Germplasm Development of Underutilized Temperate U.S. Tree Crops
Abstract
1. Introduction
2. Finding a Place for Tree Crops in the Midwest U.S.
3. Considerations for Tree Crop Development
4. Expanding the Cultivated Range of Tree Crops Using Wild Relatives

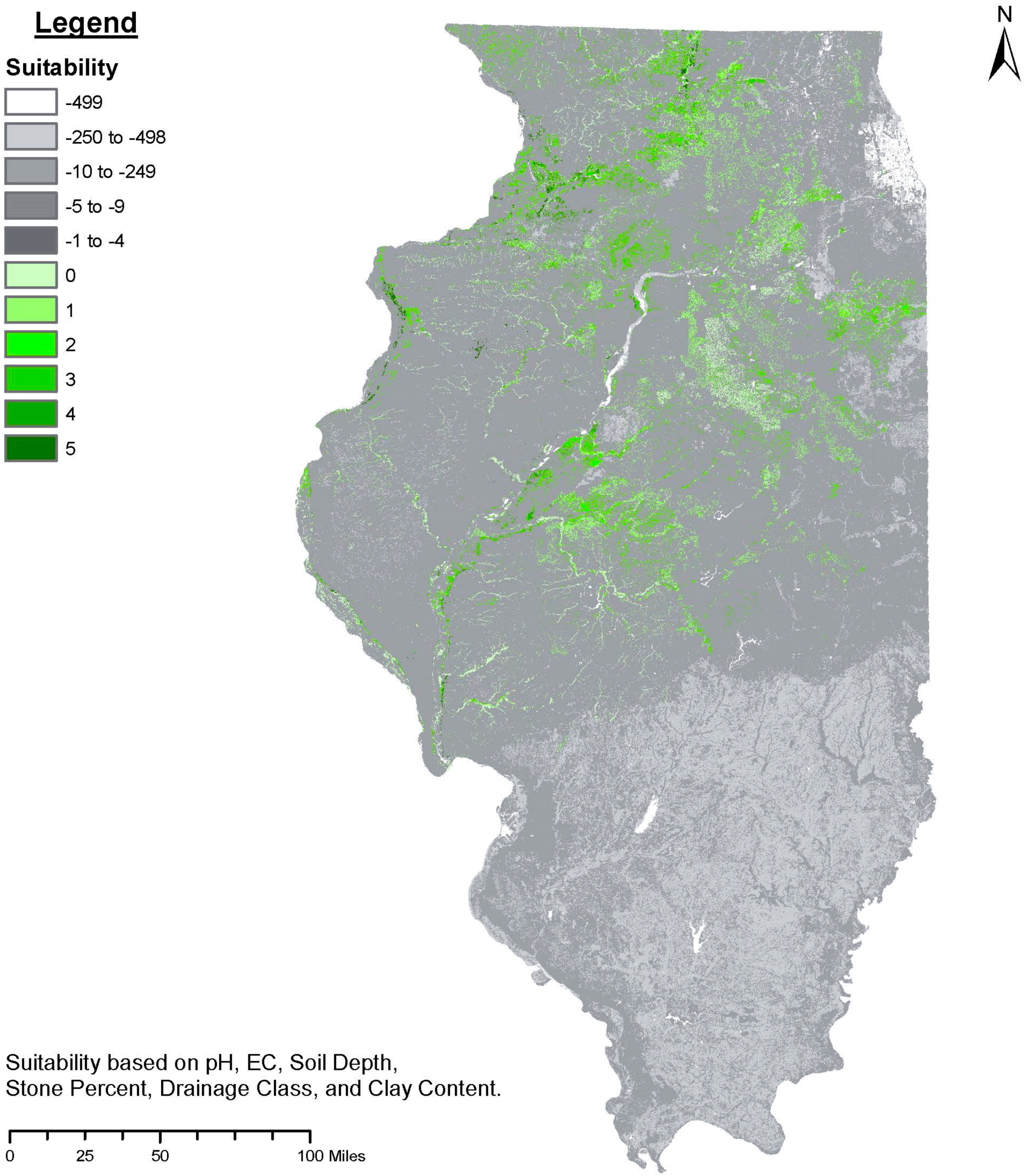
5. Defining Target Environments for Decentralized Selection
- (1)
- Spatial extent: As clusters of prospective target environments emerge, their respective sizes and the amenability of their required adaptive traits to systematic improvement can guide the priority in which they are targeted. The target environments should be focused on specific land types and regions that are abundant.
- (2)
- Productivity/profitability of row-crops: A high-resolution index (30 m × 30 m) of row-crop productivity in the Midwest U.S. is available via the National Commodity Crop Productivity Index (NCCPI) [138]. This index can be used in a comparative profitability analysis to ascertain if any of the target environments overlap with low production row-crop environments. Productivity alone, however, is insufficient in determining whether an alternative crop can outcompete row-crops under specific conditions. Instead, high-resolution profitability surfaces are now used to evaluate row-crop suitability [4,5]. In this light, it is important to note that the soil and climate suitability of tree crops and row-crops are not necessarily correlated [6].
- (3)
- Provision of regulatory ecosystem services: The prioritization of target environments could proceed based upon specific land types and regions that provide disproportionally large regulatory ecosystem services. Subsequent analysis of suitable map units could prioritize locations based upon slope, erosion, water quality, and more [5,9].
6. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Suitability Class | Soil Depth (cm) | pH (0–15cm) | EC (ds/m) (0–15cm) | Clay % (0–15 cm) | Soil Drainage Class | Stone % (>20 cm) | Rainfall, Mean August (mm) |
|---|---|---|---|---|---|---|---|
| Well suited | >50 | 6.5–6.599 | <0.15 | 30–50 | Well to moderate | <10 | <80 |
| Suited | 40–50 | 5.6–6.499 | <0.15 | 30–50 | Imperfect | 10–20 | <50 |
| Marginally suited | 30–40 | 6.6–7.199 | <0.15 | 30–50 | Imperfect | 10–20 | <50 |
| Unsuited | <30 | <5.599 or >7.2 | <0.15 | >50 or <10 | Poor to very poor | >20 | >50 |
References
- Jose, S. Agroforestry for ecosystem services and environmental benefits: An overview. Agrofor. Syst. 2009, 76, 1–10. [Google Scholar] [CrossRef]
- Torralba, M.; Fagerholm, N.; Burgess, P.J.; Moreno, G.; Plieninger, T. Do European agroforestry systems enhance biodiversity and ecosystem services? A meta-analysis. Agric. Ecosyst. Environ. 2016, 230, 150–161. [Google Scholar] [CrossRef]
- Tsonkova, P.; Böhm, C.; Quinkenstein, A.; Freese, D. Ecological benefits provided by alley cropping systems for production of woody biomass in the temperate region: A review. Agrofor. Syst. 2012, 85, 133–152. [Google Scholar] [CrossRef]
- Brandes, E.; McNunn, G.S.; Schulte, L.A.; Bonner, I.J.; Muth, D.; Babcock, B.A.; Sharma, B.; Heaton, E.A. Subfield profitability analysis reveals an economic case for cropland diversification. Environ. Res. Lett. 2016, 11, 014009. [Google Scholar] [CrossRef]
- Brandes, E.; McNunn, G.S.; Schulte, L.A.; Muth, D.J.; VanLoocke, A.; Heaton, E.A. Targeted subfield switchgrass integration could improve the farm economy, water quality, and bioenergy feedstock production. GCB Bioenergy 2018, 10, 199–212. [Google Scholar] [CrossRef]
- Wolz, K.J.; DeLucia, E.H. Black walnut alley cropping is economically competitive with row crops in the Midwest USA. Ecol. Appl. 2018. [Google Scholar] [CrossRef]
- Lovell, S.T.; Dupraz, C.; Gold, M.; Jose, S.; Revord, R.; Stanek, E.; Wolz, K.J. Temperate agroforestry research: Considering multifunctional woody polycultures and the design of long-term field trials. Agrofor. Syst. 2018, 92, 1397–1415. [Google Scholar] [CrossRef]
- Mattia, C.M.; Lovell, S.T.; Davis, A. Identifying barriers and motivators for adoption of multifunctional perennial cropping systems by landowners in the Upper Sangamon River Watershed, Illinois. Agrofor. Syst. 2018, 92, 1155–1169. [Google Scholar] [CrossRef]
- Mattia, C.M. Identifying Targets for Multifunctional Perennial Cropping Systems Implementation through a Landowner Adoption Preferences Survey and a Spatial Suitability Analysis. Master’s Thesis, University of Illinois at Urbana, Champaign, IL, USA, 2017. [Google Scholar]
- Molnar, T.J.; Kahn, P.C.; Ford, T.M.; Funk, C.J.; Funk, C.R. Tree crops, a permanent agriculture: Concepts from the past for a sustainable future. Resources 2013, 2, 457–488. [Google Scholar] [CrossRef]
- Mori, G.O.; Gold, M.; Jose, S. Specialty Crops in Temperate Agroforestry Systems: Sustainable Management, Marketing and Promotion for the Midwest Region of the USA. In Integrating Landscapes: Agroforestry for Biodiversity Conservation and Food Sovereignty; Springer: Berlin, Germany, 2017; pp. 331–366. [Google Scholar]
- Rhodes, T.K.; Aguilar, F.X.; Jose, S.; Gold, M. Factors influencing the adoption of riparian forest buffers in the Tuttle Creek Reservoir watershed of Kansas, USA. Agrofor. Syst. 2018, 92, 739–757. [Google Scholar] [CrossRef]
- Wolz, K.J.; DeLucia, E.H. Alley cropping: Global patterns of species composition and function. Agric. Ecosyst. Environ. 2018, 252, 61–68. [Google Scholar] [CrossRef]
- Wolz, K.J.; Lovell, S.T.; Branham, B.E.; Eddy, W.C.; Keeley, K.; Revord, R.S.; Wander, M.M.; Yang, W.H.; DeLucia, E.H. Frontiers in alley cropping: Transformative solutions for temperate agriculture. Glob. Chang. Biol. 2018, 24, 883–894. [Google Scholar] [CrossRef]
- Finn, C.E.; Thomas, A.L.; Byers, P.L.; Serçe, S. Evaluation of American (Sambucus canadensis) and European (S. nigra) elderberry genotypes grown in diverse environments and implications for cultivar development. Hortscience 2008, 43, 1385–1391. [Google Scholar] [CrossRef]
- Thomas, A.; Byers, P.; Avery, J.D., Jr.; Kaps, M.; Gu, S. Horticultural performance of eight American elderberry genotypes at three Missouri locations. Acta Hortic. 2015, 1061, 237–244. [Google Scholar] [CrossRef][Green Version]
- Brand, M. Aronia: Native shrubs with untapped potential. Arnoldia 2010, 67, 14–25. [Google Scholar]
- Brand, M.H. Breeding Better Aronia Plants©. Acta Hortic. 2013, 1055, 269–272. [Google Scholar] [CrossRef]
- Anagnostakis, S.L. Cultivars of chestnuts. Annu. Rep. North. Nut Grow. Assoc. 1999, 90, 16–31. [Google Scholar]
- Hunt, K.L.; Gold, M.A.; Warmund, M.R. Chinese chestnut cultivar performance in Missouri. Acta Hortic. 2004, 693, 145–148. [Google Scholar] [CrossRef]
- Coggeshall, M. Use of microsatellite markers to develop new eastern black walnut cultivars in Missouri, USA. Acta Hortic. 2010, 918, 221–226. [Google Scholar] [CrossRef]
- Reid, W.; Coggeshall, M.V.; Hunt, K.L. Cultivar evaluation and development for black walnut orchards. In Proceedings of the 6th Walnut Council Research Symposium; Gen. Tech. Rep. NC-243; U.S. Department of Agriculture, Forest Service, North Central Research Station: St. Paul, MN, USA, 2004. [Google Scholar]
- Warmund, M.; Coggeshall, M. Flowering and fruit characteristics of black walnut cultivars at the University of Missouri repository. Acta Hortic. 2009, 861, 177–182. [Google Scholar] [CrossRef]
- Molnar, T.J. Corylus. In Wild Crop Relatives: Genomic and Breeding Resources; Springer: Berlin/Heidelberg, Germany, 2011; pp. 15–48. [Google Scholar]
- Molnar, T.J.; Capik, J. Advances in hazelnut research in North America. Acta Hortic. 2012, 940, 57–65. [Google Scholar] [CrossRef]
- Molnar, T.; Lombardoni, J.; Muehlbauer, M.; Honig, J.; Mehlenbacher, S.; Capik, J. Progress breeding for resistance to eastern filbert blight in the eastern United States. Acta Hortic. 2017, 1226, 79–86. [Google Scholar] [CrossRef]
- Grauke, L.; Wood, B.W.; Harris, M.K. Crop vulnerability: Carya. Hortscience 2016, 51, 653–663. [Google Scholar] [CrossRef]
- Thompson, T.E.; Conner, P.J. Pecan. In Fruit Breeding; Springer: Berlin, Germany, 2012; pp. 771–801. [Google Scholar]
- Fischer, R.; Byerlee, D.; Edmeades, G. Crop Yields and Global Food Security; ACIAR: Canberra, Australia, 2014. [Google Scholar]
- Lobell, D.B.; Azzari, G. Satellite detection of rising maize yield heterogeneity in the US Midwest. Environ. Res. Lett. 2017, 12, 014014. [Google Scholar] [CrossRef]
- Lobell, D.B.; Roberts, M.J.; Schlenker, W.; Braun, N.; Little, B.B.; Rejesus, R.M.; Hammer, G.L. Greater sensitivity to drought accompanies maize yield increase in the U.S. Midwest. Science 2014, 344, 516–519. [Google Scholar] [CrossRef]
- Barbier, E.B. Sustaining agriculture on marginal land: A policy framework. Environ. Sci. Policy Sustain. Dev. 1989, 31, 12–40. [Google Scholar] [CrossRef]
- Kang, S.; Post, W.; Wang, D.; Nichols, J.; Bandaru, V.; West, T. Hierarchical marginal land assessment for land use planning. Land Use Policy 2013, 30, 106–113. [Google Scholar] [CrossRef]
- Wiegmann, K.; Hennenberg, K.J.; Fritsche, U.R. Degraded land and sustainable bioenergy feedstock production. In Proceedings of the Joint International Workshop on High Nature Value Criteria and Potential for Sustainable Use of Degraded Lands, Paris, France, 30 June–1 July 2008. [Google Scholar]
- Niu, X.; Duiker, S.W. Carbon sequestration potential by afforestation of marginal agricultural land in the Midwestern US. For. Ecol. Manag. 2006, 223, 415–427. [Google Scholar] [CrossRef]
- Olsen, J. Nutrient Management Guide—Hazelnut; Oregon State University Extension Service: Corvallis, OR, USA, 2001. [Google Scholar]
- Olsen, J. Orchard nutrition. In Growing Hazelnuts in the Pacific Northwest; Oregon State University Extension Service: Corvallis, OR, USA, 2013; pp. 1–5. [Google Scholar]
- Stanek, E.C.; Lovell, S.T.; Reisner, A. Designing multifunctional woody polycultures according to landowner preferences in Central Illinois. Agrofor. Syst. 2019. [Google Scholar] [CrossRef]
- Smith, J.R. Tree Crops: A Permanent Agriculture; Island Press: Washington, DC, USA, 2013. [Google Scholar]
- Technavio. Global Hazelnut Market 2017–2021; EMIS Intelligence Database; Technavio: London, UK, 2017. [Google Scholar]
- Leakey, R.R.B. The role of trees in agroecology and sustainable agriculture in the tropics. Annu. Rev. Phytopathol. 2014, 52, 113–133. [Google Scholar] [CrossRef]
- Mosquera-Losada, M.; Freese, D.; Rigueiro-Rodríguez, A. Carbon sequestration in European agroforestry systems. In Carbon Sequestration Potential of Agroforestry Systems; Springer: Berlin, Germany, 2011; pp. 43–59. [Google Scholar]
- Rigueiro-Rodríguez, A.; Fernández-Núñez, E.; González-Hernández, P.; McAdam, J.; Mosquera-Losada, M.R. Agroforestry systems in Europe: Productive, ecological and social perspectives. In Agroforestry in Europe; Springer: Berlin, Germany, 2009; pp. 43–65. [Google Scholar]
- Lovell, S.T.; Johnston, D.M. Creating multifunctional landscapes: How can the field of ecology inform the design of the landscape? Front. Ecol. Environ. 2009, 7, 212–220. [Google Scholar] [CrossRef]
- Jose, S. Agroforestry for conserving and enhancing biodiversity. Agrofor. Syst. 2012, 85, 1–8. [Google Scholar] [CrossRef]
- Schoeneberger, M.M. Agroforestry: Working trees for sequestering carbon on agricultural lands. Agrofor. Syst. 2009, 75, 27–37. [Google Scholar] [CrossRef]
- Schoeneberger, M.; Bentrup, G.; De Gooijer, H.; Soolanayakanahally, R.; Sauer, T.; Brandle, J.; Zhou, X.; Current, D. Branching out: Agroforestry as a climate change mitigation and adaptation tool for agriculture. J. Soil Water Conserv. 2012, 67, 128A–136A. [Google Scholar] [CrossRef]
- Schoeneberger, M.M.; Bentrup, G.; Patel-Weynand, T. Agroforestry: Enhancing Resiliency in US Agricultural Landscapes under Changing Conditions; Gen. Tech. Report WO-96; US Department of Agriculture, Forest Service: Washington, DC, USA, 2017.
- Udawatta, R.P.; Jose, S. Agroforestry strategies to sequester carbon in temperate North America. Agrofor. Syst. 2012, 86, 225–242. [Google Scholar] [CrossRef]
- Amadi, C.C.; Van Rees, K.C.; Farrell, R.E. Soil–atmosphere exchange of carbon dioxide, methane and nitrous oxide in shelterbelts compared with adjacent cropped fields. Agric. Ecosyst. Environ. 2016, 223, 123–134. [Google Scholar] [CrossRef]
- Kim, D.-G.; Kirschbaum, M.U.; Beedy, T.L. Carbon sequestration and net emissions of CH4 and N2O under agroforestry: Synthesizing available data and suggestions for future studies. Agric. Ecosyst. Environ. 2016, 226, 65–78. [Google Scholar] [CrossRef]
- Udawatta, R.P.; Krstansky, J.J.; Henderson, G.S.; Garrett, H.E. Agroforestry practices, runoff, and nutrient loss. J. Environ. Qual. 2002, 31, 1214–1225. [Google Scholar] [CrossRef]
- Garrett, H.; McGraw, R.; Walter, W. Alley cropping practices. In North American Agroforestry: An Integrated Science and Practice, 2nd ed.; American Society of Agronomy: Madison, WI, USA, 2009; pp. 133–162. [Google Scholar]
- Allen, S.C.; Jose, S.; Nair, P.K.R.; Brecke, B.J.; Nkedi-Kizza, P.; Ramsey, C.L. Safety-net role of tree roots: Evidence from a pecan (Carya illinoensis K. Koch)–cotton (Gossypium hirsutum L.) alley cropping system in the southern United States. For. Ecol. Manag. 2004, 192, 395–407. [Google Scholar] [CrossRef]
- Bambo, S.K.; Nowak, J.; Blount, A.R.; Long, A.J.; Osiecka, A. Soil nitrate leaching in silvopastures compared with open pasture and pine plantation. J. Environ. Qual. 2009, 38, 1870–1877. [Google Scholar] [CrossRef]
- Muth, D. Profitability versus environmental performance: Are they competing? J. Soil Water Conserv. 2014, 69, 203A–206A. [Google Scholar] [CrossRef]
- Lerch, R.; Kitchen, N.; Kremer, R.; Donald, W.; Alberts, E.; Sadler, E.; Sudduth, K.; Myers, D.; Ghidey, F. Development of a conservation-oriented precision agriculture system: Water and soil quality assessment. J. Soil Water Conserv. 2005, 60, 411–421. [Google Scholar]
- Ford-Lloyd, B.V.; Schmidt, M.; Armstrong, S.J.; Barazani, O.; Engels, J.; Hadas, R.; Hammer, K.; Kell, S.P.; Kang, D.; Khoshbakht, K. Crop wild relatives—Undervalued, underutilized and under threat? BioScience 2011, 61, 559–565. [Google Scholar] [CrossRef]
- Khoury, C.K.; Greene, S.; Wiersema, J.; Maxted, N.; Jarvis, A.; Struik, P.C. An inventory of crop wild relatives of the United States. Crop Sci. 2013, 53, 1496–1508. [Google Scholar] [CrossRef]
- Mayes, S.; Massawe, F.J.; Alderson, P.G.; Roberts, J.A.; Azam-Ali, S.N.; Hermann, M. The potential for underutilized crops to improve security of food production. J. Exp. Bot. 2011, 63, 1075–1079. [Google Scholar] [CrossRef]
- Castañeda-Álvarez, N.P.; Khoury, C.K.; Achicanoy, H.A.; Bernau, V.; Dempewolf, H.; Eastwood, R.J.; Guarino, L.; Harker, R.H.; Jarvis, A.; Maxted, N.; et al. Global conservation priorities for crop wild relatives. Nat. Plants 2016, 2, 16022. [Google Scholar] [CrossRef]
- Khoury, C.K.; Greene, S.L.; Williams, K.A.; Sosa, C.; Richards, C. Conservation Priorities for Tree Crop Wild Relatives in the United States. In Proceedings of the Workshop on Gene Conservation of Tree Species–Banking on the Future, Chicago, IL, USA, 16–19 May 2016. [Google Scholar]
- Dempewolf, H.; Baute, G.; Anderson, J.; Kilian, B.; Smith, C.; Guarino, L. Past and future use of wild relatives in crop breeding. Crop Sci. 2017, 57, 1070–1082. [Google Scholar] [CrossRef]
- Prohens, J.; Gramazio, P.; Plazas, M.; Dempewolf, H.; Kilian, B.; Díez, M.J.; Fita, A.; Herraiz, F.J.; Rodríguez-Burruezo, A.; Soler, S.; et al. Introgressiomics: A new approach for using crop wild relatives in breeding for adaptation to climate change. Euphytica 2017, 213, 158. [Google Scholar] [CrossRef]
- Cernusca, M.; Gold, M. Breaking down market barriers for elderberry growers and producers. Acta Hortic. 2013, 1061, 269–277. [Google Scholar] [CrossRef]
- Lee, J.; Finn, C.E. Anthocyanins and other polyphenolics in American elderberry (Sambucus canadensis) and European elderberry (S. nigra) cultivars. J. Sci. Food Agric. 2007, 87, 2665–2675. [Google Scholar] [CrossRef]
- Özgen, M.; Scheerens, J.C.; Reese, R.N.; Miller, R.A. Total phenolic, anthocyanin contents and antioxidant capacity of selected elderberry (Sambucus canadensis L.) accessions. Pharmacogn. Mag. 2010, 6, 198. [Google Scholar] [CrossRef]
- Charlebois, D.; Byers, P.L.; Finn, C.E.; Thomas, A.L. 4 Elderberry: Botany, Horticulture, Potential. Hortic. Rev. 2010, 37, 213. [Google Scholar]
- Chocolate Confectionery: Comparing Ferrero Group Top 20 Countries against the Market 2011–2016. Euromonitor International. 2016. Available online: Statista.com (accessed on 30 November 2017).
- Xie, M.; Zheng, J.; Wang, D. Achievements and perspective in hazelnut breeding in China. Acta Hortic. 2012, 1052, 41–43. [Google Scholar] [CrossRef]
- Grau, P.; Bastias, R. Productivity and yield efficiency of hazelnut (Corylus avellana L.) cultivars in Chile. Acta Hortic. 2004, 686, 57–64. [Google Scholar] [CrossRef]
- Ellena, M.; Sandoval, P.; Gonzalez, A.; Jequier, J.; Contreras, M.; Grau Beretta, P. Chilean hazelnut situation and perspectives. Acta Hortic. 2012, 1052, 329–342. [Google Scholar] [CrossRef]
- Melhenbacher, S. The hazelnut situation in Oregon. Acta Hortic. 2004, 686, 665–668. [Google Scholar] [CrossRef]
- Baldwin, B.J. The Growth and Productivity of Hazelnut Cultivars (Corylus avellana L.) in Australia. Ph.D. Thesis, University of Sydney, Faculty of Rural Management, Sydney, Australia, 2015. [Google Scholar]
- Gönenc, S.; Tanrıvermis, H.; Bülbül, M. Economic assessment of hazelnut production and the importance of supply management approaches in Turkey. J. Agric. Rural Dev. Trop. Subtrop. (JARTS) 2006, 107, 19–32. [Google Scholar]
- Mehlenbacher, S.A. Genetic resources for hazelnut: State of the art and future perspectives. Acta Hortic. 2008, 845, 33–38. [Google Scholar] [CrossRef]
- Ustaoğlu, B. The Effect of Climatic Conditions on Hazelnut (Corylus avellana) Yield in Giresun (Turkey). 2012. Available online: http://hdl.handle.net/11424/2685 (accessed on 20 March 2018).
- FAOSTAT, Food and Agriculture Organization of the United Nations Cropping Database; FAO: Rome, Italy, 2014.
- Dempewolf, H.; Eastwood, R.J.; Guarino, L.; Khoury, C.K.; Müller, J.V.; Toll, J. Adapting Agriculture to Climate Change: A Global Initiative to Collect, Conserve, and Use Crop Wild Relatives. Agroecol. Sustain. Food Syst. 2014, 38, 369–377. [Google Scholar] [CrossRef]
- Kole, C.; Muthamilarasan, M.; Henry, R.; Edwards, D.; Sharma, R.; Abberton, M.; Batley, J.; Bentley, A.; Blakeney, M.; Bryant, J.; et al. Application of genomics-assisted breeding for generation of climate resilient crops: Progress and prospects. Front. Plant Sci. 2015, 6, 563. [Google Scholar] [CrossRef]
- Warschefsky, E.; Varma Penmetsa, R.; Cook, D.R.; Von Wettberg, E.J.B. Back to the wilds: Tapping evolutionary adaptations for resilient crops through systematic hybridization with crop wild relatives. Am. J. Bot. 2014, 101, 1791–1800. [Google Scholar] [CrossRef]
- Miller, A.J.; Gross, B.L. From forest to field: Perennial fruit crop domestication. Am. J. Bot. 2011, 98, 1389–1414. [Google Scholar] [CrossRef]
- Molnar, T.J.; Honig, J.A.; Mayberry, A.; Revord, R.S.; Lovell, S.T.; Mehlenbacher, S.A.; Capik, J.M. Corylus americana: A valuable genetic resource for developing hazelnuts adapted to the eastern United States. In Proceedings of the IX International Congress on Hazelnut, Atakum, Samsun, Turkey, 15–19 August 2017; Tuncer, P.D.C., Ed.; IX International Congress on Hazelnut—Book of Abstracts. Atakum, Samsun, Turkey; pp. 115–122. [Google Scholar]
- Migicovsky, Z.; Myles, S. Exploiting wild relatives for genomics-assisted breeding of perennial crops. Front. Plant Sci. 2017, 8, 460. [Google Scholar] [CrossRef]
- McClure, K.A.; Sawler, J.; Gardner, K.M.; Money, D.; Myles, S. Genomics: A potential panacea for the perennial problem. Am. J. Bot. 2014, 101, 1780–1790. [Google Scholar] [CrossRef]
- Taheri, R.; Connolly, B.A.; Brand, M.H.; Bolling, B.W. Underutilized chokeberry (Aronia melanocarpa, Aronia arbutifolia, Aronia prunifolia) accessions are rich sources of anthocyanins, flavonoids, hydroxycinnamic acids, and proanthocyanidins. J. Agric. Food Chem. 2013, 61, 8581–8588. [Google Scholar] [CrossRef]
- Brand, M.H.; Connolly, B.A.; Levine, L.H.; Richards, J.T.; Shine, S.M.; Spencer, L.E. Anthocyanins, total phenolics, ORAC and moisture content of wild and cultivated dark-fruited Aronia species. Sci. Hortic. 2017, 224, 332–342. [Google Scholar] [CrossRef]
- Alexander, M.T. Conservation of Castanea Dentata Genetic Resources of the Southeastern United States; University of Tennessee at Chattanooga, Environmental Science: Chattanooga, TN, USA, 2005. [Google Scholar]
- Li, G.-T.; Ai, C.-X.; Zhang, L.-S.; Wei, H.-R.; Liu, Q.-Z. Chestnut genebank in China national clonal plant germplasm repository. Acta Hortic. 2009, 844, 199–206. [Google Scholar]
- Miller, A.C.; Woeste, K.E.; Anagnostakis, S.L.; Jacobs, D.F. Exploration of a rare population of Chinese chestnut in North America: Stand dynamics, health and genetic relationships. AoB Plants 2014, 6, plu065. [Google Scholar] [CrossRef]
- McGranahan, G.; Leslie, C. Breeding walnuts (Juglans regia). In Breeding Plantation Tree Crops: Temperate Species; Springer: Berlin, Germany, 2009; pp. 249–273. [Google Scholar]
- Npgs, U. Genetic Resources Information Service. In GRIN Taxonomy. Available online: https://npgsweb.ars-grin.gov/gringlobal/taxon/taxonomysearch.aspx (accessed on 30 May 2018).
- Thompson, M.M.; Lagerstedt, H.B.; Mehlenbacher, S.A. Hazelnuts. Fruit Breed. 1996, 3, 125–184. [Google Scholar]
- Sathuvalli, V.R.; Mehlenbacher, S.A. Characterization of American hazelnut (Corylus americana) accessions and Corylus americana × Corylus avellana hybrids using microsatellite markers. Genet. Resour. Crop Evol. 2012, 59, 1055–1075. [Google Scholar] [CrossRef]
- Demchik, M.; Kern, A.; Braun, L.; Fischbach, J.; Turnquist, K. Genetic diversity of American hazelnut in the Upper Midwest, USA. Agrofor. Syst. 2018, 92, 1507–1516. [Google Scholar] [CrossRef]
- Molnar, T.J.; Lombardoni, J.J.; Muehlbauer, M.F.; Honig, J.A.; Mehlenbacher, S.A.; Capik, J.M. Progress breeding for resistance to eastern filbert blight in the eastern United States. In Proceedings of the IX International Congress on Hazelnut, Atakum, Samsun, Turkey, 15–19 August 2017; Tuncer, P.D.C., Ed.; IX International Congress on Hazelnut—Book of Abstracts. Atakum, Samsun, Turkey, 2018; pp. 79–86. [Google Scholar]
- Drumke, J.S. A Systematic Survey of Corylus in North America; The University of Tennessee: Knoxville, TN, USA, 1964. [Google Scholar]
- Capik, J.M.; Molnar, T.J. Assessment of host (Corylus sp.) resistance to eastern filbert blight in New Jersey. J. Am. Soc. Hortic. Sci. 2012, 137, 157–172. [Google Scholar] [CrossRef]
- Fuller, A.S. The Nut Culturist; Orange Judd: New York, NY, USA, 1908. [Google Scholar]
- Weschcke, C. Growing Nuts in the North; Webb: St. Paul, MN, USA, 1953. [Google Scholar]
- Johnson, K.; Pinkerton, J. Eastern filbert blight. In Compendium of Nut Crop Diseases in Temperate Zones; APS Press: St. Paul, MN, USA, 2002; pp. 44–46. [Google Scholar]
- Muehlbauer, M.; Tobia, J.; Honig, J.; Hillman, B.; Zhang, N.; Gold, K.M.; Molnar, T. Population differentiation within Anisogramma anomala in North America. Phytopathology 2018. [Google Scholar] [CrossRef]
- Erdogan, V.; Mehlenbacher, S.A. Interspecific hybridization in hazelnut (Corylus). J. Am. Soc. Hortic. Sci. 2000, 125, 489–497. [Google Scholar] [CrossRef]
- Bhattarai, G.; Mehlenbacher, S.A.; Smith, D.C. Eastern filbert blight disease resistance from Corylus americana ‘Rush’ and selection ‘Yoder #5’ maps to linkage group 7. Tree Genet. Genomes 2017, 13, 45. [Google Scholar]
- Cornille, A.; Gladieux, P.; Smulders, M.J.; Roldan-Ruiz, I.; Laurens, F.; Le Cam, B.; Nersesyan, A.; Clavel, J.; Olonova, M.; Feugey, L. New insight into the history of domesticated apple: Secondary contribution of the European wild apple to the genome of cultivated varieties. PLoS Genet. 2012, 8, e1002703. [Google Scholar] [CrossRef]
- Myles, S.; Boyko, A.R.; Owens, C.L.; Brown, P.J.; Grassi, F.; Aradhya, M.K.; Prins, B.; Reynolds, A.; Chia, J.-M.; Ware, D. Genetic structure and domestication history of the grape. Proc. Natl. Acad. Sci. USA 2011, 108, 3530–3535. [Google Scholar] [CrossRef]
- Migicovsky, Z.; Sawler, J.; Money, D.; Eibach, R.; Miller, A.J.; Luby, J.J.; Jamieson, A.R.; Velasco, D.; von Kintzel, S.; Warner, J.; et al. Genomic ancestry estimation quantifies use of wild species in grape breeding. BMC Genom. 2016, 17, 478. [Google Scholar] [CrossRef]
- Öztürk, S.C.; Balık, H.İ.; Balık, S.K.; Kızılcı, G.; Duyar, Ö.; Doğanlar, S.; Frary, A. Molecular genetic diversity of the Turkish national hazelnut collection and selection of a core set. Tree Genet. Genomes 2017, 13, 113. [Google Scholar] [CrossRef]
- Ellstrand, N.C.; Prentice, H.C.; Hancock, J.F. Gene flow and introgression from domesticated plants into their wild relatives. Annu. Rev. Ecol. Syst. 1999, 30, 539–563. [Google Scholar] [CrossRef]
- Maxted, N.; Kell, S.; Ford-Lloyd, B.; Dulloo, E.; Toledo, Á. Toward the Systematic Conservation of Global Crop Wild Relative Diversity. Crop Sci. 2012, 52, 774–785. [Google Scholar] [CrossRef]
- Brumlop, S.; Reichenbecher, W.; Tappeser, B.; Finckh, M.R. What is the SMARTest way to breed plants and increase agrobiodiversity? Euphytica 2013, 194, 53–66. [Google Scholar] [CrossRef]
- Duminil, J.; Fineschi, S.; Hampe, A.; Jordano, P.; Salvini, D.; Vendramin, G.G.; Petit, R.J. Can population genetic structure be predicted from life-history traits? Am. Nat. 2007, 169, 662–672. [Google Scholar]
- Duminil, J.; Hardy, O.J.; Petit, R.J. Plant traits correlated with generation time directly affect inbreeding depression and mating system and indirectly genetic structure. BMC Evolut. Biol. 2009, 9, 177. [Google Scholar] [CrossRef]
- Hamrick, J.L.; Godt, M.J.W.; Sherman-Broyles, S.L. Factors influencing levels of genetic diversity in woody plant species. In Population Genetics of Forest Trees; Springer: Berlin, Germany, 1992; pp. 95–124. [Google Scholar]
- Loveless, M.D.; Hamrick, J.L. Ecological determinants of genetic structure in plant populations. Annu. Rev. Ecol. Syst. 1984, 15, 65–95. [Google Scholar] [CrossRef]
- Petit, R.J.; Hampe, A. Some Evolutionary Consequences of Being a Tree. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 187–214. [Google Scholar] [CrossRef]
- Hedrick, P.W. Genetic polymorphism in heterogeneous environments: The age of genomics. Annu. Rev. Ecol. Evol. Syst. 2006, 37, 67–93. [Google Scholar] [CrossRef]
- Kawecki, T.J.; Ebert, D. Conceptual issues in local adaptation. Ecol. Lett. 2004, 7, 1225–1241. [Google Scholar] [CrossRef]
- Linhart, Y.B.; Grant, M.C. Evolutionary significance of local genetic differentiation in plants. Annu. Rev. Ecol. Syst. 1996, 27, 237–277. [Google Scholar] [CrossRef]
- Pritchard, J.K.; Di Rienzo, A. Adaptation—Not by sweeps alone. Nat. Rev. Gen. 2010, 11, 665. [Google Scholar] [CrossRef]
- Le Corre, V.; Kremer, A. The genetic differentiation at quantitative trait loci under local adaptation. Mol. Ecol. 2012, 21, 1548–1566. [Google Scholar] [CrossRef]
- Aitken, S.N.; Yeaman, S.; Holliday, J.A.; Wang, T.; Curtis-McLane, S. Adaptation, migration or extirpation: Climate change outcomes for tree populations. Evol. Appl. 2008, 1, 95–111. [Google Scholar] [CrossRef]
- Savolainen, O.; Lascoux, M.; Merilä, J. Ecological genomics of local adaptation. Nat. Rev. Gen. 2013, 14, 807. [Google Scholar] [CrossRef]
- Neale, D.B.; Savolainen, O. Association genetics of complex traits in conifers. Trends Plant Sci. 2004, 9, 325–330. [Google Scholar] [CrossRef]
- Brown, G.R.; Gill, G.P.; Kuntz, R.J.; Langley, C.H.; Neale, D.B. Nucleotide diversity and linkage disequilibrium in loblolly pine. Proc. Natl. Acad. Sci. USA 2004, 101, 15255–15260. [Google Scholar] [CrossRef]
- Heuertz, M.; De Paoli, E.; Källman, T.; Larsson, H.; Jurman, I.; Morgante, M.; Lascoux, M.; Gyllenstrand, N. Multilocus patterns of nucleotide diversity, linkage disequilibrium and demographic history of Norway spruce (Picea abies (L.) Karst). Genetics 2006, 174, 2095–2105. [Google Scholar] [CrossRef]
- Varshney, R.K.; Terauchi, R.; McCouch, S.R. Harvesting the promising fruits of genomics: Applying genome sequencing technologies to crop breeding. PLoS Biol. 2014, 12, e1001883. [Google Scholar] [CrossRef]
- Lijavetzky, D.; Cabezas, J.A.; Ibáñez, A.; Rodríguez, V.; Martínez-Zapater, J.M. High throughput SNP discovery and genotyping in grapevine (Vitis vinifera L.) by combining a re-sequencing approach and SNPlex technology. BMC Genom. 2007, 8, 424. [Google Scholar] [CrossRef]
- Parchman, T.L.; Gompert, Z.; Mudge, J.; Schilkey, F.D.; Benkman, C.W.; Buerkle, C.A. Genome-wide association genetics of an adaptive trait in lodgepole pine. Mol. Ecol. 2012, 21, 2991–3005. [Google Scholar] [CrossRef]
- Sork, V.; Aitken, S.; Dyer, R.; Eckert, A.; Legendre, P.; Neale, D. Putting the landscape into the genomics of trees: Approaches for understanding local adaptation and population responses to changing climate. Tree Genet. Genomes 2013, 9, 901–911. [Google Scholar] [CrossRef]
- Simmonds, N. Selection for local adaptation in a plant breeding programme. Theor. Appl. Genet. 1991, 82, 363–367. [Google Scholar] [CrossRef]
- Ceccarelli, S. Efficiency of plant breeding. Crop Sci. 2015, 55, 87–97. [Google Scholar] [CrossRef]
- Atlin, G.N. Breeding for Suboptimal Environments. In Proceedings of the International Workshop on Increased Lowland Rice Production in the Mekong Region, Vientiane, Laos, 30 October–2 November 2000; Fukai, S., Basnayake, J., Eds.; ACIAR: Canberra, Australia; pp. 245–251. [Google Scholar]
- Annicchiarico, P.; Bellah, F.; Chiari, T. Defining subregions and estimating benefits for a specific-adaptation strategy by breeding programs. Crop Sci. 2005, 45, 1741–1749. [Google Scholar] [CrossRef]
- Kidd, D.; Webb, M.; Malone, B.; Minasny, B.; McBratney, A. Digital soil assessment of agricultural suitability, versatility and capital in Tasmania, Australia. Geoderma Reg. 2015, 6, 7–21. [Google Scholar] [CrossRef]
- Wallace, D.C.; Young, F.J. Black walnut suitability index: A natural resources conservation service national soil information system based interpretive model. In Proceedings of the 16th Central Hardwood Forest Conference, West Lafayette, IN, USA, 8–9 April 2008; Gen. Tech. Rep. NRS-P-24. US Department of Agriculture, Forest Service, Northern Research Station: Newtown Square, PA, USA, 2008; pp. 589–595. [Google Scholar]
- Staff, S.S. Soil Survey Geographic (SSURGO) Database for Illinois; Natural Resources Conservation Service, United States Department of Agriculture: Washington, DC, USA, 2017. [Google Scholar]
- Dobos, R.; Sinclair, H.; Hipple, K. National Commodity Crop Productivity Index (NCCPI) User Guide v2. 0; USDA NRCS National Soil Survey Center: Lincoln, NE, USA, 2012.
- Richards, B.K.; Stoof, C.R.; Cary, I.J.; Woodbury, P.B. Reporting on marginal lands for bioenergy feedstock production: A modest proposal. BioEnergy Res. 2014, 7, 1060–1062. [Google Scholar] [CrossRef]
- Bushakra, J.; Bassil, N.; Finn, C.; Hummer, K. Sambucus Genetic Resources at the US National Clonal Germplasm Repository. I Int. Symp. Elderberry 2013, 1061, 135–145. [Google Scholar]
- Byers, P.; Thomas, A. Elderberry research and production in Missouri. Proc. Mo. Small Fruit Veg. Conf. 2005, 25, 91–97. [Google Scholar]
- Wolz, K.J.; Branham, B.E.; DeLucia, E.H. Reduced nitrogen losses after conversion of row crop agriculture to alley cropping with mixed fruit and nut trees. Agric. Ecosyst. Environ. 2018, 258, 172–181. [Google Scholar] [CrossRef]
- Zomer, R.J.; Neufeldt, H.; Xu, J.; Ahrends, A.; Bossio, D.; Trabucco, A.; van Noordwijk, M.; Wang, M. Global Tree Cover and Biomass Carbon on Agricultural Land: The contribution of agroforestry to global and national carbon budgets. Sci. Rep. 2016, 6, 29987. [Google Scholar] [CrossRef]
- Udawatta, R.P.; Jose, S. Carbon sequestration potential of agroforestry practices in temperate North America. In Carbon Sequestration Potential of Agroforestry Systems; Springer: Berlin, Germany, 2011; pp. 17–42. [Google Scholar]
- Böhm, C.; Kanzler, M.; Freese, D. Wind speed reductions as influenced by woody hedgerows grown for biomass in short rotation alley cropping systems in Germany. Agrofor. Syst. 2014, 88, 579–591. [Google Scholar] [CrossRef]
- Lin, B.B. Agroforestry management as an adaptive strategy against potential microclimate extremes in coffee agriculture. Agric. For. Meteorol. 2007, 144, 85–94. [Google Scholar] [CrossRef]
- Siriri, D.; Wilson, J.; Coe, R.; Tenywa, M.; Bekunda, M.; Ong, C.; Black, C. Trees improve water storage and reduce soil evaporation in agroforestry systems on bench terraces in SW Uganda. Agrofor. Syst. 2013, 87, 45–58. [Google Scholar] [CrossRef]
- Lobell, D.B.; Hammer, G.L.; McLean, G.; Messina, C.; Roberts, M.J.; Schlenker, W. The critical role of extreme heat for maize production in the United States. Nat. Clim. Change 2013, 3, 497–501. [Google Scholar] [CrossRef]
- Anderson, S.H.; Udawatta, R.P.; Seobi, T.; Garrett, H.E. Soil water content and infiltration in agroforestry buffer strips. Agrofor. Syst. 2009, 75, 5–16. [Google Scholar] [CrossRef]
- Verchot, L.V.; Van Noordwijk, M.; Kandji, S.; Tomich, T.; Ong, C.; Albrecht, A.; Mackensen, J.; Bantilan, C.; Anupama, K.; Palm, C. Climate change: Linking adaptation and mitigation through agroforestry. Mitig. Adapt. Strateg. Glob. Change 2007, 12, 901–918. [Google Scholar] [CrossRef]
| Tree Crop | Cultivated Species | Breeding Objective | Breeding Stage | Adapted Regions | References |
|---|---|---|---|---|---|
| Elderberry | Sambucus nigra L. subspecies canadensis R. Bolli | Identify adapted varieties, selection for site specific conditions, fruit quality and yield, reduced inter-annual variability, and late bud break. | Multi-location trials of traditional varieties and new germplasm. | MO. | [15,16] |
| Aronia | Aronia melanocarpa (Michx.) Elliot | Identify adapted varieties, narrower and shorter growth habit, total phenolics and anthocyanins, total yield, and low chilling. | Trials of traditional varieties and new germplasm. | CT, the NE. | [17,18] |
| Chinese chestnut | Castanea mollissima Blume | Identify adapted varieties, kernel size and quality, yield quantity and consistency. | Variety trials; initiated pedigree breeding. | CT; MO. | [11,19,20] |
| Eastern black walnut | Juglans nigra L. | Identify adapted varieties, kernel quality and yield (e.g., nutmeat/shell ratio), alternate bearing, define host resistance to pests and disease, early flowering, and spur-type growth habit. | Multi-location variety trials; pedigree breeding. | MO; KS. | [21,22,23] |
| European hazelnut | Corylus avellana | Eastern filbert blight resistance, cold hardiness, commercial kernel quality and yield. | Screening wild germplasm; modified backcrossing. | NJ; NY. | [24,25,26] |
| Northern Pecan | Carya illinoinensis (Wangenh) K. Koch | High yield, perocity, kernel quality, kernel percentage, ease of shelling, disease resistance, and reduced masting. | Multi-location trials; controlled crossing. | GA; MO; TX. | [27,28] |
| Species | Known Traits of Value in Wild Relatives 1 |
|---|---|
| Sambucus nigra L. subspecies canadensis R. Bolli | Commercial yields, late flowering, short ripening period, [15], high phenolics and anthocyanins, acylated forms of cyanidin-glycosides, and regional and local adaptation [16]. |
| Aronia arbutifolia (L.) Pers. | Lower chill hours, fruit ripening date, fruit size, and ripe fruit color [18,86]. |
| Aronia melanocarpa (Michx.) Elliot | High anthocyanin and unique profiles [86,87], plant habit (e.g., prostrate), fruit ripening date, fruit size [18], and diverse microclimate adaptation [17]. |
| Aronia prunifolia (Marshall) Rehder | High phenolics [86,87]. |
| Castanea dentata (Marshall) Borkh. | Regional and local adaptation along with rare genetic diversity [88]. |
| Castanea mollissima Blume 2 | Resistance to chestnut blight, winter hardiness, kernel size and quality, yield quantity, late bud break, flowering date, early nut maturity [20,89], naturalize populations [90], and some regional and local adaptation [20]. |
| Juglans nigra L. | Local adaptation, late flowering, anthracnose resistance [91], and rootstock [92]. |
| Corylus americana Walter | Eastern filbert blight resistance [93], local adaptation [24], cold hardiness [94], heat and drought tolerance [24], and genetic diversity [94,95]. |
| Carya illinoinensis (Wangenh) K. Koch | Disease resistance, drought and heat tolerance, cold hardiness, and tree size reduction [27]. |
| Species | Accessions | States 1 Represented in Endemic Range | States Represented in Germplasm Collections | ||
|---|---|---|---|---|---|
| USDA-ARS 2 | University 3 | Populations Collected | |||
| >10 | 1 to 10 | ||||
| Sambucus nigra L. subspecies canadensis R. Bolli | 38 4 | 55 5 | 52 | 1 | 8 |
| Aronia arbutifolia (L.) Pers. | 20 | 19 6 | 29 | 0 | 13 |
| Aronia melanocarpa (Michx.) Elliot | 50 | 57 6 | 36 | 1 | 16 |
| Aronia prunifolia (Marshall) Rehder | 28 | 41 6 | 31 | 2 | 6 |
| Castanea dentata (Marshall) Borkh. | 1 | Not published 7 | 30 | - | - |
| Castanea mollissima Blume 8 | 239 9 | 65 10 | N/A | N/A | N/A |
| Juglans nigra L. | 27 | 64 | 44 | 0 | 8 |
| Corylus americana Walter | 43 | ~100 11 | 39 | 2 | 27 |
| Carya illinoinensis (Wangenh) K. Koch | 3615 | 21 | Data not available | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Revord, R.; Lovell, S.; Molnar, T.; Wolz, K.J.; Mattia, C. Germplasm Development of Underutilized Temperate U.S. Tree Crops. Sustainability 2019, 11, 1546. https://doi.org/10.3390/su11061546
Revord R, Lovell S, Molnar T, Wolz KJ, Mattia C. Germplasm Development of Underutilized Temperate U.S. Tree Crops. Sustainability. 2019; 11(6):1546. https://doi.org/10.3390/su11061546
Chicago/Turabian StyleRevord, Ronald, Sarah Lovell, Thomas Molnar, Kevin J. Wolz, and Chloé Mattia. 2019. "Germplasm Development of Underutilized Temperate U.S. Tree Crops" Sustainability 11, no. 6: 1546. https://doi.org/10.3390/su11061546
APA StyleRevord, R., Lovell, S., Molnar, T., Wolz, K. J., & Mattia, C. (2019). Germplasm Development of Underutilized Temperate U.S. Tree Crops. Sustainability, 11(6), 1546. https://doi.org/10.3390/su11061546





