Responses of Microbial Communities and Interaction Networks to Different Management Practices in Tea Plantation Soils
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Site and Soil Sampling
2.2. Physicochemical Properties of Soils
2.3. DNA Extraction and Amplicon Sequencing
2.4. Sequence Preprocessing and Bioinformatic Approaches
2.5. Ecological and Statistical Analysis
2.6. Network Analysis
3. Results
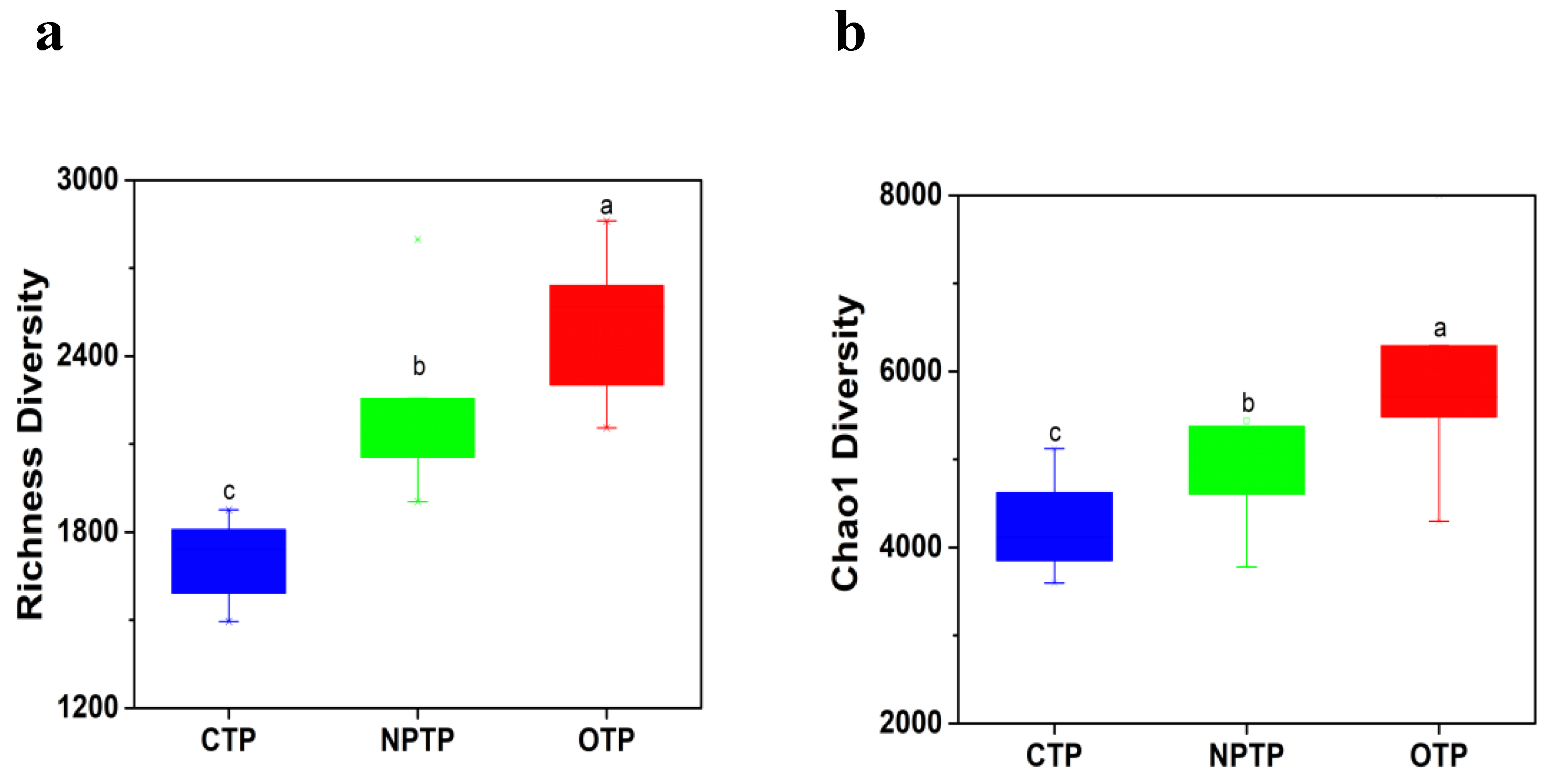
3.1. Microbial Diversity
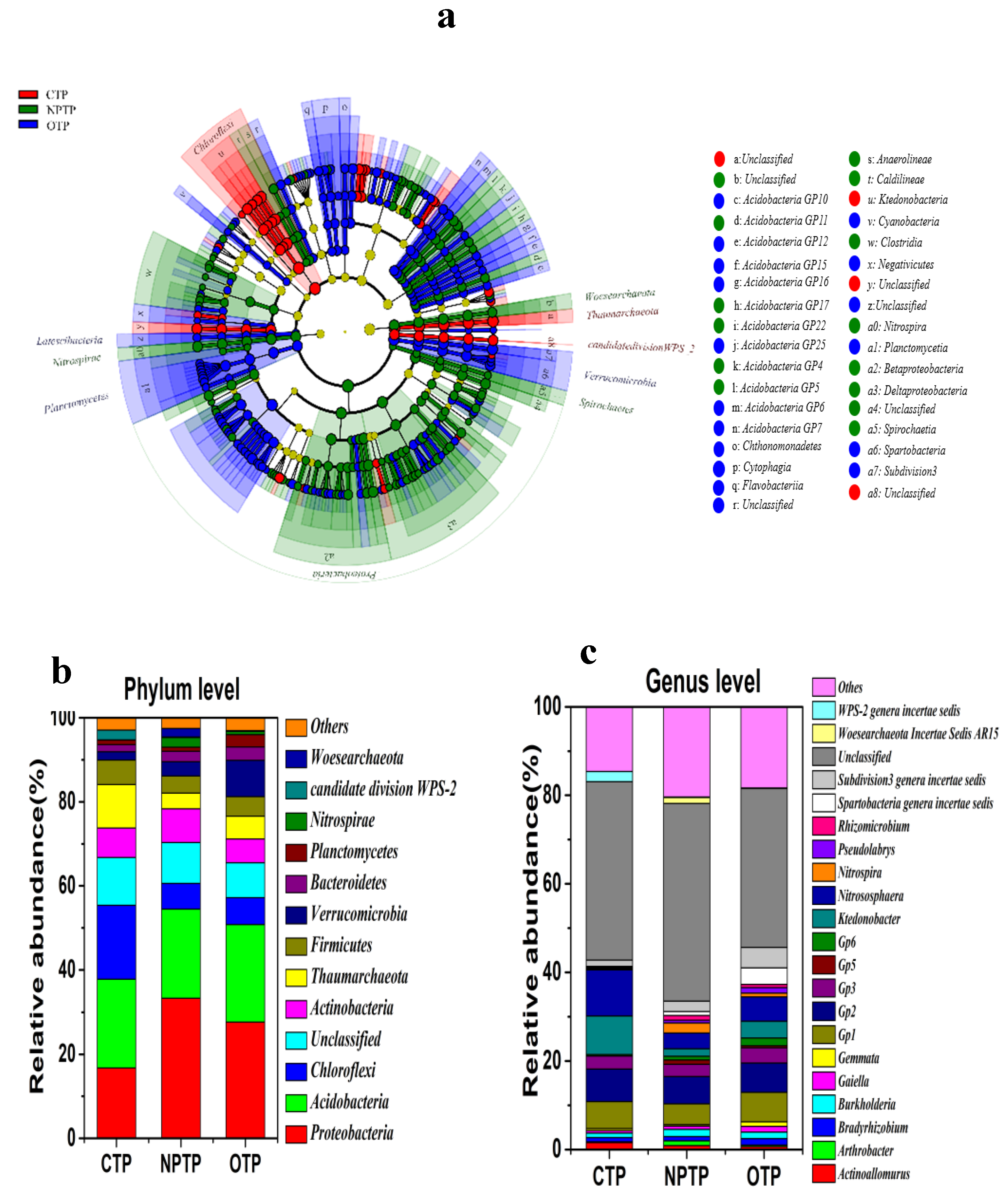
3.2. Microbial Community Composition
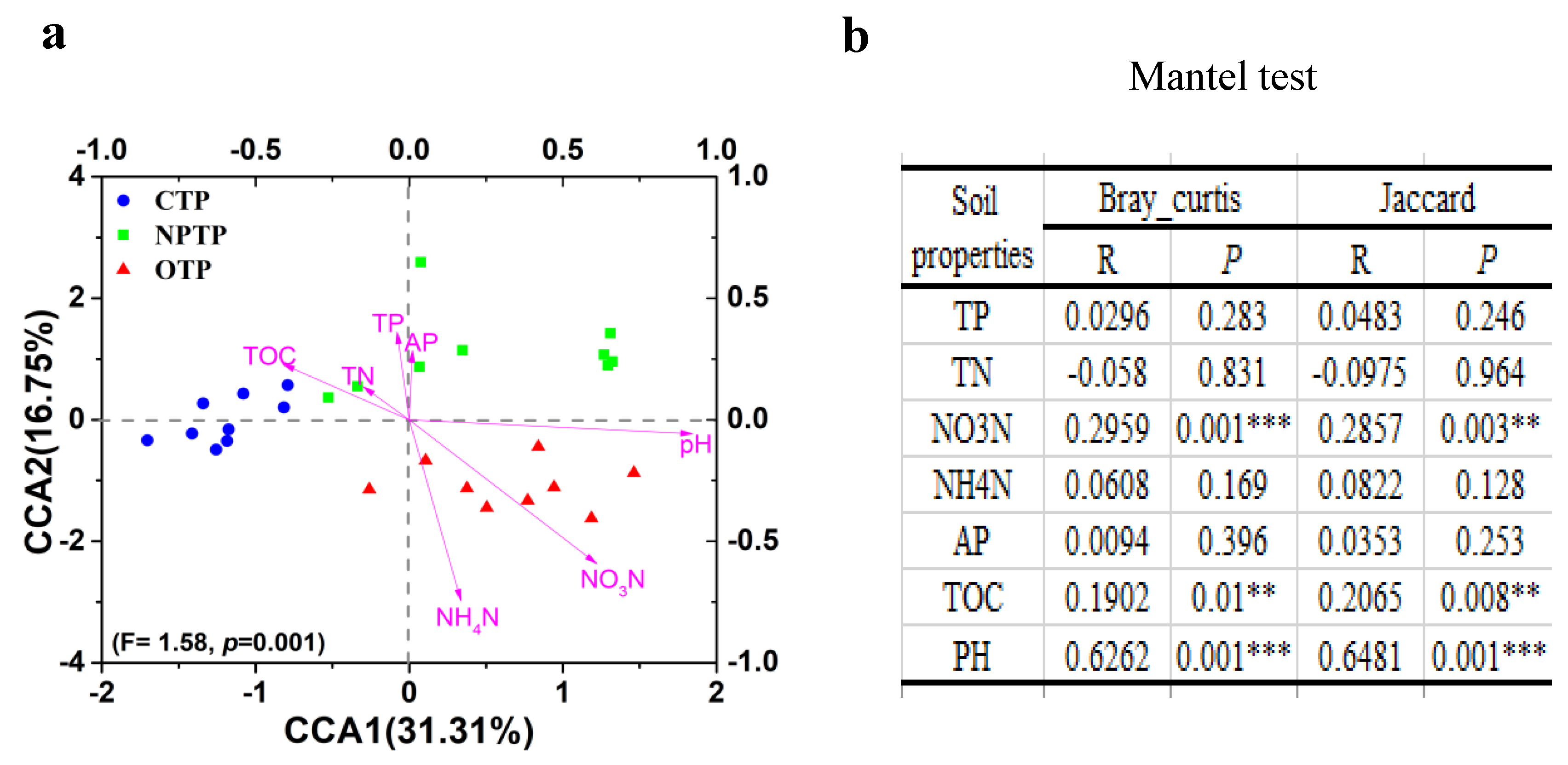
3.3. Correlation Analysis between Soil Properties and Bacterial Community Structures
3.4. Molecular Ecology Networks
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Liu, Z.H.; Gao, L.Z.; Chen, Z.M.; Zeng, X.X.; Huang, J.A.; Gong, Y.S.; Li, Q.; Liu, S.; Lin, Y.; Cai, S.X.; et al. Leading progress on genomics, health benefits and utilization of tea resources in China. Nature 2019, 7742, e566. [Google Scholar]
- Adesemoye, A.O.; Torbert, H.A.; Kloepper, J.W. Plant growth-promoting rhizobacteria allow reduced application rates of chemical fertilizers. Microb. Ecol. 2009, 85, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Savci, S. An agricultural pollutant: Chemical fertilizer international. J. Environ. Sci. Dev. 2012, 3, 77–80. [Google Scholar]
- Nafees, M.; Jan, M.R. Residues of cypermethrin and endosulfan in soils of Swat valley. Soil Environ. 2009, 28, 113–118. [Google Scholar]
- Chowdhury, A.; Pradhan, S.; Saha, M.; Sanya, N. Impact of pesticides on soil microbiological parameters and possible bioremediation strategies. Indian J. Microbiol. 2008, 48, 114–127. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, M.; Frey, B.; Mayer, J.; Maeder, P.; Widmer, F. Distinct soil microbial diversity under long-term organic and conventional farming. ISME J. 2015, 9, 1177–1194. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, C.Y.; Tu, C.; Hoyt, G.D.; DeForest, J.L.; Hu, S.J. Long-term no-tillage and organic input management enhanced the diversity and stability of soil microbial community. Sci. Total Environ. 2017, 609, 341–347. [Google Scholar] [CrossRef]
- Francioli, D.; Schulz, E.; Lentendu, G.; Wubet, T.; Buscot, F.; Reitz, T. Mineral vs. organic amendments: Microbial community structure, activity and abundance of agriculturally relevant microbes are driven by long-term fertilization strategies. Front. Microbiol. 2016, 7, e1446. [Google Scholar] [CrossRef]
- Tuomisto, H.L.; Hodge, I.D.; Riordan, P.; Macdonald, D.W. Does organic farming reduce environmental impacts? A meta-analysis of European research. J. Environ. Manag. 2012, 112, 309–320. [Google Scholar] [CrossRef]
- Xie, S.W.; Feng, H.X.; Yang, F.; Zhao, Z.D.; Hu, X.D.; Wei, C.Y.; Liang, T.; Li, H.T.; Geng, Y.B. Does dual reduction in chemical fertilizer and pesticides improve nutrient loss and tea yield and quality? A pilot study in a green tea garden in Shaoxing, Zhejiang Province, China. Environ. Sci. Pollut. R 2019, 26, 2464–2476. [Google Scholar] [CrossRef]
- Shi, W.C.; Li, M.C.; Wei, G.S.; Tian, R.M.; Li, C.P.; Wang, B.; Lin, R.S.; Shi, C.Y.; Chi, X.L.; Zhou, B.; et al. The occurrence of potato common scab correlates with the community composition and function of the geocaulosphere soil microbiome. Microbiome 2019, 7, e14. [Google Scholar] [CrossRef] [PubMed]
- Raaijmakers, J.M.; Mazzola, M. Soil immune responses. Science 2016, 352, 1392–1393. [Google Scholar] [CrossRef] [PubMed]
- Bisi-Johnson, M.A.; Obi, C.L.; Ekosse, G.E. Microbiological and health related perspectives of geophagia: An overview. Afr. J. Biotechnol. 2010, 9, 5784–5791. [Google Scholar]
- Walters, W.A.; Jin, Z.; Youngblut, N.; Wallace, J.G.; Sutter, J.; Zhang, W.; González-Peña, A.; Peiffer, J.; Koren, O.; Shi, Q.J.; et al. Large-scale replicated field study of maize rhizosphere identifies heritable microbes. Pro. Natl. Acad. Sci. USA 2018, 115, 7368–7373. [Google Scholar] [CrossRef] [PubMed]
- Mendes, L.W.; Raaijmakers, J.M.; De Hollander, M.; Mendes, R.; Tsai, S.M. Influence of resistance breeding in common bean on rhizosphere microbiome composition and function. ISME J. 2018, 12, 212–224. [Google Scholar] [CrossRef] [PubMed]
- Lareen, A.; Burton, F.; Schafer, P. Plant root-microbe communication in shaping root microbiomes. Plant Mol. Biol. 2016, 90, 575–587. [Google Scholar] [CrossRef] [PubMed]
- Schulz, B.J.E.; Boyle, C.J.C.; Sieber, T.N. Microbial Root Endophytes; Springer: Dordercht, NL, USA; Berlin, Germany, 2006. [Google Scholar]
- Mendes, L.W.; Kuramae, E.E.; Navarrete, A.A.; van Veen, J.A.; Tsai, S.M. Taxonomical and functional microbial community selection in soybean rhizosphere. ISME J. 2014, 8, 1577–1587. [Google Scholar] [CrossRef] [PubMed]
- Mendes, R.; Garbeva, P.; Raaijmakersm, J.M. The rhizosphere microbiome: Significance of plant beneficial, plant pathogenic, and human pathogenic microorganisms. Fems Microbiol. Rev. 2013, 37, 634–663. [Google Scholar] [CrossRef]
- Ji, L.F.; Wu, Z.D.; You, Z.M.; Yi, X.Y.; Ni, K.; Guo, S.W.; Ruan, J.Y. Effects of organic substitution for synthetic N fertilizer on soil bacterial diversity and community composition: A 10-year field trial in a tea plantation. Agr. Ecosyst. Environ. 2018, 268, 124–132. [Google Scholar] [CrossRef]
- Wang, S.Q.; Li, T.X.; Zheng, Z.C.; Chen, H.Y.H. Soil aggregate-associated bacterial metabolic activity and community structure in different aged tea plantations. Sci. Total Environ. 2019, 654, 1023–1032. [Google Scholar] [CrossRef]
- Kong, X.; Jin, D.C.; Jin, S.L.; Wang, Z.G.; Yin, H.Q.; Xu, M.Y.; Deng, Y. Responses of bacterial community to dibutyl phthalate (DBP) pollution in a soil-vegetable ecosystem. J. Hazar. Mater. 2018, 353, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Feng, K.; Zhang, Z.J.; Cai, W.W.; Liu, W.Z.; Xu, M.Y.; Yin, H.Q.; Wang, A.J.; He, Z.L.; Deng, Y. Biodiversity and species competition regulate the resilience of microbial biofilm community. Mol. Ecol. 2017, 26, 6170–6182. [Google Scholar] [CrossRef] [PubMed]
- Magoc, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Chao, A. Nonparametric Estimation of the Number of Classes in a Population. Scand. J. Stat. 1984, 11, 265–270. [Google Scholar]
- Lozupone, C.; Knight, R. UniFrac: A New Phylogenetic Method for Comparing Microbial Communities. Appl. Environ. Microbiol. 2005, 71, 8228–8235. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Jiang, Y.H.; Yang, Y.F.; He, Z.L.; Luo, F.; Zhou, J.Z. Molecular ecological network analyses. BMC Bioinformatics 2012, 13, e113. [Google Scholar] [CrossRef]
- Zhou, J.Z.; Deng, Y.; Luo, F.; He, Z.L.; Yang, Y.F. Phylogeneticmolecular ecological network of soil microbial communities inresponse to elevated CO2. MBIO 2011, 2, e00122-11. [Google Scholar] [CrossRef]
- Luo, F.; Yang, Y.F.; Zhong, J.X.; Gao, H.; Khan, L.; Thompson, D.K.; Zhou, J.Z. Constructing gene co-expression networks andpredicting functions of unknown genes by random matrix theory. BMC Bioinformatics 2007, 8, e299. [Google Scholar] [CrossRef]
- Shannon, P.; Markiel, A.; Ozier, O.; Baliga, N.S.; Wang, J.T.; Ramage, D.; Amin, N.; Schwikowski, B.; Ideker, T. Cytoscape: A software environment for integrated models of biomolecular interaction networks. Genome Res. 2003, 13, 2498–2504. [Google Scholar] [CrossRef]
- Newman, M.E.J. Modularity and community structure in networks. Pro. Natl. Acad. Sci. USA 2016, 103, 8577–8582. [Google Scholar] [CrossRef] [PubMed]
- Watts, D.J.; Strogatz, S.H. Collective dynamics of ‘small-world’ networks. Nature 1998, 393, 440–442. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.S.; Gupta, V.K. Soil microbial biomass: A key soil driver in management of ecosystem functioning. Sci. Total Enviro. 2018, 634, 497–500. [Google Scholar] [CrossRef]
- Ji, B.Y.; Hu, H.; Zhao, Y.L.; Mu, X.Y.; Liu, K.; Li, C.H. Effects of deep tillage and straw teturning on soil microorganism and enzyme activities. Sci. World J. 2014, e451493. [Google Scholar]
- Aktar, M.W.; Sengupta, D.; Chowdhury, A. Impact of pesticides use in agriculture: Their benefits and hazards. Interdisc. Toxicol. 2009, 2, 1–12. [Google Scholar] [CrossRef]
- Bilsborrow, P.; Cooper, J.; Tetard-Jones, C.; Srednicka-Tober, D.; Baraniski, M.; Eyre, M.; Schmidt, C.; Shotton, P.; Volakakis, N.; Cakmak, I.; et al. The effect of organic and conventional management on the yield and quality of wheat grown in a long-term field trial. Eur. J. Agron. 2013, 51, 71–80. [Google Scholar] [CrossRef]
- Chin, F.S.; Ho, T.Y.; Chong, K.P.; Jalloh, M.B.; Wong, N.K. Organic versus conventional farming of tea plantation. J. Sci. Biotechnol. 2010, 26, 19–26. [Google Scholar]
- Diacono, M.; Montemurro, F. Long-term effects of organic amendments on soil fertility. A review. Agron. Sustain. Dev. 2010, 30, 401–422. [Google Scholar] [CrossRef]
- Araújo, A.S.F.; Monterio, R.T.R.; Abarkeli, R.B. Effect of glyphosate on the microbial activity of two brazilian soils. Chemosphere 2003, 52, 799–804. [Google Scholar] [CrossRef]
- Devare, M.H.; Jones, C.M.; Thies, J.E. Effect of Cry3B transgenic corn and tefluthrin on the soil microbial community: Biomass, activity and diversity. J. Environ. Qual. 2004, 33, 837–843. [Google Scholar] [CrossRef]
- SeVerstraete, W.; Top, E.M. Effect of long-term herbicide applications on the bacterial community structure and function in an agricultural soil. Fems Microbiol. Ecol. 2003, 46, 139–146. [Google Scholar]
- Gupta, S.; Gupta, R.; Sharma, S. Impact of chemical- and bio-pesticides on bacterial diversity in rhizosphere of Vigna radiata. Ecotoxicology 2013, 22, 1479–1489. [Google Scholar] [CrossRef] [PubMed]
- Lori, M.; Symnaczik, S.; Mäder, P.; De Deyn, G.; Gattinger, A. Organic farming enhances soil microbial abundance and activity–a meta-analysis and meta-regression. PLoS ONE 2017, 12, e018044. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S. Network analysis reveals functional redundancy and key stone taxa amongst bacterial and fungal communities during organic matter decomposition in an arable soil. Soil Biol. Biochem. 2016, 97, 188–198. [Google Scholar] [CrossRef]
- Bhattacharyya, P.; Roy, K.S.; Nayak, A.K.; Shahid, M.; Lal, B.; Gautam, P.; Mohapatra, T. Metagenomic assessment of methane production-oxidation and nitrogen metabolism of long term manured systems in lowland rice paddy. Sci. Total Environ. 2017, 586, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Lücker, S.; Wagner, M.; Maixner, F.; Pelletier, E.; Koch, H.; Vacherie, B.; Ratteie, T.; Damstéf, J.S.S.; Spieckg, E.; Paslier, D.L.; et al. A Nitrospira metagenome illuminates the physiology and evolution of globally important nitrite-oxidizing bacteria. Proc. Natl. Acad. Sci. USA 2010, 107, 13479–13484. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.F.; Dong, J.; Shen, Z.Q.; Mou, R.; Zhou, Y.X.; Chen, X.M.; Fu, X.Y.; Yang, C.P. Nitrogen removal of naerobically digested swine wastewater by pilot-scale tidal flow constructed wetland based on in-situ iological regeneration of zeolite. Chemosphere 2019, 217, 364–373. [Google Scholar] [CrossRef]
- Zhang, X.X.; Zhang, R.J.; Gao, J.S.; Ma, X.T.; Yin, H.Q.; Zhang, C.W.; Feng, K.; Deng, Y. Thirty-one years of rice-ricegreen manure rotations shape the rhizosphere microbial community and enrich beneficial bacteria. Soil Biol. Biochem. 2017, 104, 208–217. [Google Scholar] [CrossRef]
- Fox, J.E.; Gulledge, J.; Engelhaupt, E.; Burow, M.E.; McLachlan, J.A. Pesticides reduce symbiotic efficiency of nitrogen-fixing rhizobia and host plants. Proc. Natl. Acad. Sci. USA 2007, 104, 10282–10287. [Google Scholar] [CrossRef]
- Vieira, R.F.; Silva, C.M.M.S.; Silveira, A.P.D. Soil microbial biomass C and symbiotic processes associated with soybean after sulfentrazone herbicide application. Plant Soil 2007, 300, 95–103. [Google Scholar] [CrossRef]
- Kampfer, P.; Young, C.C.; Arun, A.B.; Shen, F.T.; Jäckel, U.; Rossello´-Mora, R.; Lai, W.A.; Rekha, P.D. Pseudolabrys taiwanensis gen. nov., sp. nov., an alphaproteobacterium isolated from soil. Int. J. Syst. Evol. Micr. 2006, 56, 2469–2472. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Fu, S.; Mathew, R.P.; Lawrence, K.S.; Feng, Y. Soil microbial community structure and activity in a 100-year-old fertilization and crop rotation experiment. J. Plant Ecol. 2015, 8, 623–632. [Google Scholar] [CrossRef]
- Birkhofer, K.; Bezemer, T.M.; Bloem, J.; Bonkowski, M.; Christensen, S.; Dubois, D.; Ekelund, F.; Fliessbach, A.; Gunst, L.; Hedlund, K.; et al. Long-term organic farming fosters below and aboveground biota: Implications for soil quality, biological control and productivity. Soil Biol. Biochem. 2008, 40, 2297–2308. [Google Scholar] [CrossRef]
- Bulluck, L.R.; Ristaino, J. Effffect of synthetic and organic soil fertility amendments on southern blight, soil microbial communities, and yield of processing tomatoes. Phytopathology 2002, 92, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Bunemann, E.K.; Schwenke, G.D.; Van Zwieten, L. Impact of agricultural inputs on soil organisms a review. Aust. J. Soil Res. 2006, 44, 379–406. [Google Scholar] [CrossRef]
- Singh, B.K.; Walker, A. Microbial degradation of organophosphorus compounds. Fems Microbiol. Rev. 2006, 30, 428–471. [Google Scholar] [CrossRef] [PubMed]
- Barberán, A.; Bates, S.T.; Casamayor, E.O.; Fierer, N. Using network analysis to explore co-occurrence patterns in soil microbial communities. ISME J. 2012, 6, 343–351. [Google Scholar] [CrossRef]
- de Menezes, A.B.; Prendergast-Miller, M.T.; Richardson, A.E.; Toscas, P.; Farrell, M.; Macdonald, L.M.; Baker, G.; Wark, T.; Thrall, P.H. Network analysis reveals that bacteria and fungi form modules that correlate independently with soil parameters. Environ. Microbiol. 2015, 17, 2677–2689. [Google Scholar] [CrossRef]
- Williams, R.J.; Howe, A.; Hofmockel, K. Demonstrating microbial co-occurrence pattern analyses within and between ecosystems. Front. Microbiol. 2014, 5, e358. [Google Scholar] [CrossRef]
- Chen, S.; Qi, G.F.; Luo, T.; Zhang, H.C.; Jiang, Q.K.; Wang, R.; Zhao, X.Y. Continuous-cropping tobacco caused variance of chemical properties and structure of bacterial network in soils. Land Degrad. Dev. 2018, 29, 4106–4120. [Google Scholar] [CrossRef]


| Samples | TP (mg kg−1) | TN (mg kg−1) | NO3-N (mg kg−1) | NH4-N (mg kg−1) | AP (mg kg−1) | TOC (%) | pH |
|---|---|---|---|---|---|---|---|
| CTP | 245.11 a | 1050.22 b | 48.56 c | 10.86 b | 10.72 b | 1.21 b | 4.32 b |
| NPTP | 352.56 a | 1134.56 a | 51.61 b | 4.96 c | 24.97 a | 0.94 c | 5.17 ab |
| OTP | 283.33 a | 1054.44 b | 60.03 a | 23.26 a | 24.06 a | 1.82 a | 5.23 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, L.; Gu, S.; Li, S.; Ren, Z.; Deng, Y.; Liu, Z.; Gong, Z.; Xiao, W.; Hu, Q. Responses of Microbial Communities and Interaction Networks to Different Management Practices in Tea Plantation Soils. Sustainability 2019, 11, 4428. https://doi.org/10.3390/su11164428
Tan L, Gu S, Li S, Ren Z, Deng Y, Liu Z, Gong Z, Xiao W, Hu Q. Responses of Microbial Communities and Interaction Networks to Different Management Practices in Tea Plantation Soils. Sustainability. 2019; 11(16):4428. https://doi.org/10.3390/su11164428
Chicago/Turabian StyleTan, Lin, Songsong Gu, Shi Li, Zuohua Ren, Ye Deng, Zhonghua Liu, Zhihua Gong, Wenjun Xiao, and Qiulong Hu. 2019. "Responses of Microbial Communities and Interaction Networks to Different Management Practices in Tea Plantation Soils" Sustainability 11, no. 16: 4428. https://doi.org/10.3390/su11164428
APA StyleTan, L., Gu, S., Li, S., Ren, Z., Deng, Y., Liu, Z., Gong, Z., Xiao, W., & Hu, Q. (2019). Responses of Microbial Communities and Interaction Networks to Different Management Practices in Tea Plantation Soils. Sustainability, 11(16), 4428. https://doi.org/10.3390/su11164428





