You Can’t See the Woods for the Trees: Invasive Acer negundo L. in Urban Riparian Forests Harms Biodiversity and Limits Recreation Activity
Abstract
1. Introduction
2. Materials and Methods
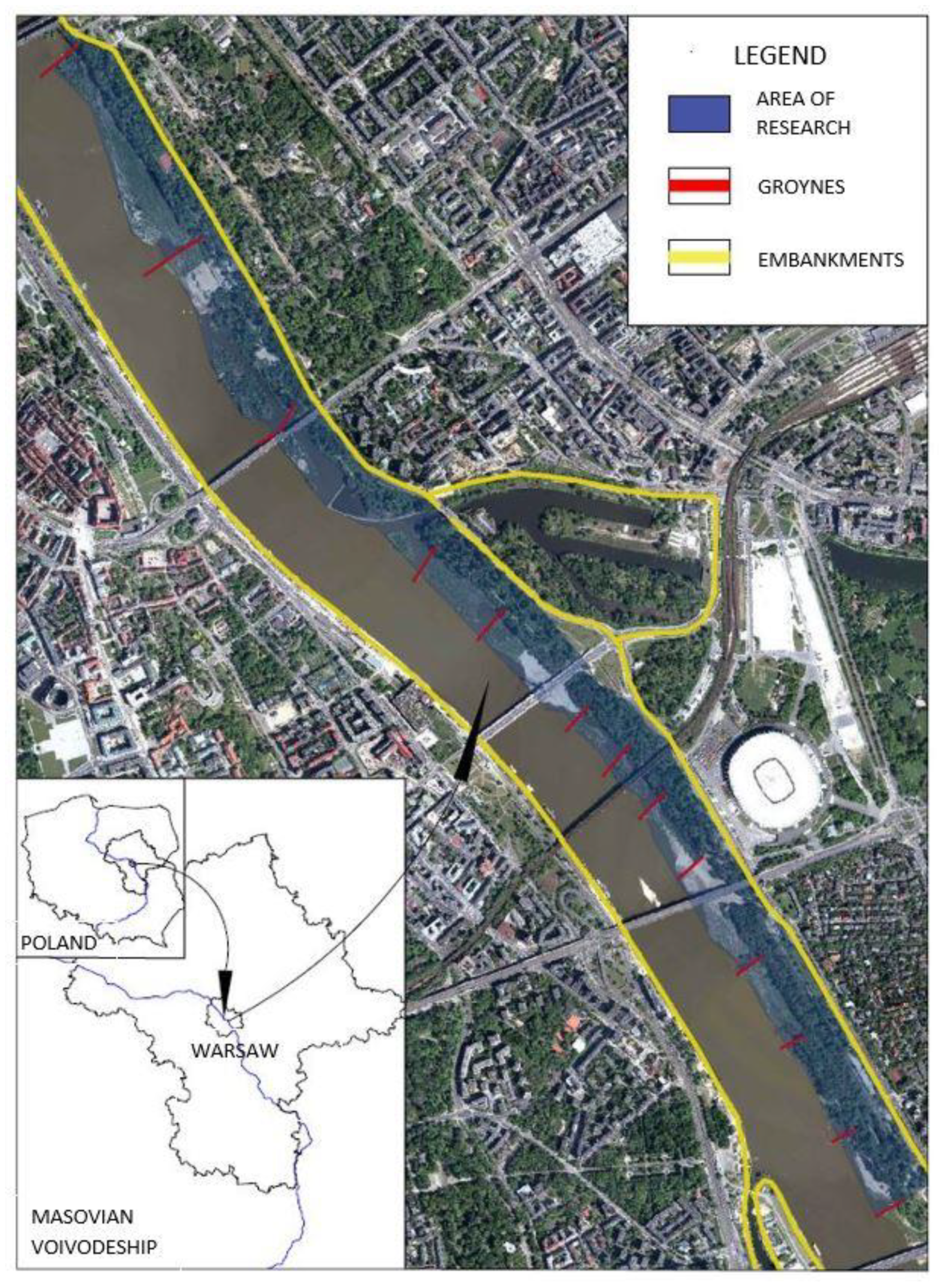
2.1. Study Area
2.2. Canopy Tree Inventory
2.3. The Effect of A. negundo on Biodiversity
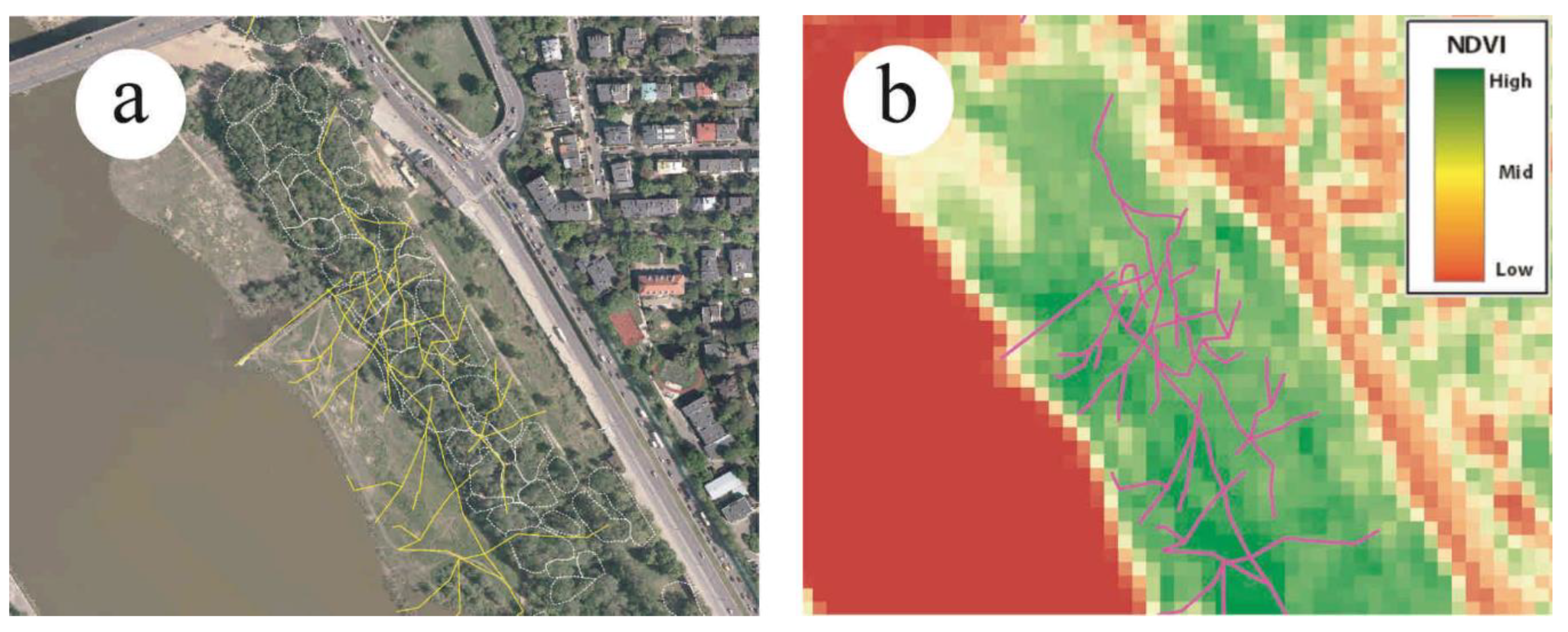
2.4. Off Trail Activity as an Indicator of Recreational Ecosystem Services of Riparian Forests
3. Results
3.1. Invasion Intensity into Different Forest Types
3.2. The Effect of A. negundo on Biodiversity
3.3. Effect of A. negundo on Recreation Ecosystem Services
4. Discussion
5. Conclusions
- A. negundo is a permanent and abundant component of the urban riparian forests in the Vistula river valley in Warsaw, and it was found to be more abundant in willow forests stands than in poplar forests.
- The abundance of A. negundo was found to be significantly higher in younger stands than in older ones, the differences were manifested in both number of stems and their volume per plot.
- Occurrence of A. negundo in riparian forests negatively affects biodiversity, shrub layer, and herbaceous vegetation, the more invaded stands were poorer in species diversity.
- An increased share of A. negundo was found to be related to decreased activity of visitors in the forest, but the effect was slightly stronger in the willow forests. Presence of ash-leaved maple plays an important role in providing recreation possibilities for the city dwellers.
- Factors explaining the recreational activity by users in the riparian forests, expressed in soil compaction of informal tracks, were the distance from the main track and the volume of A. negundo trunks.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tzoulas, K.; Korpela, K.; Venn, S.; Yli-Pelkonen, V.; Kazmierczak, A.; Niemela, J.; James, P. Promoting ecosystem and human health in urban areas using green infrastructure: A literature review. Landsc. Urban Plan. 2007, 81, 167–178. [Google Scholar] [CrossRef]
- Breuste, J.; Niemela, J.; Snep, R.P.H. Applying landscape ecological principles in urban environments. Landsc. Ecol. 2008, 23, 1139–1142. [Google Scholar] [CrossRef]
- Vandermeulen, V.; Verspecht, A.; Vermeire, B.; van Huylenbroeck, G.; Gellynck, X. The use of economic valuation to create public support for green infrastructure investments in urban areas. Landsc. Urban Plan. 2011, 103, 198–206. [Google Scholar] [CrossRef]
- Gómez-Baggethun, E.; Barton, D.N. Classifying and valuing ecosystem services for urban planning. Ecol. Econ. 2013, 86, 235–245. [Google Scholar] [CrossRef]
- Schäffler, A.; Swilling, M. Valuing green infrastructure in an urban environment under pressure—The Johannesburg Case. Ecol. Econ. 2013, 86, 246–257. [Google Scholar] [CrossRef]
- Breuste, J.; Rahimi, A. Many public urban parks, but who profits from them? The example of Tabriz, Iran. Ecol. Process. 2015, 4, 6. [Google Scholar] [CrossRef]
- Richards, D.R.; Warren, P.H.; Moggridge, H.L.; Maltby, L. Spatial variation in the impact of dragonflies and debris on recreational ecosystem services in a floodplain wetland. Ecosyst. Serv. 2015, 15, 113–121. [Google Scholar] [CrossRef]
- Shanahan, D.F.; Lin, B.B.; Gaston, K.J.; Bush, R.; Fuller, R.A. What is the role of trees and remnant vegetation in attracting people to urban parks? Landsc. Ecol. 2015, 30, 153–165. [Google Scholar] [CrossRef]
- Liquete, C.; Kleeschulte, S.; Dige, G.; Maes, J.; Grizzetti, B.; Olah, B.; Zulian, G. Mapping green infrastructure based on ecosystem services and ecological networks: A Pan-European case study. Environ. Sci. Policy 2016, 54, 268–280. [Google Scholar] [CrossRef]
- Pennington, R.T.; Lavin, M.; Oliveira-Filho, A. Woody plant diversity, evolution, and ecology in the tropics: Perspectives from seasonally dry tropical forests. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 437–457. [Google Scholar] [CrossRef]
- Haase, D. Holocene floodplains and their distribution in urban areas—Functionality indicators for their retention potentials. Landsc. Urban Plan. 2003, 66, 5–18. [Google Scholar] [CrossRef]
- Juutinen, A.; Koseniusc, A.K.; Ovaskainen, V. Estimating the benefits of recreation-oriented management in state-owned commercial forests in Finland: A choice experiment. J. For. Econ. 2014, 20, 396–412. [Google Scholar] [CrossRef]
- Bayley, P.B. Understanding large river: Floodplain ecosystems. BioScience 1995, 45, 153–158. [Google Scholar] [CrossRef]
- Barbier, E.B.; Thompson, J.R. The value of water: Floodplain versus large-scale irrigation benefits in northern Nigeria. Ambio 1998, 27, 434–440. [Google Scholar]
- Tockner, K.; Stanford, J.A. Riverine flood plains: Present state and future trends. Environ. Conserv. 2002, 29, 308–330. [Google Scholar] [CrossRef]
- Grygoruk, M.; Mirosław-Świątek, D.; Chrzanowska, W.; Ignar, S. How much for water? Economic assessment and mapping of floodplain water storage as a catchment-scale ecosystem service of wetlands. Water 2013, 5, 1760–1779. [Google Scholar] [CrossRef]
- van Looy, K.; Tormos, T.; Souchon, Y.; Gilvear, D. Analyzing riparian zone ecosystem services bundles to instruct river management. Ecosyst. People 2017, 13, 330–341. [Google Scholar]
- Fitter, A.; Elmqvist, T.; Haines-Young, R.; Potschin, M.; Rinaldo, A.; Setälaä, H.; Stoll-Kleemann, S.; Zobel, M.; Murlis, J. An assessment of ecosystem services and biodiversity in Europe. Environ. Sci. Technol. 2010, 30, 1–28. [Google Scholar]
- Suding, K.N. Toward and area of restoration in ecology: Successes, failures and opportunities ahead. Annu. Rev. Ecol. Evol. Syst. 2011, 42, 465–487. [Google Scholar] [CrossRef]
- Lachowycz, K.; Jones, A.P. Towards a better understanding of the relationship between greenspace and health: Development of a theoretical framework. Landsc. Urban Plan. 2013, 118, 62–69. [Google Scholar] [CrossRef]
- White, M.P.; Alcock, I.; Wheeler, B.W.; Depledge, M.H. Would you be happier living in a greener urban area? A Fixed-effects Analysis of Panel Data. Psychol. Sci. 2013, 24, 920–928. [Google Scholar] [CrossRef] [PubMed]
- Alcock, I.; White, M.P.; Wheeler, B.W.; Fleming, L.E.; Depledge, M.H. Longitudinal effects on mental health of moving to greener and less green urban areas. Environ. Sci. Technol. 2014, 48, 1247–1255. [Google Scholar] [CrossRef] [PubMed]
- Gascon, M.; Triguero-Mas, M.; Martinez, D.; Dadvand, P.; Forns, J.; Plasencia, A. Mental health benefits of long-term exposure to residential green and blue spaces: A systematic review. Int. J. Environ. Res. Public Health 2015, 12, 4354–4379. [Google Scholar] [CrossRef] [PubMed]
- Gascon, M.; Triguero-Mas, M.; Martínez, D. Residential green spaces and mortality: A systematic review. Environ. Int. 2016, 86, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Rupprecht, C.D.D.; Furuya, K. Residents’ perception of informal green space—A case study of Ichikawa City, Japan. Land 2018, 7, 102. [Google Scholar] [CrossRef]
- Sikorski, P.; Wińska-Krysiak, M.; Chormański, J.; Sikorska, D. Low-maintenance green tram tracks as a socially acceptable solution to greening a city. Urban For. Urban Green. 2018, 35, 148–164. [Google Scholar] [CrossRef]
- Charkes, H.; Dukes, J.S. Impacts of invasive species on ecosystem services. In Biological Invasions; Nentwig, W., Ed.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 217–237. [Google Scholar]
- Vilà, M.; Hulme, P.E. Non-native species, ecosystem services, and human well-being. In Impact of Biological Invasions on Ecosystem Services; Vilà, M., Hulme, P.E., Eds.; Springer: Cham, Switzerland, 2017; pp. 1–14. [Google Scholar]
- Sladonja, B.; Poljuha, D.; Uzelac, M. Non-native invasive species as ecosystem service providers. In Ecosystem Services and Global Ecology; Hufnagel, L., Ed.; IntechOpen: London, UK, 2018; pp. 39–59. [Google Scholar]
- Pejchar, L.; Mooney, H.A. Invasive species, ecosystem services and human well-being. Trends Ecol. Evol. 2009, 24, 497–504. [Google Scholar] [CrossRef]
- Antia, R.; Regoes, R.R.; Koella, J.C.; Bergstrom, C.T. The role of evolution in the emergence of infectious diseases. Nature 2003, 426, 658–660. [Google Scholar] [CrossRef]
- Pysek, P.; Richardson, D.M. Invasive species, environmental change and management, and health. Annu. Rev. Environ. Resour. 2010, 35, 25–55. [Google Scholar] [CrossRef]
- Nienhuis, C.; Stout, J. Effectiveness of native bumblebees as pollinators of the alien invasive plant Impatiens glandulifera (Balsaminaceae) in Ireland. J. Pollinat. Ecol. 2009, 1, 1–11. [Google Scholar] [CrossRef]
- Preston, I.R.; le Maitre, D.C.; Blignaut, J.N.; Louw, L.; Palmer, C.G. Impact of invasive alien plants on water provision in selected catchments. Water SA 2018, 44, 719–729. [Google Scholar] [CrossRef]
- Greenwood, P.; Baumann, P.; Pulley, S.; Kuhn, N.J. The invasive alien plant, Impatiens glandulifera (Himalayan Balsam), and increased soil erosion: Causation or association? Case studies from a river system in Switzerland and the UK. J. Soils Sediment 2018, 18, 3463–3477. [Google Scholar] [CrossRef]
- Kapitza, K.; Zimmermann, H.; Martín-López, B.; von Wehrden, H. Research on the social perception of invasive species: A systematic literature review. NeoBiota 2019, 43, 47–68. [Google Scholar] [CrossRef]
- Newig, J.; Fritsch, O. Environmental governance: Participatory, multi-level—And effective? Environ. Policy Gov. 2009, 19, 197–214. [Google Scholar] [CrossRef]
- Schnitzler, J.; Benzler, J.; Altmann, D.; Mucke, I.; Krause, G. Survey on the population’s needs and the public health response during floods in Germany 2002. J. Public Health Manag. Pract. 2007, 13, 461–464. [Google Scholar] [CrossRef]
- Höfle, R.; Dullinger, S.; Essl, F. Different factors affect the local distribution, persistence and spread of alien tree species in floodplain forests. Basic Appl. Ecol. 2014, 15, 426–434. [Google Scholar] [CrossRef]
- Dyderski, M.K.; Gdula, A.K.; Jagodziński, A.M. “The rich get richer” concept in riparian woody species—A case study of the Warta River Valley (Poznan, Poland). Urban For. Urban Green. 2015, 14, 107–114. [Google Scholar] [CrossRef]
- Rieger, I.; Kowarik, I.; Cherubini, P.; Cierjacks, A. A novel dendrochronological approach reveals drivers of carbon sequestration in tree species of riparian forests across spatiotemporal scales. Sci. Total Environ. 2017, 574, 1261–1275. [Google Scholar] [CrossRef]
- Foster, J.; Sandberg, L. Friends or foe? Invasive species and public green space in Toronto. Geogr. Rev. 2004, 94, 178–198. [Google Scholar] [CrossRef]
- Shackleton, R.T.; Richardson, D.M.; Shackleton, C.M.; Bennett, B.; Crowley, S.L.; Dehnen-Schmutz, K.; Estevez, R.A.; Fischer, A.; Kueffer, C.; Kull, C.A.; et al. Explaining people’s perceptions of invasive alien species: A conceptual framework. J. Environ. Manag. 2018, 229, 10–26. [Google Scholar] [CrossRef]
- Potgieter, L.J.; Gaertner, M.; O’Farrell, P.J.; Richardson, D.M. Perceptions of impact: Invasive alien plants in the urban environment. J. Environ. Manag. 2018, 229, 76–87. [Google Scholar] [CrossRef]
- Hoyle, H.; Hitchmough, J.; Jorgensen, A. Attractive, climate-adapted and sustainable? Public perception of non-native planting in the designed urban landscape. Landsc. Urban Plan. 2017, 164, 49–63. [Google Scholar] [CrossRef]
- Sikorska, D.; Sikorski, P.; Richard, H.J. High biodiversity of green infrastructure does not contribute to recreational ecosystem services. Sustainability 2017, 9, 334. [Google Scholar] [CrossRef]
- Szymanowski, T. Kiedy zostały wprowadzone obce gatunki drzew do uprawy w Polsce? Rocz. Sekc. Dendrol. PTB 1960, 14, 81–99. [Google Scholar]
- Kowarik, I. Einführung und Ausbreitug Nichteinheimischer Gehölzarten in Berlin und Brandenburg; Botanischer Verein von Berlin und Brandenburg e.V.: Berlin, Germany, 1992. [Google Scholar]
- Pyšek, P.; Chytrý, M.; Pergl, J.; Sádlo, J.; Wild, J. Plant invasions in the Czech Republic: Current state, introduction dynamics, invasive species and invaded habitats. Preslia 2012, 84, 575–629. [Google Scholar]
- Tokarska-Guzik, B.; Dajdok, Z.; Zając, M.; Zając, A.; Urbisz, A.; Danielewicz, W.; Hołdyński, C. Plants of Foreign Origin in Poland with Particular Emphasis on Invasive Species; GDOŚ: Warszawa, Poland, 2012; pp. 1–197. (In Polish) [Google Scholar]
- Tutin, T.G.; Heywood, V.H.; Burges, N.A.; Moore, D.M.; Valentine, D.H.; Walters, S.M.; Webb, D.A. Flora Europaea; Cambridge University Press: Cambridge, UK, 2010; Volume 2, pp. 1–500. [Google Scholar]
- Gudžinskas, Z. Conspectus of alien plant species of Lithuania. 8. Aceraceae, Balsaminaceae, Elaeagnaceae, Geraniaceae, Hippocastanaceae, Linaceae, Lythraceae, Onagraceae, Oxalidaceae, Rutaceae, and Vitaceae. Bot. Lith. 1998, 4, 363–377. [Google Scholar]
- Mędrzycki, P. NOBANIS—Invasive Alien Species Fact Sheet—Acer negundo. 2011. Available online: https://www.nobanis.org/globalassets/speciesinfo/a/acer-negundo/acer_negundo.pdf (accessed on 10 October 2019).
- Zisenis, M. Alien plant species: A real fear for urban ecosystems in Europe? Urban Ecosyst. 2015, 18, 355–370. [Google Scholar] [CrossRef]
- Degórska, B.; Degórski, M. Green infrastructure as a very important quality factor in urban areas—Warsaw case study. Europa XXI 2017, 32, 51–70. [Google Scholar] [CrossRef]
- Sikorski, P.; Parafjańczuk, S.; Wierzba, M.; Sikorska, D.; Borowski, J.; Kosić, I.V. The phenomenon of illegal dispersion in riparian forests under high tourist pressure. In Problems of Water Management in Forest, Urban and Non-Urbanized Areas; Kałuża, T., Strzeliński, P., Eds.; Wydawnictwo Naukowe Bogucki: Poznań, Poland, 2014; pp. 131–144. (In Polish) [Google Scholar]
- Matuszkiewicz, W.; Sikorski, P.; Szwed, W.; Wierzba, M.; Danielewicz, W.; Wysocki, C.; Kiciński, P. Vegetation of Poland. Illustrated Guide: Forests and Shrubs; PWN: Warszawa, Poland, 2012; pp. 189–207. (In Polish) [Google Scholar]
- Leuschner, C.; Ellenberg, H. Vegetation Ecology of Central Europe, 6th ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 652–688. [Google Scholar]
- Xue, J.; Su, B. Significant remote sensing vegetation indices: A review of developments and applications. J. Sens. 2017, 2017, 13536911. [Google Scholar] [CrossRef]
- Carlson, T.; Ripley, D. On the relationship between NDVI, fractional vegetation cover, and leaf area index. Remote Sens. Environ. 1997, 62, 241–252. [Google Scholar] [CrossRef]
- Coppes, J.; Braunisch, V. Managing visitors in nature areas: Where do they leave the trails? A spatial model. Wildl. Biol. 2013, 19, 1–12. [Google Scholar] [CrossRef]
- Abbott, A. City living marks the brain. Nature 2011, 474, 429. [Google Scholar] [CrossRef] [PubMed]
- Arnberger, A.; Schneider, I.E.; Ebenberger, M.; Eder, R.; Venette, R.C.; Snyder, S.A.; Gobster, P.H.; Choi, A.; Cottrell, S. Emerald ash borer impacts on visual preferences for urban forest recreation settings. Urban For. Urban Green. 2017, 27, 235–245. [Google Scholar] [CrossRef]
- Kaplan, R.; Kaplan, S. The Experience of Nature: A Psychological Perspective; Cambridge University Press: New York, NY, USA, 1989; pp. 1–368. [Google Scholar]
- Schnitzler, A.; Hale, B.W.; Alsum, E.M. Examining native and exotic species diversity in European riparian forests. Biol. Conserv. 2007, 138, 146–156. [Google Scholar] [CrossRef]
- Kowarik, I. Novel urban ecosystems, biodiversity and conservation. Environ. Pollut. 2011, 159, 1974–1983. [Google Scholar] [CrossRef]
- Krevš, A.; Kučinskiene, A. Influence of invasive Acer negundo leaf litter on benthic microbial abundance and activity in the littoral zone of a temperate river in Lithuania. Knowl. Manag. Aquat. Ecosyst. 2017, 418, 26. [Google Scholar] [CrossRef][Green Version]
- Marozas, V.; Cekstere, G.; Laivins, M.; Straigyte, L. Comparison of neophyte communities of Robinia pseudoacacia L. and Acer negundo L. in the eastern Baltic Sea region cities of Riga and Kaunas. Urban For. Urban Green. 2015, 14, 826–834. [Google Scholar] [CrossRef]
- Säumel, I.; Kowarik, I. Urban rivers as dispersal corridors for primarily wind-dispersed invasive tree species. Landsc. Urban Plan. 2010, 94, 244–249. [Google Scholar] [CrossRef]
- Kostina, M.V.; Minkova, N.O.; Yasinskaya, O.I. Some biological features of Acer negundo L. in green plantations of Moscow. Russ. J. Biol. Invasions 2014, 5, 21–28. [Google Scholar] [CrossRef]
- Sunga, C.Y.; Li, M.-H.; Rogers, G.O.; Volder, A.; Wang, Z. Investigating alien plant invasion in urban riparian forests in a hot and semi-arid region. Landsc. Urban Plan. 2011, 100, 278–286. [Google Scholar] [CrossRef]
- Pyšek, P.; Jarošík, V.; Hulme, P.E.; Pergl, J.; Hejda, M.; Schaffner, U.; Vilà, M. A global assessment of invasive plant impacts on resident species, communities and ecosystems: The interaction of impact measures, invading species’ traits and environment. Glob. Chang. Biol. 2012, 18, 1725–1737. [Google Scholar]
- Dyakov, N.; Zhelev, P. Alien species invasion and diversity of riparian forest according to environmental gradients and disturbance regime. Appl. Ecol. Environ. Res. 2013, 11, 249–272. [Google Scholar] [CrossRef]
- Saccone, P.; Girel, J.; Pages, J.P.; Brun, J.J.; Michalet, R. Ecological resistance to Acer negundo invasion in a European riparian forest: Relative importance of environmental and biotic drivers. Appl. Veg. Sci. 2013, 16, 184–192. [Google Scholar] [CrossRef]
- Merceron, N.R.; Lamarque, L.J.; Delzon, S.; Porté, A.J. Killing it softly: Girdling as an efficient eco-friendly method to locally remove invasive Acer negundo. Ecol. Rest. 2016, 34, 297–305. [Google Scholar] [CrossRef]
- Saccone, P.; Pagès, J.P.; Girel, J.; Brun, J.J.; Michalet, R. Acer negundo invasion along a successional gradient: Early direct facilitation by native pioneers and late indirect facilitation by conspecifics. New Phytol. 2010, 187, 831–842. [Google Scholar] [CrossRef]
- Pennington, D.N.; Hansel, J.R.; Gorchov, D.L. Urbanization and riparian forest woody communities: Diversity, composition, and structure within a metropolitan landscape. Biol. Conserv. 2010, 143, 182–194. [Google Scholar] [CrossRef]
- Straigytė, L.; Cekstere, G.; Laivins, M.; Marozas, V. The spread, intensity and invasiveness of the Acer negundo in Riga and Kaunas. Dendrobiology 2015, 74, 157–168. [Google Scholar] [CrossRef]
- Vakhlamova, T.; Rusterholz, H.P.; Kamkin, V.; Baur, B. Recreational use of urban and suburban forests affects plant diversity in a Western Siberian city. Urban For. Urban Green. 2016, 17, 92–103. [Google Scholar] [CrossRef]
- Sikorski, P. Influence of the Urban Park Nature on the Floristic Diversity of Undergrowth and Park Lawns; Wyd. Wieś Jutra: Warsaw, Poland, 2013; pp. 1–108. (In Polish) [Google Scholar]
- Mihalič, T. Performance of environmental resources of a tourist destination: Concept and application. J. Travel Res. 2013, 52, 614–630. [Google Scholar] [CrossRef]
- Kenwick, R.A.; Shammin, M.R.; Sullivanc, W.C. Preferences for riparian buffers. Landsc. Urban Plan. 2009, 91, 88–96. [Google Scholar] [CrossRef]
- Gonzalez, E.; Martínez-Fernandez, V.; Shafroth, P.B.; Sher, A.A.; Henry, A.L.; Garofano-Gomez, V.; Corenblit, D. Regeneration of Salicaceae riparian forests in the Northern Hemisphere: A new framework and management tool. J. Environ. Manag. 2018, 218, 374–387. [Google Scholar] [CrossRef]
- Cole, D.N.; Marion, J.L. Recreation impacts in some riparian forests of the Eastern United States. Environ. Manag. 1988, 12, 99–107. [Google Scholar] [CrossRef]
- Bötsch, Y.; Tablado, Z.; Scherl, D.; Kéry, M.; Graf, R.F.; Jenni, L. Effect of recreational trails on forest birds: Human presence matters. Front. Ecol. Evol. 2018, 6, 175. [Google Scholar] [CrossRef]
- Sharp, R.L.; Larson, L.R.; Green, G.T. Factors influencing public preferences for invasive alien species management. Biol. Conserv. 2011, 144, 2097–2104. [Google Scholar] [CrossRef]


| Number of Trunks per Patch | Total Tree Basal Area [cm2/ha] | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Type forest and average age of trees [years] | invasive maples | poplars | willows | invasive maple % in total number of trunks | invasive maples | poplars | willows | invasive maple % in total basal area | NDVI |
| willow forest | 165.0 | 17.8 | 162.3 | 47.8 | 245.9 | 877.6 | 4649.8 | 4.3 | 0.55 |
| poplar forest | 96.8 | 163.9 | 28.3 | 33.5 | 157.1 | 6907.9 | 836.3 | 2.0 | 0.60 |
| p-value | 0.09 | <0.01 * | <0.01 * | 0.01 * | <0.01 * | <0.01 * | <0.01 * | ||
| willow forest | |||||||||
| <10 | 633.1a | 3.9a | 223.6 | 73.6 | 454.5a | 79.0a | 2606.3a | 14.5 | 0.50a |
| 10–20 | 150.1b | 5.3ab | 149.3 | 49.3 | 209.9b | 164.7a | 3898.6ab | 4.9 | 0.52ab |
| 20–30 | 77.4b | 25.3b | 160.2 | 29.4 | 194.9b | 949.9ab | 3838.0ab | 3.9 | 0.55ab |
| >30 | 102.9b | 22.1ab | 152.7 | 37.1 | 239.4b | 1361.6b | 6082.1b | 3.1 | 0.57b |
| p-value | <0.01 * | 0.01 * | 0.71 | <0.01 * | <0.01 * | 0.01 * | 0.01 * | ||
| poplar forest | |||||||||
| <10 | 240.5a | 59.2 | 30.1a | 72.9 | 309.5a | 3402.7 | 1030.4 | 6.5 | 0.59 |
| 10–20 | 200.4ab | 125.1 | 25.1a | 57.2 | 229.0ab | 6341.2 | 524.5 | 3.2 | 0.58 |
| 20–30 | 81.4bc | 175.3 | 50.5b | 26.5 | 101.2b | 7260.5 | 1247.7 | 1.2 | 0.60 |
| >30 | 61.8c | 177.2 | 15.7c | 24.3 | 158.0ab | 7132.9 | 664.4 | 2.0 | 0.61 |
| p-value | <0.01 * | 0.31 | <0.01 * | 0.03 * | 0.44 | 0.40 | 0.72 | ||
| Average | Standard Deviation | r | p-Value | |
|---|---|---|---|---|
| Tree layer [%] | 53.74 | 21.99 | −0.236 | 0.13 |
| Shrubs [%] | 48.93 | 23.32 | 0.708 | <0.01 * |
| Undergrowth [%] | 67.95 | 17.62 | −0.495 | <0.01 * |
| Number of species | 11.44 | 3.87 | −0.439 | <0.01 * |
| Effect | Willow Forest | Poplar Forest | ||
|---|---|---|---|---|
| F | p | F | p | |
| A. negundo basal area | 21.46 | <0.01 * | 19.37 | <0.01 * |
| distance from the main path | 7.59 | 0.01 * | 6.91 | 0.01 * |
| stand age | 1.46 | 0.23 | 0.91 | 0.34 |
| NDVI | 0.00 | 0.96 | 0.00 | 0.96 |
| distance to the nearest entrance | 1.99 | 0.16 | 0.02 | 0.31 |
| distance to the river | 2.55 | 0.11 | 2.70 | 0.10 |
| number of residents in the 500 m buffer | 0.08 | 0.78 | 1.12 | 0.27 |
| all trees basal area | 0.00 | 0.96 | 0.00 | 0.98 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sikorska, D.; Sikorski, P.; Archiciński, P.; Chormański, J.; Hopkins, R.J. You Can’t See the Woods for the Trees: Invasive Acer negundo L. in Urban Riparian Forests Harms Biodiversity and Limits Recreation Activity. Sustainability 2019, 11, 5838. https://doi.org/10.3390/su11205838
Sikorska D, Sikorski P, Archiciński P, Chormański J, Hopkins RJ. You Can’t See the Woods for the Trees: Invasive Acer negundo L. in Urban Riparian Forests Harms Biodiversity and Limits Recreation Activity. Sustainability. 2019; 11(20):5838. https://doi.org/10.3390/su11205838
Chicago/Turabian StyleSikorska, Daria, Piotr Sikorski, Piotr Archiciński, Jarosław Chormański, and Richard J. Hopkins. 2019. "You Can’t See the Woods for the Trees: Invasive Acer negundo L. in Urban Riparian Forests Harms Biodiversity and Limits Recreation Activity" Sustainability 11, no. 20: 5838. https://doi.org/10.3390/su11205838
APA StyleSikorska, D., Sikorski, P., Archiciński, P., Chormański, J., & Hopkins, R. J. (2019). You Can’t See the Woods for the Trees: Invasive Acer negundo L. in Urban Riparian Forests Harms Biodiversity and Limits Recreation Activity. Sustainability, 11(20), 5838. https://doi.org/10.3390/su11205838





