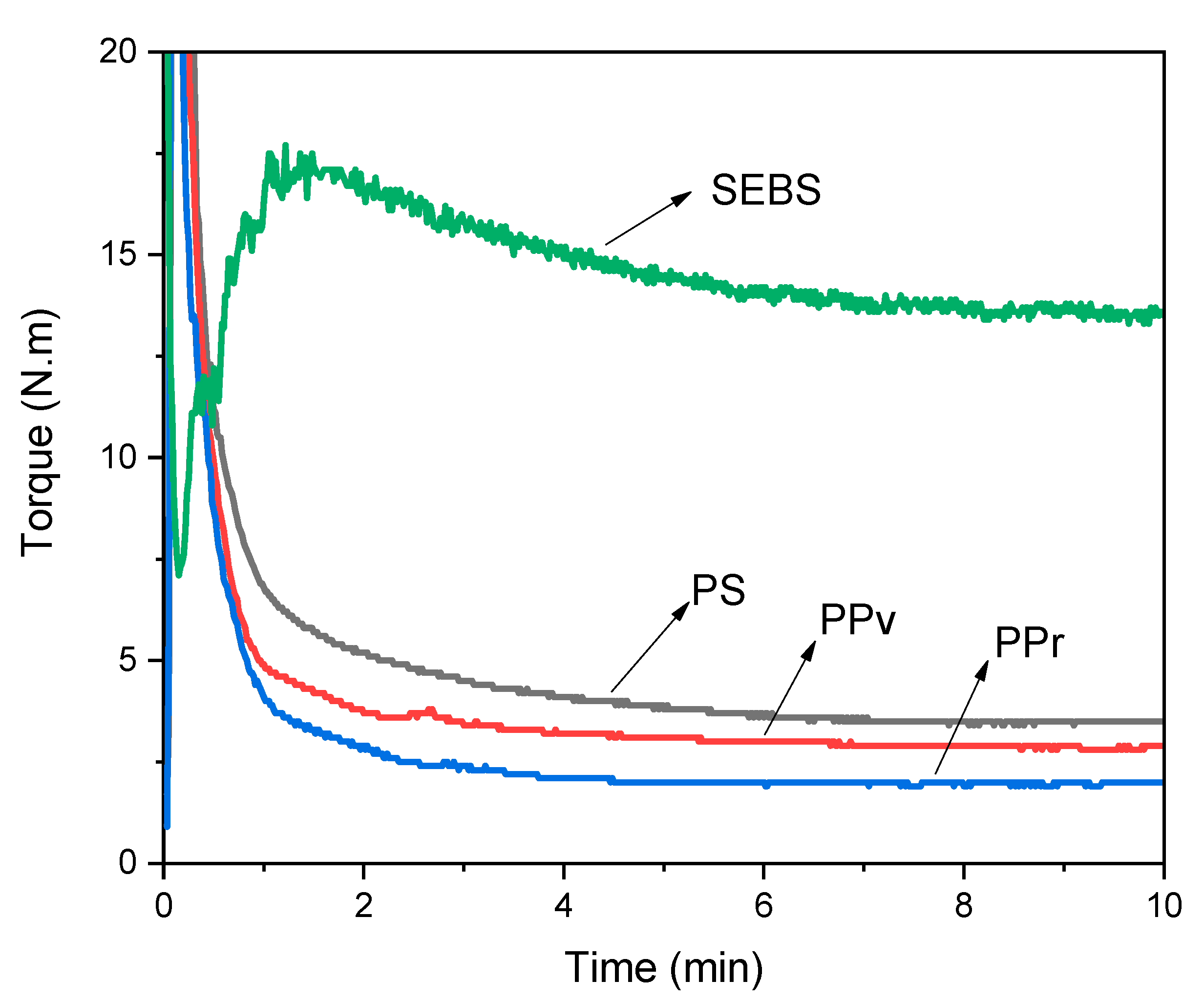
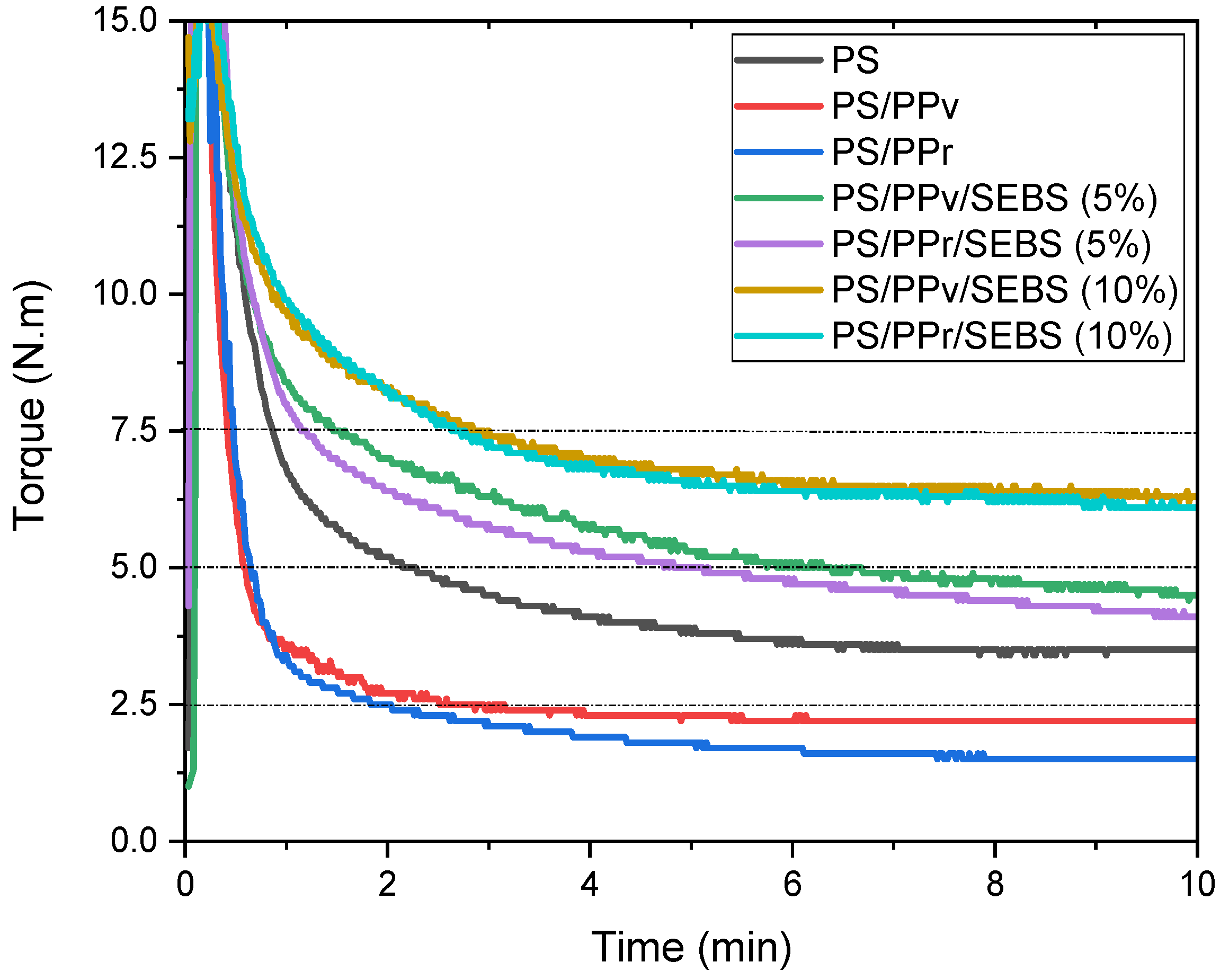
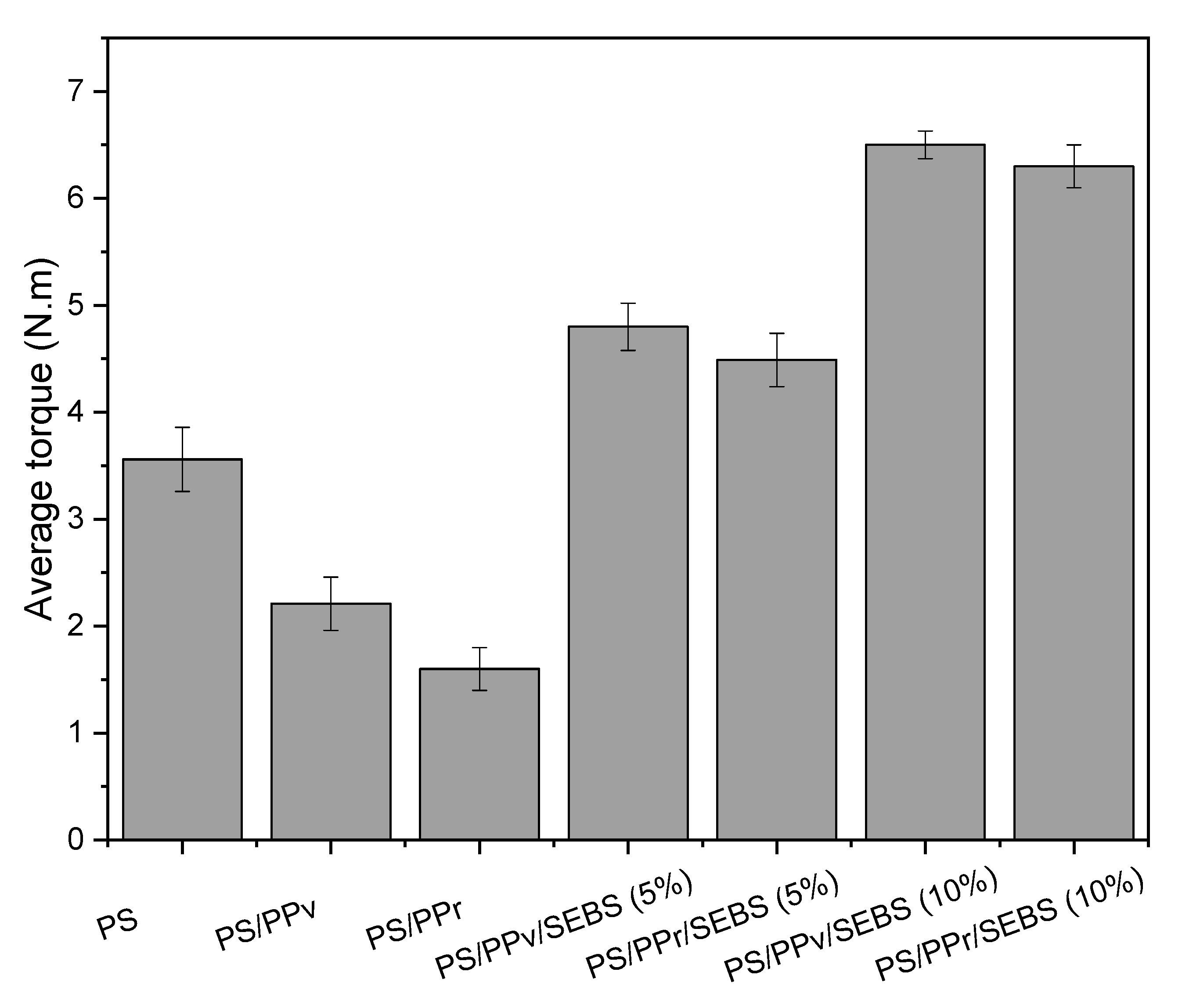
3.1. Torque Rheometry
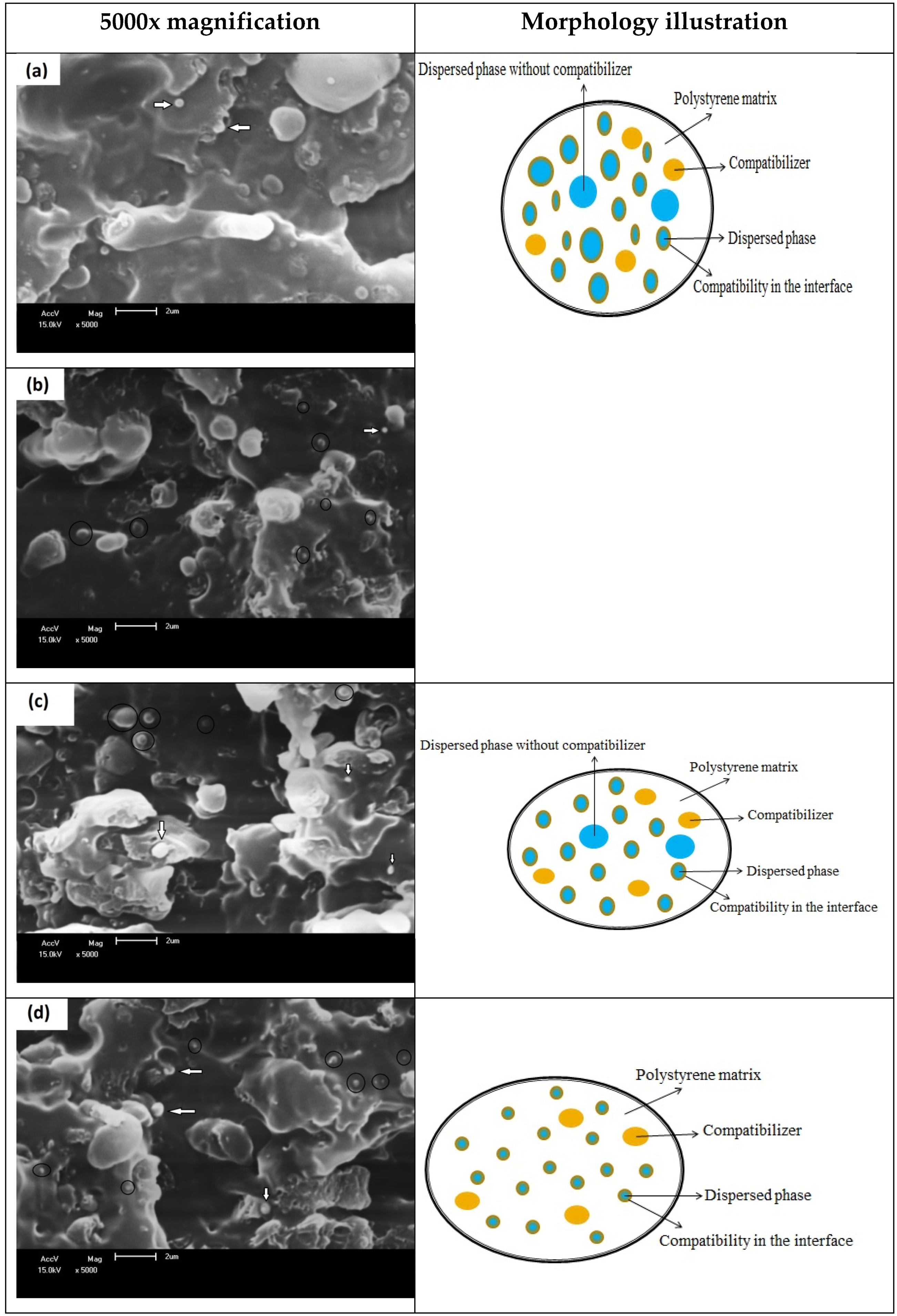
3.2. Scanning Electron Microscopy (SEM)
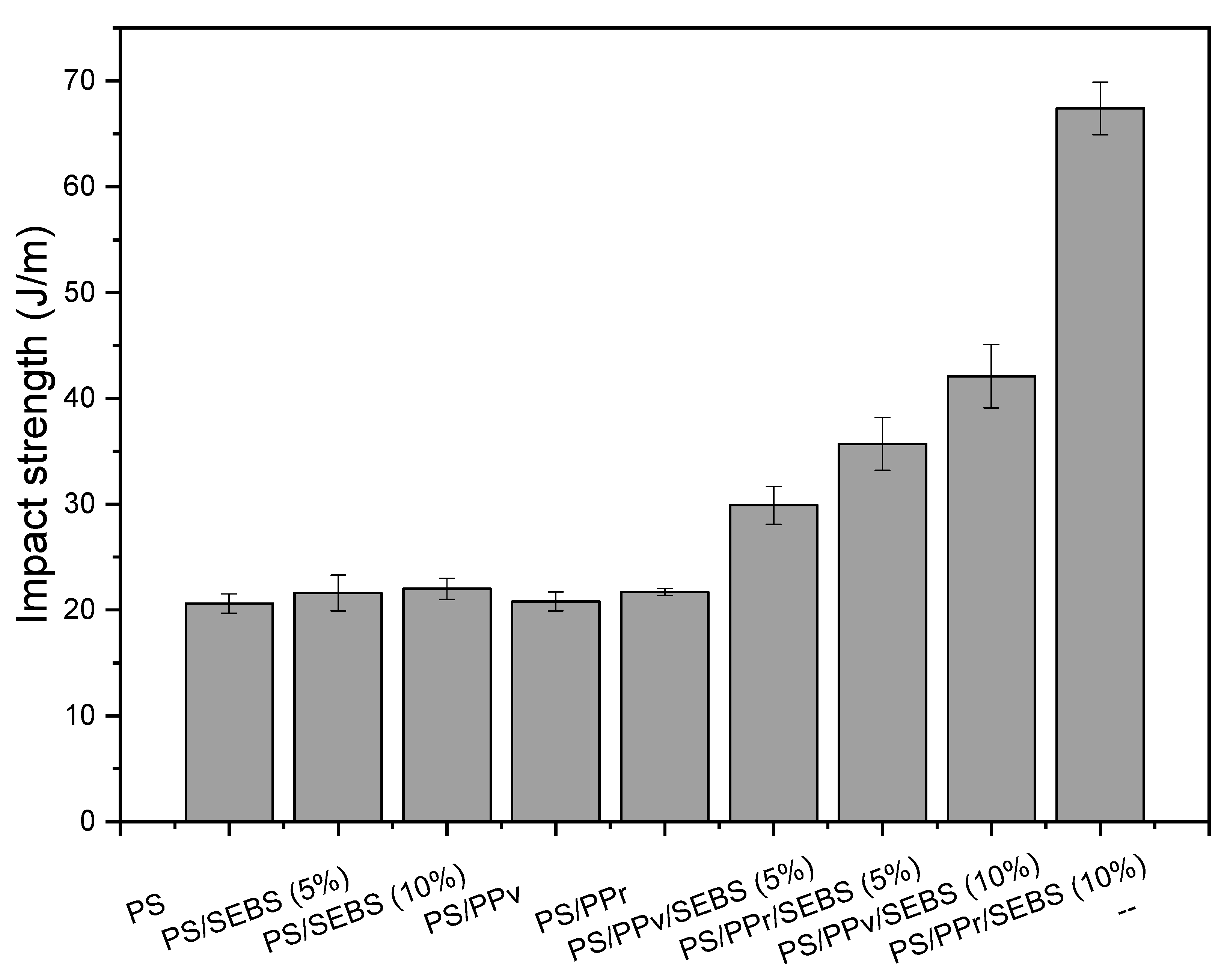
3.3. Impact Strength
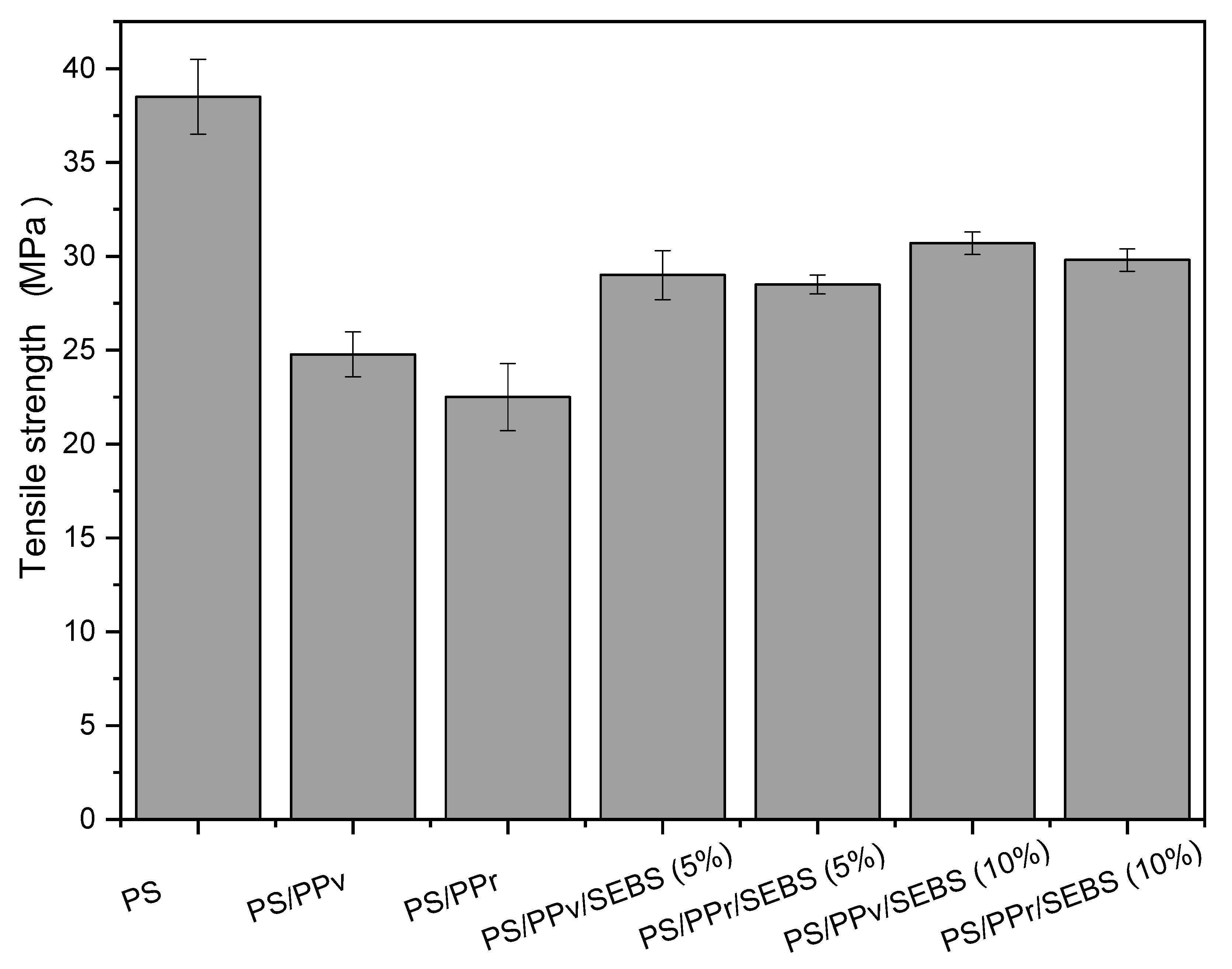
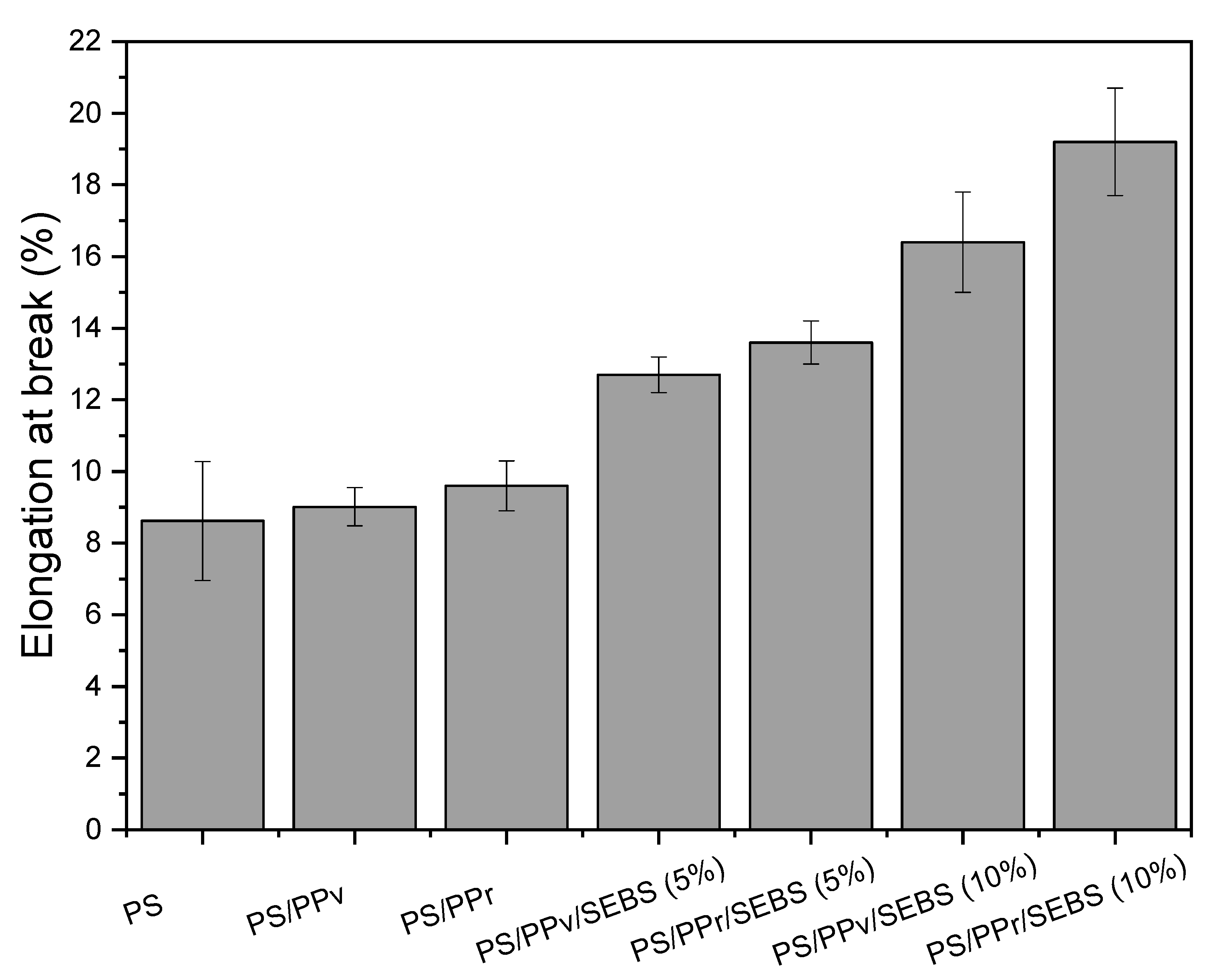
3.4. Tensile Strength
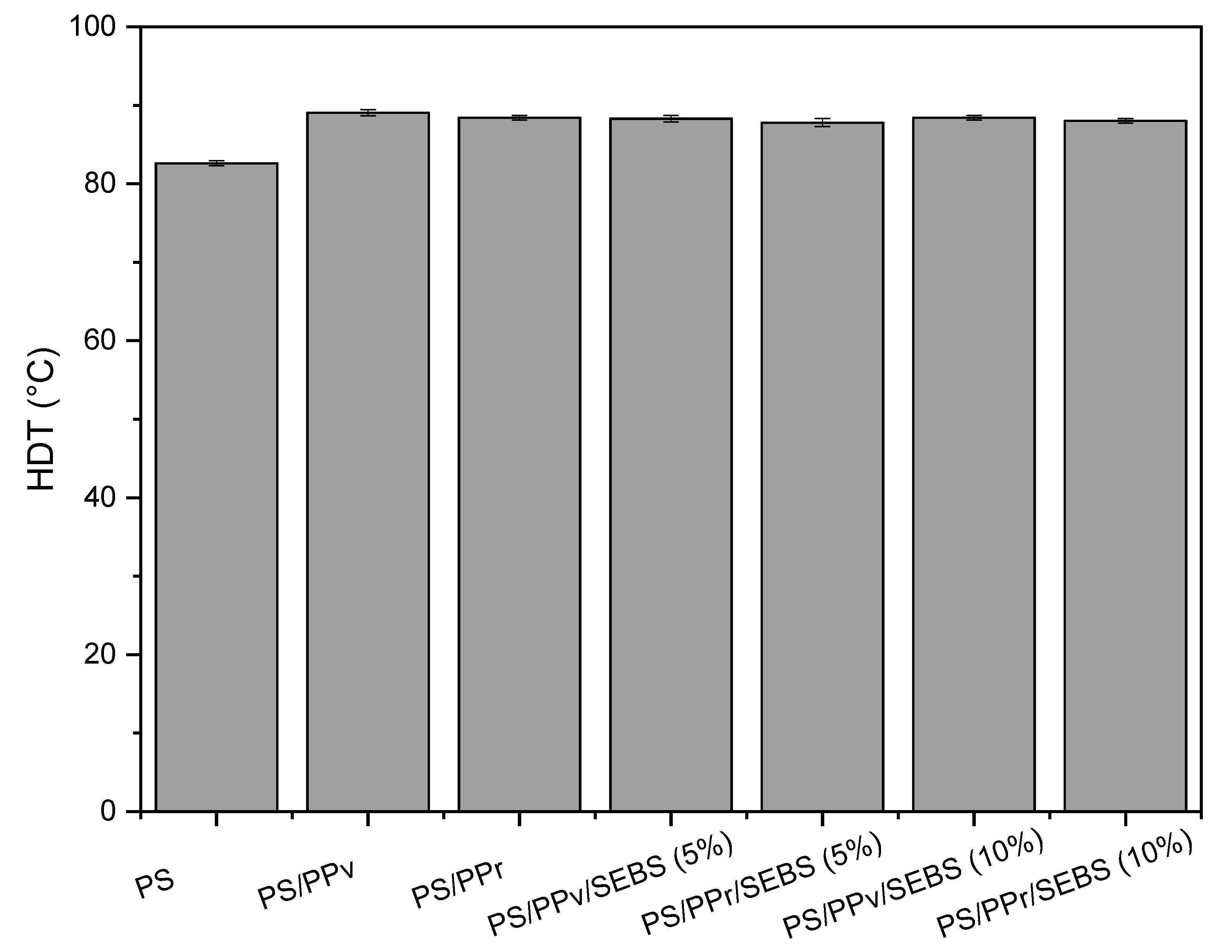
3.5. Heat Deflection Temperature (HDT)
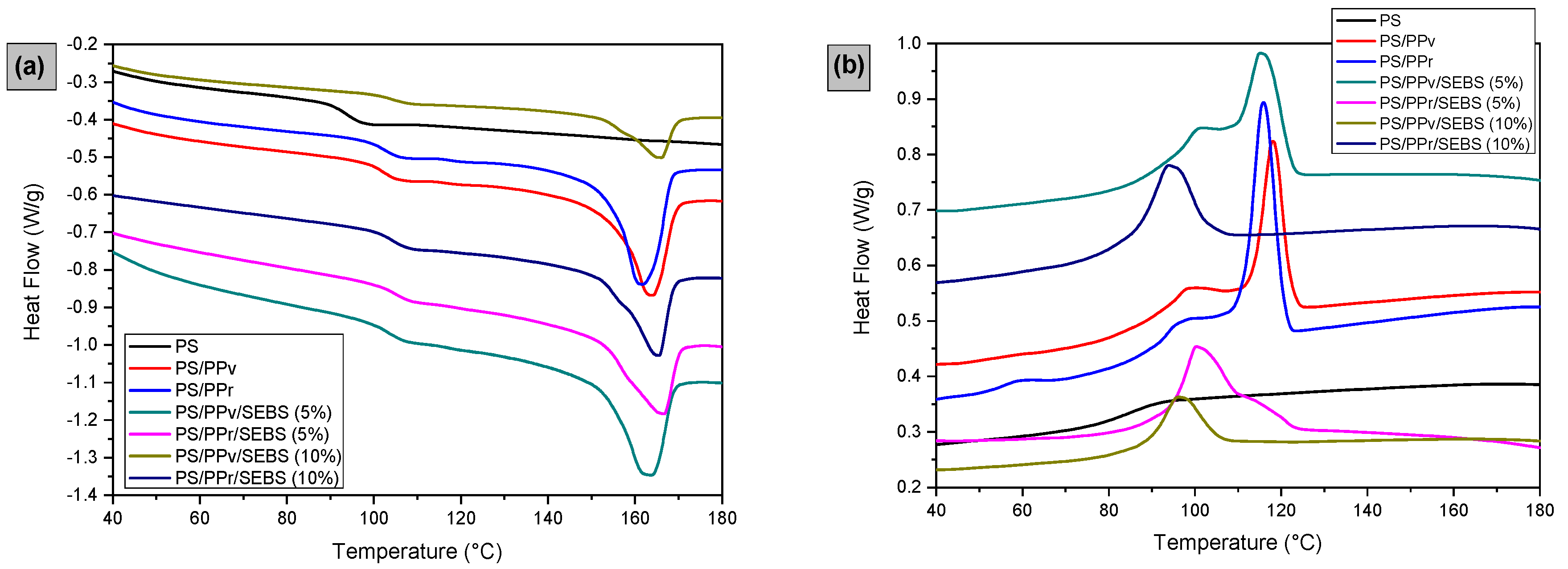
3.6. Differential Scanning Calorimetry (DSC)
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rossino, L.S.; Oliveira, R.C.; Ferreira, R.; Moreto, J.A.; Manfrinato, M.D.; Paiva, J.M.F. Estudo comparativo da resistência à flexão da madeira plástica, fibra de madeira de densidade média, pinus e polipropileno submetidos aos condicionamentos de secagem, saturação de umidade e radiação UV. Rev. Eletrônica Mater. Process. 2019, 14, 11–18. [Google Scholar]
- Samper, M.D.; Bertomeu, D.; Arrieta, M.P.; Ferri, J.M.; Martínez, J.L. Interference of biodegradable plastics in the polypropylene recycling process. Materials 2018, 11, 886. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.H.; Ferreira, E.S.B.; Bezerra, E.B.; Araújo, E.M.; Wellen, R.M.R. Influência da incorporação de resíduos de tecido de Big Bag nas propriedades mecânicas do polipropileno. Rev. Eletrônica Mater. Process. 2018, 13, 18–22. [Google Scholar]
- De Camargo, R.V.; Saron, C. Mechanical–Chemical Recycling of Low-Density Polyethylene Waste with Polypropylene. J. Polym. Environ. 2019, 28, 794–802. [Google Scholar] [CrossRef]
- Cavalcanti, R.S.D.F.B.; Rabello, M.S. The Effect of Red Pigment and Photo Stabilizers on the Photo Degradation of Polypropylene Films. Mater. Res. 2019, 22, 1–8. [Google Scholar] [CrossRef]
- Graziano, A.; Jaffer, S.A.; Sain, M. Review on modification strategies of polyethylene/polypropylene immiscible thermoplastic polymer blends for enhancing their mechanical behavior. J. Elastomers Plast. 2018, 51, 291–336. [Google Scholar] [CrossRef]
- Ferreira, E.D.S.B.; Pereira, C.H.O.; Araújo, E.M.; Bezerra, E.B.; Siqueira, D.D.; Wellen, R.M.R. Properties and Morphology of Polypropylene/Big Bags Compounds. Mater. Res. 2019, 22, 1–8. [Google Scholar] [CrossRef]
- Pereira, C.; Pisanu, L.; Viana, J.; Azevedo, J.B.; Almeida, T.; Fook, M.; Wellen, R.M.R.; Canedo, E. Heterophasic polypropylene and wood flour composites: Processing and properties. Mater. Res. Express 2019, 6, 085321. [Google Scholar] [CrossRef]
- Pereira, C.A.B.; Azevedo, J.B.; Almeida, T.G.; Canedo, E.L. Estudo do processamento de compósitos de polipropileno/pó de madeira em extrusora de dupla rosca corrotacional. Rev. Eletrônica Mater. Process. 2018, 13, 23–36. [Google Scholar]
- Baltá-Calleja, F.J.; Cagiao, M.; Adhikari, R.; Michler, G. Relating microhardness to morphology in styrene/butadiene block copolymer/polystyrene blends. Polymer 2004, 45, 247–254. [Google Scholar] [CrossRef]
- Luna, C.B.B.; Araújo, E.M.; Siqueira, D.D.; Morais, D.D.D.S.; Filho, E.A.D.S.; Fook, M.V.L. Incorporation of a recycled rubber compound from the shoe industry in polystyrene: Effect of SBS compatibilizer content. J. Elastomers Plast. 2019, 52, 3–28. [Google Scholar] [CrossRef]
- Araújo, E.M.; Carvalho, L.H.; Fook, M.V.L.; D’Almeida, J.R.M. Mechanical properties of PS/rubber waste blends—Effects of concentration, particle size and moulding procedures. Polímeros 1997, 7, 45–52. [Google Scholar] [CrossRef]
- Luna, C.B.B.; Siqueira, D.D.; Araújo, E.M.; Morais, D.D.D.S.; Bezerra, E.B. Toughening of polystyrene using styrene-butadiene rubber (SBRr) waste from the shoe industry. REM Int. Eng. J. 2018, 71, 253–260. [Google Scholar] [CrossRef]
- Ibrahim, B.A.; Kadum, K.M. Morphology studies and mechanical properties for PS/SBS blends. Int. J. Eng. Technol. IJET-IJENS 2012, 12, 19–27. [Google Scholar]
- Wen, Y.; Lei, F. Study on mechanical properties of styrene-ethylene/butylene-styrene/polystyrene blends. Adv. Mater. Res. 2011, 396–398, 1422–1425. [Google Scholar]
- Libio, I.C.; Grassi, V.G.; Pizzol, M.F.D.; Nachtigall, S. Toughened polystyrene with improved photoresistance: Effects of the compatibilizers. J. Appl. Polym. Sci. 2012, 126, 179–185. [Google Scholar] [CrossRef]
- Parameswaranpillai, J.; Joseph, G.; Chellappan, R.V.; Zachariah, A.K.; Hameed, N. The effect of polypropylene-graft-maleic anhydride on the morphology and dynamic mechanical properties of polypropylene/polystyrene blends. J. Polym. Res. 2015, 22, 1–11. [Google Scholar] [CrossRef]
- Omonov, T.; Harrats, C.; Groeninckx, G.; Moldenaers, P. Anisotropy and instability of the co-continuous phase morphology in uncompatibilized and reactively compatibilized polypropylene/polystyrene blends. Polymer 2007, 48, 5289–5302. [Google Scholar] [CrossRef]
- Pleska, A.; Hlavatá, D.; Horák, Z.; Hromádková, J.; Lednický, F. Compatabilization of polystyrene/polypropylene blends by styrene-butadiene block copolymers with differing polystyrene block lengths. J. Polym. Sci. Part B Polym. Phys. 1999, 37, 1647–1656. [Google Scholar]
- Samsudin, S.A.; Hassan, A.; Mokhtar, M.; Jamaluddin, S.M.S. Chemical resistance evaluation of polystyrene/polypropylene blends: Effect of blend compositions and SEBS content. Malays. Polym. J. (MPJ) 2006, 1, 11–24. [Google Scholar]
- Halimatudahliana, H.I.; Nasir, M. The effect of various compatibilizers on mechanical properties of polystyrene/polypropylene blend. Polym. Test. 2002, 21, 163–170. [Google Scholar]
- Latreche, L.; Haddaoui, N.; Cagiao, M.E. The effect of various compatibilizers on thermal, mechanical, and morphological properties of polystyrene/polypropylene blends. Russ. J. Appl. Chem. 2016, 89, 1713–1721. [Google Scholar] [CrossRef]
- Luna, C.B.B.; Siqueira, D.D.; Araújo, E.M.; Wellen, R.M.R. Tailoring PS/PPrecycled blends compatibilized with SEBS. Evaluation of rheological, mechanical, thermomechanical and morphological characters. Mater. Res. Express 2019, 6, 075316. [Google Scholar] [CrossRef]
- Hammani, S.; Moulai-Mostefa, N.; Samyn, P.; Bechelany, M.; Dufresne, A.; Barhoum, A. Morphology, Rheology and Crystallization in Relation to the Viscosity Ratio of Polystyrene/Polypropylene Polymer Blends. Materials 2020, 13, 926. [Google Scholar] [CrossRef]
- Horák, Z.; Kolařík, J.; Šípek, M.; Hynek, V.; Večerka, F. Gas permeability and mechanical properties of polystyrene–polypropylene blends. J. Appl. Polym. Sci. 1998, 69, 2615–2623. [Google Scholar] [CrossRef]
- Oliveira, R.; Ferreira, C.I.; Peixoto, L.J.F.; Bianchi, O.; Silva, P.A.; Demori, R.; Da Silva, R.P.; Veronese, V.B. Mistura polipropileno/poliestireno: Um exemplo da relação processamento-estrutura-propriedade no ensino de polímeros. Polímeros 2013, 23, 91–96. [Google Scholar] [CrossRef][Green Version]
- Luna, C.B.B.; Ferreira, E.D.S.B.; Da Silva, L.J.M.D.; Silva, W.; Araújo, E.M.; Melo, J.B.D.C.A.D. Blends with technological potential of copolymer polypropylene with polypropylene from post-consumer industrial containers. Mater. Res. Express 2019, 6, 125319. [Google Scholar] [CrossRef]
- Lima, J.C.C.; Araújo, J.P.; Agrawal, P.; Mélo, T.J.A. Efeito do teor do copolímero SEBS no comportamento reológico da blenda PLA/SEBS. Rev. Eletrônica Mater. Process. 2016, 11, 10–17. [Google Scholar]
- González-González, V.; Neira-Velázquez, G.; Angulo-Sanchez, J. Polypropylene chain scissions and molecular weight changes in multiple extrusion. Polym. Degrad. Stab. 1998, 60, 33–42. [Google Scholar] [CrossRef]
- Bretas, R.E.S.; D’Ávila, M.A.D. Reologia de Polímeros Fundidos, 2nd ed.; EDUFSCar: São Carlos, Brazil, 2010. [Google Scholar]
- Diaz, M.F.; Barbosa, S.E.; Capiati, N.J. Addition compatibilization of PP/PS blends by tailor-made copolymers. Polym. Eng. Sci. 2006, 46, 329–336. [Google Scholar] [CrossRef]
- Kong, Y.; Li, Y.; Hu, G.; Cao, N.; Ling, Y.; Pan, D.; Shao, Q.; Guo, Z. Effects of polystyrene-b-poly(ethylene/propylene)-bpolystyrene compatibilizer on the recycled polypropylene and recycled high-impact polystyrene blends. Polym. Adv. Technol. 2018, 29, 2344–2351. [Google Scholar] [CrossRef]
- Aghavi, S.K.; Shahrajabian, H.; Hosseini, H.M. Detailed comparison of compatibilizers MAPE and SEBS-g-MA on the mechanical/thermal properties, and morphology in ternary blend of recycled PET/HDPE/MAPE and recycled PET/HDPE/SEBS-g-MA. J. Elastomers Plast. 2017, 50, 13–35. [Google Scholar] [CrossRef]
- Debbah, I.; Krache, R.; Aranburu, N.; Fernández, M.; Etxeberria, A. Effect of SEBS-g-MAH addition on the mechanical, rheological, and morphological properties of polycarbonate/acrylonitrile–butadiene–styrene blends. J. Elastomers Plast. 2018, 50, 611–633. [Google Scholar] [CrossRef]
- Omonov, T.; Harrats, C.; Moldenaers, P.; Groeninckx, G. Phase continuity detection and phase inversion phenomena in immiscible polypropylene/polystyrene blends with different viscosity ratios. Polymer 2007, 48, 5917–5927. [Google Scholar] [CrossRef]
- Zhao, X.; Huang, Y.; Kong, M.; Yang, Q.; Li, G. Assessment of compatibilization efficiency of SEBS in the PP/PS blend. J. Appl. Polym. Sci. 2018, 135, 46244. [Google Scholar] [CrossRef]
- De Mello, F.B.; Nachtigall, S.M.B.; Salles, C.D.A.; Amico, S.C. Compatibilization and mechanical properties of compression-molded polypropylene/high-impact polystyrene blends. Prog. Rubber Plast. Recycl. Technol. 2018, 34, 117–127. [Google Scholar] [CrossRef]
- Melo, T.; De Carvalho, L.H.; Calumby, R.B.; Brito, K.G.Q.; D’Almeida, J.R.M.; Spieth, E. Propriedades mecânicas e morfologia de uma blenda polimérica de PP/HIPS compatibilizada com SEBS. Polímeros 2000, 10, 82–89. [Google Scholar] [CrossRef]
- Luna, C.B.B.; Siqueira, D.D.; Ferreira, E.D.S.B.; Araújo, E.M.; Wellen, R.M.R. Reactive compatibilization as a proper tool to improve PA6 toughness. Mater. Res. Express 2020, 6, 125367. [Google Scholar] [CrossRef]
- Coutinho, F.M.B.; Costa, M.P.M.; Guimarães, M.J.O.C.; Soares, B.G. Comparative study of different types of polybutadiene on the toughening of polystyrene. Polímeros 2007, 17, 318–324. [Google Scholar] [CrossRef][Green Version]
- Agrawal, P.; Araújo, E.M.; Mélo, T.J.A. Torque rheometry, mechanical properties and morphology of compatibilized PA6/HDPE blends. Polímeros 2008, 18, 152–157. [Google Scholar] [CrossRef]
- Bedjaoui, K.; Krache, R.; Marcos-Fernández, A.; Guessoum, M. The effect of compatibilizer SEBS on the mechanical, morphological and thermal properties of the polystyrene/poly (styrene-co-acrylonitrile) copolymer blends. Mater. Res. Express 2019, 6, 105334. [Google Scholar] [CrossRef]
- Al-Saleh, M.H.; Sundararaj, U. Mechanical properties of carbon black-filled polypropylene/polystyrene blends containing styrene-butadiene-styrene copolymer. Polym. Eng. Sci. 2009, 49, 693–702. [Google Scholar] [CrossRef]
- Bezerra, E.B.; França, D.C.; Morais, D.D.D.S.; Siqueira, D.D.; Araújo, E.M.; Wellen, R.M.R. Toughening of bio-PE upon addition of PCL and PEgAA. REM Int. Eng. J. 2019, 72, 469–478. [Google Scholar] [CrossRef]
- Hanken, R.B.; Arimatéia, R.R.; Farias, G.M.; Agrawal, P.; Santana, L.N.; Freitas, D.M.; Melo, T. Effect of natural and expanded vermiculite clays on the properties of eco-friendly biopolyethylene-vermiculite clay biocomposites. Compos. Part B Eng. 2019, 175, 1–12. [Google Scholar] [CrossRef]
- Ferreira, E.D.S.B.; Luna, C.B.B.; Araújo, E.M.; Siqueira, D.D.; Wellen, R.M.R. Polypropylene/wood powder composites: Evaluation of PP viscosity in thermal, mechanical, thermomechanical, and morphological characters. J. Thermoplast Compos. Mater. 2019. [Google Scholar] [CrossRef]
- Alves, A.M.; Araújo, E.M.; Agrawal, P.; Cavalcanti, S.N.; Arimatéria, R.R.; Fretas, N.L.; Mélo, T.J.A. Influence of the alumina synthesized by combustion reaction on the properties of composites with polypropylene matrix. Matéria (Rio J.) 2018, 23, e12230. [Google Scholar]
- Silva, D.F.; Luna, C.B.B.; Silva, G.; Araújo, E.M.; Mélo, T.J.A. Avaliação das propriedades mecânicas de blendas de poliestireno/composto de borracha reciclada (SBRr). Rev. Eletrônica Mater. Process. 2014, 9, 92–97. [Google Scholar]
- Ferreira, L.A.S.; Pessan, L.A.; Júnior, E.H. Mechanical and thermo-mechanical behavior of PBT/ABS polymer blends. Polímeros 1997, 7, 67–72. [Google Scholar] [CrossRef]
- Anjos, E.G.R.; Backes, E.H.; Marini, J.; Pessan, L.A.; Montagna, L.S.; Passador, F.R. Effect of LLDPE-g-MA on the rheological, thermal, mechanical properties and morphological characteristic of PA6/LLDPE blends. J. Polym. Res. 2019, 134, 1–10. [Google Scholar] [CrossRef]
- Bin Rusayyis, M.A.; Schiraldi, D.A.; Maia, J. Property/Morphology Relationships in SEBS-Compatibilized HDPE/Poly(phenylene ether) Blends. Macromolecules 2018, 51, 6513–6523. [Google Scholar] [CrossRef]
- Luna, C.B.B.; Siqueira, D.D.; Ferreira, E.D.S.B.; Araújo, E.M.; Wellen, R.M.R. Reactive compatilization of PCL/WP upon addition of PCL-MA. Smart option for recycling industry. Mater. Res. Express 2019, 6, 125317. [Google Scholar] [CrossRef]
- Takemori, M.T. Towards an understanding of the heat distortion temperature of thermoplastics. Polym. Eng. Sci. 1979, 19, 1104–1109. [Google Scholar] [CrossRef]
- Grassi, V.G.; Forte, M.M.C.; Pizzol, M.F.D. Morphologic aspects and structure-properties relations of high impact polystyrene. Polímeros 2001, 11, 158–168. [Google Scholar] [CrossRef]
- Luna, C.B.B.; Silva, D.F.; Araújo, E.M. Compatibilização e reaproveitamento da borracha de SBR no processo de tenacificação do poliestireno. Rev. Eng. Tecnol. 2015, 7, 29–36. [Google Scholar]
- Rieger, J. The glass transition temperature of polystyrene. J. Therm. Anal. Calorim. 1996, 46, 965–972. [Google Scholar] [CrossRef]
- Thirtha, V.; Lehman, R.L.; Nosker, T. Glass transition phenomena in melt-processed polystyrene/polypropylene blends. Polym. Eng. Sci. 2005, 45, 1187–1193. [Google Scholar] [CrossRef]
- Zander, N.E.; Gillan, M.; Burckhard, Z.; Gardea, F. Recycled polypropylene blends as novel 3D printing materials. Addit. Manuf. 2019, 25, 122–130. [Google Scholar] [CrossRef]
- Líbano, E.V.D.G.; Visconte, L.L.Y.; Pacheco, E.B.A.V. Propriedades térmicas de compósitos de polipropileno e bentonita organofílica. Polímeros 2012, 22, 430–435. [Google Scholar] [CrossRef]
- Luna, C.B.B.; Ferreira, E.D.S.B.; Siqueira, D.D.; Silva, W.; Araújo, E.M.; Wellen, R.M.R. Tailoring performance of PP/HIPS/SEBS through blending design. Mater. Res. Express 2019, 6, 115321. [Google Scholar] [CrossRef]
- Aurrekoetxea, J.; Sarrionandia, M.; Urrutibeascoa, I.; Maspoch, M.L. Effects of recycling on the microstructure and the mechanical properties of isotactic polypropylene. J. Mater. Sci. 2001, 36, 2607–2613. [Google Scholar] [CrossRef]





| Sample | Proportion (%wt.) |
|---|---|
| PS | 100 |
| PS/SEBS | 95/5 |
| PS/SEBS | 90/10 |
| PS/PPv | 70/30 |
| PS/PPr | 70/30 |
| PS/PPv/SEBS | 70/25/5 |
| PS/PPr/SEBS | 70/25/5 |
| PS/PPv/SEBS | 70/20/10 |
| PS/PPr/SEBS | 70/20/10 |
| Samples * | Tg (°C) | Tm (°C) | Tc (°C) | ΔHm (J/g) | Xc (%) |
|---|---|---|---|---|---|
| PS | 98.0 | - | - | - | - |
| PS/PPv | 106.1 | 164.1 | 118.1 | 12.1 | 29.2 |
| PS/PPr | 106.3 | 163.9 | 115.9 | 17.2 | 41.5 |
| PS/PPv/SEBS (5%) | 106.3 | 164.0 | 115.1 | 14.5 | 42.0 |
| PS/PPr/SEBS (5%) | 107.3 | 166.6 | 109.5 | 10.5 | 30.4 |
| PS/PPv/SEBS (10%) | 107.9 | 165.2 | 96.1 | 5.7 | 20.6 |
| PS/PPr/SEBS (10%) | 107.5 | 165.5 | 94.0 | 10.7 | 38.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luna, C.B.B.; Siqueira, D.D.; Ferreira, E.d.S.B.; Silva, W.A.d.; Nogueira, J.A.d.S.; Araújo, E.M. From Disposal to Technological Potential: Reuse of Polypropylene Waste from Industrial Containers as a Polystyrene Impact Modifier. Sustainability 2020, 12, 5272. https://doi.org/10.3390/su12135272
Luna CBB, Siqueira DD, Ferreira EdSB, Silva WAd, Nogueira JAdS, Araújo EM. From Disposal to Technological Potential: Reuse of Polypropylene Waste from Industrial Containers as a Polystyrene Impact Modifier. Sustainability. 2020; 12(13):5272. https://doi.org/10.3390/su12135272
Chicago/Turabian StyleLuna, Carlos Bruno Barreto, Danilo Diniz Siqueira, Eduardo da Silva Barbosa Ferreira, Wallisson Alves da Silva, Jessika Andrade dos Santos Nogueira, and Edcleide Maria Araújo. 2020. "From Disposal to Technological Potential: Reuse of Polypropylene Waste from Industrial Containers as a Polystyrene Impact Modifier" Sustainability 12, no. 13: 5272. https://doi.org/10.3390/su12135272
APA StyleLuna, C. B. B., Siqueira, D. D., Ferreira, E. d. S. B., Silva, W. A. d., Nogueira, J. A. d. S., & Araújo, E. M. (2020). From Disposal to Technological Potential: Reuse of Polypropylene Waste from Industrial Containers as a Polystyrene Impact Modifier. Sustainability, 12(13), 5272. https://doi.org/10.3390/su12135272





