Extent of Bollworm and Sucking Pest Damage on Modern and Traditional Cotton Species and Potential for Breeding in Organic Cotton
Abstract
:1. Introduction
2. Materials and Methods
2.1. Location and Climatic Conditions
2.2. Experimental Design
2.3. Assessment of Damage by Bollworm and Sucking Pest Incidence
2.4. Statistical Analysis
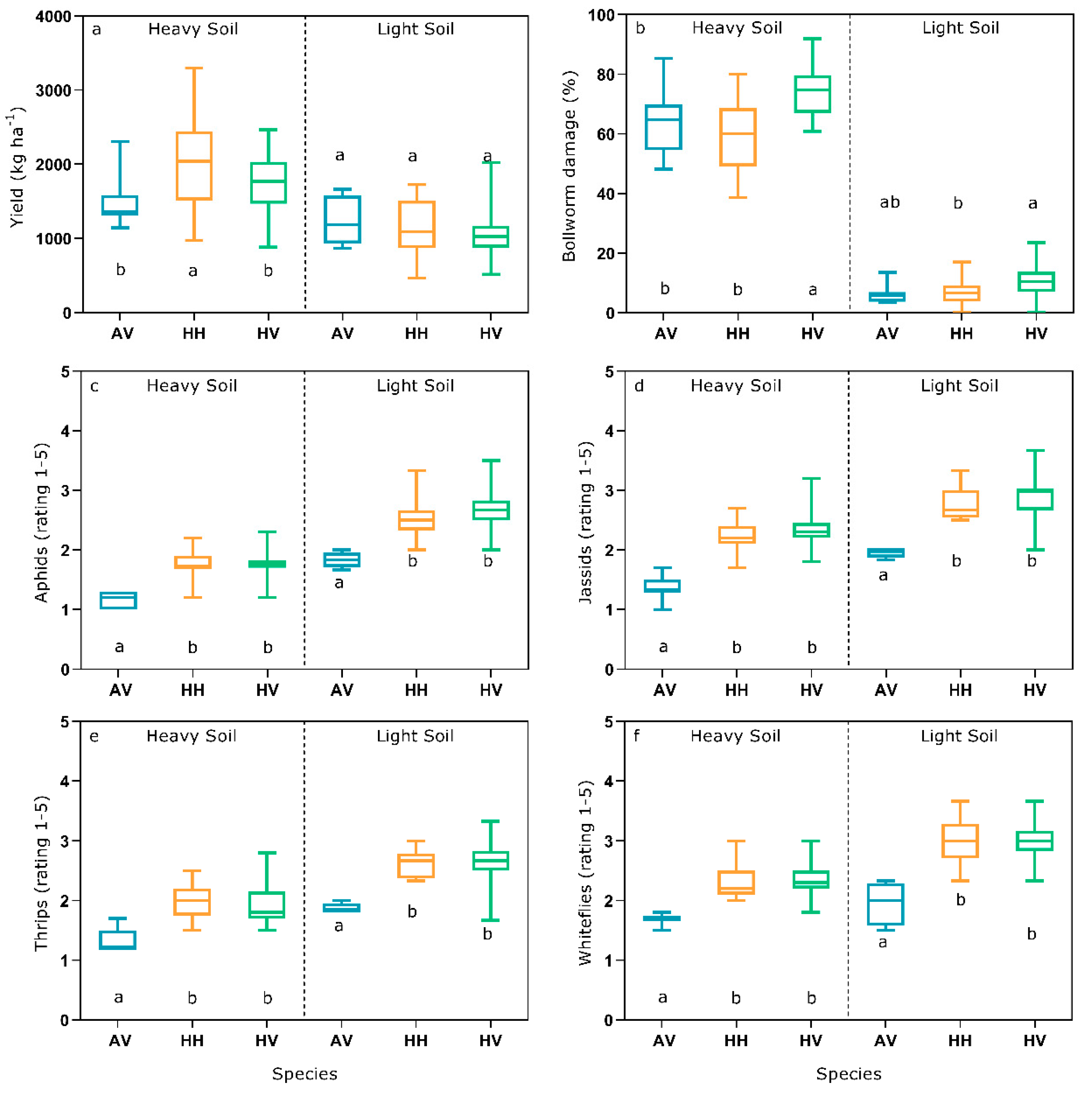
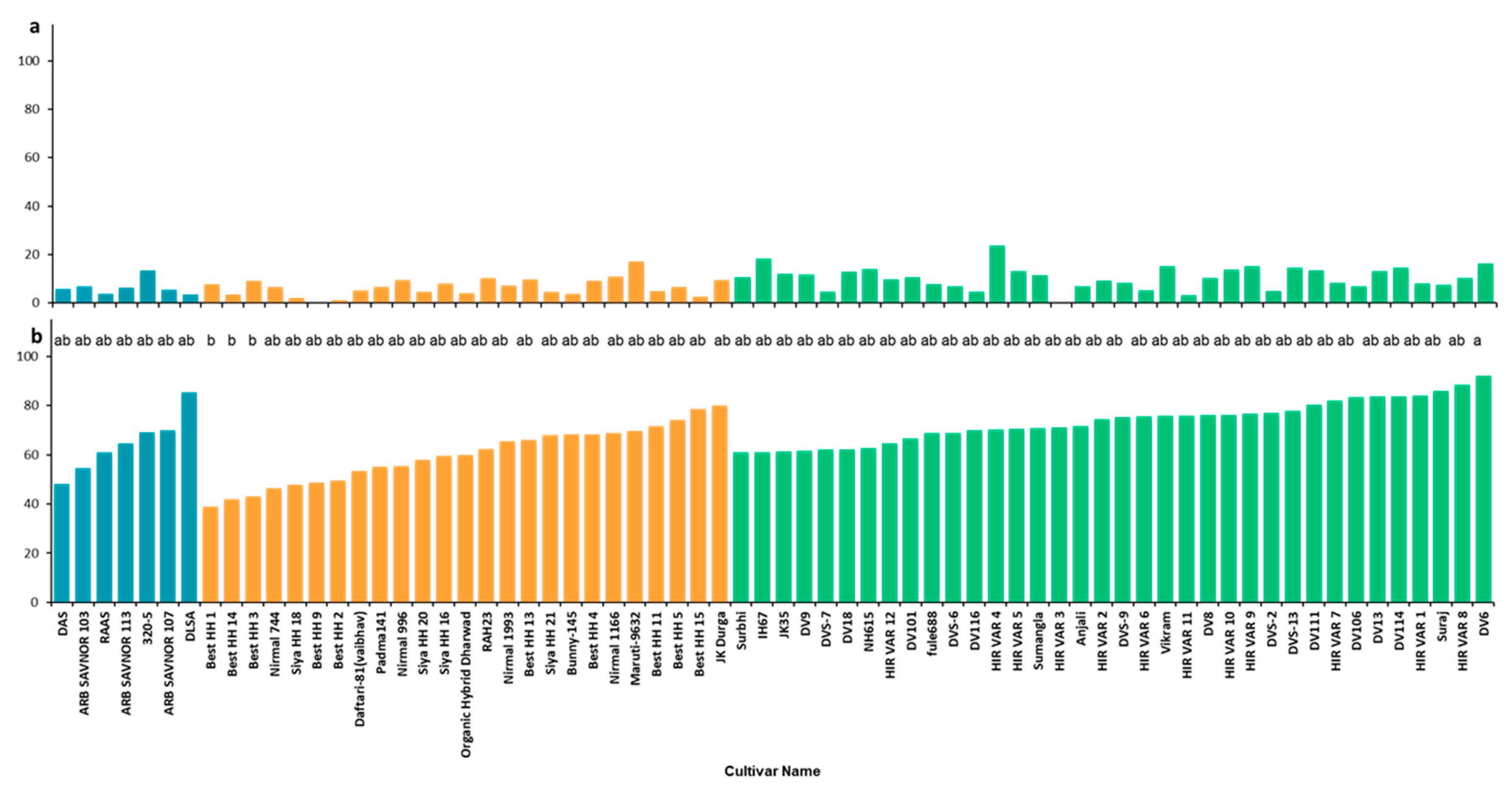
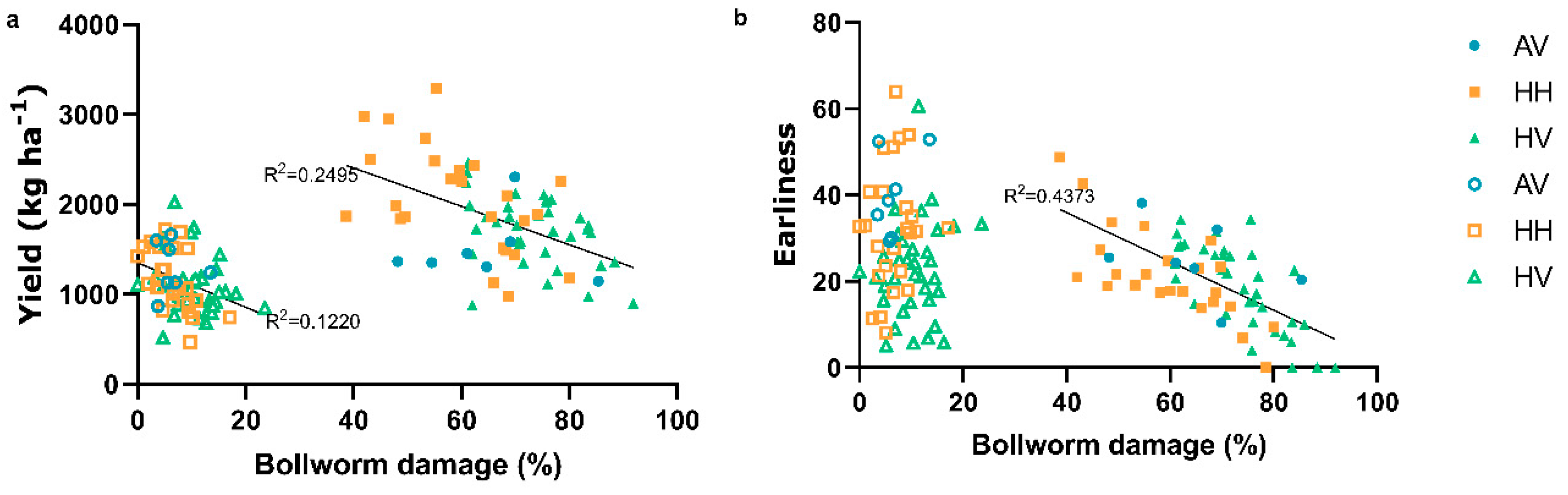
3. Results
4. Discussion
4.1. Importance of Bollworm Attack and Damage at Contrasting Sites
4.2. Potential of Different Cotton Species and Cultivar Types for Organic Production
4.3. Indirect Parameters for Selection of Resistance to Bollworm and Sucking Pests
4.4. Selection of Cultivars and Breeding Lines for Cultivation and Participatory Breeding
5. Conclusions and Outlook
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- ICAC, The International Cotton Advisory Committee. Organic Cotton Production IV; The International Cotton Advisory Committee Recorder: Washington, DC, USA, 1998; Volume XVI. [Google Scholar]
- Forster, D.; Andres, C.; Verma, R.; Zundel, C.; Messmer, M.M.; Mäder, P. Yield and economic performance of organic and conventional cotton-based farming systems–results from a field trial in India. PLoS ONE 2013, 8, e81039. [Google Scholar] [CrossRef] [PubMed]
- TextileExchange. 2018 Organic Cotton Market Report; TextileExchagne: Milano, Italy, 2018. [Google Scholar]
- Fayet, L.; Vermeulen, W.J.V. Supporting Smallholders to Access Sustainable Supply Chains: Lessons from the Indian Cotton Supply Chain. Sustain. Dev. 2014, 22, 289–310. [Google Scholar] [CrossRef]
- Riar, A.; Mandloi, L.S.; Poswal, R.S.; Messmer, M.M.; Bhullar, G.S. A diagnosis of biophysical and socio-economic factors influencing farmers’ choice to adopt organic or conventional farming systems for cotton production. Front. Plant Sci. 2017, 8, 1289. [Google Scholar] [CrossRef] [PubMed]
- Coventry, D.R.; Poswal, R.S.; Yadav, A.; Riar, A.S.; Zhou, Y.; Kumar, A.; Chand, R.; Chhokar, R.S.; Sharma, R.K.; Yadav, V.K.; et al. A comparison of farming practices and performance for wheat production in Haryana, India. Agric. Syst. 2015, 137, 139–153. [Google Scholar] [CrossRef]
- Gutierrez, A.P.; Ponti, L.; Herren, H.; Baumgärtner, J.; Kenmore, P. Deconstructing Indian cotton: Weather, yields, and suicides. Environ. Sci. Eur. 2015, 27, 1–17. [Google Scholar] [CrossRef]
- Fand, B.B.; Nagrare, V.; Gawande, S.; Nagrale, D.; Naikwadi, B.; Deshmukh, V.; Gokte-Narkhedkar, N.; Waghmare, V. Widespread infestation of pink bollworm, Pectinophora gossypiella (Saunders) (Lepidoptera: Gelechidae) on Bt cotton in Central India: A new threat and concerns for cotton production. Phytoparasitica 2019, 47, 1–13. [Google Scholar] [CrossRef]
- Kranthi, K.R. Bt-Cotton Question and Answer: Indian Society for Cotton Improvement; Central Institute for Cotton Research: Nagpur, India, 2012. [Google Scholar]
- Carrière, Y.; Crickmore, N.; Tabashnik, B.E. Optimizing pyramided transgenic Bt crops for sustainable pest management. Nat. Biotechnol. 2015, 33, 161. [Google Scholar] [CrossRef] [PubMed]
- Ranjith, M.T.; Prabhuraj, A.; Srinivasa, Y.B. Survival and reproduction of natural populations of Helicoverpa armigera on Bt-cotton hybrids in Raichur, India. Curr. Sci. (Bangalore) 2010, 99, 1602–1606. [Google Scholar]
- Bennett, R.M.; Ismael, Y.; Kambhampati, U.; Morse, S. Economic impact of genetically modified cotton in India. AgBioForum 2005, 7, 96–100. [Google Scholar]
- Sethi, K.; Siwach, P.; Verma, S.K. Assessing genetic diversity among six populations of Gossypium arboreum L. using microsatellites markers. Physiol. Mol. Biol. Plants 2015, 21, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Tan, W.; Li, Q.; Chen, X.; Wang, J.; Liu, X.; Ye, W.; Yin, Z. Genome-wide characterization and expression profiling of the MAPKKK genes in Gossypium arboreum L. Genome 2019, 62, 609–622. [Google Scholar] [CrossRef] [PubMed]
- Roner, T. Participatory Cotton Breeding for Organic and Low Input Farming in Central India; Cuvillier Verlag: Gottingen, Germany, 2012. [Google Scholar]
- Kulkarni, V.N.; Khadi, B.M.; Maralappanavar, M.S.; Deshapande, L.A.; Narayanan, S. The worldwide gene pools of Gossypium arboreum L. and G. herbaceum L. and their improvement. In Genetics and Genomics of Cotton; Springer: Berlin/Heidelberg, Germany, 2009; pp. 69–97. [Google Scholar]
- Miyazaki, J.; Stiller, W.N.; Wilson, L.J. Identification of host plant resistance to silverleaf whitefly in cotton: Implications for breeding. Field Crop. Res. 2013, 154, 145–152. [Google Scholar] [CrossRef]
- Dilnur, T.; Peng, Z.; Pan, Z.; Palanga, K.K.; Jia, Y.; Gong, W.; Du, X. Association analysis of salt tolerance in Asiatic cotton (Gossypium arboretum) with SNP markers. Int. J. Mol. Sci. 2019, 20, 2168. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Erpelding, J.E.; Stetina, S.R. Genome-wide association study of Gossypium arboreum resistance to reniform nematode. BMC Genet. 2018, 19, 52. [Google Scholar] [CrossRef] [PubMed]
- Dietzel, N.; Kahle, J. Local Knowledge of Pests and Pest Management in the Nimar Region in Madhya Pradesh, India; Research Insititute of Organic Agriculture (FiBL): Frick, Switzerland, 2012. [Google Scholar]
- Hofs, J.-L.; Hau, B.; Marais, D.; Fok, M. Boll distribution patterns in Bt and non-Bt cotton cultivars: II. Study on small-scale farming systems in South Africa. Field Crop. Res. 2006, 98, 210–215. [Google Scholar] [CrossRef]
- Makvana, D.K.; Chudasama, K.A.; Balas, T.K. Estimation of Yield Losses Due to Major Sucking Insect Pests of Bt Cotton. Int. J. Curr. Microbiol. Appl. Sci. 2018, 7, 956–959. [Google Scholar] [CrossRef]
- Eyhorn, F.; Ratter, S.G.; Ramakrishnan, M. Organic Cotton Crop Guide. A Manual for Practitioners in the Tropics; Research Institute of Organic Agriculture (FibL): Frick, Switzerland, 2005. [Google Scholar]
- Meena, R.; Monga, D.; Singh, V.; Mahala, S. Screening of cotton (Gossypium hirsutum and G. arboreum) germplasm for economical characters. Indian J. Agric. Sci. 2004, 74, 21–23. [Google Scholar]
- Chandra, M.; Sreenivasan, S. Studies on improved Gossypium arboreum cotton: Part II–Yarn and fabric quality parameters. Indian J. Fibre Text. Res. 2011, 36, 158–167. [Google Scholar]
- OCCW, Cotton Plant & Fibre, Organic Cotton Community Worldwide. Available online: http://www.organiccotton.org/oc/Cotton-general/Plant-and-fibres/Cotton-lint.php (accessed on 16 July 2015).
- Blaise, D.; Kranthi, K.R. Cry1Ac expression in transgenic Bt cotton hybrids is influenced by soil moisture and depth. Curr. Sci. 2011, 101, 783–786. [Google Scholar]
- Esquivel, J.F.; Hinze, L.L. The cotton boll: The relationship of species and genotype with temporal development of boll wall thickness. Crop Sci. 2019, 59, 1200–1210. [Google Scholar] [CrossRef]
- Zweifel, D.J. Assessment of Options and Tools for Pest Monitoring in Organic Cotton Production Systems of the Nimar Region, Madhya Pradesh, India; University of Applied Sciences ZHAW: Zurich, Switzerland, 2014. [Google Scholar]
- Keshav, A.; Shera, P.; Singh, J. Morphological basis of resistance to spotted bollworm, Earias vittella (Fabricius) in Asiatic cotton. Phytoparasitica 2013, 41, 235–240. [Google Scholar] [CrossRef]
- Aslam, M.; Saeed, N.A. Comparative resistance of different cotton genotypes against suckinginsect pest complex of cotton. Sarhad J. Agric. 2004, 23, 141–143. [Google Scholar]
- Raza, A.; Afzal, M.; Sarfraz, M.; Bashir, M.; Gogi, M.; Sarwar, M. Physico-morphic plant characters in relation to resistance against sucking insect pests in some new cotton genotypes. Pakistan Entomol. 2000, 22, 73–77. [Google Scholar]
- Gomti Grover, B.K.; Pathak, D.; Kumar, V. Genetic variation for leaf trichome density and its association with sucking insect-pests incidence in Asiatic cotton. Indian J. Genet. 2016, 76, 365–368. [Google Scholar] [CrossRef]
- Wan, Q.; Zhang, Z.; Hu, M.; Chen, L.; Liu, D.; Chen, X.; Wang, W.; Zheng, J. T 1 locus in cotton is the candidate gene affecting lint percentage, fiber quality and spiny bollworm (Earias spp) resistance. Euphytica 2007, 158, 241–247. [Google Scholar] [CrossRef]
- Sankeshwar, M.; Jadhav, M.; Adiger, S.; Patil, R.S.; Katageri, I. Mapping of QTLs for traits related to leaf pubescence, jassid resistance and yield in cotton (Gossypium spp). Indian J. Genet. Plant Breed. 2018, 78, 252–260. [Google Scholar] [CrossRef]
- Ahuja, S.; Banerjee, S.; Singh, J.; Singh, P.; Singh, V.; Monga, D.; Tuteja, O. Development of Gossypium hirsutum lines with cytoplasmic bollworm tolerance from Gossypium arboreum and Gossypium tomentosum. Plant Breed. 2009, 128, 712–715. [Google Scholar] [CrossRef]

| Parameter | Observation Basis | Unit | Number of Observation Timing | |
|---|---|---|---|---|
| Bollworm damage | five marked plants | number | 3 | |
| Yield data | Yield per plot | Plot | g | 3 |
| No. of harvested bolls | Plot | number | 3 | |
| No. of plants per plot | Plot | number | 3 | |
| Earliness | First picking (%) | Plot | percentage | 1 |
| Sucking pest | Aphid | Plot | Score 1-51 | 3 |
| Jassid | Plot | Score 1-51 | 3 | |
| Thrips | Plot | Score 1-51 | 3 | |
| Whitefly | Plot | Score 1-51 | 3 | |
| Morphological traits | Leaf hairiness | five single plants per plot | Score 1-52 | 1 |
| Stem hairiness | five single plants per plot | Score 1-52 | 1 | |
| Parameter | Bollworm Damage | Yield | Earliness | Jassids | Aphids | Whiteflies | Thrips | Leaf Hairiness | Stem Hairiness |
|---|---|---|---|---|---|---|---|---|---|
| Bollworm damage | −0.15 | 0.02 | −0.06 | 0.05 | 0.13 | 0.11 | 0.11 | 0.15 | |
| Yield | −0.37*** | −0.38*** | −0.08 | −0.02 | 0.07 | −0.08 | −0.06 | −0.14 | |
| First picking | −0.62*** | 0.13 | −0.23* | −0.15 | −0.13 | −0.19* | 0.01 | 0.08 | |
| Jassids | −0.01 | −0.03 | 0.03 | 0.36*** | 0.41*** | 0.45*** | 0.21* | 0.09 | |
| Aphids | −0.09 | 0.11 | 0.12 | 0.66*** | 0.28** | 0.28** | 0.23* | −0.00 | |
| Whiteflies | −0.13 | −0.07 | 0.13 | 0.61*** | 0.35*** | 0.30** | 0.21* | 0.03 | |
| Thrips | −0.01 | 0.20* | −0.04 | 0.65*** | 0.60*** | 0.37*** | 0.07 | 0.04 | |
| Leaf hairiness | 0.20* | −0.13 | 0.02 | 0.12 | 0.19* | 0.18* | 0.14 | 0.62*** | |
| Stem hairiness | 0.19* | −0.30 | −0.07 | 0.07 | 0.10 | 0.12 | 0.06 | 0.62*** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vonzun, S.; Messmer, M.M.; Boller, T.; Shrivas, Y.; Patil, S.S.; Riar, A. Extent of Bollworm and Sucking Pest Damage on Modern and Traditional Cotton Species and Potential for Breeding in Organic Cotton. Sustainability 2019, 11, 6353. https://doi.org/10.3390/su11226353
Vonzun S, Messmer MM, Boller T, Shrivas Y, Patil SS, Riar A. Extent of Bollworm and Sucking Pest Damage on Modern and Traditional Cotton Species and Potential for Breeding in Organic Cotton. Sustainability. 2019; 11(22):6353. https://doi.org/10.3390/su11226353
Chicago/Turabian StyleVonzun, Seraina, Monika M. Messmer, Thomas Boller, Yogendra Shrivas, Shreekant S. Patil, and Amritbir Riar. 2019. "Extent of Bollworm and Sucking Pest Damage on Modern and Traditional Cotton Species and Potential for Breeding in Organic Cotton" Sustainability 11, no. 22: 6353. https://doi.org/10.3390/su11226353
APA StyleVonzun, S., Messmer, M. M., Boller, T., Shrivas, Y., Patil, S. S., & Riar, A. (2019). Extent of Bollworm and Sucking Pest Damage on Modern and Traditional Cotton Species and Potential for Breeding in Organic Cotton. Sustainability, 11(22), 6353. https://doi.org/10.3390/su11226353






