Process Design Characteristics of Syngas (CO/H2) Separation Using Composite Membrane
Abstract
:1. Introduction
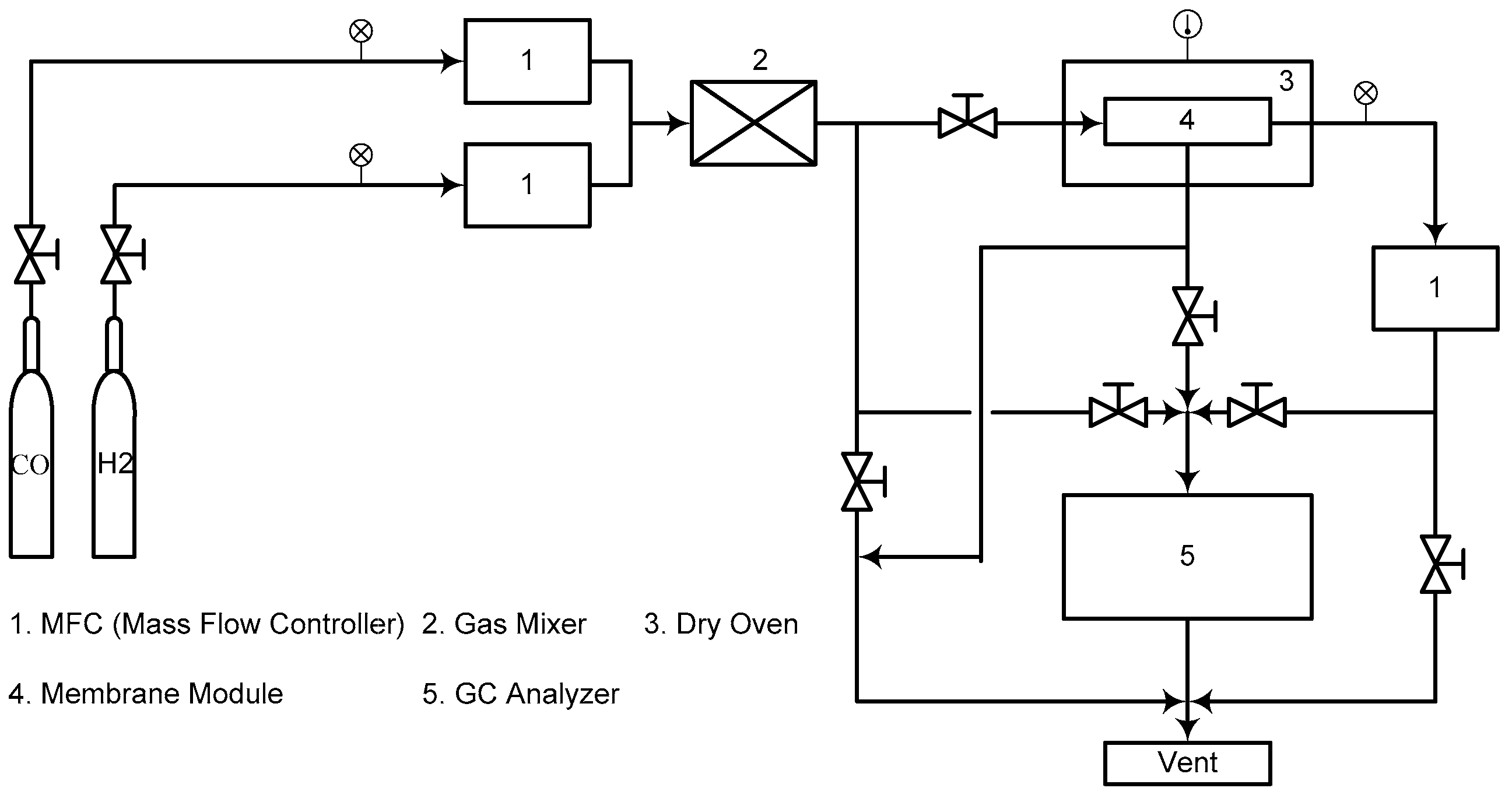
2. Experimental
3. Results and Discussions
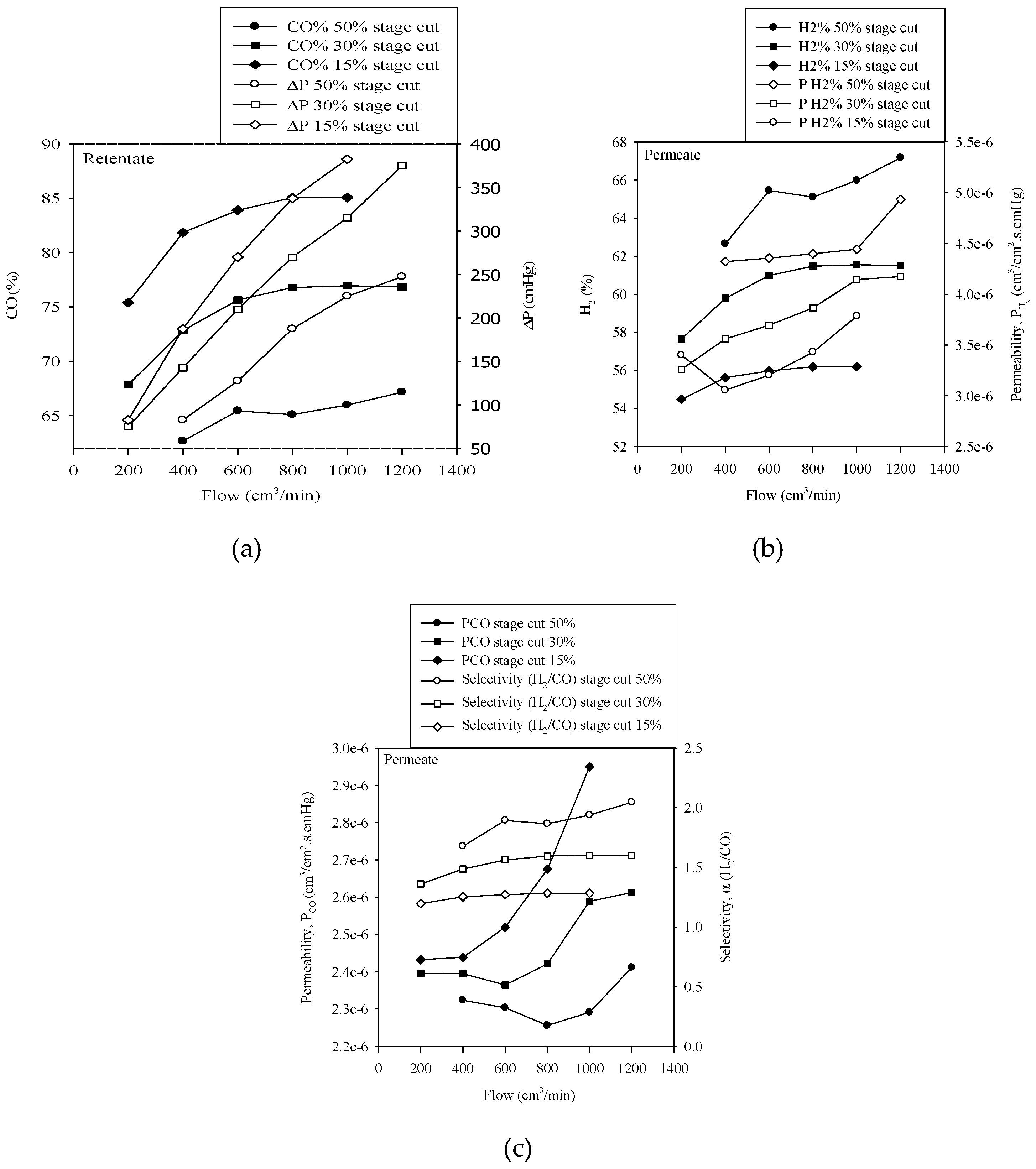
3.1. Effect of Varying Gas Flow Rate for Various Stage Cut
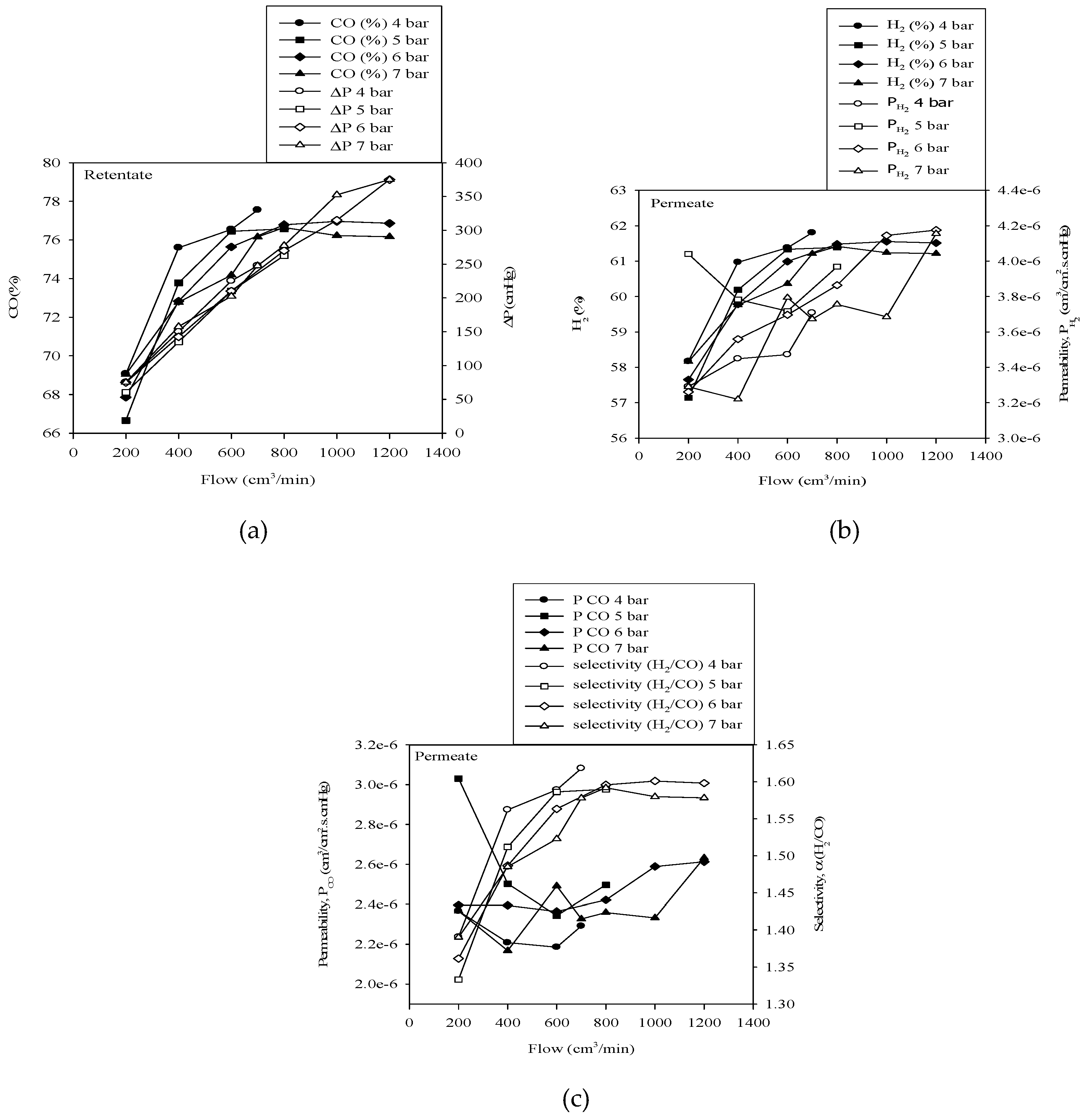
3.2. Effect of Varying Gas Flow Rate for Various Gas Inlet Pressures
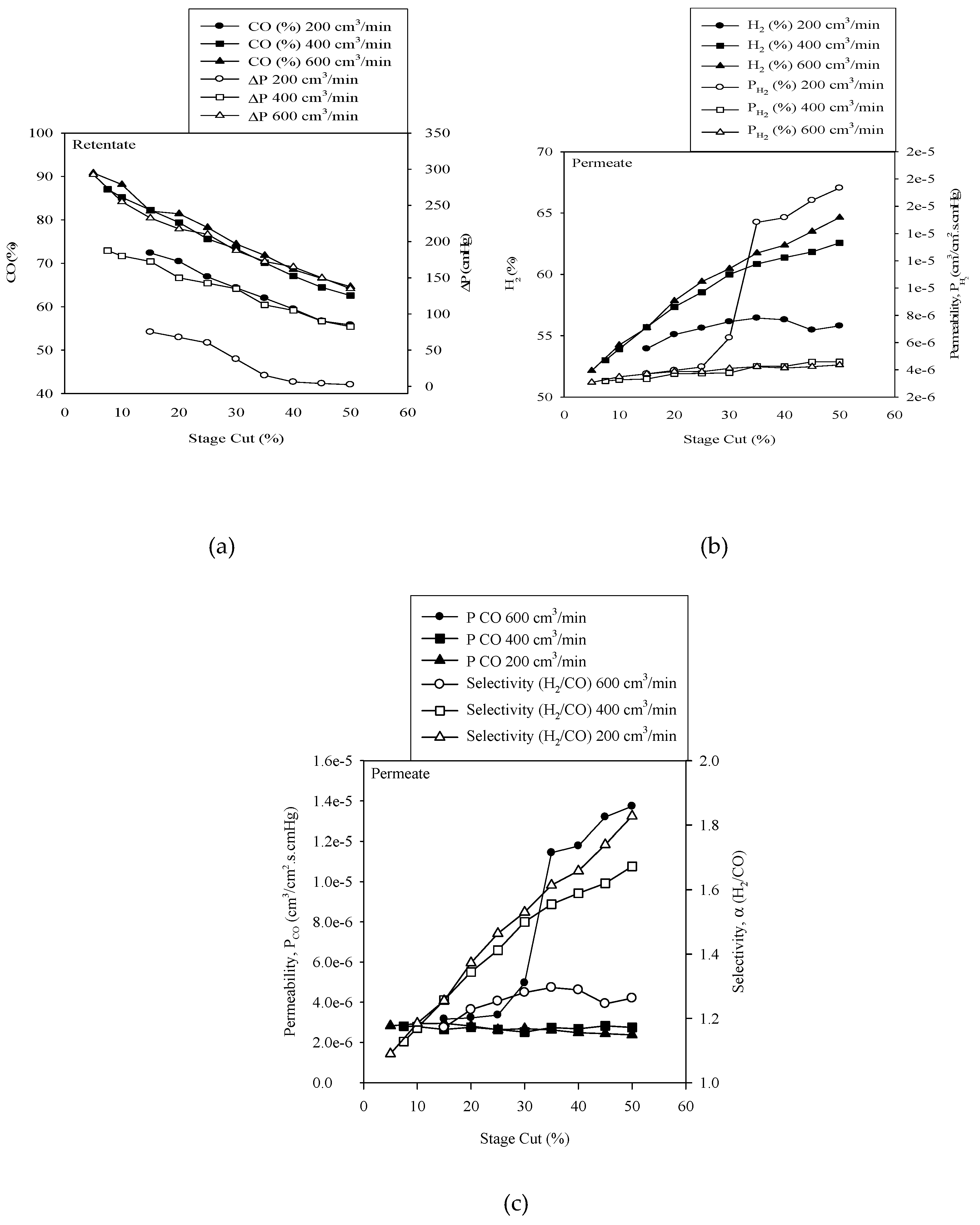
3.3. Effect of Varying the Stage Cut for Various Gas Flow Rate
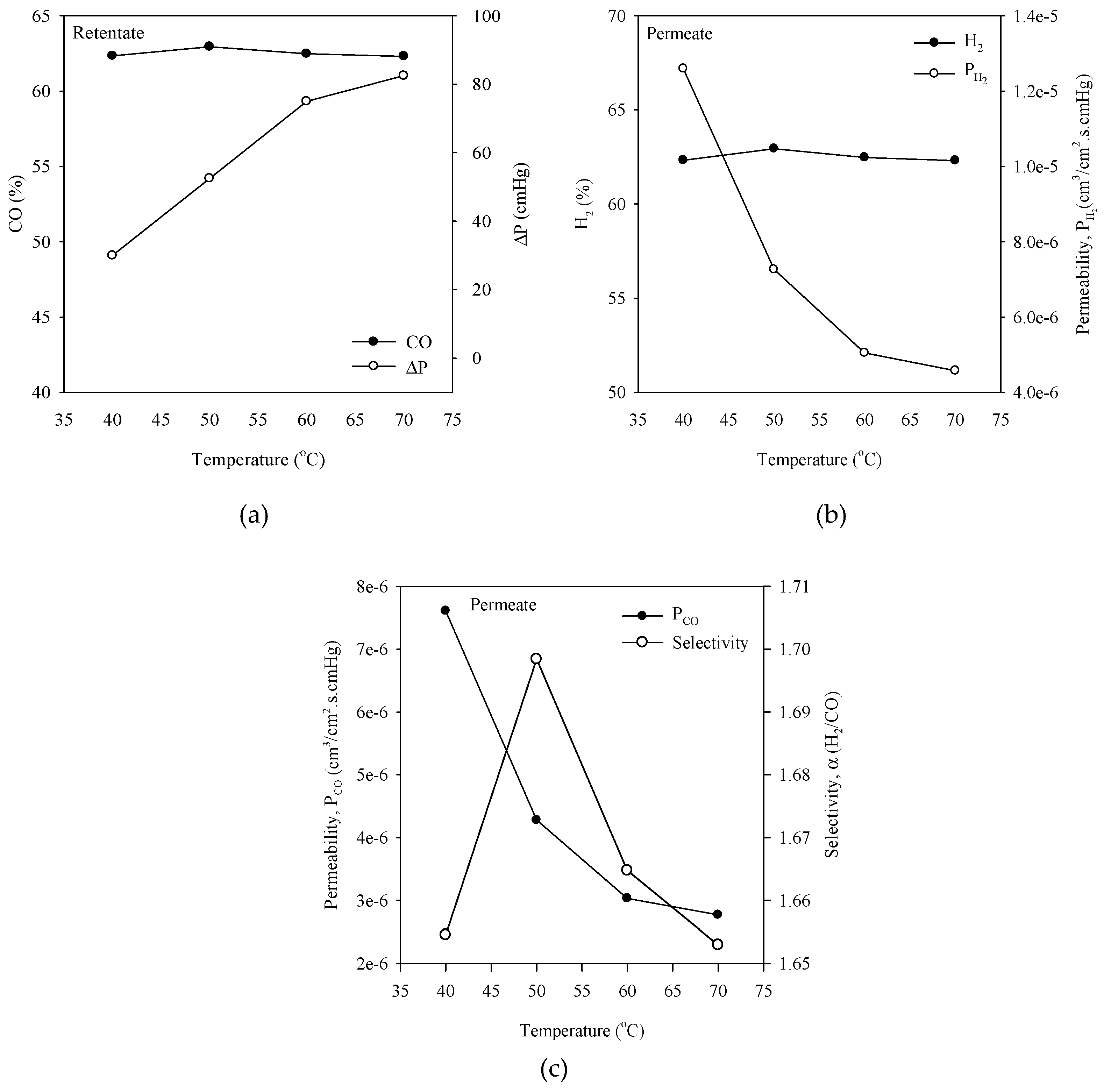
3.4. Effect of Varying Membrane Module Temperature
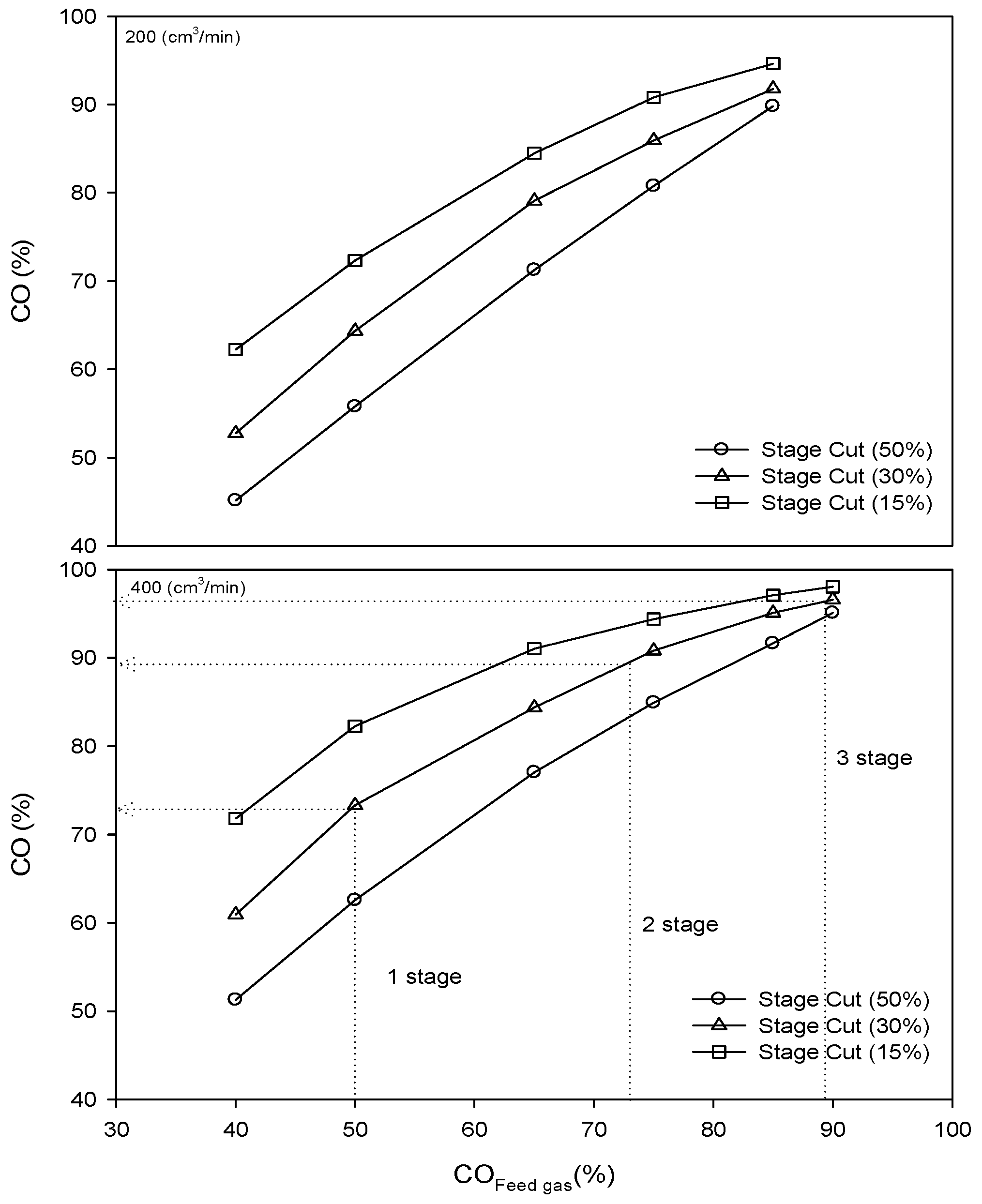
3.5. Estimation of Multistage Membrane Required for CO Purity
4. Conclusions and Future Outlooks
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Kwak, T.; Maken, S.; Lee, S.; Park, J.; Min, B.; Yoo, Y.D. Environmental Aspects of Gasification of Korean Municipal Solid Waste in a Pilot Plant. Fuel 2006, 85, 2012–2017. [Google Scholar] [CrossRef]
- Sakai, S.; Sawell, S.; Chandler, A.; Eighmy, T.; Kosson, D.; Vehlow, J.; Van der Sloot, H.; Hartlen, J.; Hjelmar, O. World Trends in Municipal Solid Waste Management. Waste Manag. 1996, 16. [Google Scholar] [CrossRef]
- Tchobanoglous, G.; Theisen, H.; Vigil, S.A.; Alaniz, V.M. Integrated Solid Waste Management: Engineering Principles and Management Issues; McGraw-Hill: New York, NY, USA, 1993. [Google Scholar]
- Murphy, J.D.; McKeogh, E. Technical, Economic and Environmental Analysis of Energy Production from Municipal Solid Waste. Renew. Energy 2004, 29, 1043–1057. [Google Scholar] [CrossRef]
- Alzate-Gaviria, L.M.; Sebastian, P.; Pérez-Hernández, A.; Eapen, D. Comparison of Two Anaerobic Systems for Hydrogen Production from the Organic Fraction of Municipal Solid Waste and Synthetic Wastewater. Int. J. Hydrogen Energy 2007, 32, 3141–3146. [Google Scholar] [CrossRef]
- Luo, S.; Xiao, B.; Hu, Z.; Liu, S.; Guan, Y.; Cai, L. Influence of Particle Size on Pyrolysis and Gasification Performance of Municipal Solid Waste in a Fixed Bed Reactor. Bioresour. Technol. 2010, 101, 6517–6520. [Google Scholar] [CrossRef] [PubMed]
- Desideri, U. Developments and Innovation in Carbon Dioxide (CO2) Capture and Storage Technology; Woodhead Publishing: Cambridge, UK, 2010. [Google Scholar]
- Nikolaidis, G.N.; Kikkinides, E.S.; Georgiadis, M.C. A Model-Based Approach for the Evaluation of New Zeolite 13X-Based Adsorbents for the Efficient Post-Combustion CO2 Capture using P/VSA Processes. Chem. Eng. Res. Des. 2018, 131, 362–374. [Google Scholar] [CrossRef]
- Cavenati, S.; Grande, C.A.; Rodrigues, A.E. Separation of CH4/CO2/N2 Mixtures by Layered Pressure Swing Adsorption for Upgrade of Natural Gas. Chem. Eng. Sci. 2006, 61, 3893–3906. [Google Scholar] [CrossRef]
- DiMartino, S.; Glazer, J.; Houston, C.; Schott, M. Hydrogen/Carbon Monoxide Separation with Cellulose Acetate Membranes. Gas Sep. Purif. 1988, 2, 120–125. [Google Scholar] [CrossRef]
- Khanipour, M.; Mirvakili, A.; Bakhtyari, A.; Farniaei, M.; Rahimpour, M.R. Enhancement of Synthesis Gas and Methanol Production by Flare Gas Recovery Utilizing a Membrane Based Separation Process. Fuel Process. Technol. 2017, 166, 186–201. [Google Scholar] [CrossRef]
- Dutta, N.; Patil, G. Developments in CO Separation. Gas Sep. Purif. 1995, 4, 277–283. [Google Scholar] [CrossRef]
- Hsieh, H. Inorganic Membranes for Separation and Reaction; Elsevier: Amsterdam, The Netherlands, 1996. [Google Scholar]
- Koros, W.J.; Mahajan, R. Pushing the Limits on Possibilities for Large Scale Gas Separation: Which Strategies? J. Membr. Sci. 2000, 175, 181–196. [Google Scholar] [CrossRef]
- Ho, W.; Sirkar, K. Membrane Handbook; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Bhadra, S.; Farooq, S. Separation of Methane–nitrogen Mixture by Pressure Swing Adsorption for Natural Gas Upgrading. Ind. Eng. Chem. Res. 2011, 50, 14030–14045. [Google Scholar] [CrossRef]
- Corbett, L. Industrial Engineering Chemistry Process Design and Development; American Chemical Society: Washington, DC, USA, 1975; Volume 13, p. 181. [Google Scholar]
- Lu, G.Q.; Da Costa, J.D.; Duke, M.; Giessler, S.; Socolow, R.; Williams, R.H.; Kreutz, T. Inorganic membranes for hydrogen production and purification: A critical review. J. Colloid Interface Sci. 2007, 314, 589–603. [Google Scholar] [CrossRef] [PubMed]
- Basile, A.; Gallucci, F.; Tosti, S. Synthesis, characterization and applications of palladium membranes. Membr. Sci. Technol. 2008, 13, 255–323. [Google Scholar]
- Sanchez, J.M.; Barreiro, MM.; Marono, M. Hydrogen enrichment and separation from synthesis gas by the use of a membrane reactor. Biomass Bioenergy 2011, 35, S132–S144. [Google Scholar] [CrossRef]
- Shimekit, B.; Mukhtar, H. Natural Gas Purification Technologies-Major Advances for CO2 Separation and Future Directions; INTECH Open Access Publisher: London, UK, 2012; pp. 235–270. [Google Scholar]
- Robeson, L.M. The upper bound revisited. J. Membr. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]
- Noble, R.D. Perspectives on mixed matrix membranes. J. Membr. Sci. 2011, 15, 393–397. [Google Scholar] [CrossRef]
- Scholes, C.A. Membrane gas separation applications in natural gas processing. Fuel 2012, 96, 15–28. [Google Scholar] [CrossRef]
- Meulenberg, W.A.; Hansch, R.; Buchkremer, H.P.; Stöver, D. Device for Gas Separation and Method for Producing such a System. U.S. Patent 8,016,924, 13 September 2011. [Google Scholar]
- David, O.C.; Gorri, D.; Urtiaga, A.; Ortiz, I. Mixed Gas Separation Study for the Hydrogen Recovery from H2/CO/N2/CO2 Post Combustion Mixtures using a Matrimid Membrane. J. Membr. Sci. 2011, 378, 359–368. [Google Scholar] [CrossRef]
- Koros, W.J.; Fleming, G. Membrane-Based Gas Separation. J. Membr. Sci. 1993, 83, 1–80. [Google Scholar] [CrossRef]
- Javaid, A. Membranes for Solubility-Based Gas Separation Applications. Chem. Eng. J. 2005, 112, 219–226. [Google Scholar] [CrossRef]
- Hines, A.L.; Maddox, R.N. Mass Transfer: Fundamentals and Applications; Prentice-Hall: Englewood Cliffs, NJ, USA, 1985. [Google Scholar]
- Lin, H.; Freeman, B.D. Gas Solubility, Diffusivity and Permeability in Poly (Ethylene Oxide). J. Membr. Sci. 2004, 239, 105–117. [Google Scholar] [CrossRef]
- Reid, R.C.; Prausnitz, J.M.; Poling, B.E. The Properties of Gases and Liquids; Mcgraw-Hill: New York, NY, USA, 1987. [Google Scholar]
- Robeson, L.M.; Smith, Z.P.; Freeman, B.D.; Paul, D.R. Contributions of Diffusion and Solubility Selectivity to the Upper Bound Analysis for Glassy Gas Separation Membranes. J. Membr. Sci. 2014, 453, 71–83. [Google Scholar] [CrossRef]
- El-Azzami, L.A.; Grulke, E.A. Dual Mode Model for Mixed Gas Permeation of CO2, H2, and N2 through a Dry Chitosan Membrane. J. Polym. Sci. Part B Polym. Phys. 2007, 45, 2620–2631. [Google Scholar] [CrossRef]
- Tanaka, K.; Kita, H.; Okamoto, K.; Nakamura, A.; Kusuki, Y. Gas Permeability and Permselectivity in Polyimides Based on 3,3′,4,4′-Biphenyltetracarboxylic Dianhydride. J. Membr. Sci. 1989, 47, 203–215. [Google Scholar] [CrossRef]
- Peer, M.; Mehdi Kamali, S.; Mahdeyarfar, M.; Mohammadi, T. Separation of Hydrogen from Carbon Monoxide using a Hollow Fiber Polyimide Membrane: Experimental and Simulation. Chem. Eng. Technol. 2007, 30, 1418–1425. [Google Scholar] [CrossRef]
- Marković, A.; Stoltenberg, D.; Enke, D.; Schlünder, E.; Seidel-Morgenstern, A. Gas Permeation through Porous Glass Membranes: Part I. Mesoporous Glasses—Effect of Pore Diameter and Surface Properties. J. Membr. Sci. 2009, 336, 17–31. [Google Scholar]
- Zarca, G.; Urtiaga, A.; Biegler, L.T.; Ortiz, I. An Optimization Model for Assessment of Membrane-Based Post-Combustion Gas Upcycling into Hydrogen or Syngas. J. Membr. Sci. 2018, 563, 83–92. [Google Scholar] [CrossRef]
- Bernardo, P.; Drioli, E.; Golemme, G. Membrane Gas Separation: A Review/State of the Art. Ind. Eng. Chem. Res. 2009, 48, 4638–4663. [Google Scholar] [CrossRef]
- Drioli, M.S.B.; Barbieri, G. CO2-CH4 Membrane Separation. Available online: http://dx.medra.org/10.17374/CI.2015.97.5.14 (accessed on 15 September 2017).

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poudel, J.; Choi, J.H.; Oh, S.C. Process Design Characteristics of Syngas (CO/H2) Separation Using Composite Membrane. Sustainability 2019, 11, 703. https://doi.org/10.3390/su11030703
Poudel J, Choi JH, Oh SC. Process Design Characteristics of Syngas (CO/H2) Separation Using Composite Membrane. Sustainability. 2019; 11(3):703. https://doi.org/10.3390/su11030703
Chicago/Turabian StylePoudel, Jeeban, Ja Hyung Choi, and Sea Cheon Oh. 2019. "Process Design Characteristics of Syngas (CO/H2) Separation Using Composite Membrane" Sustainability 11, no. 3: 703. https://doi.org/10.3390/su11030703
APA StylePoudel, J., Choi, J. H., & Oh, S. C. (2019). Process Design Characteristics of Syngas (CO/H2) Separation Using Composite Membrane. Sustainability, 11(3), 703. https://doi.org/10.3390/su11030703




