Impact of Leachate from Northern Landfill Site in Bloemfontein on Water and Soil Quality: Implications for Water and Food Security
Abstract
1. Introduction
2. Materials and Methods
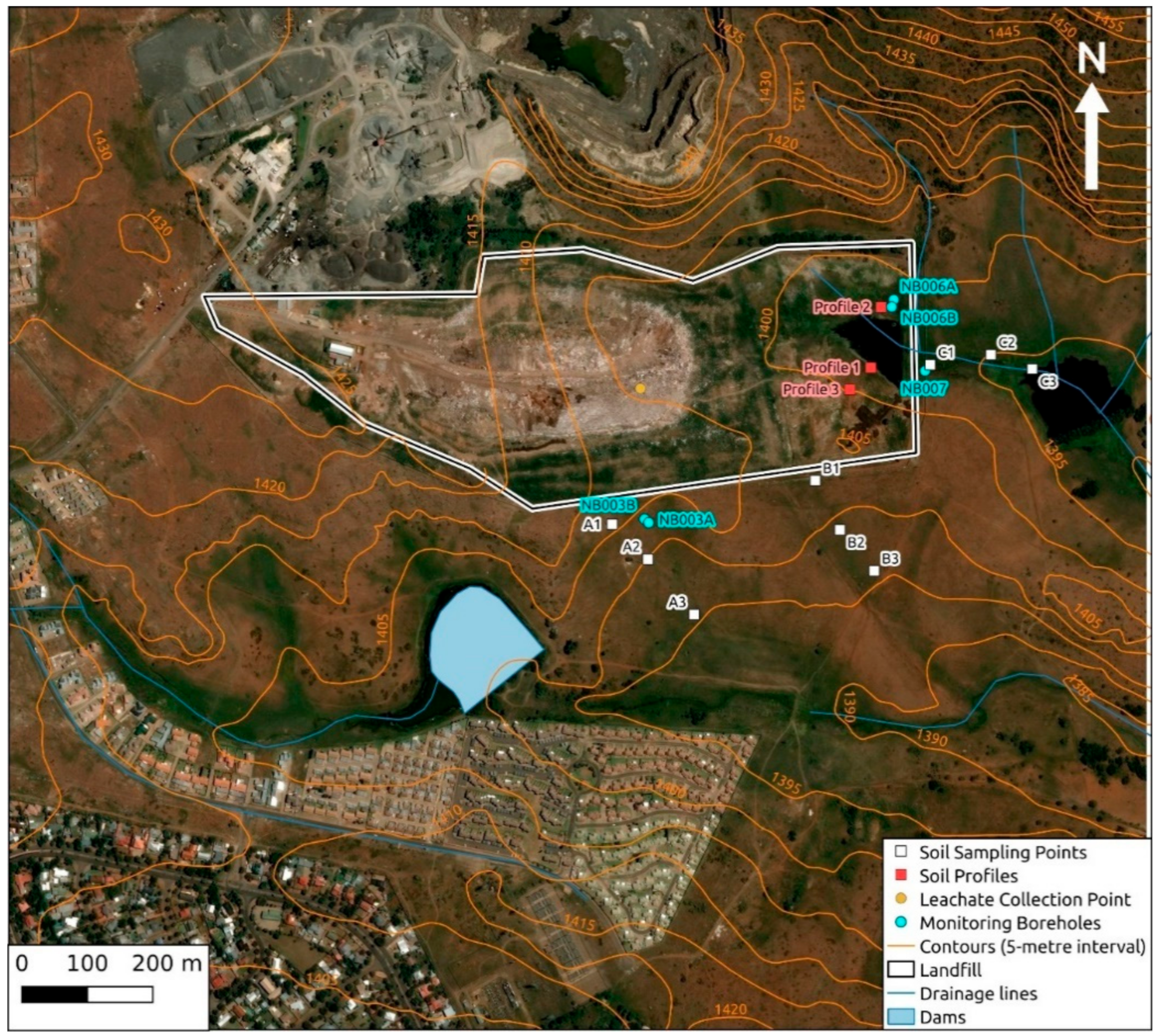
2.1. Study Area
2.2. Groundwater, Surface Water, and Leachate Sampling
2.3. Soil Drainage and Soil Profile Sampling
2.4. Hydrogeochemistry and Geochemical Speciation
3. Results
3.1. Soil Physical and Chemical Properties
3.2. Physicochemical Characteristics of the Leachate
3.3. Water Quality Characteristics of Surface and Groundwater
3.4. Bacteriological Characteristics of Water Samples
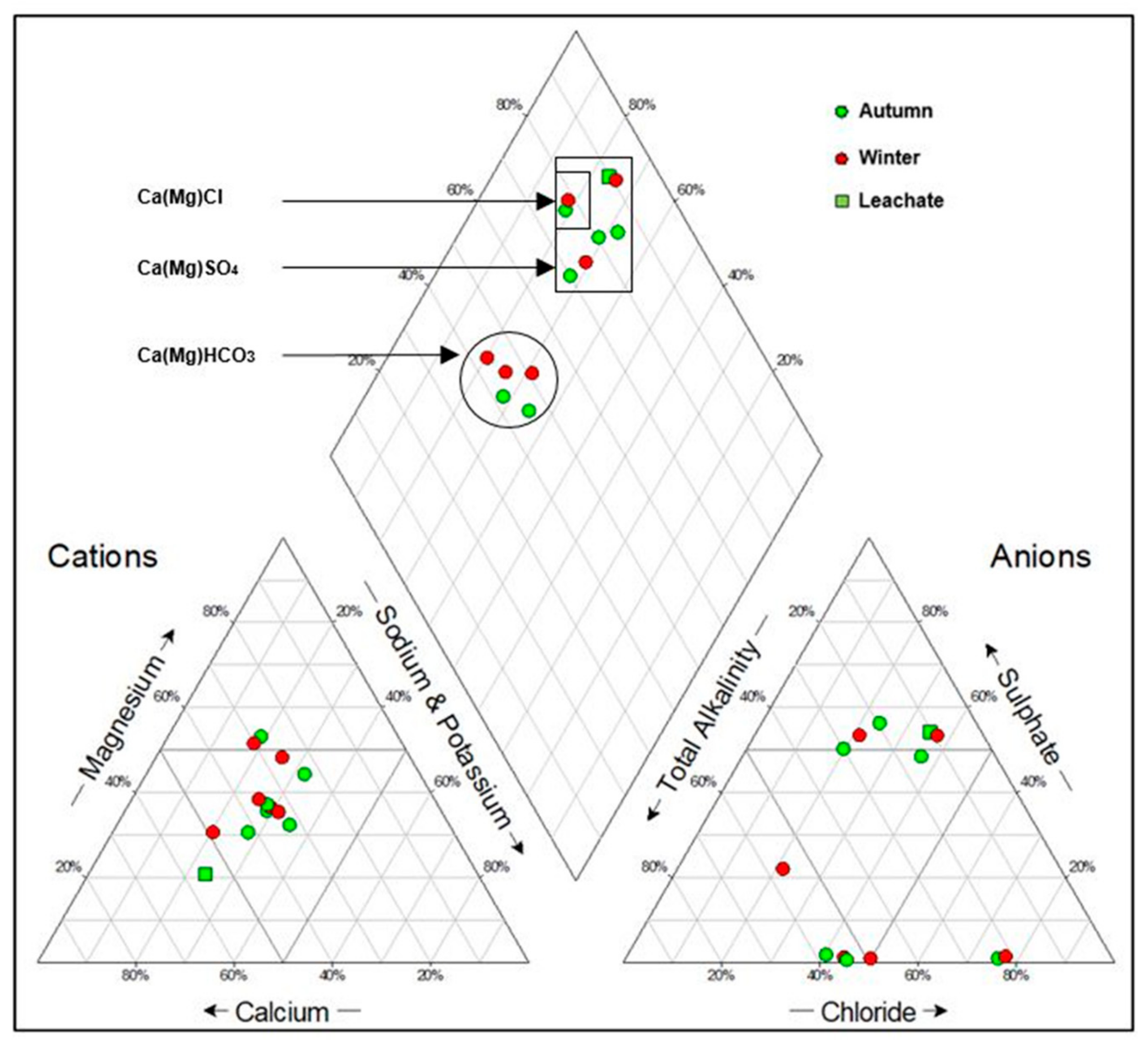
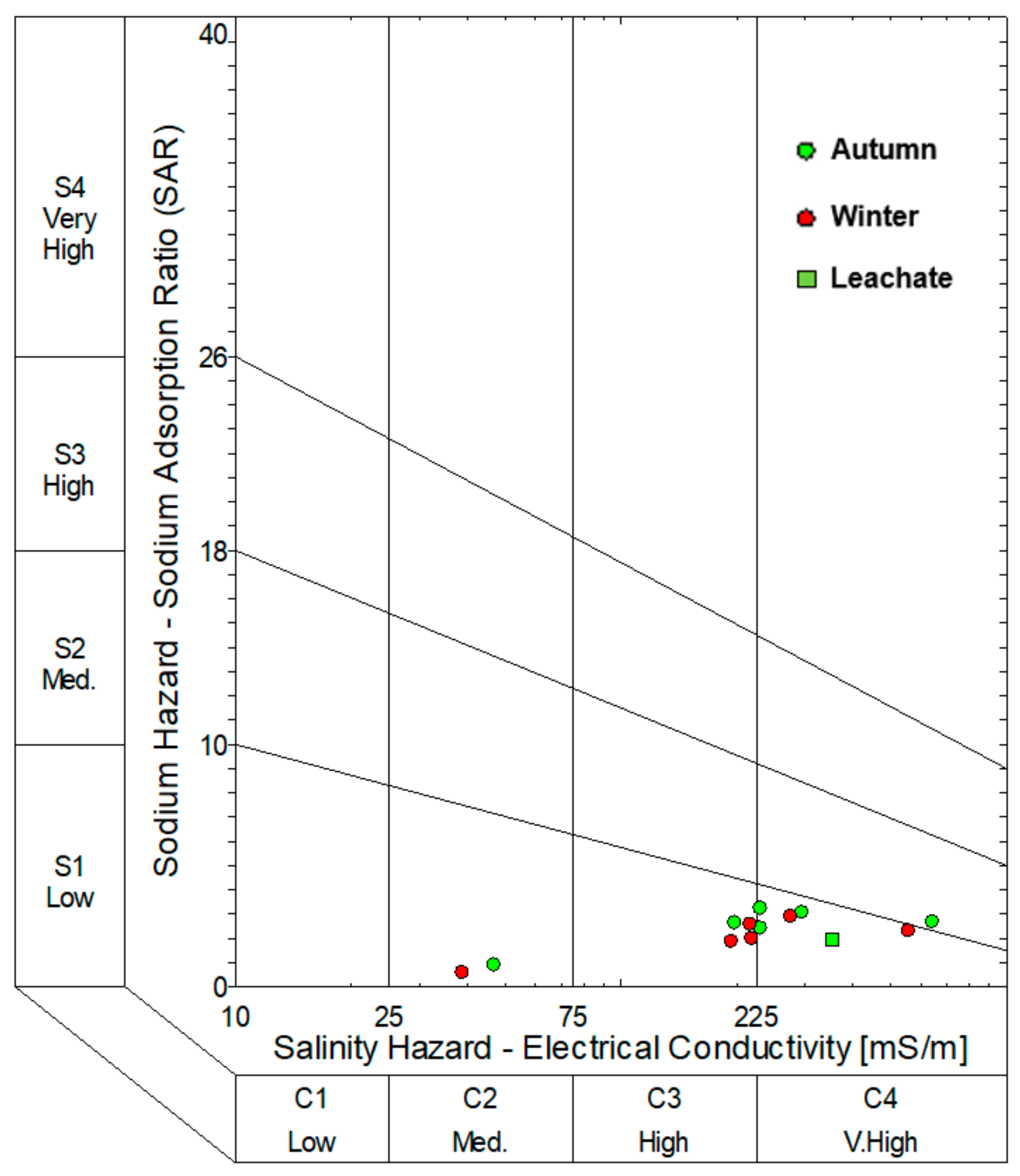
3.5. Hydrochemical Facies
3.6. Geochemical Speciation
4. Discussion
Implications of Water and Soil Quality on Food and Water Security
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Omran, A.; Gavrilescu, M. Municipal solid waste management in developing countries: A perspective on Vietnam. Environ. Eng. Manag. J. 2008, 7, 469–478. [Google Scholar] [CrossRef]
- DEA (Department of Environmental Affairs). National Waste Management Strategy, Government Gazette 35306; Government Notice 344 of 4 May 2012; Department of Environmental Affairs: Pretoria, South Africa, 2012.
- South African Waste Management Series, 2015. Available online: https://www.environment.gov.za/sites/default/.../environmentoutlook_chapter13.pdf (accessed on 25 June 2018).
- Sholichin, M. Field investigation of groundwater contamination from solid waste landfill in Malang, Indonesia. Int. J. Civ. Environ. Eng. 2012, 12, 74–81. [Google Scholar]
- Aderemi, A.O.; Oriaku, A.V.; Adewumi, G.A.; Otitoloju, A.A. Assessment of groundwater contamination by leachate near municipal solid waste landfill. Afr. J. Environ. Sci. Tech. 2011, 5, 933–940. [Google Scholar]
- El-Gohary, F.A.; Kamel, G. Characterization and biological treatment of pre-treated landfill leachate. Ecol. Eng. 2016, 94, 268–274. [Google Scholar] [CrossRef]
- Krčmar, D.; Tenodi, S.; Grba, N.; Kerkez, D.; Watson, M.; Rončević, S.; Dalmacija, B. Preremedial assessment of the municipal landfill pollution impact on soil and shallow groundwater in Subotica, Serbia. Sci. Total Environ. 2018, 615, 1341–1354. [Google Scholar] [CrossRef] [PubMed]
- Aljaradin, M.; Persson, K.M. Environmental impact of municipal solid waste landfills in semi-arid climates: Case study-Jordan. Open Waste Manag. J. 2012, 5, 28–39. [Google Scholar] [CrossRef]
- Dharmarathne, N.; Gunatilake, J. Leachate characterisation and surface groundwater pollution at municipal at municipal solid waste landfill of Gohagoda, Sri Lanka. Int. J. Sci. Res. Publ. 2013, 3, 1–7. [Google Scholar]
- Magaji, J.Y. Effects of waste dump on the quality of plants cultivated around Mpape dumpsite FCT Abuja, Nigeria. Ethiop. J. Environ. Stud. Manag. 2012, 5. [Google Scholar] [CrossRef]
- Swati, P.G.; Das, M.T.; Thakur, I.S. In vitro toxicity evaluation of organic extract of landfill soil and its detoxification by indigenous pyrene-degrading Bacillus sp. ISTPY1. Int. Biodeterior. Biodegrad. 2014, 90, 145–151. [Google Scholar] [CrossRef]
- Shaikh, P.R.; Bhosle, A.B.; Yannawar, V.B. The impact of landfill on soil and groundwater quality of the Nanded city, Maharashtra. Rese 2012, 4, 56–63. [Google Scholar]
- Mohammadi, S.; Heidari, M.; Shahmoradi, B.; Hosseini, G. Assessing landfill leachate heavy metal effect on the surface water quality: A case of Gheshlagh River, Sanandaj City, Iran. J. Adv. Environ. Health Res. 2014, 2, 38–43. [Google Scholar]
- Vaverková, M.D.; Adamcová, D. Evaluation of landfill leachate pollution: Findings from a monitoring study at municipal waste landfill. J. Ecol. Eng. 2015, 16, 19–32. [Google Scholar] [CrossRef][Green Version]
- Adamcová, D.; Vaverková, M.D.; Bartoň, S.; Havlíček, Z.; Břoušková, E. Soil contamination in landfills: A case study of a landfill in Czech Republic. Solid Earth 2016, 7, 239–247. [Google Scholar] [CrossRef]
- Kanmani, S.; Gandhimathi, R. Assessment of heavy metal contamination in soil due to leachate migration from an open dumping site. Appl. Water Sci. 2013, 3, 193–205. [Google Scholar] [CrossRef]
- Lin, A.Y.; Huang, S.T.; Wahlgvist, M.L. Waste management to improve food safety and security for health advancement. Asia Pac. J. Clin. Nutr. 2009, 18, 538–545. [Google Scholar] [PubMed]
- Frone, D.-F.; Frone, S. The importance of water security for sustainable development in the Romanian Agri-Food sector. Agric. Agric. Sci. Proc. 2015, 6, 674–681. [Google Scholar] [CrossRef]
- Ololade, O.O. Understanding the nexus between energy and water: A basis for human survival in South Africa. Dev. South. Afr. 2018, 35, 1–16. [Google Scholar] [CrossRef]
- Musvoto, C.; Nortje, K.; De Wet, B.; Mahumani, B.K.; Nahman, A. Imperatives for an agricultural green economy in South Africa. S. Afr. J. Sci. 2015, 111, 1–8. [Google Scholar] [CrossRef]
- Mangaung Metropolitan Municipality. Integrated Waste Management Plan: Review of Integrated Waste Management Plan; final report; Mangaung Metropolitan Municipality: Bloemfontein, South Africa, 2016.
- Hartzenberg, S. Northern Dumping Site Causing Unhappiness in Bloemfontein. OFM News. Available online: https://www.ofm.co.za/article/local-news/265257/northern-dumping-site-causing-unhappiness-in-bloemfontein (accessed on 24 May 2019).
- Verhagen, B.T.; Butler, M.J.; Levin, M.; Walton, D.J. Investigation of Ground Water Pollution Associated with Waste Disposal: Development of an Environmental Isotope Approach: Report to the Water Research Commission; WRC Project No 311/1/01; Water Research Commission: Pretoria, South Africa, 1996; ISBN 1-86845-739-7.
- Naveen, B.P.; Sumalatha, J.; Malik, R.K. A study on contamination of ground and surface water bodies by leachate leakage from a landfill in Bangalore, India. Int. J. Geo-Eng. 2018, 9, 1–20. [Google Scholar] [CrossRef]
- Dingaan, M.N.V.; Du Preez, P.J. Grassland communities of urban open spaces in Bloemfontein, Free State, South Africa. Koedoe 2013, 55, 1–8. Available online: http://dx.doi.org/10.4102/koedoe.v55i1.1075 (accessed on 30 September 2017). [CrossRef][Green Version]
- DWA. Development of Water Reconciliation Strategies for the Luvuvhu and Letaba Water Supply System: Literature Review Report. Prepared by WRP Consulting Engineers (Pty) Ltd, in association with DMM Development Consultants cc, Golder Associates Africa, Kwezi V3 Engineers and Zitholele Consulting; Department of Water Affairs: Pretoria. 2012. Available online: https://www.academia.edu/7903476/Development_of_A_Reconciliation_Strategy_For_The_Luvuvhu_And_Letaba_Water_Supply_System_Literature_Review_Report (accessed on 8 September 2018).
- Hensley, M.; Le Roux, P.; Du Preez, C.; Van Huyssteen, C.; Kotze, E.; Van Rensburg, L. Soils: The Free State’s Agricultural Base. S. Afr. Geog. J. 2006, 88, 11–21. [Google Scholar] [CrossRef]
- Botha, J.F.; Cloot, A.H. Karoo Aquifers: Deformations, Hydraulic and Mechanical Properties; Report to the Water Research Commission by the Institute for Groundwater Studies; University of the Free State: Bloemfontein, South Africa, 2004. [Google Scholar]
- Butler, M.J. Groundwater Pollution at Sanitary Landfill Sites; Geohydrological, Environmental Isotope and Hydrochemical Studies. Master’s Thesis, University of the Free State, Bloemfontein, South Africa, 1998. [Google Scholar]
- APHA; AWWA; WEF. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association; American Water Works Association; Water Environment Federation; Government Printing Office: Washington, DC, USA, 2005.
- Papuga, K.; Kaszubkiewicz, J.; Wilczewski, W.; Staś, M.; Belowski, J.; Kawałko, D. Soil grain size analysis by the dynamometer method—A comparison to the pipette and hydrometer method. Soil Sci. Annu. 2018, 69, 17–27. [Google Scholar] [CrossRef]
- Non Affiliated Soil Analysis Work Committee. Handbook of Standard Soil Testing Methods for Advisory Purposes; Soil Science Society of South Africa: Pretoria, South Africa, 1990. [Google Scholar]
- Parkhurst, D.L.; Appelo, C.A.J. User’s Guide to PHREEQC (version 3)—A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations; U.S. Geol. Survey, Water-Resources Inv. Report 99–4259; USGS: Reston, VA, USA, 2017.
- Voigt, M.; Marieni, C.; Clark, D.E.; Gislason, S.R.; Oelkers, E.H. Evaluation and refinement of thermodynamic databases for mineral carbonation. Energy Procedia 2018, 146, 81–91. [Google Scholar] [CrossRef]
- Blasco, M.; Gimeno, M.J.; Auque, L.F. Comparison of different thermodynamic database used in a geothermometrical modelling calculation. Proceedia Earth Planet. Sci. 2017, 17, 120–123. [Google Scholar] [CrossRef]
- Shock, E.L.; Helgeson, H.C. Calculation of the thermodynamic and transport properties of aqueous species at high pressures and temperatures: Standard partial molal properties of organic species. Geochim. Cosmochim. Acta 1990, 54, 915–945. [Google Scholar] [CrossRef]
- Government Gazette. Draft National Norms and Standards for the Remediation of Contaminated Land and Soil Quality (Notice No. 233, 2012); Government Gazette 35160:561, March 19 (Regulation Gazette No. 59, 2008); DEAT: Pretoria, South Africa, 2012.
- Tótha, G.; Hermannb, T.; Da Silvac, M.R.; Montanarella, L. Heavy metals in agricultural soils of the European Union with implications for food safety. Environ. Int. 2016, 88, 299–309. [Google Scholar] [CrossRef]
- DWA. Guidelines for Leachate Control; Department of Water Affairs: Pretoria, South Africa, 1999.
- DWA, Department of Water Affairs, South Africa. Revision of General Authorisations in Terms of Section 39 of the National Water Act, 1998; Government Gazette 36820; Government Printer: Pretoria, South Africa, 2013.
- Sabahi, E.A.; Rahim, S.A.; Zuhairi, W.Y.; Nozaily, F.A.; Alshaebi, F. The characteristics of leachate and groundwater pollution at municipal solid waste landfill of Ibb city, Yemen. Am. J. Environ. Sci. 2009, 5, 256–266. [Google Scholar] [CrossRef]
- Kumar, P.J.S.; James, E.J. Physicochemical parameters and their sources in groundwater in the Thirupathur region, Tamil Nadu, South India. Appl. Water Sci. 2013, 3, 219–228. [Google Scholar] [CrossRef]
- South African National Standard 241-1: Drinking Water, Part 1: Microbiological, Physical, Aesthetic and Chemical Determinants; 241-2:2015 drinking water; SABS: Pretoria, South Africa, 2015.
- World Health Organization (WHO). Guidelines for Drinking Water Quality; Final Task Group Meeting; WHO: Geneva, Switzerland, 2015. [Google Scholar]
- Elisante, E.; Muzuka, A.N.N. Sources and seasonal variation of coliform bacteria abundance in groundwater around the slopes of Mount Meru, Arusha, Tanzania. Environ. Monit. Assess. 2016, 188, 395. [Google Scholar] [CrossRef]
- DWAF. South African Water Quality Guidelines, 2nd ed.; Volume 4: Agricultural use: Irrigation; Department of Water Affairs and Forestry: Pretoria, South Africa, 1996; 196p.
- Oke, S.; Vermeulen, D. Geochemical modeling and remediation of heavy metals and trace elements from artisanal mines discharge. Soil Sediment Contam. Int. J. 2017, 26, 84–95. [Google Scholar] [CrossRef]
- Wuana, R.A.; Okieimen, F.E. Heavy metals in contaminated soils: A review of sources, chemistry, risks and best available strategies for remediation. Int. Sch. Res. Network Ecol. 2011, 2011, 1–20. [Google Scholar] [CrossRef]
- Ngabirano, H.; Byamugisha, D.; Ntambi, E. Effects of seasonal variations in physical parameters on quality of gravity flow water in kyanamira sub-county, Kabale District, Uganda. J. Water Resour. Prot. 2016, 8, 1297–1309. [Google Scholar] [CrossRef]
- Patil, P.N.; Sawant, D.V.; Deshmukh, R.N. Physicochemical parameters for testing of water; A review. Int. J. Environ. Sci. 2012, 3, 1194–1207. [Google Scholar]
- Sasakova, N.; Gregova, G.; Takacova, D.; Mojzisova, J.; Papajova, I.; Venglovsky, J.; Szaboova, T.; Kovacova, S. Pollution of surface and ground water by sources related to agricultural activities. Front. Sustain. Food Syst. 2018, 2, 42. [Google Scholar] [CrossRef]
- FAO. Water Quality for Agriculture. Rome: Food and Agriculture Organisation. Available online: http://www.fao.org/docrep/003/t0234e/T0234E01.htm#ch1.4 (accessed on 5 June 2019).
- Ahmad, M. Iron and Manganese Removal from Groundwater: Geochemical Modelling of the Vyredox Method. Master’s Thesis, University of Oslo, Oslo, Norway, 2012. [Google Scholar]
- Kumar, P.J. Interpretation of groundwater chemistry using piper and chadha’s diagrams: A comparative study from Perambalur, taluk. Elixir Int. J. 2013, 54, 12208–12211. [Google Scholar]
- Khatri, N.; Tyagi, S. Influences of natural and anthropogenic factors on surface and groundwater quality in rural and urban areas. Front. Life Sci. 2015, 8, 23–39. [Google Scholar] [CrossRef]
- Hossain, M.L.; Satyajit Roy Das, S.D.; Hossain, M.K. Impact of landfill leachate on surface and ground water quality. J. Environ. Sci. Tech. 2014, 7, 337–346. [Google Scholar] [CrossRef]
- Sanders, E.C.; Yuan, Y.; Pitchford, A. Faecal coliform and E. coli concentrations in effluent-dominated streams of the upper Santa Cruz watershed. Water 2013, 5, 243–261. [Google Scholar] [CrossRef]
- Nagarajan, R.; Thirumalaisamy, S.; Lakshumanan, E. Impact of leachate on groundwater pollution due to non-engineered municipal solid waste landfill sites of erode city; Tamil Nadu, India. Iran. J. Environ. Health Sci. Eng. 2012, 9, 1–12. [Google Scholar] [CrossRef]
- Talukder, M.S.U.; Shirazi, S.M.; Niazuddin, M.; Ghosh, A.K. Groundwater quality and its impact on soil and crop. Bangladesh J. Agric. Sci. 1999, 26, 153–160. [Google Scholar]
- Kumar, K.; Chopra, A.K. Monitoring of physico-chemical and microbiological characteristics of municipal wastewater at treatment plant, Haridwar City (Uttarakhand) India. J. Environ. Sci. Tech. 2012, 5, 109–118. [Google Scholar] [CrossRef]
- Fitsanakis, V.A.; Zhang, N.; Garcia, S.; Aschner, M. Manganese (Mn) and Iron (Fe): Interdependency of Transport and Regulation. Neurotox Res. 2010, 18, 124–131. [Google Scholar] [CrossRef]
- Brown, L.R. Outgrowing the Earth: The Food Security Challenge in an Age of Falling Water Tables and Rising Temperatures; Taylor and Francis: London, UK, 2012; p. 57. [Google Scholar]
- Beck, M.B.; Walker, R.V. On water security, sustainability, and the water food-energy nexus. Front. Environ. Sci. Eng. 2013, 7, 626–639. [Google Scholar] [CrossRef]
- Ololade, O.O. Sustainable development: An overview. In Environmental Science and Engineering Vol 1: Sustainable Development; Rao, S.Y., Gurjar, B.R., Govil, J.N., Eds.; Studium Press LLC: Houston, TX, USA, 2017; 17p, ISBN 1-62699-089-1. [Google Scholar]
| Exchangeable Cations (mg/kg) | Heavy Metals (mg/kg) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Drainage | Distance [m] | pH (KCl) | % C | Ca | Mg | Na | K | Cu | Mn | Fe | Zn |
| A1 | 0 | 7.49 | 0.24 | 938 | 190 | 148 | 115 | 1.5 | 2.9 | 11.9 | 0.77 |
| A2 | 50 | 7.03 | 0.26 | 883 | 333 | 148 | 88 | 2.5 | 10.6 | 18.2 | 0.89 |
| A3 | 100 | 7.36 | 0.26 | 2020 | 688 | 163 | 263 | 2.8 | 17.5 | 18.5 | 1.23 |
| Mean | 7.29 | 0.25 | 1280 | 403 | 153 | 155 | 2.26 | 10.3 | 18.2 | 0.96 | |
| B1 | 0 | 7.31 | 1.16 | 1325 | 413 | 155 | 155 | 2.8 | 14.8 | 17.3 | 1.25 |
| B2 | 50 | 7.02 | 0.44 | 523 | 260 | 155 | 105 | 3.0 | 49.4 | 70.7 | 1.21 |
| B3 | 100 | 7.16 | 0.65 | 1693 | 948 | 173 | 283 | 2.7 | 42.5 | 24.7 | 0.99 |
| Mean | 7.16 | 0.75 | 1180 | 540 | 160 | 180 | 2.8 | 35.5 | 37.5 | 1.15 | |
| C1 | 0 | 7.61 | 1.11 | 4978 | 808 | 210 | 175 | 21.4 | 79.2 | 229.7 | 2.31 |
| C2 | 50 | 7.58 | 1.75 | 2415 | 198 | 180 | 73 | 23.7 | 51.1 | 128.0 | 2.60 |
| C3 | 100 | 7.16 | 2.83 | 5250 | 405 | 195 | 118 | 29.4 | 47.6 | 156.5 | 2.02 |
| Mean | 7.45 | 1.9 | 4242 | 470 | 195 | 121.7 | 24.81 | 59.27 | 171.4 | 2.31 | |
| Norms | SA | - | - | - | - | - | - | 16 | 740 | - | 240 |
| Exchangeable Cations (mg/kg) | Heavy Metals (mg/kg) | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Soil Profile | Depth (cm) | pH (KCl) | Ca | Mg | Na | K | S | P | Cd | Cr | Cu | Fe | Mn | Ni | Zn | Clay% | Silt% | Sand% | Bulk Density |
| SP 1 | 20 | 6.58 | 4780 | 1430 | 484.4 | 248 | 203 | 5.2 | 0.1 | 2.01 | 13 | 84.56 | 163 | 7.6 | 1.48 | 44 | 25 | 31 | 1.79 |
| 40 | 6.94 | 2500 | 1956 | 372.4 | 97 | 40 | 3.6 | 0.04 | 0.26 | 4.4 | 36.78 | 20 | 0.6 | 0.39 | 35 | 16 | 48 | 1.85 | |
| 80 | 7.03 | 2536 | 2734 | 681 | 140 | 208 | 6.4 | 0.05 | 0.29 | 7.7 | 32.28 | 17 | 2 | 0.46 | 41 | 26 | 33 | 1.87 | |
| 120 | 6.98 | 1224 | 1036 | 335.4 | 56 | 33 | 3.8 | 0.05 | 0.19 | 2.8 | 30.04 | 17 | 1.3 | 0.53 | 12 | 7 | 80 | 1.67 | |
| SP 2 | 20 | 6.51 | 2428 | 828 | 209.2 | 129 | 29 | 8.8 | 0.06 | 0.14 | 8.1 | 83.76 | 56 | 2.2 | 0.7 | 15 | 4 | 81 | 1.1 |
| 60 | 6.36 | 3270 | 2058 | 425 | 201 | 31 | 8.2 | 0.05 | 0.13 | 9.6 | 42.54 | 25 | 1.2 | 0.38 | 11 | 6 | 83 | 1.58 | |
| 100 | 7.06 | 3512 | 2022 | 525.6 | 147 | 201 | 4.4 | 0.04 | 0.19 | 6.5 | 24.76 | 13 | 0.6 | 0.3 | 36 | 26 | 38 | 1.64 | |
| SP 3 | 10 | 6.98 | 886 | 262 | 125.6 | 126 | 13 | 5.4 | 0.03 | 0.33 | 2.2 | 32.14 | 28 | 1.8 | 0.49 | 15 | 8 | 76 | 1.59 |
| 20 | 6.38 | 1854 | 722 | 162 | 66 | 26 | 6.6 | 0.04 | 0.22 | 5.9 | 30.7 | 13 | 0.8 | 0.29 | 15 | 8 | 76 | 1.55 | |
| 45 | 6.26 | 2046 | 864 | 170.2 | 33 | 13 | 3 | 0.04 | 0.03 | 2.8 | 19.3 | 13 | 0.5 | 0.21 | 19 | 9 | 72 | 1.43 | |
| Norms | SA | - | - | - | - | - | - | - | 7.5 | 6.5 | 16 | - | 740 | 91 | 240 | ||||
| EU | >7 | - | - | - | - | - | - | 1.5 | - | 100 | - | - | - | 200 | |||||
| Autumn Season | Winter Season | Water Quality Guidelines | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sampling Site | Leachate | Surface | NB03A | NB03B | NB07 | NB06A | NB06B | Surface | NB03A | NB03B | NB07 | NB06A | NB06B | SANS 2015 | DWAF 1998 | WHO 2011 | DWA 1999 |
| pH | 7.6 | 7.7 | 7.6 | 7.8 | 7.4 | 7.3 | 7.5 | 7.9 | 7.8 | 7.7 | 7.4 | 7.4 | 7.4 | 5–9.7 | 6.5–8.4 | 6.5–8.5 | 5–9.5 |
| TDS | 2825 | 361 | 1449 | 2435 | 4756.5 | 1537 | 1647 | 280.5 | 1364.5 | 2415 | 3432 | 1343.5 | 1417 | ≤1200 | - | 500 | - |
| EC | 355 | 46 | 197.5 | 295 | 643.5 | 230.5 | 230 | 38.8 | 193 | 275.5 | 557 | 218 | 216.5 | ≤170 | <40 | 150 | 150 |
| COD | 441 | 91 | 191.5 | 96.5 | 299.5 | 72 | 52 | 31.5 | 37 | 77 | 261 | 82 | 51 | - | - | - | 75 |
| TOC | 120 | 27 | 8.3 | 18 | 82.7 | 11.5 | 13 | 9.2 | 8.7 | 14.6 | 77 | 10 | 12 | ≤10 | - | - | - |
| Ca | 489 | 43 | 107 | 282.5 | 450.5 | 180 | 176 | 41 | 103 | 241 | 391 | 153 | 150 | 300 | - | 75 | |
| Mg | 110 | 19 | 122.5 | 171.5 | 520 | 117 | 106 | 16 | 112 | 155 | 405 | 100 | 97 | 100 | - | 30 | |
| Na | 180 | 28 | 168 | 264 | 351.5 | 169 | 219.5 | 17 | 116 | 235 | 272 | 128 | 165 | ≤200 | 0–70 | 200 | |
| K | 110 | 8 | 3 | 4.5 | 8 | 2 | 1 | 5 | 3 | 6 | 8 | 1 | 0.5 | 100 | - | 300 | |
| HCO3 | 229 | 74 | 174 | 362.5 | 939.5 | 699.5 | 708.5 | 129 | 98 | 478 | 647 | 596 | 569 | - | - | - | |
| SO4 | 1147 | 119 | 535 | 998 | 31 | 19 | 5 | 48 | 536 | 975 | 34 | 11 | 8 | ≤500 | - | 500 | |
| Cl | 559 | 35 | 300 | 318.5 | 2190 | 349 | 421 | 35 | 279 | 292 | 1656 | 347 | 412 | ≤300 | 0–105 | 250 | 0.25 |
| Br | 1.4 | 0.5 | 2.0 | 1.9 | 11 | 1 | 2 | 0 | 2 | 2 | 8 | 2 | 3 | ≤3 | - | ||
| Mn | 1 | 0 | 0 | 0.9 | 5 | 0 | 0 | 0 | 0 | 0 | 2 | 1 | 0 | ≤0.400 | ≤10 | 0.1 | |
| Total coliform | 2420 | 2420 | 98.5 | 2420 | 2420 | 23 | 517 | 1552 | 2420 | 2420 | 2420 | 38.5 | 36 | ≤10 | varies | 10 | 1000 |
| E. coli | 2420 | 613 | 0 | 0 | 0 | 0 | 0 | 18 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Leachate (SI) | Autumn (SI) | Winter (SI) | ||||
|---|---|---|---|---|---|---|
| Mineral | Formula | NB03B | NB07 | NB03B | NB07 | |
| Al(OH)3 | Al(OH)3 | −1.39 | - | - | −1.15 | −1.40 |
| Alunite | KAl3(SO4)2(OH) | 1.08 | - | - | −1.91 | −3.47 |
| Anhydrite | CaSO4 | −0.54 | −0.81 | −2.34 | −0.87 | −2.26 |
| Aragonite | CaCO3 | 0.18 | 0.95 | 1.10 | 1.00 | 0.54 |
| Calcite | CaCO3 | 0.32 | 1.09 | 1.24 | 1.14 | 0.68 |
| CdSO4 | CdSO4 | −10.63 | - | - | - | −11.68 |
| Cd(OH)2 | Cd(OH)2 | −7.93 | - | - | - | −7.30 |
| Dolomite | CaMg (CO3)2 | 0.33 | 2.30 | 2.92 | 2.43 | 1.74 |
| Fe(OH)3 | Fe(OH)3 | 0.27 | 1.82 | 1.52 | 2.12 | 0.39 |
| Gibbsite | Al(OH)3 | 1.30 | - | - | 1.54 | 1.29 |
| Goethite | FeOOH | 6.16 | 7.71 | 7.42 | 8.01 | 6.28 |
| Gypsum | CaSO4.2H20 | −0.24 | −0.50 | −2.04 | −0.56 | −1.98 |
| Halite | NaCl | −5.67 | −5.73 | −4.82 | −5.82 | −5.04 |
| Hausmannite | Mn3O4 | −12.81 | - | −8.07 | −9.07 | −11.90 |
| Hematite | Fe2O3 | 14.34 | 17.43 | 16.84 | 18.03 | 14.57 |
| Jarosite | KFe3(SO4)2 (OH) | −3.86 | −3.02 | −5.98 | −2.02 | −8.02 |
| Manganite | MnOOH | −5.60 | - | −3.89 | −4.09 | −5.30 |
| Melanterite | FeSO4.7H2O | −7.06 | −7.95 | −8.78 | −7.64 | −8.63 |
| Otavite | CdCO3 | −1.97 | - | - | - | −0.92 |
| Pyrochroite | Mn (OH)2 | −6.46 | - | −5.1 | −5.75 | −6.16 |
| Pyrolusite | MnO2.H20 | −10.64 | - | −8.53 | −8.33 | −10.34 |
| Rhodochrosite | MnCO3 | 0.08 | - | 1.55 | 0.31 | 0.80 |
| Siderite | FeCO3 | −1.71 | −1.57 | −0.71 | −1.15 | −1.18 |
| Smithsonite | ZnCO3 | −2.83 | - | - | - | −2.60 |
| Sylvite | KCl | −5.45 | −7.02 | −6.03 | −1.15 | −6.15 |
| Zn(OH)2 | Zn(OH)2 | −4.59 | - | - | - | −4.73 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ololade, O.O.; Mavimbela, S.; Oke, S.A.; Makhadi, R. Impact of Leachate from Northern Landfill Site in Bloemfontein on Water and Soil Quality: Implications for Water and Food Security. Sustainability 2019, 11, 4238. https://doi.org/10.3390/su11154238
Ololade OO, Mavimbela S, Oke SA, Makhadi R. Impact of Leachate from Northern Landfill Site in Bloemfontein on Water and Soil Quality: Implications for Water and Food Security. Sustainability. 2019; 11(15):4238. https://doi.org/10.3390/su11154238
Chicago/Turabian StyleOlolade, Olusola O., Sabelo Mavimbela, Saheed A. Oke, and Rinae Makhadi. 2019. "Impact of Leachate from Northern Landfill Site in Bloemfontein on Water and Soil Quality: Implications for Water and Food Security" Sustainability 11, no. 15: 4238. https://doi.org/10.3390/su11154238
APA StyleOlolade, O. O., Mavimbela, S., Oke, S. A., & Makhadi, R. (2019). Impact of Leachate from Northern Landfill Site in Bloemfontein on Water and Soil Quality: Implications for Water and Food Security. Sustainability, 11(15), 4238. https://doi.org/10.3390/su11154238





