COVID-19 Healthcare Planning: Predicting Mortality and the Role of the Herd Immunity Barrier in the General Population
Abstract
1. Introduction
2. Materials and Methods
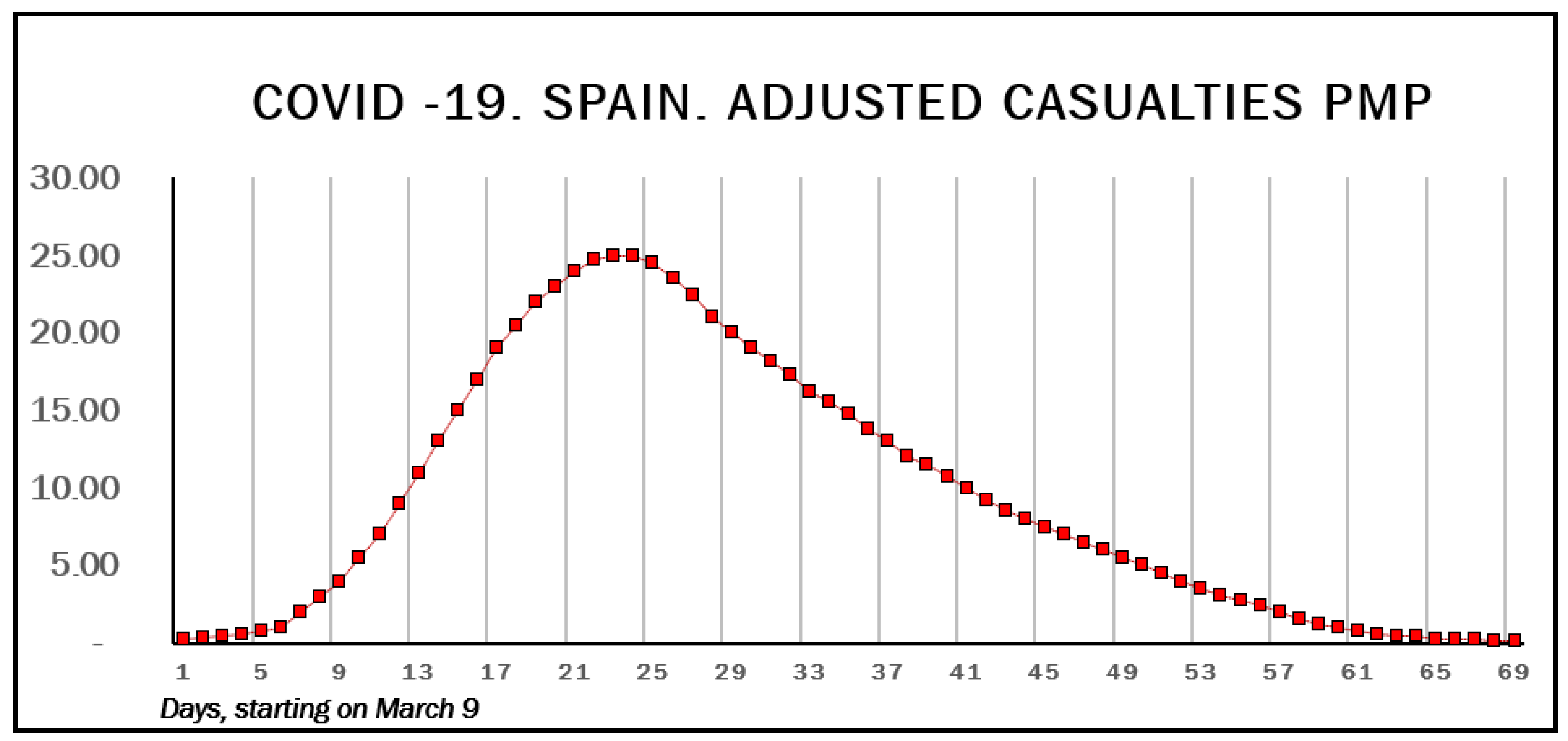
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baker, T. Mathematical modeling draws more accurate picture of coronavirus cases 2020. Available online: https://www.eurekalert.org/pub_releases/2020-04/mcog-mmd041020.php (accessed on 20 June 2020).
- Red Nacional de Vigilancia Epidemiológica-[Spanish epidemiological surveillance network]. Informe sobre la situación de COVID-19 en España-[Report on the situation of COVID-19 in Spain]. 2020. Available online: https://www.isciii.es/QueHacemos/Servicios/VigilanciaSaludPublicaRENAVE/EnfermedadesTransmisibles/Documents/INFORMES/Informes%20COVID-19/Informe%20nº%2022.%20Situación%20de%20COVID-19%20en%20España%20a%2013%20de%20abril%20de%202020.pdf (accessed on 20 June 2020).
- Ministerio de Justicia [Spanish Ministry of Justice]. MoMo-Vigilancia de los excesos de mortalidad por todas las causas [Surveillance of excess mortality from all causes (MoMo)]; Ministerio de Justicia [Spanish Ministry of Justice]: Madrid, Spain, 2020; Available online: https://www.isciii.es/QueHacemos/Servicios/VigilanciaSaludPublicaRENAVE/EnfermedadesTransmisibles/MoMo/Documents/informesMoMo2020/MoMo_Situacion%20a%2011%20de%20mayo_CNE.pdf (accessed on 20 June 2020).
- Flaxman, S.; Mishra, S.; Gandy, A.; Unwin, J.T.; Coupland, H.; Mellan, T.A.; Zhu, H.; Berah, T.; Eaton, J.W.; Guzman, P.N.P.; et al. Estimating the number of infections and the impact of non-pharmaceutical interventions on COVID-19 in 11 European countries. Imp. Coll. Lond. 2020, 1–35. [Google Scholar] [CrossRef]
- Jung, S.; Akhmetzhanov, A.R.; Hayashi, K.; Linton, N.M.; Yang, Y.; Yuan, B.; Kobayashi, T.; Kinoshita, R.; Nishiura, H. Real-time estimation of the risk of death from novel coronavirus (covid-19) infection: Inference using exported cases. J. Clin. Med. 2020, 9, 523. [Google Scholar] [CrossRef] [PubMed]
- Linton, N.M.; Kobayashi, T.; Yang, Y.; Hayashi, K.; Akhmetzhanov, A.R.; Jung, S.; Yuan, B.; Kinoshita, R.; Nishiura, H. Incubation period and other epidemiological characteristics of 2019 Novel Coronavirus infections with right truncation: A Statistical analysis of publicly available case data. J. Clin. Med. 2020, 9, 538. [Google Scholar] [CrossRef] [PubMed]
- Salje, H.; Kiem, C.T.; Lefrancq, N.; Courtejoie, N.; Paireau, J.; Andronico, A.; Hoze, N.; Richet, J.; Dubost, C.; Salje, H.; et al. Estimating the Burden of SARS-CoV-2 in France to Cite this Version: HAL Id: Pasteur-02548181; Institute Pasteur: Paris, France, 2020; Available online: https://hal-pasteur.archives-ouvertes.fr/pasteur-02548181/document (accessed on 20 June 2020).
- Instituto Nacional de Estadística (INE) [National Institute of Statistics of Spain]. Defunciones según la Causa de Muerte año 2018. Notas de prensa. Defunciones por causa muerte 2018 [Death by cause death, 2018] 2019, 2018, 1–8. Available online: https://www.ine.es/prensa/edcm_2018.pdf (accessed on 20 June 2020).
- Streeck, H.; Hartmann, G.; Exner, M.; Schmid, M. Vorläufiges Ergebnis und Schlussfolgerungen der COVID-19 Case-Cluster-Study (Gemeinde Gangelt) [Preliminary results and conclusions of the COVID-19 case group study (Gangelt municipality); University Hospital Bonn: Bonn, Germany, 2020; Available online: https://www.land.nrw/sites/default/files/asset/document/zwischenergebnis_covid19_case_study_gangelt.pdf (accessed on 20 June 2020).
- Rajgor, D.D.; Lee, M.H.; Archuleta, S.; Bagdasarian, N.; Quek, S.C. The many estimates of the COVID-19 case fatality rate. Lancet Infect. Dis. 2020, 3099, 30244. [Google Scholar] [CrossRef]
- Mizumoto, K.; Kagaya, K.; Zarebski, A.; Chowell, G. Estimating the asymptomatic proportion of coronavirus disease 2019 (COVID-19) cases on board the Diamond Princess cruise ship, Yokohama, Japan, 2020. Eurosurveillance 2020, 25, 2000180. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.T.; Leung, K.; Bushman, M.; Kishore, N.; Niehus, R.; de Salazar, P.M.; Cowling, B.J.; Lipsitch, M.; Leung, G.M. Estimating clinical severity of COVID-19 from the transmission dynamics in Wuhan, China. Nat. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Riou, J.; Hauser, A.; Counotte, M.J.; Althaus, C.L. Adjusted age-specific case fatality ratio during the COVID-19 epidemic in Hubei, China, January and February 2020. medRxiv 2020. [Google Scholar] [CrossRef]
- Russell, T.W.; Hellewell, J.; Jarvis, C.I.; van Zandvoort, K.; Abbott, S.; Ratnayake, R.; Flasche, S.; Eggo, R.M.; Edmunds, W.J.; Kucharski, A.J. Estimating the infection and case fatality ratio for coronavirus disease (COVID-19) using age-adjusted data from the outbreak on the Diamond Princess cruise ship, February 2020. Eurosurveillance 2020, 25, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a report of 72314 Cases from the Chinese Center for Disease Control and Prevention. JAMA J. Am. Med. Assoc. 2020, 323. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Wang, X.; Ma, Z.; Ning, Y.; Chen, C.; Chen, R.; Chen, Q.; Zhang, H.; Li, C.; He, Y.; Wang, T.; et al. Estimating the case fatality ratio of the COVID-19 epidemic in China. medRxiv 2020. [Google Scholar] [CrossRef]
- Oke, J.; Heneghan, C. Global Covid-19 Case Fatality Rates; Centre for Evidence-Based Medicine, Oxford University: Oxford, UK, 2020; Available online: https://www.cebm.net/covid-19/global-covid-19-case-fatality-rates/ (accessed on 20 June 2020).
- Italian Task Force COVID-19. Epidemia COVID-19 [Covid-19 epidemic]. 2020. Available online: https://www.epicentro.iss.it/coronavirus/bollettino/Bollettino-sorveglianza-integrata-COVID-19_26-marzo%202020.pdf (accessed on 20 June 2020).
- Weaver, M. UK coronavirus death toll reaches 1,789 amid data reporting concerns |World news| The Guardian. Available online: https://www.theguardian.com/world/2020/mar/31/uk-coronavirus-death-toll-reaches-1789-amid-data-reporting-concerns (accessed on 7 May 2020).
- London Business School. How Many People Really Die From Covid-19? Lessons from Italy. Forbes, 6 April 2020. [Google Scholar]
- Wu, J.; McCann, A.; Peltier, E. Coronavirus-missing-deaths. Available online: https://www.nytimes.com/interactive/2020/04/21/world/coronavirus-missing-deaths.html (accessed on 10 May 2020).
- Lipsitch, M.; Cohen, T.; Cooper, B.; Robins, J.M.; Ma, S.; James, L.; Gopalakrishna, G.; Chew, S.K.; Tan, C.C.; Samore, M.H.; et al. Transmission dynamics and control of severe acute respiratory syndrome. Science 2003, 300, 1966–1970. [Google Scholar] [CrossRef] [PubMed]
- Sanche, S.; Lin, Y.T.; Xu, C.; Romero-Severson, E.; Hengartner, N.; Ke, R. High Contagiousness and rapid spread of severe acute respiratory syndrome coronavirus 2. Emerg. Infect. Dis. 2020, 26. [Google Scholar] [CrossRef] [PubMed]
- Holmes, E.C. The Evolution and Emergence of RNA Viruses; Oxford University Press: Oxford, UK, 2010; Volume 16, ISBN 9780199211135. [Google Scholar]
- World Health Organization. Consensus Document on The Epidemiology of Severe Acute Respiratory Syndrome (SARS); WHO Publishing: Geneva, Switzerland, 2003; pp. 1–47. Available online: https://www.who.int/csr/sars/WHOconsensus.pdf?ua=1 (accessed on 7 May 2020).
- Boseley, S. WHO warns that few have developed antibodies to Covid-19. The Guardian. 2020. Available online: https://www.theguardian.com/society/2020/apr/20/studies-suggest-very-few-have-had-covid-19-without-symptoms (accessed on 20 June 2020).
- World Health Organization (WHO) Tech Guid #2. Health Systems Respond to COVID-19 Technical Guidance #2 Creating surge capacity for acute and intensive care Recommendations for the WHO European Region (6 April 2020). JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef]
- Verity, R.; Okell, L.C.; Dorigatti, I.; Winskill, P.; Whittaker, C.; Imai, N.; Cuomo-Dannenburg, G.; Thompson, H.; Walker, P.; Fu, H.; et al. Estimates of the severity of COVID-19 disease. medRxiv 2020. [Google Scholar] [CrossRef]
- Wong, J.Y.; Kelly, H.; Ip, D.K.M.; Wu, J.T.; Leung, G.M.; Cowling, B.J. Case fatality risk of influenza a (H1N1pdm09): A systematic review. Epidemiology 2013, 24. [Google Scholar] [CrossRef] [PubMed]
- Cámara-Larrauri, A.; Jiménez-Jorge, S.; Simón-Méndez, L.; de Mateo-Ontañón, S. Vigilancia de la pandemia de la gripe 2009 (H1N1) en España [Surveillance of 2009 flu (H1N1) pandemic in Spain]. Rev. Esp. Salud Publica 2010, 24, 569–588. [Google Scholar]
- Centro de coordinación de alertas y emergencias sanitarias; R98-[Spanish centre of health alerts and emergencies. R98]. Actualización no 98. Enfermedad por el coronavirus (COVID-19)- [Update No 98. Coronavirus (Covid-19)]; Centro de coordinación de alertas y emergencias sanitarias: Madrid, Spain, 2020; Available online: https://www.mscbs.gob.es/profesionales/saludPublica/ccayes/alertasActual/nCov-China/documentos/Actualizacion_98_COVID-19.pdf (accessed on 7 May 2020).
- Bendavid, E.; Mulaney, B.; Sood, N.; Shah, S.; Ling, E.; Bromley-Dulfano, R.; Lai, C.; Weissberg, Z.; Saavedra, R.; Tedrow, J.; et al. COVID-19 Antibody Seroprevalence in Santa Clara County, California. medRxiv 2020. [Google Scholar] [CrossRef]
- Regalado, A. Blood tests show 14% of people are now immune to covid-19 in one town in Germany. MIT Technology Review, 9 April 2020. [Google Scholar]
- Wu, Z.; McGoogan, J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China. JAMA 2020, 323, 1239–1242. [Google Scholar] [CrossRef] [PubMed]
- Meyerowitz-Katz, G. Here’s Why Herd Immunity Won’t Save Us From The COVID-19 Pandemic. Science Alert, 30 March 2020. [Google Scholar]
| Total Cases | Expected Cases | Unexpected | Reported COVID-19 | Report Under-Estimation | |
|---|---|---|---|---|---|
| 63,676 | 37,769 | 25,907 | 20,043 | 5864 | 29% |
| Regions (CCAA) | Total Casualties | CFR PMP | Regions (CCAA) | Total Casualties | CFR PMP |
|---|---|---|---|---|---|
| Andalucía | 1501 | 178.7 | Valencia | 1563 | 312.7 |
| Aragón | 939 | 708.6 | Extremadura | 556 | 523.4 |
| Asturias | 328 | 321.8 | Galicia | 519 | 192.4 |
| Baleares | 231 | 192.5 | Madrid | 10,442 | 1562.0 |
| Canarias | 173 | 77.8 | Melilla | 3 | 31.3 |
| Cantabria | 241 | 414.5 | Murcia | 167 | 112.0 |
| Castilla La Mancha | 3071 | 1506.7 | Navarra | 568 | 870.7 |
| Castilla y León | 2196 | 913.9 | Pais Vasco | 1621 | 743.1 |
| Cataluña | 6019 | 790.0 | La Rioja | 415 | 1.320.3 |
| Ceuta | 5 | 62.4 | |||
| Spain (Overall) | 30,568 | 649.0 |
| Sample Size | PCR Positive | Over Infected | Over PCR + | Over Sample | |||
| True Asymp | Symp. | IFR | CFR | Casualties | |||
| 21% | 18% | 82% | 1.11% | 1.35% | 0.23% | ||
| (n) | 3063 | 630 | 113.3 | 516.7 | 7 | ||
| Age | Casualties | Cases | % CFR (95% CI) |
|---|---|---|---|
| ≤9 years | 0 | 416 | 0 |
| 10 to 19 years | 1 | 549 | 0.18 [0.03–1.02] |
| 20 to 49 years | 63 | 19,790 | 0.32 [0.25–0.41] |
| 50 to 59 years | 130 | 10,008 | 1.3 [1.1–1.5] |
| 60 to 69 years | 309 | 8583 | 3.6 [3.2–4.0] |
| 70 to 79 years | 312 | 3918 | 8.0 [7.2–8.9] |
| ≥80 years | 208 | 1408 | 14.8 [13.0–16.7] |
| Overall | 1023 | 44,415 | 2.30 |
| Total Cases | |||||
| Age Band | # Cases | % Age Band | # Death | % Death/ Age Band | % CFR |
| 0–9 | 424 | 0.6 | - | 0.0 | 0.00 |
| 10–19 | 510 | 0.7 | - | 0.0 | 0.00 |
| 20–29 | 2713 | 3.7 | - | 0.0 | 0.00 |
| 30–30 | 4959 | 6.8 | 17 | 0.2 | 0.34 |
| 40-49 | 9167 | 12.6 | 67 | 1.0 | 0.73 |
| 50–59 | 14,335 | 19.7 | 243 | 3.6 | 1.70 |
| 60–69 | 13,149 | 18.1 | 761 | 11.2 | 5.79 |
| 70–79 | 14,090 | 19.4 | 2403 | 35.3 | 17.05 |
| 80–89 | 10,929 | 15.0 | 2702 | 39.7 | 24.72 |
| ≥90 | 2517 | 3.5 | 608 | 8.9 | 24.16 |
| Total | 72,793 | 100.0 | 6801 | 100.0 | 9.34 |
| Males | |||||
| Age Band | # Cases | % Gender | # Death | % Death/ Gender | % CFR |
| 0–9 | 244 | 57.5 | - | 0.0 | 0.00 |
| 10–19 | 261 | 51.2 | - | 0.0 | 0.00 |
| 20–29 | 1203 | 44.3 | - | 0.0 | 0.00 |
| 30–30 | 2465 | 49.7 | 14 | 82.4 | 0.57 |
| 40–49 | 4597 | 50.1 | 49 | 73.1 | 1.07 |
| 50–59 | 7998 | 55.8 | 190 | 78.2 | 2.38 |
| 60–69 | 8755 | 66.6 | 606 | 79.6 | 6.92 |
| 70–79 | 9309 | 66.1 | 1846 | 76.8 | 1.83 |
| 80–89 | 6195 | 56.7 | 1808 | 66.9 | 29.18 |
| ≥90 | 877 | 34.8 | 273 | 44.9 | 31.13 |
| Total | 41,904 | 57.6 | 4786 | 70.4 | 11.42 |
| Females | |||||
| Age Band | # Cases | % Gender | # Death | % Death/ Gender | % CFR |
| 0–9 | 180 | 42.5 | - | 0.0 | 0.00 |
| 10–19 | 249 | 48.8 | - | 0.0 | 0.00 |
| 20–29 | 1510 | 55.7 | - | 0.0 | 0.00 |
| 30–30 | 2494 | 50.3 | 3 | 17.6 | 0.12 |
| 40-49 | 4570 | 49.9 | 18 | 26.9 | 0.39 |
| 50–59 | 6337 | 44.2 | 52 | 21.4 | 0.82 |
| 60-69 | 4394 | 33.4 | 154 | 20.2 | 3.50 |
| 70–79 | 4781 | 33.9 | 555 | 23.1 | 11.61 |
| 80-89 | 4734 | 43.3 | 894 | 33.1 | 18.88 |
| ≥90 | 1640 | 65.2 | 334 | 54.9 | 20.37 |
| Total | 30889 | 42.4 | 2010 | 29.6 | 6.51 |
| Category | Risk Ratio | 95% CI |
|---|---|---|
| Age 30–39 | 0.06 | [0.038–0.10] |
| Age 40–49 | 0.14 | [0.11–0.17] |
| Age 50–59 | 0.31 | [0.27–0.35] |
| Age 60–69 (reference) | 1 | - |
| Age 70–79 | 2.95 | [2.7–3.2] |
| Age 80–89 | 4.47 | [4.1–4.8] |
| Age 90+ | 4.83 | [4.4–5.3] |
| Female | 1 | - |
| Male | 1.66 | [1.58–1.74] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marco-Franco, J.E.; Guadalajara-Olmeda, N.; González-de Julián, S.; Vivas-Consuelo, D. COVID-19 Healthcare Planning: Predicting Mortality and the Role of the Herd Immunity Barrier in the General Population. Sustainability 2020, 12, 5228. https://doi.org/10.3390/su12135228
Marco-Franco JE, Guadalajara-Olmeda N, González-de Julián S, Vivas-Consuelo D. COVID-19 Healthcare Planning: Predicting Mortality and the Role of the Herd Immunity Barrier in the General Population. Sustainability. 2020; 12(13):5228. https://doi.org/10.3390/su12135228
Chicago/Turabian StyleMarco-Franco, Julio Emilio, Natividad Guadalajara-Olmeda, Silvia González-de Julián, and David Vivas-Consuelo. 2020. "COVID-19 Healthcare Planning: Predicting Mortality and the Role of the Herd Immunity Barrier in the General Population" Sustainability 12, no. 13: 5228. https://doi.org/10.3390/su12135228
APA StyleMarco-Franco, J. E., Guadalajara-Olmeda, N., González-de Julián, S., & Vivas-Consuelo, D. (2020). COVID-19 Healthcare Planning: Predicting Mortality and the Role of the Herd Immunity Barrier in the General Population. Sustainability, 12(13), 5228. https://doi.org/10.3390/su12135228







