Effects of an Integrated Carbide Slag-Mushroom Dreg-Calcium Superphosphate Amendment on the Stabilization Process of Pb, Cu, Zn and Cd in Contaminated Soils
Abstract
1. Introduction
2. Experimental
2.1. Soil and Binders
2.2. Total Heavy Metal Content Determination
2.3. Selection of Proportion by Box–Behnken Design
2.4. Speciation Identification of Heavy Metals
3. Results and Discussion
3.1. Proportion Optimization of CS–MD–CSP Integration Amendments
3.1.1. Model Fitting and Analysis of Variance (ANOVA)
3.1.2. Prediction of Immobilization Efficiency by RSM Model
3.1.3. Contour Analysis
3.1.4. Validation of the Models
3.2. Evaluation of the Immobilization Ability Using CS–MD–CSP Integration Amendments
3.2.1. BCR Specification Analysis of Targeted Heavy Metals
3.2.2. Recovery Rate of Targeted Metals
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tang, X.; Li, X.; Liu, X.; Hashmi, M.Z.; Xu, J.; Brookes, P.C. Effects of inorganic and organic amendments on the uptake of lead and trace elements by Brassica chinensis grown in an acidic red soil. Chemosphere 2015, 119, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, S.; Xu, X.; Zhong, Q.; Zhang, C.; Jia, Y.; Li, T.; Deng, O.; Li, Y. Heavy metal removal by GLDA washing: Optimization, redistribution, recycling, and changes in soil fertility. Sci. Total. Environ. 2016, 569, 557–568. [Google Scholar] [CrossRef] [PubMed]
- Huber, M.; Welker, A.; Helmreich, B. Critical review of heavy metal pollution of traffic area runoff: Occurrence, influencing factors, and partitioning. Sci. Total. Environ. 2015, 541, 895–919. [Google Scholar] [CrossRef] [PubMed]
- Dominguez, M.T.; Alegre, J.; Madejón, P.; Madejón, E.; Burgos, P.; Cabrera, F.; Marañón, T.; Murillo, J. River banks and channels as hotspots of soil pollution after large-scale remediation of a river basin. Geoderma 2016, 261, 133–140. [Google Scholar] [CrossRef]
- Adrees, M.; Ali, S.; Rizwan, M.; Zia-Ur-Rehman, M.; Ibrahim, M.; Abbas, F.; Farid, M.; Qayyum, M.F.; Irshad, M.K. Mechanisms of silicon-mediated alleviation of heavy metal toxicity in plants: A review. Ecotoxicol. Environ. Saf. 2015, 119, 186–197. [Google Scholar] [CrossRef]
- Lee, S.S.; Lim, J.E.; El-Azeem, S.A.; Choi, B.; Oh, S.E.; Moon, D.H.; Ok, Y.S. Heavy metal immobilization in soil near abandoned mines using eggshell waste and rapeseed residue. Environ. Sci. Pollut. Res. 2013, 20, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Ruley, A.T.; Sharma, N.C.; Sahi, S.V.; Singh, S.R.; Sajwan, K.S. Effects of lead and chelators on growth, photosynthetic activity and Pb uptake in Sesbania drummondii grown in soil. Environ. Pollut. 2006, 144, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, N.C.; Hatzidaki, E.G.; Belivanis, S.; Tzanakakis, G.N.; Tsatsakis, A.M. Lead toxicity update. A brief review. Med. Sci. Monit. 2005, 11, 329–336. [Google Scholar]
- Inaba, T.; Kobayashi, E.; Suwazono, Y.; Uetani, M.; Oishi, M.; Nakagawa, H.; Nogawa, K. Estimation of cumulative cadmium intake causing Itai–itai disease. Toxicol. Lett. 2005, 159, 192–201. [Google Scholar] [CrossRef]
- Huo, X.; Peng, L.; Xu, X.; Zheng, L.; Qiu, B.; Qi, Z.; Zhang, B.; Han, D.; Piao, Z. Elevated Blood Lead Levels of Children in Guiyu, an Electronic Waste Recycling Town in China. Environ. Heal. Perspect. 2007, 115, 1113–1117. [Google Scholar] [CrossRef]
- Mahar, A.; Wang, P.; Li, R.; Zhang, Z. Immobilization of Lead and Cadmium in Contaminated Soil Using Amendments: A Review. Pedosphere 2015, 25, 555–568. [Google Scholar] [CrossRef]
- Clemente, R.; Pardo, T.; Madejón, P.; Madejón, E.; Bernal, M.P. Food byproducts as amendments in trace elements contaminated soils. Food Res. Int. 2015, 73, 176–189. [Google Scholar] [CrossRef]
- Agnello, A.; Bagard, M.; Van Hullebusch, E.; Esposito, G.; Huguenot, D. Comparative bioremediation of heavy metals and petroleum hydrocarbons co-contaminated soil by natural attenuation, phytoremediation, bioaugmentation and bioaugmentation-assisted phytoremediation. Sci. Total. Environ. 2016, 563, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Rijkenberg, M.J.; DePree, C.V. Heavy metal stabilization in contaminated road-derived sediments. Sci. Total. Environ. 2010, 408, 1212–1220. [Google Scholar] [CrossRef] [PubMed]
- Basta, N.; Gradwohl, R.; Snethen, K.; Schroder, J. Chemical Immobilization of Lead, Zinc, and Cadmium in Smelter-Contaminated Soils Using Biosolids and Rock Phosphate. J. Environ. Qual. 2001, 30, 1222. [Google Scholar] [CrossRef]
- Walker, D.J.; Clemente, R.; Bernal, M. Contrasting effects of manure and compost on soil pH, heavy metal availability and growth of Chenopodium album L. in a soil contaminated by pyritic mine waste. Chemosphere 2004, 57, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Xu, R.; Hou, L.; Song, M.; Liu, J. Stabilization/Solidification of Nitrobenzene Contaminated Soil based on Hydrophobilized CaO. Procedia Environ. Sci. 2016, 31, 288–294. [Google Scholar] [CrossRef][Green Version]
- Udovic, M.; Mcbride, M.B. Influence of compost addition on lead and arsenic bioavailability in reclaimed orchard soil assessed using Porcellio scaber bioaccumulation test. J. Hazard. Mater. 2012, 205–206, 144–149. [Google Scholar] [CrossRef]
- Basta, N.; McGowen, S. Evaluation of chemical immobilization treatments for reducing heavy metal transport in a smelter-contaminated soil. Environ. Pollut. 2004, 127, 73–82. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, F.-J.; Meharg, A.A.; Raab, A.; Feldmann, J.; McGrath, S.P. Mechanisms of Arsenic Hyperaccumulation in Pteris vittata. Uptake Kinetics, Interactions with Phosphate, and Arsenic Speciation1. Plant Physiol. 2002, 130, 1552–1561. [Google Scholar] [CrossRef]
- Chlopecka, A.; Adriano, D. Influence of zeolite, apatite and Fe-oxide on Cd and Pb uptake by crops. Sci. Total. Environ. 1997, 207, 195–206. [Google Scholar] [CrossRef]
- Chlopecka, A.; Adriano, D.C. Mimicked In-Situ Stabilization of Metals in a Cropped Soil: Bioavailability and Chemical Form of Zinc. Environ. Sci. Technol. 1996, 30, 3294–3303. [Google Scholar] [CrossRef]
- Cheng, S.-F.; Hseu, Z.-Y. In-Situ Immobilization of Cadmium and Lead by Different Amendments in Two Contaminated Soils. Water Air Soil Pollut. 2002, 140, 73–84. [Google Scholar] [CrossRef]
- Brallier, S.; Harrison, R.B.; Henry, C.L.; Dongsen, X. Liming effects on availability of Cd, Cu, Ni and Zn in a soil amended with sewage sludge 16 years previously. Water Air Soil Pollut. 1996, 86, 195–206. [Google Scholar] [CrossRef]
- Keller, C.; Marchetti, M.; Rossi, L.; Lugon-Moulin, N. Reduction of Cadmium Availability to Tobacco (Nicotiana tabacum) Plants using Soil Amendments in Low Cadmium-contaminated Agricultural Soils: A Pot Experiment. Plant Soil 2005, 276, 69–84. [Google Scholar] [CrossRef]
- Mendez, A.; Gómez, A.; Paz-Ferreiro, J.; Gascó, G. Effects of sewage sludge biochar on plant metal availability after application to a Mediterranean soil. Chemosphere 2012, 89, 1354–1359. [Google Scholar] [CrossRef] [PubMed]
- Forsberg, L.S.; Dan, B.K.; Greger, M.; Ledin, S. Effects of sewage sludge on solution chemistry and plant uptake of Cu in sulphide mine tailings at different weathering stages. Appl. Geochem. 2008, 24, 475–482. [Google Scholar] [CrossRef]
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L. Effects of biochar and greenwaste compost amendments on mobility, bioavailability and toxicity of inorganic and organic contaminants in a multi-element polluted soil. Environ. Pollut. 2010, 158, 2282–2287. [Google Scholar] [CrossRef] [PubMed]
- Kandil, H.; Elkherbawy, M.I.; Ibrahim, S.; Abdelfattah, A.; Elmoez, M.R.A.; Badawy, S.H. Effect of different sources and rates of some organic manure on content of some heavy metals in different soils and plants grown therein: II. Effect on corn plants. Soil Form. Factors Process. Temp. Zone 2012, 11, 9–18. [Google Scholar]
- Wu, L.H.; Li, Z.; Akahane, I.; Liu, L.; Han, C.L.; Makino, T.; Luo, Y.M. Effects of organic amendments on Cd, Zn and Cu bioavailability in soil with repeated phytoremediation by Sedum plumbizincicola. Intern. J. Phytoreme. 2012, 14, 1024–1038. [Google Scholar] [CrossRef]
- Iżewska, A. The impact of manure, municipal sewage sludge and compost prepared from municipal sewage sludge on crop yield and content of Mn, Zn, Cu, Ni, Pb, Cd in spring rape and spring triticale. J. Elementol. 2009, 14, 449–456. [Google Scholar]
- Park, J.H.; Choppala, G.K.; Bolan, N.S.; Chung, J.W.; Chuasavathi, T. Biochar reduces the bioavailability and phytotoxicity of heavy metals. Plant Soil 2011, 348, 439–451. [Google Scholar] [CrossRef]
- Weggler-Beaton, K.; McLaughlin, M.J.; Graham, R.D. Salinity increases cadmium uptake by wheat and Swiss chard from soil amended with biosolids. Soil Res. 2000, 38, 37–46. [Google Scholar] [CrossRef]
- Brown, S.L.; Chaney, R.L.; Angle, J.S.; Ryan, J.A. The Phytoavailability of Cadmium to Lettuce in Long-Term Biosolids-Amended Soils. J. Environ. Qual. 1998, 27, 1071. [Google Scholar] [CrossRef]
- Sakanakura, H. Formation and Durability of Dithiocarbamic Metals in Stabilized Air Pollution Control Residue from Municipal Solid Waste Incineration and Melting Processes. Environ. Sci. Technol. 2007, 41, 1717–1722. [Google Scholar] [CrossRef] [PubMed]
- Bolan, N.; Kunhikrishnan, A.; Thangarajan, R.; Kumpiene, J.; Park, J.; Makino, T.; Kirkham, M.B.; Scheckel, K. Remediation of heavy metal(loid)s contaminated soils—To mobilize or to immobilize? J. Hazard. Mater. 2014, 266, 141–166. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, F.A.; Fernandes, H.C.; Pileggi, R.G.; Cincotto, M.A.; John, V.M. Carbide lime and industrial hydrated lime characterization. Powder Technol. 2009, 195, 143–149. [Google Scholar] [CrossRef]
- Yi, Y.; Zheng, X.; Liu, S.; Al-Tabbaa, A. Comparison of reactive magnesia- and carbide slag-activated ground granulated blastfurnace slag and Portland cement for stabilisation of a natural soil. Appl. Clay Sci. 2015, 111, 21–26. [Google Scholar] [CrossRef]
- Wang, R.; Guo, J.; Xu, Y.; Ding, Y.; Shen, Y.; Zheng, X.; Feng, R. Evaluation of silkworm excrement and mushroom dreg for the remediation of multiple heavy metal/metalloid contaminated soil using pakchoi. Ecotoxicol. Environ. Saf. 2016, 124, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Gimeno-García, E.; Andreu, V.; Boluda, R. Heavy metals incidence in the application of inorganic fertilizers and pesticides to rice farming soils. Environ. Pollut. 1996, 92, 19–25. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Hua, F.; Su, M.; Zhao, Y. Trans-membrane transport of fluoranthene by Rhodococcus sp. BAP-1 and optimization of uptake process. Bioresour. Technol. 2014, 155, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Lundstedt, T.; Seifert, E.; Abramo, L.; Thelin, B.; Nyström, Å.; Pettersen, J.; Bergman, R. Experimental design and optimization. Chemom. Intell. Lab. Syst. 1998, 42, 3–40. [Google Scholar] [CrossRef]
- Wang, F.-H.; Zhang, F.; Chen, Y.-J.; Gao, J.; Zhao, B. A comparative study on the heavy metal solidification/stabilization performance of four chemical solidifying agents in municipal solid waste incineration fly ash. J. Hazard. Mater. 2015, 300, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Nemati, K.; Abu Bakar, N.K.; Abas, M.R.; Sobhanzadeh, E. Speciation of heavy metals by modified BCR sequential extraction procedure in different depths of sediments from Sungai Buloh, Selangor, Malaysia. J. Hazard. Mater. 2011, 192, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Ghevariya, C.M.; Bhatt, J.K.; Dave, B.P. Enhanced chrysene degradation by halotolerant Achromobacter xylosoxidans using Response Surface Methodology. Bioresour. Technol. 2011, 102, 9668–9674. [Google Scholar] [CrossRef] [PubMed]
- Kostić, M.D.; Bazargan, A.; Stamenković, O.S.; Veljković, V.B.; McKay, G. Optimization and kinetics of sunflower oil methanolysis catalyzed by calcium oxide-based catalyst derived from palm kernel shell biochar. Fuel 2016, 163, 304–313. [Google Scholar] [CrossRef]
- Mohammadi, R.; Mohammadifar, M.A.; Mortazavian, A.M.; Rouhi, M.; Ghasemi, J.B.; Delshadian, Z. Extraction optimization of pepsin-soluble collagen from eggshell membrane by response surface methodology (RSM). Food Chem. 2016, 190, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Wu, Z.; Zhou, J.; Zhao, J.; Ruan, X.; Liu, J.; Qian, G. Chemical characteristics and risk assessment of typical municipal solid waste incineration (MSWI) fly ash in China. J. Hazard. Mater. 2013, 261, 269–276. [Google Scholar] [CrossRef]
- Branzini, A.; Zubillaga, M.S. Comparative Use of Soil Organic and Inorganic Amendments in Heavy Metals Stabilization. Appl. Environ. Soil Sci. 2012, 2012, 1–7. [Google Scholar] [CrossRef]
- Feng, N.-C.; Guo, X.-Y. Characterization of adsorptive capacity and mechanisms on adsorption of copper, lead and zinc by modified orange peel. Trans. Nonferrous Met. Soc. China 2012, 22, 1224–1231. [Google Scholar] [CrossRef]
- Cao, X.; Ma, L.Q.; Rhue, D.R.; Appel, C.S. Mechanisms of lead, copper, and zinc retention by phosphate rock. Environ. Pollut. 2004, 131, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Ning, X.-A.; Liao, X.; Lin, M.; Liu, J.; Wang, J. Characterization and environmental risk assessment of heavy metals found in fly ashes from waste filter bags obtained from a Chinese steel plant. Ecotoxicol. Environ. Saf. 2013, 95, 130–136. [Google Scholar] [CrossRef] [PubMed]
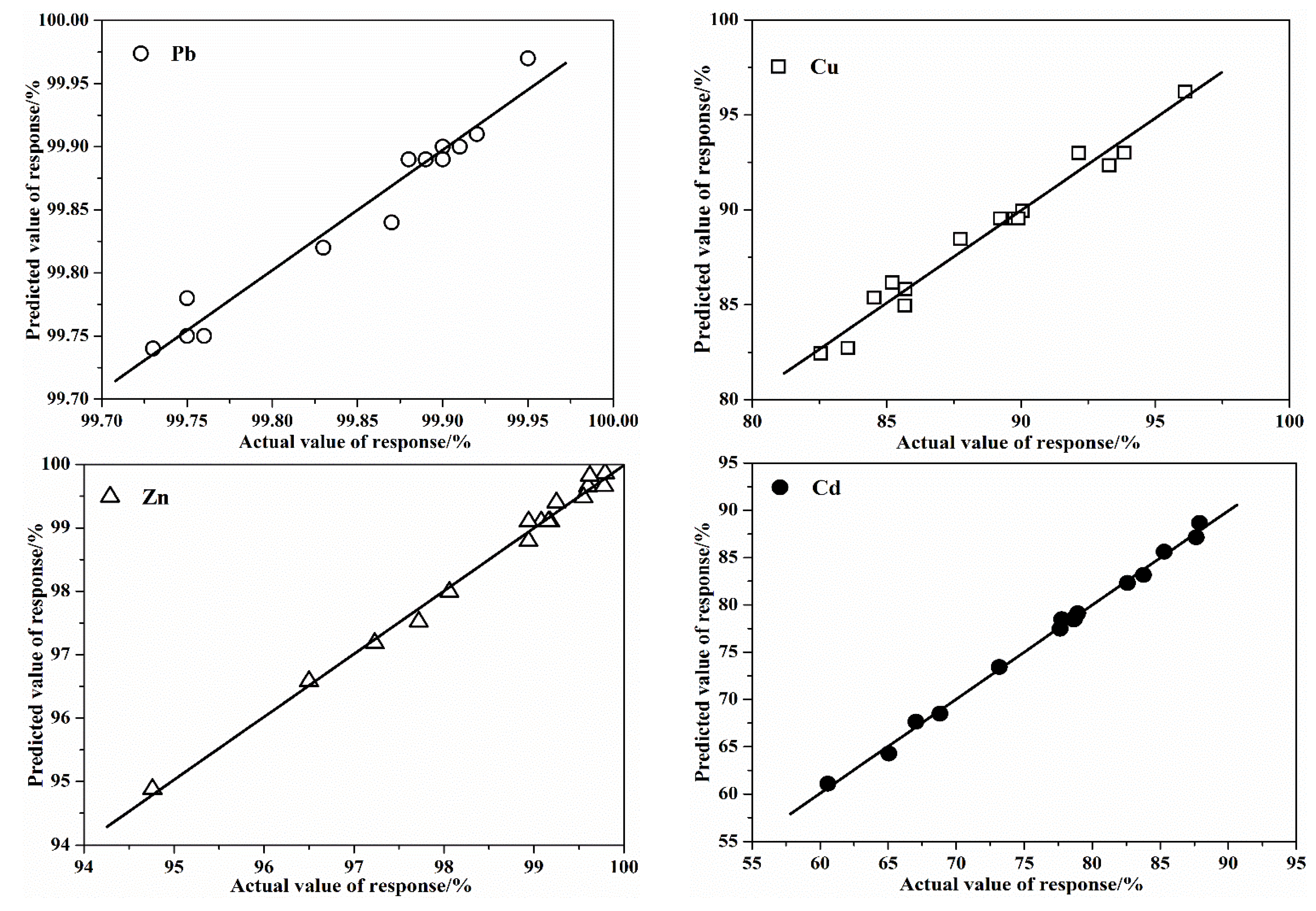
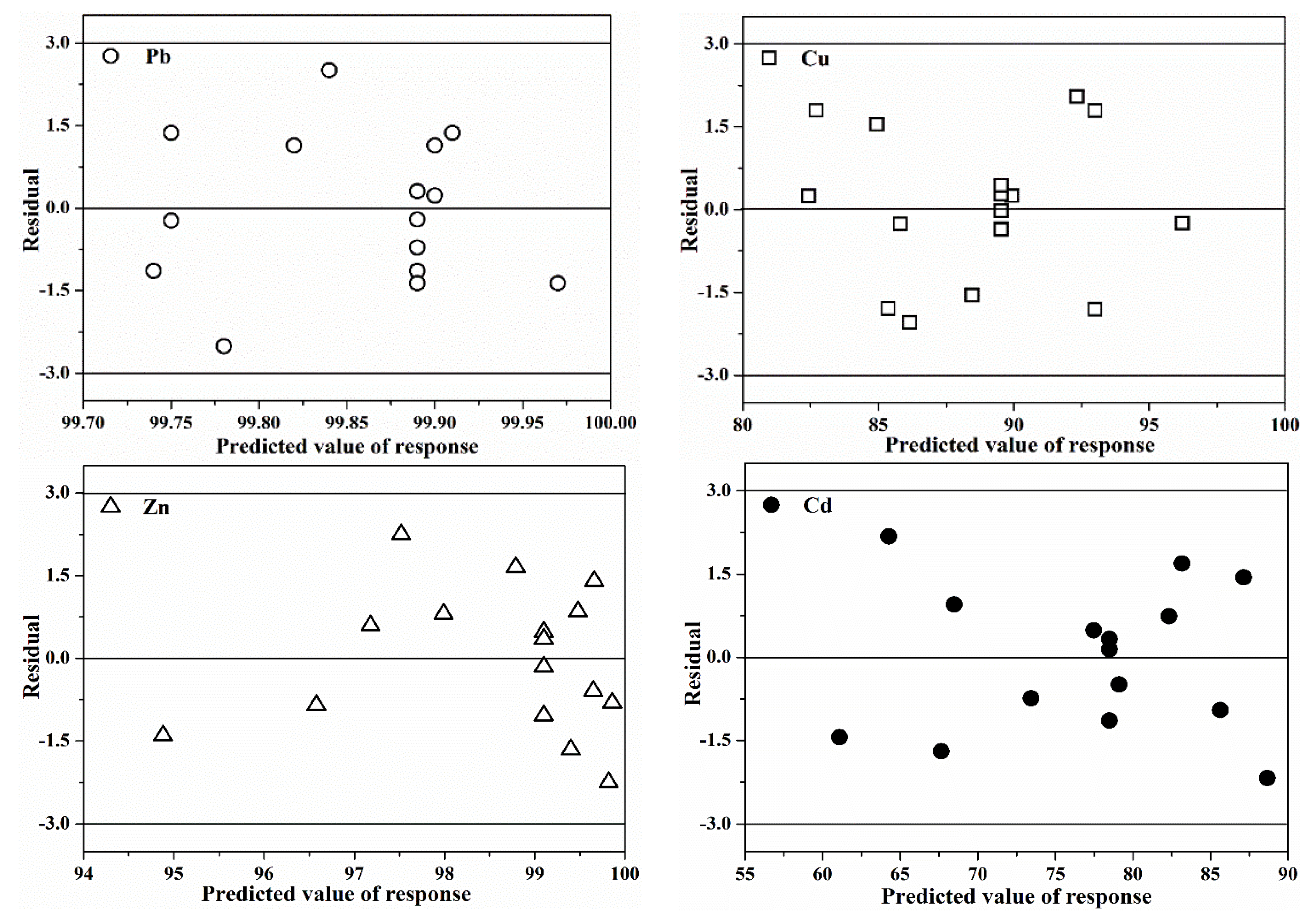
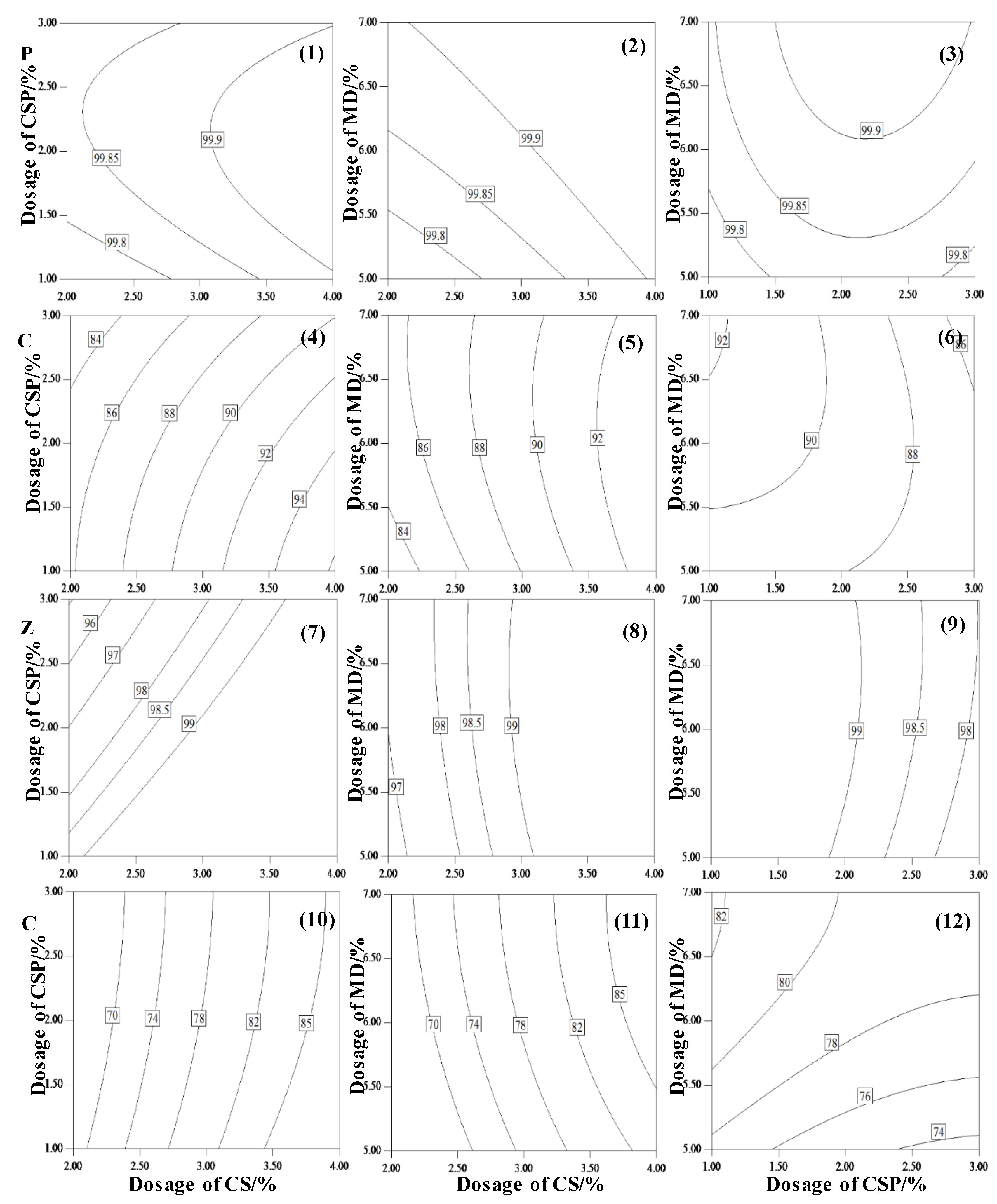
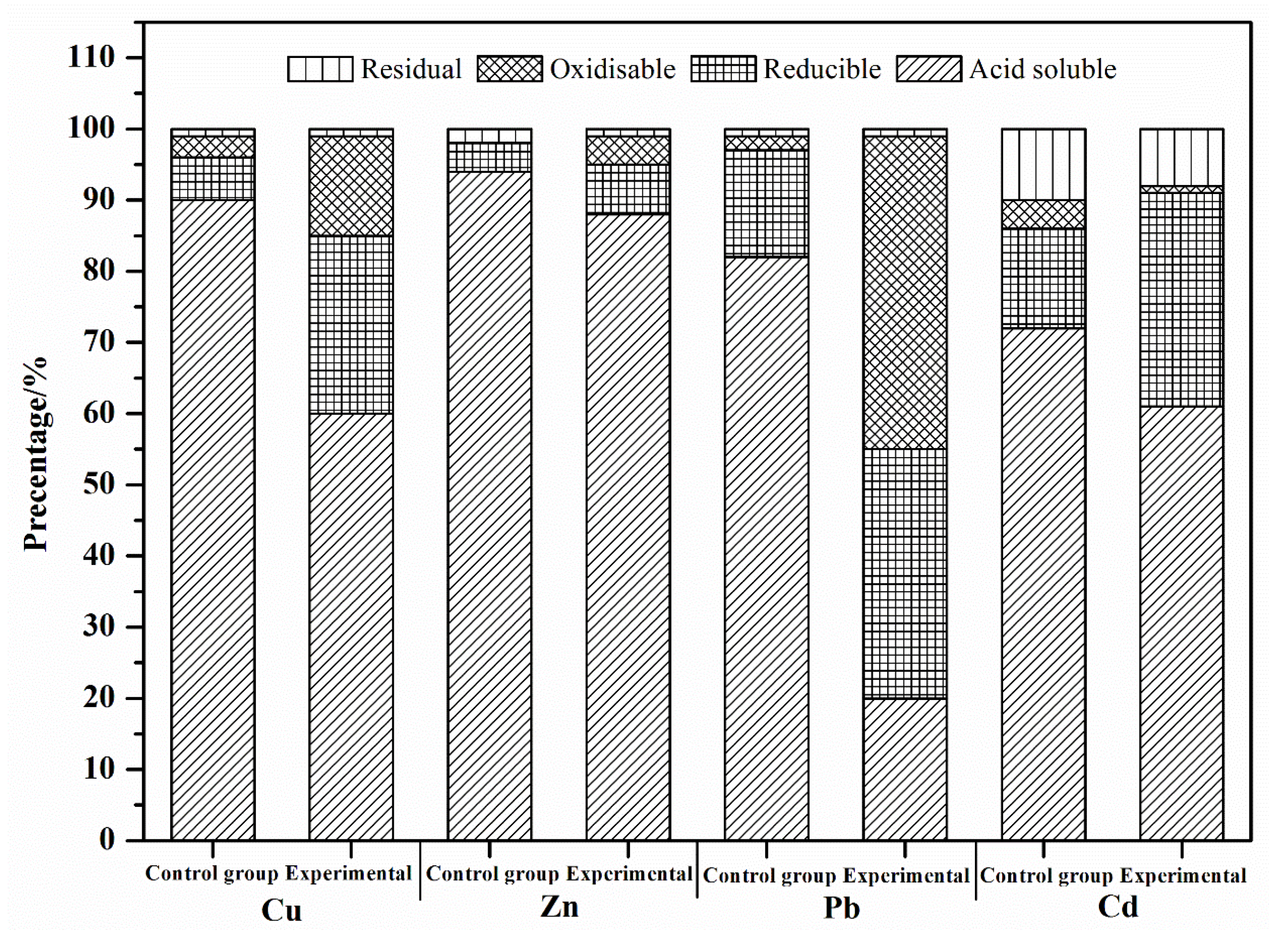
| Parameters | pH | Total Heavy Metal Content/(mg·kg−1 DW *) | |||
|---|---|---|---|---|---|
| Zn | Cu | Pb | Cd | ||
| Fresh soil | 5.22 | 74.8 | 38.1 | 45.3 | 0.77 |
| Experimental soil | 3.97 | 5040 | 3600 | 4080 | 49.6 |
| Materials | CaO | SiO2 | Al2O3 | FeO | Fe2O3 | S | P2O5 | MgO | K2O | Loss on Ignition |
|---|---|---|---|---|---|---|---|---|---|---|
| /% | 67.8 | 3.51 | 2.06 | 0.55 | 0.36 | 0.05 | <0.05 | 0.074 | <0.01 | 25.4 |
| Parameters | pH | Heavy Metals’ Leaching Toxicity Concentrations/(mg·L−1) | |||
|---|---|---|---|---|---|
| Zn | Cu | Pb | Cd | ||
| Experimental soil | 3.97 | 600 | 380 | 398 | 5.33 |
| CS | 8.84 | - | 0.031 | - | 0.002 |
| Mushroom dreg (MD) | 6.82 | 0.038 | 0.065 | 0.016 | 0.002 |
| Calcium superphosphate (CSP) | 1.94 | 4.73 | 1.02 | 0.055 | 0.127 |
| No. | Variables | Level | |||
|---|---|---|---|---|---|
| −1 | 0 | +1 | |||
| X1 | Dosage of CS/(by % weight) | 2 | 3 | 4 | |
| X2 | Dosage of CSP/(by % weight) | 1 | 2 | 3 | |
| X3 | Dosage of MD/(by % weight) | 5 | 6 | 7 | |
| Standard Order | Experimental Values | Immobilization Efficiency/% | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Observed | Predicted | ||||||||||
| X1 | X2 | X3 | Pb | Cu | Zn | Cd | Pb | Cu | Zn | Cd | |
| 1 | 2.00 | 1.00 | 6.00 | 99.7 | 85.7 | 98.9 | 68.8 | 99.7 | 85.8 | 98.8 | 68.5 |
| 2 | 4.00 | 1.00 | 6.00 | 99.9 | 96.1 | 99.8 | 87.9 | 99.9 | 96.2 | 99.7 | 88.7 |
| 3 | 2.00 | 3.00 | 6.00 | 99.8 | 82.5 | 94.8 | 65.1 | 99.8 | 82.4 | 94.9 | 64.3 |
| 4 | 4.00 | 3.00 | 6.00 | 99.9 | 90.1 | 99.3 | 85.3 | 99.9 | 89.9 | 99.4 | 85.6 |
| 5 | 2.00 | 2.00 | 5.00 | 99.8 | 83.6 | 96.5 | 60.6 | 99.8 | 82.7 | 96.6 | 61.1 |
| 6 | 4.00 | 2.00 | 5.00 | 99.9 | 93.8 | 99.6 | 83.8 | 99.9 | 93.0 | 99.7 | 83.2 |
| 7 | 2.00 | 2.00 | 7.00 | 99.9 | 84.5 | 97.2 | 67.1 | 99.9 | 85.4 | 97.2 | 67.7 |
| 8 | 4.00 | 2.00 | 7.00 | 100.0 | 92.2 | 99.6 | 87.6 | 100.0 | 93.0 | 99.5 | 87.1 |
| 9 | 3.00 | 1.00 | 5.00 | 99.8 | 87.7 | 99.8 | 77.6 | 99.8 | 88.5 | 99.9 | 77.5 |
| 10 | 3.00 | 3.00 | 5.00 | 99.8 | 85.2 | 97.7 | 73.2 | 99.8 | 86.2 | 97.5 | 73.4 |
| 11 | 3.00 | 1.00 | 7.00 | 99.9 | 93.3 | 99.6 | 82.6 | 99.8 | 92.3 | 99.8 | 82.3 |
| 12 | 3.00 | 3.00 | 7.00 | 99.9 | 85.7 | 98.1 | 78.9 | 99.9 | 85.0 | 98.0 | 79.1 |
| 13 | 3.00 | 2.00 | 6.00 | 99.9 | 89.5 | 99.2 | 78.6 | 99.9 | 89.5 | 99.1 | 78.5 |
| 14 | 3.00 | 2.00 | 6.00 | 99.9 | 89.8 | 99.1 | 78.7 | 99.9 | 89.5 | 99.1 | 78.5 |
| 15 | 3.00 | 2.00 | 6.00 | 99.9 | 89.3 | 99.2 | 78.7 | 99.9 | 89.5 | 99.1 | 78.5 |
| 16 | 3.00 | 2.00 | 6.00 | 99.9 | 89.2 | 99.2 | 78.7 | 99.9 | 89.5 | 99.1 | 78.5 |
| 17 | 3.00 | 2.00 | 6.00 | 99.9 | 89.9 | 98.9 | 77.8 | 99.9 | 89.5 | 99.1 | 78.5 |
| Factor | df | Sum of Squares | F-Value | p-Value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pb | Cu | Zn | Cd | Pb | Cu | Zn | Cd | Pb | Cu | Zn | Cd | ||
| Model | 9 | 0.071 | 227 | 29.5 | 982 | 16.5 | 28.9 | 105 | 214 | 0.0006 | 0.0001 | <0.0001 | <0.0001 |
| X1 | 1 | 0.026 | 161 | 14.5 | 863 | 54.9 | 184 | 462 | 1690 | 0.0001 | <0.0001 | <0.0001 | <0.0001 |
| X2 | 1 | 3.20 × 10−3 | 46.8 | 8.69 | 26.2 | 6.65 | 53.7 | 278 | 51.3 | 0.0366 | 0.0002 | <0.0001 | 0.0002 |
| X3 | 1 | 0.022 | 3.50 | 0.090 | 55.4 | 45.8 | 4.01 | 2.89 | 109 | 0.0003 | 0.0852 | 0.1330 | <0.0001 |
| X1×2 | 1 | 1.23 × 10−3 | 2.12 | 3.33 | 0.35 | 2.54 | 2.43 | 107 | 0.68 | 0.155 | 0.163 | <0.0001 | 0.436 |
| X1X3 | 1 | 2.03 × 10−3 | 1.78 | 0.15 | 1.68 | 4.21 | 2.05 | 4.87 | 3.29 | 0.0794 | 0.196 | 0.0632 | 0.113 |
| X2X3 | 1 | 2.25 × 10−4 | 6.45 | 0.065 | 0.17 | 0.47 | 7.40 | 2.08 | 0.34 | 0.516 | 0.0297 | 0.193 | 0.579 |
| X12 | 1 | 1.29 × 10−5 | 0.16 | 2.37 | 26.9 | 0.027 | 0.19 | 75.7 | 52.7 | 0.875 | 0.678 | <0.0001 | 0.0002 |
| X22 | 1 | 0.014 | 2.30 | 0.12 | 2.68 | 29.7 | 2.64 | 3.98 | 5.26 | 0.0010 | 0.149 | 0.0861 | 0.0554 |
| X32 | 1 | 1.40 × 10−3 | 2.83 | 0.076 | 6.10 | 2.91 | 3.25 | 2.44 | 12.0 | 0.132 | 0.115 | 0.163 | 0.0106 |
| Residual | 7 | 3.37 × 10−3 | 6.10 | 0.22 | 3.57 | ||||||||
| Lack of fit | 3 | 3.05 × 10−3 | 5.74 | 0.18 | 2.90 | 12.7 | 21.1 | 6.05 | 5.74 | 0.0164 | 0.0065 | 0.0573 | 0.0623 |
| Pure error | 4 | 3.20 × 10−3 | 0.36 | 0.040 | 0.67 | ||||||||
| Total | 16 | 0.075 | 233 | 29.7 | 986 | ||||||||
| Pb: R2 = 0.955, R2adj. = 0.897, C.V. (%) = 0.022, Adeq Precision = 13.2. | Cu: R2 = 0.974, R2adj. = 0.940, C.V. (%) = 1.05, Adeq Precision = 19.3. | ||||||||||||
| Zn: R2 = 0.993, R2adj. = 0.983, C.V. (%) = 0.180, Adeq Precision = 36.7. | Cd: R2 = 0.996, R2adj. = 0.992, C.V. (%) = 0.930, Adeq Precision = 50.3. | ||||||||||||
| Fractions | Extraction Fluid | Steps |
|---|---|---|
| Step1—F1 | HOAc (0.11 mol·L−1) | In this step, 32 mL of HOAc was added to 0.8 g of sample in a centrifuge tube and shaken at 25 °C for 16 h. The supernatant was separated and tested. |
| Step2—F2 | NH2OH·HCl (0.11 mol·L−1) | In this step, 32 mL of NH2OH·HCl was added to the residue from Step 1 and shaken at 25 °C for 16 h. The supernatant was separated and tested. |
| Step3—F3 | H2O2 (30%, pH = 2) CH3COONH4 (1 mol·L−1, pH = 2) | In this step, 8 mL of H2O2 was added to the residue in Step 2, digested at 85 °C for 1 h in a water bath until the volume reduced to approximately 2–3 mL. Then, 32 mL of CH3COONH4 was added to the tube and digested with a cover at 85 °C for another 1 h and shaken at 25 °C for 16 h. The supernatant was separated and tested. |
| Step4—F4 | HCl/HNO3 + HClO4 | In this step, the residue from Step 3 was determined by the method mentioned in 2.2. |
| Elements | Total Concentration | F1 | F2 | F3 | F4 | Recovery/% |
|---|---|---|---|---|---|---|
| Pb | 4080 | 948 | 1430 | 1650 | 28.6 | 99.5 |
| Cu | 3600 | 2180 | 899 | 502 | 14.1 | 98.5 |
| Zn | 5040 | 4320 | 398 | 103 | 18.9 | 95.9 |
| Cd | 49.6 | 33.8 | 13.4 | 1.57 | 2.90 | 104 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, L.; Nong, J.; Zeng, D.; Li, Y. Effects of an Integrated Carbide Slag-Mushroom Dreg-Calcium Superphosphate Amendment on the Stabilization Process of Pb, Cu, Zn and Cd in Contaminated Soils. Sustainability 2019, 11, 4957. https://doi.org/10.3390/su11184957
Wu L, Nong J, Zeng D, Li Y. Effects of an Integrated Carbide Slag-Mushroom Dreg-Calcium Superphosphate Amendment on the Stabilization Process of Pb, Cu, Zn and Cd in Contaminated Soils. Sustainability. 2019; 11(18):4957. https://doi.org/10.3390/su11184957
Chicago/Turabian StyleWu, Lieshan, Jiajing Nong, Dongmei Zeng, and Yi Li. 2019. "Effects of an Integrated Carbide Slag-Mushroom Dreg-Calcium Superphosphate Amendment on the Stabilization Process of Pb, Cu, Zn and Cd in Contaminated Soils" Sustainability 11, no. 18: 4957. https://doi.org/10.3390/su11184957
APA StyleWu, L., Nong, J., Zeng, D., & Li, Y. (2019). Effects of an Integrated Carbide Slag-Mushroom Dreg-Calcium Superphosphate Amendment on the Stabilization Process of Pb, Cu, Zn and Cd in Contaminated Soils. Sustainability, 11(18), 4957. https://doi.org/10.3390/su11184957




