Influence of Ripening on Chemical Characteristics of a Traditional Italian Cheese: Provolone del Monaco
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cheese-Making
2.2. Sampling
2.3. pH and Moisture Content
2.4. Titratable Acidity
2.5. Nitrogen Content
2.6. Fat Content
2.7. Gas Chromatographic Analysis of Fatty Acids
2.8. Volatile Organic Compounds Analysis
2.9. Statistical Analysis
3. Results
3.1. Moisture, pH, and Titratable Acidity
3.2. Nitrogen Content
3.3. Fat Content
3.4. Fatty Acid Profile
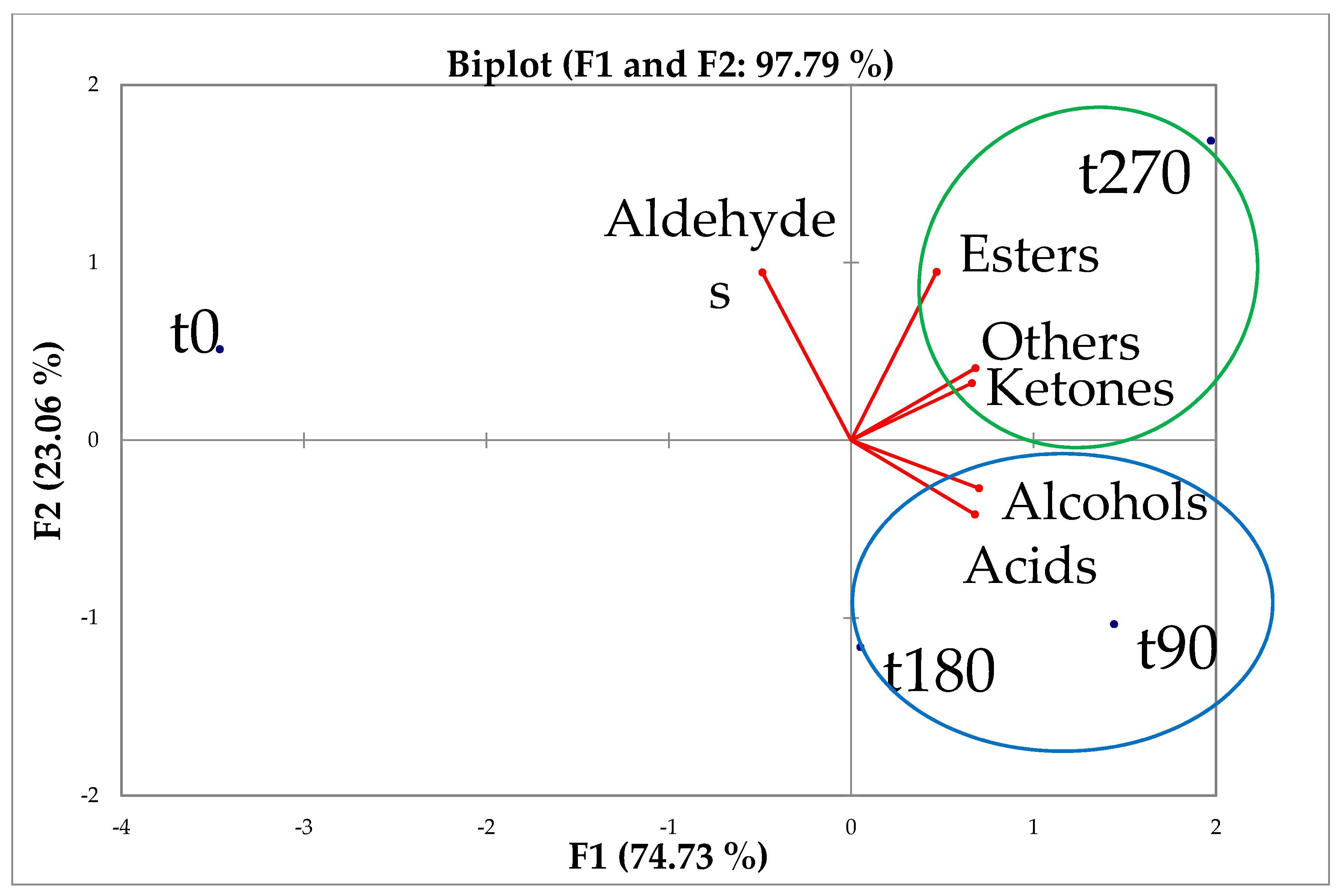
3.5. Volatile Organic Compounds
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- DOP Italian Regulation 10A02770, 2010, n.56 of 09.03.2010. Available online: https://www.gazzettaufficiale.it/do/atto/serie_generale/caricaPdf?cdimg=10A0277000100010110012&dgu=2010-03-09&art.dataPubblicazioneGazzetta=2010-03-09&art.codiceRedazionale=10A02770&art.num=1&art.tiposerie=SG (accessed on 25 March 2019).
- Aponte, M.; Fusco, V.; Andolfi, R.; Coppola, S. Lactic acid bacteria occurring during manufacture and ripening of Provolone del Monaco cheese: Detection by different analytical approaches. Int. Dairy J. 2008, 18, 403–413. [Google Scholar] [CrossRef]
- Romano, R.; Boriello, I.; Macaldi, C.; Giordano, A.; Spagna Musso, S. Influenza del grado di stagionatura sulla composizione in acidi grassi, ω-3 e CLA del Provolone del Monaco. Prog. Nutr. 2008, 10, 165–173. [Google Scholar]
- Romano, R.; Masucci, F.; Giordano, A.; Spagna Musso, S.; Naviglio, D.; Santini, A. Effect of tomato by-products in the diet of Comisana sheep on composition and conjugated linoleic acid content of milk fat. Int. Dairy J. 2010, 20, 858–862. [Google Scholar] [CrossRef]
- Di Monaco, R.; Di Marzo, S.; Cavella, S.; Masi, P. Valorization of traditional foods: The case of Provolone del Monaco cheese. Br. Food J. 2005, 107, 98–110. [Google Scholar] [CrossRef]
- Delgado, F.J.; González-Crespo, J.; Ladero, L.; Cava, R.; Ramírez, R. Free fatty acids and oxidative changes of a Spanish soft cheese (PDO ‘Torta del Casaro’) during ripening. Int. J. Food Sci. Technol. 2009, 44, 1721–1728. [Google Scholar] [CrossRef]
- Romano, R.; Giordano, A.; Le Grottaglie, L.; Manzo, N.; Paduano, A.; Sacchi, R.; Santini, A. Volatile compounds in intermittent frying by gas chromatography and nuclear magnetic resonance. Eur. J. Lipid Sci. Technol. 2013, 115, 764–773. [Google Scholar] [CrossRef]
- Thierry, A.; Collins, Y.F.; Mukdsi, M.C.A.; Mcsweeney, P.L.H.; Wilkinson, M.G.; Spinnler, H.E. Lipolysis and Metabolism of Fatty Acids in Cheese. In Cheese, Chemistry, Physics and Microbiology, 4th ed.; Academic Press—Elsevier: San Diego, CA, USA, 2017; ISBN 9780124170124. [Google Scholar]
- Hassan, F.A.M.; Abd El-Gawad, M.A.M.; Enab, A.K. Flavour Compounds in Cheese (Review). Res. Prec. Instr. Mach. 2013, 2, 15–29. [Google Scholar] [CrossRef]
- Aydin, C.M. Formation of volatile and non-volatile compounds in cheese. Int. J. Sci. Technol. Res. 2017, 6, 252–263. [Google Scholar]
- McSweeney, P.L.; Sousa, M.J. Biochemical pathways for the production of flavour compounds in cheeses during ripening: A review. Lait 2000, 80, 293–324. [Google Scholar] [CrossRef]
- Bradley, R.L.; Vanderwarn, A.A. Determination of moisture in cheese and cheese products. AOAC Int. 2001, 84, 570–592. [Google Scholar]
- D.M. Official Method of Cheese Analysis. Ministry Regulation April 21 1986. Available online: https://www.gazzettaufficiale.it/do/gazzetta/serie_generale/3/pdfPaginato?dataPubblicazioneGazzetta=19861002&numeroGazzetta=229&tipoSerie=SG&tipoSupplemento=SO&numeroSupplemento=88&progressivo=0&numPagina=4&edizione=0&elenco30giorni= (accessed on 27 March 2019).
- Nota, G.; Spagna Musso, S.; Romano, R.; Naviglio, D.; Improta, C. Idrolisi rapida degli esteri degli steroli nei grassi. Riv. Italiana Sostanze Grasse 1995, 72, 315–316. [Google Scholar]
- Lee, J.H.; Diono, R.; Kim, G.Y.; Min, D.B. Optimization of solid phase microextraction analysis for the headspace volatile compounds of Parmesan cheese. J. Agric. Food Chem. 2003, 51, 1136–1140. [Google Scholar] [CrossRef]
- Fox, P.F.; Singh, T.K.; McSweeney, P.L.H. Proteolysis in cheese during ripening. Food Rev. Int. 2005, 4, 457–509. [Google Scholar]
- McSweeney, P.L.H. Biochemistry of cheese ripening. Int. J. Food Microbiol. 2004, 57, 127–144. [Google Scholar] [CrossRef]
- Akın, N.; Aydemir, S.; Koçak, C.; Yıldız, M.A. Changes of free fatty acid contents and sensory properties of white pickled cheese during ripening. Food Chem. 2003, 80, 77–83. [Google Scholar] [CrossRef]
- Bintsis, T.; Papademas, P. An overview of the cheesemaking process. In Global Cheesemaking Technology: Cheese Quality and Characteristics, 1st ed.; Papademas, P., Bintsis, T., Eds.; John Wiley & Sons, Ltd.: New York, NY, USA, 2017; pp. 120–156. [Google Scholar]
- Gobbetti, M.; Morea, M.; Baruzzi, F.; Corbo, M.R.; Matarante, A.; Considine, T.; Fox, P.F. Microbiological, compositional, biochemical and textural characterisation of Caciocavallo Pugliese cheese during ripening. Int. Dairy J. 2002, 12, 511–523. [Google Scholar] [CrossRef]
- Almena-Aliste, M.; Mietton, B. Cheese classification, characterization and categorization: A global perspective. In Cheese and Microbes, 1st ed.; Donnelly, C.W., Ed.; ASM Press: Herndon, VA, USA, 2014; pp. 39–72. [Google Scholar]
- Naviglio, D.; Romano, R.; Pizzolongo, F.; Santini, A.; De Vivo, A.; Schiavo, L.; Nota, G.; Spagna Musso, S. Rapid determination of esterified glycerol and glycerides in triglycerides fats and oils by means of periodate method after transesterfication. Food Chem. 2007, 102, 399–405. [Google Scholar] [CrossRef]
- Cornu, A.; Rabiau, N.; Kondjoyan, N.; Verdier-Metz, I.; Pradel, P.; Tournayre, P.; Berdagué, J.L.; Martin, B. Odour-active compound profiles in Cantal-type cheese: Effect of cow diet, milk pasteurization and cheese ripening. Int. Dairy J. 2009, 19, 588–594. [Google Scholar] [CrossRef]
- Esposito, G.; Masucci, F.; Napolitano, F.; Braghieri, A.; Romano, R.; Manzo, N.; Di Francia, A. Fatty acid and sensory profiles of Caciocavallo cheese as affected by management system. J. Dairy Sci. 2014, 97, 1918–1928. [Google Scholar] [CrossRef]
- Scerra, M.; Chies, L.; Caparra, P.; Cilione, C.; Foti, F. Effect of only pasture on fatty acid composition of cow milk and Ciminà caciocavallo cheese. J. Food Res. 2016, 5, 20–28. [Google Scholar] [CrossRef]
- Rodríguez-Alcalá, L.M.; Castro-Gómez, M.P.; Pimentel, L.L.; Fontec, J. Milk fat components with potential anticancer activity—A review. Biosci. Rep. 2017, 37, BSR20170705. [Google Scholar] [CrossRef]
- den Hartigh, L.J. Conjugated Linoleic Acid Effects on Cancer, Obesity, and Atherosclerosis: A Review of Pre-Clinical and Human Trials with Current Perspectives. Nutrients 2019, 11, 370. [Google Scholar] [CrossRef]
- Zhang, J.; Yi, M.; Zha, L.; Chen, S.; Li, Z.; Li, C.; Gong, M.; Deng, H.; Chu, X.; Chen, J.; et al. Sodium butyrate induces endoplasmic reticulum stress and autophagy in colorectal cells: Implications for apoptosis. PLoS ONE 2016, 11, e0147218. [Google Scholar] [CrossRef]
- Borycka-Kiciak, K.; Banasiewicz, T.; Rydzewska, G. Butyric acid—A well-known molecule revisited. Gastroenterol. Rev. 2017, 12, 83–89. [Google Scholar] [CrossRef]
- Deeth, H.C. Lipoprotein lipase and lipolysis in milk. Int. Dairy J. 2006, 16, 555–562. [Google Scholar] [CrossRef]
- Singh, T.K.; Drake, M.A.; Cadwallader, K.R. Flavor of Cheddar cheese: A chemical and sensory perspective. Compr. Rev. Food Sci. Food Saf. 2003, 2, 166–189. [Google Scholar] [CrossRef]
- Karagul-Yuceer, Y.; Drake, M.A.; Cadwallader, K.R. Aroma-active components of liquid Cheddar whey. J. Food Sci. 2003, 68, 1215–1219. [Google Scholar] [CrossRef]
- Karagul-Yuceer, Y.; Vlahovich, K.N.; Drake, M.A.; Cadwallader, K.R. Characteristic aroma compounds of rennet casein. J. Agric. Food Chem. 2003, 51, 6797–6801. [Google Scholar] [CrossRef]
- Marilley, L.; Casey, M.G. Flavours of cheese products: Metabolic pathways, analytical tools and identification of producing strains. Int. J. Food Microbiol. 2004, 90, 139–159. [Google Scholar] [CrossRef]
- Lecanu, L.; Ducruet, V.; Jouquand, C.; Gratadoux, J.J.; Feigenbaum, A. Optimization of headspace solid-phase microextraction (SPME) for the odour analysis of surface-ripened cheese. J. Agric. Food Chem. 2002, 50, 3810–3817. [Google Scholar] [CrossRef]
- Kaminarides, S.; Stamou, P.; Massouras, T. Changes of organic acids, volatile aroma compounds and sensory characteristics of Halloumi cheese kept in brine. Food Chem. 2007, 100, 219–225. [Google Scholar] [CrossRef]
- Varming, C.; Agerlin, M.; Thomas, P.; Ardö, S. Challenges in quantitative analysis of aroma compounds in cheeses with different fat content and maturity level. Int. Dairy J. 2013, 29, 15–20. [Google Scholar] [CrossRef]
- Sádecká, J.; Kolek, E.; Pangallo, D.; Valík, L.; Kuchta, T. Principal volatile odorants and dynamics of their formation during the production of May Bryndza cheese. Food Chem. 2014, 150, 301–306. [Google Scholar] [CrossRef]
- Di Marzo, S.; Di Monaco, R.; Cavella, S.; Romano, R.; Borriello, I.; Masi, P. Correlation between sensory and instrumental properties of Canestrato Pugliese slices packed in biodegradable films. Trends Food Sci. Technol. 2006, 17, 169–176. [Google Scholar] [CrossRef]
- Romano, R.; Santini, A.; Le Grottaglie, L.; Manzo, N.; Visconti, A.; Ritieni, A. Identification markers based on fatty acid composition to differentiate between roasted Arabica and Canephora (Robusta) coffee varieties in mixtures. J. Food Compos. Anal. 2014, 35, 1–9. [Google Scholar] [CrossRef]
- Pannico, A.; Schouten, R.E.; Basile, B.; Romano, R.; Woltering, E.J.; Cirillo, C. Non-destructive detection of flawed hazelnut kernels and lipid oxidation assessment using NIR spectroscopy. J. Food Eng. 2015, 160, 42–48. [Google Scholar] [CrossRef]
- Durazzo, A.; Kiefer, J.; Lucarini, M.; Marconi, S.; Lisciani, S.; Camilli, E.; Gambelli, L.; Gabrielli, P.; Aguzzi, A.; Finotti, E.; et al. An innovative and integrated food research approach: Spectroscopy applications to milk and a case study of a milk-based dish. Braz. J. Anal. Chem. 2018, 5, 12–27. [Google Scholar] [CrossRef]
- Durazzo, A.; Kiefer, J.; Lucarini, M.; Camilli, E.; Marconi, S.; Gabrielli, P.; Aguzzi, A.; Gambelli, L.; Lisciani, S.; Marletta, L. Qualitative Analysis of Traditional Italian Dishes: FTIR Approach. Sustainability 2018, 10, 4112. [Google Scholar] [CrossRef]

| Samples | Ripening Days | pH | Moisture % | Titratable Acidity (Lact. ac. %) | Total N (g/100 g dw) | Soluble N (g/100 g dw) | SN/TN*100 | Fat (g/100 g dw) |
|---|---|---|---|---|---|---|---|---|
| C3 | 0 | 5.03 b ± 0.25 | 46.20 a ± 1.34 | 0.17 ± 0.02 | 7.17 ± 0.15 | 0.49 c ± 0.01 | 6.87 c ± 0.05 | 42.96 ± 2.81 |
| 90 | 5.63 a,b ± 0.02 | 40.03 b ± 0.85 | 0.14 ± 0.01 | 6.82 ± 0.13 | 1.00 b ± 0.01 | 14.74 b ± 0.15 | 41.65 ± 1.13 | |
| 180 | 5.77 a ± 0.18 | 40.61 b ± 0.54 | 0.22 + 0.03 | 7.02 ± 0.03 | 1.08 b ± 0.03 | 15.36 b ± 0.42 | 43.45 ± 0.13 | |
| 270 | 5.68 a,b ± 0.10 | 38.35 b ± 0.13 | 0.22 ± 0.03 | 7.06 ± 0.18 | 1.70 a ± 0.08 | 24.02 a ± 0.59 | 39.89 ± 2.09 | |
| C5 | 0 | 5.30 c ± 0.04 | 47.05 a ± 0.92 | 0.16 a,b ± 0.01 | 7.10 ± 0.23 | 0.50 d ± 0.02 | 6.99 c ± 0.58 | 42.84 ± 1.83 |
| 90 | 5.79 b ± 0.03 | 42.93 b ± 0.64 | 0.13 b ± 0.01 | 7.11 ± 0.08 | 1.12 c ± 0.06 | 15.70 b ± 0.61 | 42.65 ± 1.26 | |
| 180 | 5.97 a,b ± 0.01 | 42.11 b,c ± 1.05 | 0.21 a ± 0.01 | 7.12 ± 0.24 | 1.30 b ± 0.04 | 18.33 b ± 1.14 | 43.51 ± 1.17 | |
| 270 | 6.02 a ± 0.08 | 39.51 c ± 0.27 | 0.22 a ± 0.03 | 7.17 ± 0.03 | 2.04 a ± 0.06 | 28.46 a ± 0.88 | 41.88 ± 1.24 | |
| S3 | 0 | 5.23 b ± 0.06 | 43.80 a ± 1.89 | 0.18 b ± 0.01 | 7.01 ± 0.56 | 0.40 c ± 0.01 | 5.73 c ± 0.27 | 41.67 ± 3.28 |
| 90 | 5.49 a,b ± 0.03 | 31.63 b ± 1.18 | 0.13 c ± 0.01 | 6.62 ± 0.12 | 0.80 b ± 0.01 | 12.11 b ± 0.40 | 39.40 ± 2.39 | |
| 180 | 5.70 a ± 0.11 | 31.92 b ± 3.03 | 0.24 a ± 0.01 | 7.15 ± 0.25 | 1.03 a ± 0.04 | 14.44 a ± 0.06 | 42.10 ± 3.35 | |
| 270 | 5.69 a ± 0.06 | 29.35 b ± 0.61 | 0.21 a,b ± 0.01 | 7.03 ± 0.04 | 1.14 a ± 0.06 | 16.16 a ± 0.84 | 39.84 ± 0.17 | |
| S5 | 0 | 5.29 c ± 0.02 | 45.76 a ± 0.02 | 0.18 b ± 0.01 | 7.53 a ± 0.04 | 0.47 b ± 0.01 | 6.30 b ± 0.12 | 42.91 ± 3.22 |
| 90 | 5.46 b ± 0.01 | 36.89 b ± 2.69 | 0.13 c ± 0.01 | 6.74 c ± 0.04 | 1.29 a ± 0.09 | 19.12 a ± 1.28 | 41.39 ± 1.86 | |
| 180 | 5.73 a ± 0.02 | 34.81 b ± 2.33 | 0.22 a ± 0.01 | 7.36 b,c ± 0.21 | 1.18 a ± 0.04 | 16.06 a ± 0.11 | 42.18 ± 4.72 | |
| 270 | 5.77 a ± 0.08 | 31.69 b ± 0.31 | 0.21 a,b ± 0.01 | 7.02 b,c ± 0.12 | 1.21 a ± 0.11 | 17.25 a ± 1.34 | 37.18 ± 0.53 |
| C | S | |||||||
|---|---|---|---|---|---|---|---|---|
| 0 | 90 | 180 | 270 | 0 | 90 | 180 | 270 | |
| (a) | ||||||||
| C4:0 | 3.04 ± 0.05 | 3.07 ± 0.00 | 3.13 ± 0.02 | 3.10 ± 0.04 | 2.81 ± 0.18 | 3.00 ± 0.18 | 3.01 ± 0.28 | 3.22 ± 0.22 |
| C6:0 | 1.93 a,b ± 0.05 | 1.84 b ± 0.05 | 2.03 a,b ± 0.05 | 2.13 a,b ± 0.07 | 1.93 a,b ± 0.07 | 1.97 a,b ± 0.09 | 2.05 a,b ± 0.11 | 2.19 a ± 0.13 |
| C8:0 | 1.18 ± 0.10 | 1.14 ± 0.00 | 1.23 ± 0.02 | 1.42 ± 0.24 | 1.18 ± 0.08 | 1.27 ± 0.08 | 1.20 ± 0.12 | 1.26 ± 0.13 |
| C10:0 | 2.49 c ± 0.04 | 2.51 b,c ± 0.01 | 2.78 a,b ± 0.05 | 2.83 a ± 0.07 | 2.65 a,b,c ± 0.08 | 2.63 a,b,c ± 0.02 | 2.55 a,b,c ± 0.11 | 2.53 b,c ± 0.12 |
| C12:0 | 2.94 ± 0.01 | 2.88 ± 0.01 | 2.97 ± 0.08 | 3.00 ± 0.05 | 3.00 ± 0.06 | 2.98 ± 0.03 | 2.93 ± 0.07 | 2.88 ± 0.07 |
| C14:0 | 10.90 b ± 0.16 | 10.94 a,b ± 0.01 | 11.23 a ± 0.06 | 10.77 b ± 0.04 | 11.04 a,b ± 0.08 | 11.01 a,b ± 0.09 | 10.97 a,b ± 0.02 | 10.75 b ± 0.04 |
| C14:1 | 0.95 ± 0.00 | 0.94 ± 0.00 | 0.96 ± 0.01 | 0.96 ± 0.01 | 0.97 ± 0.02 | 0.97 ± 0.00 | 0.96 ± 0.01 | 0.95 ± 0.01 |
| C15:0 | 1.07 b ± 0.02 | 1.10 a,b ± 0.01 | 1.08 a,b ± 0.00 | 1.09 a,b ± 0.01 | 1.12 a ± 0.00 | 1.12 a,b ± 0.00 | 1.11 a,b ± 0.01 | 1.11 a,b ± 0.02 |
| C16:0 | 29.52 ± 0.10 | 29.63 ± 0.03 | 29.28 ± 0.07 | 28.93 ± 0.05 | 29.34 ± 0.29 | 29.31 ± 0.05 | 29.31 ± 0.39 | 29.21 ± 0.36 |
| C16:1 | 1.49 b,c ± 0.02 | 1.48 c ± 0.03 | 1.48 c ± 0.01 | 1.53 a,b,c ± 0.01 | 1.55 a,b ± 0.02 | 1.51 a,b,c ± 0.00 | 1.52 a,b,c ± 0.01 | 1.56 a ± 0.00 |
| C17:0 | 0.89 b ± 0.01 | 0.94 a,b ± 0.01 | 0.92 a,b ± 0.02 | 0.95 a,b ± 0.04 | 0.97 a,b ± 0.04 | 0.98 a ± 0.00 | 0.99 a ± 0.03 | 1.01 a ± 0.02 |
| C17:1 | 0.22 d ± 0.00 | 0.23 c,d ± 0.00 | 0.23 b,c,d ± 0.01 | 0.25 a,b,c ± 0.01 | 0.25 a,b,c ± 0.01 | 0.25 a,b ± 0.00 | 0.25 a ± 0.00 | 0.25 a,b ± 0.00 |
| C18:0 | 15.26 ± 0.07 | 15.26 ± 0.04 | 14.96 ± 0.10 | 15.05 ± 0.10 | 15.07 ± 0.17 | 15.13 ± 0.01 | 15.08 ± 0.17 | 15.16 ± 0.22 |
| C18:1n9t | 0.45 ± 0.03 | 0.48 ± 0.04 | 0.42 ± 0.01 | 0.47 ± 0.04 | 0.45 ± 0.01 | 0.48 ± 0.01 | 0.45 ± 0.04 | 0.51 ± 0.02 |
| C18:1n9c | 21.24 a ± 0.11 | 21.18 a,b ± 0.01 | 20.96 b,c ± 0.02 | 21.03 a,b,c ± 0.09 | 20.99 b,c ± 0.05 | 20.82 c ± 0.01 | 20.96 b,c ± 0.08 | 20.86 c ± 0.01 |
| C18:2n6t | 0.52 b ± 0.01 | 0.51 b ± 0.00 | 0.51 b ± 0.01 | 0.52 b ± 0.00 | 0.57 a ± 0.02 | 0.54 a,b ± 0.00 | 0.54 a,b ± 0.00 | 0.53 b ± 0.01 |
| C18:2n6c | 3.62 a ± 0.03 | 3.55 a,b ± 0.01 | 3.52 b ± 0.04 | 3.59 a,b ± 0.02 | 3.60 a ± 0.02 | 3.61 a ± 0.00 | 3.61 a ± 0.00 | 3.58 a,b ± 0.01 |
| C20:0 | 0.21 ± 0.01 | 0.21 ± 0.01 | 0.20 ± 0.01 | 0.22 ± 0.01 | 0.22 ± 0.00 | 0.13 ± 0.00 | 0.23 ± 0.00 | 0.23 ± 0.00 |
| C18:3n3 | 0.63 ± 0.02 | 0.62 ± 0.00 | 0.63 ± 0.02 | 0.65 ± 0.02 | 0.66 ± 0.01 | 0.67 ± 0.00 | 0.67 ± 0.01 | 0.66 ± 0.00 |
| C9t11 | 0.64 a,b ± 0.00 | 0.63 a,b ± 0.00 | 0.61 b ± 0.01 | 0.65 a ± 0.02 | 0.65 a ± 0.00 | 0.65 a ± 0.00 | 0.65 a ± 0.00 | 0.66 a ± 0.01 |
| C20:3n6 | 0.17 c ± 0.00 | 0.17 c ± 0.00 | 0.17 b,c ± 0.00 | 0.17 b,c ± 0.00 | 0.19 a,b ± 0.01 | 0.19 a ± 0.00 | 0.19 a ± 0.00 | 0.18 a,b,c ± 0.00 |
| C20:3n3 | 0.10 c ± 0.00 | 0.10 b,c ± 0.00 | 0.10 b,c ± 0.01 | 0.11 a,b,c ± 0.00 | 0.11 a,b,c ± 0.00 | 0.11 a,b,c ± 0.00 | 0.11 a,b ± 0.00 | 0.12 a ± 0.00 |
| SFAs | 69.76 ± 0.14 | 69.94 ± 0.07 | 70.21 ± 0.14 | 69.87 ± 0.21 | 69.77 ± 0.06 | 69.94 ± 0.03 | 69.84 ± 0.10 | 69.91 ± 0.02 |
| MUFAs | 24.45 ± 0.10 | 24.38 ± 0.05 | 24.14 ± 0.06 | 24.33 ± 0.15 | 24.32 ± 0.06 | 24.15 ± 0.01 | 24.25 ± 0.11 | 24.23 ± 0.01 |
| PUFAs | 5.77 a,b,c ± 0.04 | 5.66 b,c ± 0.02 | 5.63 c ± 0.09 | 5.79 a,b,c ± 0.06 | 5.88 a ± 0.01 | 5.87 a ± 0.01 | 5.86 a ± 0.01 | 5.82 a,b ± 0.01 |
| UFAs | 30.21 ± 0.15 | 30.04 ± 0.07 | 29.77 ± 0.14 | 30.12 ± 0.21 | 30.20 ± 0.05 | 30.02 ± 0.02 | 30.12 ± 0.10 | 30.05 ± 0.02 |
| n3 | 0.75 ± 0.02 | 0.74 ± 0.01 | 0.74 ± 0.03 | 0.78 ± 0.03 | 0.78 ± 0.01 | 0.80 ± 0.01 | 0.80 ± 0.00 | 0.79 ± 0.00 |
| n6 | 4.38 a,b,c ± 0.02 | 4.30 c,d ± 0.01 | 4.27 d ± 0.05 | 4.35 b,c,d ± 0.02 | 4.45 a ± 0.00 | 4.42 a,b ± 0.00 | 4.42 a,b ± 0.01 | 4.37 a,b,c ± 0.00 |
| n3/n6 | 0.17 ± 0.00 | 0.17 ± 0.00 | 0.17 ± 0.01 | 0.18 ± 0.01 | 0.18 ± 0.00 | 0.18 ± 0.00 | 0.18 ± 0.00 | 0.18 ± 0.00 |
| SCFAs (4–8) | 6.15 ± 0.20 | 6.06 ± 0.04 | 6.39 ± 0.01 | 6.65 ± 0.35 | 5.92 ± 0.32 | 6.24 ± 0.35 | 6.25 ± 0.51 | 6.67 ± 0.49 |
| MCFs (10–16) | 49.49 a,b ± 0.24 | 49.58 a,b ± 0.05 | 49.90 a ± 0.26 | 49.23 a,b ± 0.02 | 49.81 a,b ± 0.06 | 49.66 a,b ± 0.18 | 49.48 a,b ± 0.19 | 49.11 b ± 0.22 |
| LCFAs (17–24) | 44.36 ± 0.03 | 44.36 ± 0.00 | 43.71 ± 0.25 | 44.12 ± 0.37 | 44.27 ± 0.26 | 44.10 ± 0.16 | 44.27 ± 0.32 | 44.23 ± 0.26 |
| (b) | ||||||||
| C4:0 | 2.81 a,b ± 0.08 | 2.72 a,b ± 0.24 | 2.75 a,b ± 0.25 | 2.77 a,b ± 0.08 | 2.84 a,b ± 0.12 | 2.62 b ± 0.13 | 2.99 a,b ± 0.21 | 3.32 a ± 0.19 |
| C6:0 | 1.84 b ± 0.04 | 1.90 a,b ± 0.05 | 1.87 b ± 0.07 | 1.85 b ± 0.06 | 1.90 b ± 0.04 | 1.76 b ± 0.06 | 2.00 a,b ± 0.16 | 2.21 a ± 0.05 |
| C8:0 | 1.13 ± 0.02 | 1.15 ± 0.09 | 1.18 ± 0.04 | 1.15 ± 0.02 | 1.16 ± 0.00 | 1.13 ± 0.02 | 1.16 ± 0.04 | 1.30 ± 0.08 |
| C10:0 | 2.52 ± 0.03 | 2.46 ± 0.07 | 2.58 ± 0.01 | 2.51 ± 0.01 | 2.49 ± 0.00 | 2.50 ± 0.01 | 2.54 ± 0.10 | 2.52 ± 0.02 |
| C12:0 | 2.89 ± 0.04 | 2.85 ± 0.02 | 2.91 ± 0.03 | 2.89 ± 0.02 | 2.86 ± 0.00 | 2.89 ± 0.01 | 2.94 ± 0.12 | 3.08 ± 0.13 |
| C14:0 | 10.93 ± 0.01 | 10.86 ± 0.00 | 10.93 ± 0.08 | 10.92 ± 0.06 | 10.89 ± 0.02 | 10.96 ± 0.04 | 10.99 ± 0.15 | 10.94 ± 0.04 |
| C14:1 | 0.95 ± 0.00 | 0.95 ± 0.01 | 0.97 ± 0.00 | 0.97 ± 0.00 | 0.95 ± 0.00 | 0.97 ± 0.00 | 0.97 ± 0.02 | 0.98 ± 0.02 |
| C15:0 | 1.10 ± 0.01 | 1.12 ± 0.00 | 1.11 ± 0.00 | 1.12 ± 0.00 | 1.11 ± 0.00 | 1.13 ± 0.01 | 1.12 ± 0.01 | 1.10 ± 0.03 |
| C16:0 | 29.40 ± 0.13 | 29.33 ± 0.23 | 29.29 ± 0.09 | 29.34 ± 0.14 | 29.37 ± 0.16 | 29.44 ± 0.05 | 29.19 ± 0.22 | 29.11 ± 0.07 |
| C16:1 | 1.53 a,b ± 0.01 | 1.58 a ± 0.02 | 1.54 a,b ± 0.01 | 1.55 a,b ± 0.00 | 1.53 a,b ± 0.03 | 1.59 a ± 0.01 | 1.54 a,b ± 0.01 | 1.52 b ± 0.01 |
| C17:0 | 0.97 ± 0.01 | 0.99 ± 0.01 | 0.98 ± 0.00 | 0.99 ± 0.00 | 1.00 ± 0.01 | 1.01 ± 0.02 | 0.99 ± 0.04 | 0.94 ± 0.05 |
| C17:1 | 0.25 ± 0.00 | 0.26 ± 0.00 | 0.25 ± 0.00 | 0.25 ± 0.00 | 0.27 ± 0.03 | 0.26 ± 0.00 | 0.25 ± 0.00 | 0.25 ± 0.01 |
| C18:0 | 15.31 a,b ± 0.09 | 15.32 a,b ± 0.01 | 15.16 a,b ± 0.02 | 15.28 a,b ± 0.06 | 15.32 a,b ± 0.08 | 15.46 a ± 0.03 | 15.16 a,b ± 0.25 | 14.96 b ± 0.15 |
| C18:1n9t | 0.49 ± 0.00 | 0.52 ± 0.00 | 0.46 ± 0.00 | 0.51 ± 0.02 | 0.51 ± 0.00 | 0.50 ± 0.00 | 0.49 ± 0.05 | 0.45 ± 0.05 |
| C18:1n9c | 21.30 a,b ± 0.12 | 21.29 a,b ± 0.13 | 21.39 a ± 0.13 | 21.19 a,b ± 0.09 | 21.19 a,b ± 0.01 | 21.00 a,b,c ± 0.03 | 20.96 b,c ± 0.16 | 20.77 c ± 0.00 |
| C18:2n6t | 0.52 ± 0.00 | 0.55 ± 0.01 | 0.52 ± 0.01 | 0.53 ± 0.01 | 0.54 ± 0.01 | 0.54 ± 0.00 | 0.55 ± 0.03 | 0.52 ± 0.02 |
| C18:2n6c | 3.63 a,b ± 0.02 | 3.67 a ± 0.05 | 3.64 a,b ± 0.01 | 3.69 a ± 0.02 | 3.61 a,b ± 0.00 | 3.65 a,b ± 0.01 | 3.58 b ± 0.02 | 3.61 a,b ± 0.01 |
| C20:0 | 0.22 ± 0.00 | 0.24 ± 0.00 | 0.22 ± 0.00 | 0.23 ± 0.00 | 0.23 ± 0.00 | 0.24 ± 0.01 | 0.23 ± 0.01 | 0.22 ± 0.02 |
| C18:3n3 | 0.66 ± 0.00 | 0.68 ± 0.01 | 0.66 ± 0.00 | 0.68 ± 0.01 | 0.66 ± 0.00 | 0.68 ± 0.01 | 0.66 ± 0.01 | 0.65 ± 0.02 |
| C9t11 | 0.66 ± 0.00 | 0.67 ± 0.01 | 0.66 ± 0.01 | 0.66 ± 0.00 | 0.66 ± 0.01 | 0.67 ± 0.00 | 0.66 ± 0.01 | 0.64 ± 0.01 |
| C20:3n6 | 0.18 b ± 0.00 | 0.19 b ± 0.00 | 0.18 b ± 0.00 | 0.19 b ± 0.00 | 0.18 b ± 0.00 | 0.27 a ± 0.00 | 0.18 b ± 0.00 | 0.18 b ± 0.00 |
| C20:3n3 | 0.11 ± 0.00 | 0.12 ± 0.00 | 0.11 ± 0.00 | 0.12 ± 0.00 | 0.11 ± 0.00 | 0.12 ± 0.00 | 0.11 ± 0.00 | 0.11 ± 0.00 |
| SFAs | 69.48 a,b ± 0.17 | 69.29 b ± 0.22 | 69.39 b ± 0.12 | 69.45 a,b ± 0.13 | 69.56 a,b ± 0.08 | 69.52 a,b ± 0.05 | 69.77 a,b ± 0.27 | 70.08 a ± 0.12 |
| MUFAs | 24.62 a ± 0.13 | 24.70 a ± 0.14 | 24.70 a ± 0.11 | 24.56 a ± 0.09 | 24.55 a ± 0.06 | 24.41 a,b ± 0.04 | 24.35 a,b ± 0.18 | 24.07 b ± 0.05 |
| PUFAs | 5.86 a,b ± 0.03 | 5.97 a,b ± 0.08 | 5.87 a,b ± 0.01 | 5.95 a,b ± 0.03 | 5.85 a,b ± 0.01 | 6.03 a ± 0.01 | 5.84 a,b ± 0.09 | 5.81 b ± 0.07 |
| UFAs | 30.48 a,b ± 0.17 | 30.67 a ± 0.22 | 30.57 a ± 0.12 | 30.51 a ± 0.13 | 30.40 a,b ± 0.07 | 30.44 a,b ± 0.05 | 30.19 a,b ± 0.27 | 29.88 b ± 0.12 |
| n3 | 0.79 ± 0.00 | 0.82 ± 0.01 | 0.79 ± 0.00 | 0.81 ± 0.01 | 0.79 ± 0.01 | 0.82 ± 0.01 | 0.79 ± 0.02 | 0.78 ± 0.02 |
| n6 | 4.41 a,b ± 0.03 | 4.49 a,b ± 0.06 | 4.42 a,b ± 0.01 | 4.48 a,b ± 0.03 | 4.40 b ± 0.00 | 4.54 a ± 0.02 | 4.39 b ± 0.06 | 4.39 b ± 0.03 |
| n3/n6 | 0.18 ± 0.00 | 0.18 ± 0.00 | 0.18 ± 0.00 | 0.18 ± 0.00 | 0.18 ± 0.00 | 0.18 ± 0.00 | 0.18 ± 0.00 | 0.18 ± 0.00 |
| SCFAs(4–8) | 5.78 a,b ± 0.01 | 5.78 a,b ± 0.38 | 5.80 a,b ± 0.35 | 5.77 a,b ± 0.15 | 5.89 a,b ± 0.16 | 5.50 b ± 0.21 | 6.16 a,b ± 0.41 | 6.83 a ± 0.32 |
| MCFAs (10–16) | 49.43 ± 0.04 | 49.28 ± 0.16 | 49.45 ± 0.18 | 49.42 ± 0.22 | 49.33 ± 0.16 | 49.60 ± 0.11 | 49.42 ± 0.17 | 49.39 ± 0.08 |
| LCFAs (17–24) | 44.78 a,b ± 0.03 | 44.94 a ± 0.23 | 44.75 a,b ± 0.17 | 44.81 a,b ± 0.07 | 44.78 a,b ± 0.00 | 44.89 a ± 0.10 | 44.42 a,b ± 0.58 | 43.78 b ± 0.40 |
| Samples | Ripening Days | Acids | Alcohols | Ketones | Aldehydes | Esters | Others |
|---|---|---|---|---|---|---|---|
| C3 | 0 | 642.86 c ± 163.29 | 28.37 c ± 11.47 | 21.28 a ± 0.19 | 34.08 a ± 7.34 | n.d. | 12.88 b,c ± 0.97 |
| 90 | 2658.35 b,c ± 502.10 | 49.53 c ± 3.88 | 3.77 c ± 0.06 | n.d. | n.d. | 7.44 c ± 1.29 | |
| 180 | 7105.93 a ± 2043.31 | 195.40 a ± 0.60 | 14.25 a,b ± 3.54 | n.d. | n.d. | 25.77 a,b ± 5.50 | |
| 270 | 5029.01 a,b ± 256.45 | 110.03 b ± 18.95 | 6.50 b,c ± 2.55 | 21.26 a ± 0.88 | 68.87 a ± 4.01 | 35.99 a ± 2.99 | |
| C5 | 0 | 529.72 c ± 54.28 | 42.47 b ± 5.77 | 26.00 a ± 1.17 | 39.84 a ± 3.17 | n.d. | 9.38 b ± 1.79 |
| 90 | 9822.30 a ± 327.61 | 384.37 a ± 56.89 | 6.81 b ± 0.52 | n.d. | 46.78 a,b ± 12.61 | 26.48 a ± 7.10 | |
| 180 | 6459.75 b ± 290.65 | 279.10 a ± 43.78 | 10.03 b ± 1.58 | n.d. | 47.24 a,b ± 7.57 | 14.41 a,b ± 0.34 | |
| 270 | 5172.55 b ± 915.88 | 343.78 a ± 54.10 | 9.41 b ± 4.08 | 12.64 b ± 2.97 | 69.17 a ± 22.67 | 27.94 a ± 2.83 | |
| S3 | 0 | 1659.50 b ± 34.48 | 22.72 c ± 2.33 | 46.96 b ± 6.17 | 74.28 a ± 4.15 | n.d. | 22.30 ± 7.87 |
| 90 | 9171.68 a,b ± 41.58 | 287.42 a,b ± 18.37 | 251.86 b ± 5.08 | n.d. | n.d. | 39.45 ± 15.89 | |
| 180 | 4147.72 a,b ± 526.24 | 154.72 b,c ± 10.36 | 76.98 b ± 12.68 | n.d. | n.d. | 14.45 ± 1.95 | |
| 270 | 16,051.11 a ± 6335.85 | 360.26 a ± 74.44 | 604.81 a ± 110.20 | 50.30 b ± 0.26 | 123.33 a ± 0.41 | 68.57 ± 20.44 | |
| S5 | 0 | 441.49 b ± 141.81 | 21.38 b ± 4.69 | 26.47 ± 1.97 | 34.74 a ± 2.80 | n.d. | 12.67 b ± 0.08 |
| 90 | 17,539.56 a ± 2130.94 | 360.20 a ± 88.95 | 511.24 ± 253.21 | n.d. | n.d. | 49.49 a ± 6.08 | |
| 180 | 15,607.76 a ± 2303.45 | 271.80 a ± 28.88 | 219.30 ± 13.33 | 17.50 a,b ± 2.15 | n.d. | 44.82 a,b ± 6.04 | |
| 270 | 7128.88 b ± 2002.45 | 209.47 a,b ± 45.13 | 255.68 ± 67.22 | 22.09 a ± 9.31 | 62.71 a ± 5.76 | 26.57 a,b ± 13.52 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzo, N.; Santini, A.; Pizzolongo, F.; Aiello, A.; Marrazzo, A.; Meca, G.; Durazzo, A.; Lucarini, M.; Romano, R. Influence of Ripening on Chemical Characteristics of a Traditional Italian Cheese: Provolone del Monaco. Sustainability 2019, 11, 2520. https://doi.org/10.3390/su11092520
Manzo N, Santini A, Pizzolongo F, Aiello A, Marrazzo A, Meca G, Durazzo A, Lucarini M, Romano R. Influence of Ripening on Chemical Characteristics of a Traditional Italian Cheese: Provolone del Monaco. Sustainability. 2019; 11(9):2520. https://doi.org/10.3390/su11092520
Chicago/Turabian StyleManzo, Nadia, Antonello Santini, Fabiana Pizzolongo, Alessandra Aiello, Andrea Marrazzo, Giuseppe Meca, Alessandra Durazzo, Massimo Lucarini, and Raffaele Romano. 2019. "Influence of Ripening on Chemical Characteristics of a Traditional Italian Cheese: Provolone del Monaco" Sustainability 11, no. 9: 2520. https://doi.org/10.3390/su11092520
APA StyleManzo, N., Santini, A., Pizzolongo, F., Aiello, A., Marrazzo, A., Meca, G., Durazzo, A., Lucarini, M., & Romano, R. (2019). Influence of Ripening on Chemical Characteristics of a Traditional Italian Cheese: Provolone del Monaco. Sustainability, 11(9), 2520. https://doi.org/10.3390/su11092520










