Exploring Potential Soil Bacteria for Sustainable Wheat (Triticum aestivum L.) Production
Abstract
1. Introduction
2. Results
2.1. Plant Growth-Promoting (PGP) Activity of Soil Bacteria
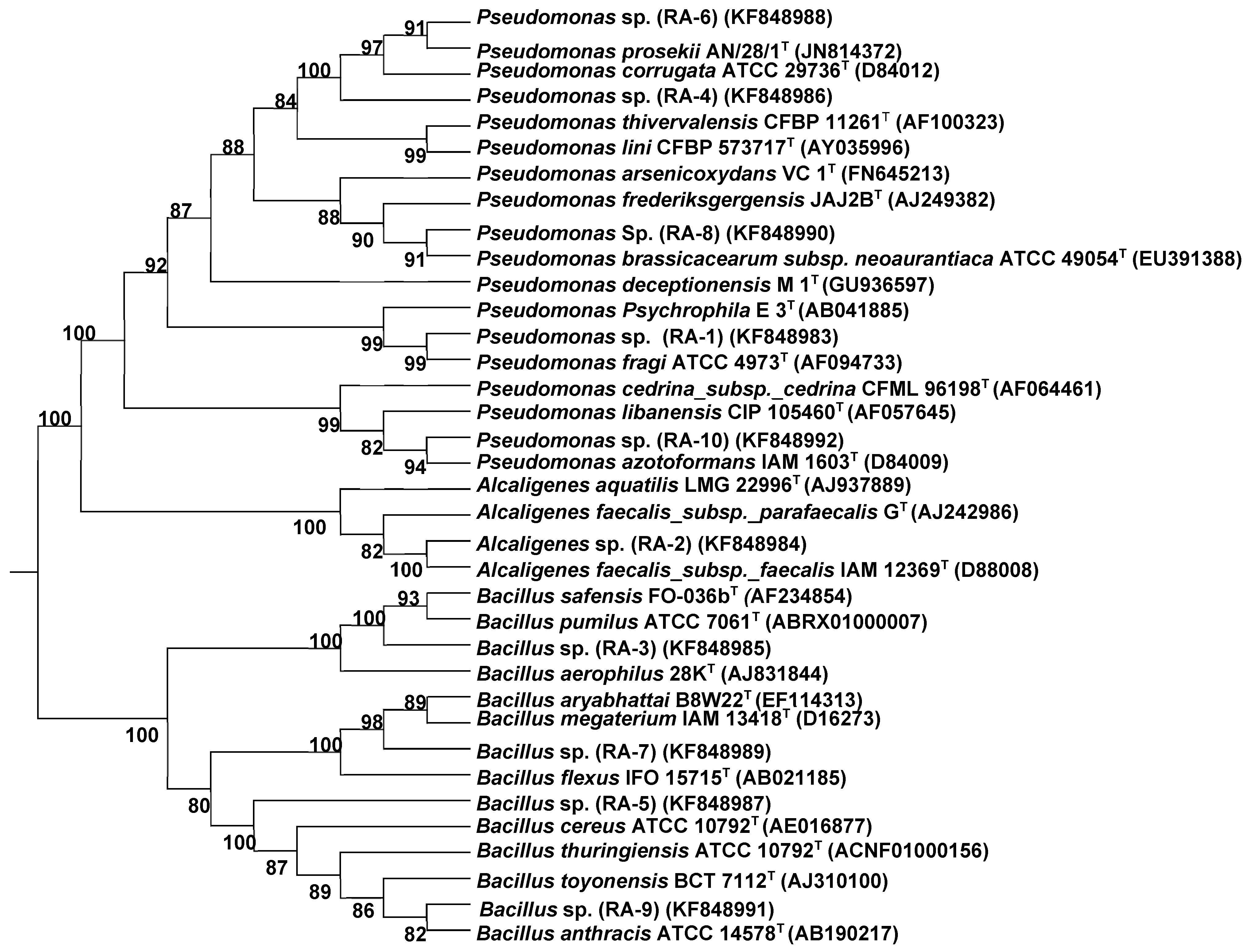
2.2. 16S rRNA Gene Sequence Identification of Bacterial Strains
2.3. Response of Wheat to Soil Bacteria under Controlled and Field Conditions
3. Discussion
4. Conclusions
5. Material and Methods
5.1. Isolation and Screening of Soil Bacteria
5.2. Plant Growth-Promoting Assay and Biochemical Characterization of Soil Bacteria
5.3. Identification of Bacterial Strains Using 16S rRNA Gene Sequencing
5.4. The Effect of Potential Soil Bacteria on Wheat Crops under Controlled and Field Conditions
5.5. Whole-Cell Fatty Acid Analysis
5.6. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hayat, R.; Ali, S.; Amara, U.; Khalid, R.; Ahmed, I. Soil beneficial bacteria and their role in plant growth promotion: A review. Ann. Microbiol. 2010, 60, 579–598. [Google Scholar] [CrossRef]
- Deubel, A.; Merbach, W. Influence of microorganisms on phosphorus bioavailability in soils. In Microorganisms in Soils: Roles in Genesis and Functions; Buscot, F., Varma, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; pp. 177–191. [Google Scholar]
- Vessey, J.K. Plant growth promoting rhizobacteria as biofertilizers. Plant Soil 2003, 255, 571–586. [Google Scholar] [CrossRef]
- Canbolat, M.Y.; Bilen, S.; Çakmakçı, R.; Şahin, F.; Aydın, A. Effect of plant growth-promoting bacteria and soil compaction on barley seedling growth, nutrient uptake, soil properties and rhizosphere microflora. Biol. Fertil. Soils 2006, 42, 350–357. [Google Scholar] [CrossRef]
- Rodríguez, H.; Fraga, R. Phosphate solubilizing bacteria and their role in plant growth promotion. Biotechnol. Adv. 1999, 17, 319–339. [Google Scholar] [CrossRef]
- Ullah, M.A.; Tariq, A.N.; Razzaq, A. Effect of Rice Bean (Vigna umbellata) Inter-cropping on the Yield of Perennial Grass, Panicum maximum CV. Gaton under Rain-fed Conditions. J. Agric. Soc. Sci. 2007, 3, 70–72. [Google Scholar]
- Wu, S.; Jia, S.; Sun, D.; Chen, M.; Chen, X.; Zhong, J.; Huan, L. Purification and characterization of two novel antimicrobial peptides Subpeptin JM4-A and Subpeptin JM4-B produced by Bacillus subtilis JM4. Curr. Microbiol. 2005, 51, 292–296. [Google Scholar] [CrossRef]
- Egamberdiyeva, D. The effect of plant growth promoting bacteria on growth and nutrient uptake of maize in two different soils. Appl. Soil Ecol. 2007, 36, 184–189. [Google Scholar] [CrossRef]
- Adesemoye, A.O.; Torbert, H.A.; Kloepper, J. Plant growth-promoting rhizobacteria allow reduced application rates of chemical fertilizers. Microb. Ecol. 2009, 58, 921–929. [Google Scholar] [CrossRef]
- Kaschuk, G.; Leffelaar, P.A.; Giller, K.E.; Alberton, O.; Hungria, M.; Kuyper, T.W. Responses of legumes to rhizobia and arbuscular mycorrhizal fungi: A metaanalysis of potential photosynthate limitation of symbioses. Soil Biol. Biochem. 2010, 42, 125–127. [Google Scholar] [CrossRef]
- Van Loon, L. Plant responses to plant growth-promoting rhizobacteria. Eur. J. Plan Pathol. 2007, 119, 243–254. [Google Scholar] [CrossRef]
- Prashant, S.D.; Rane, M.R.; Chaudhari, B.L.; Chincholkar, S.B. Siderophoregenic Acinetobacter calcoaceticus isolated from wheat rhizosphere with strong PGPR activity. Malays. J. Microbiol. 2009, 5, 6–12. [Google Scholar]
- Afzal, A.; Ashraf, M.; Asad, S.A.; Farooq, M. Effect of phosphate solubilizing microorganisms on phosphorus uptake, yield and yield traits of wheat (Triticum aestivum L.) in rainfed area. Int. J. Agric. Biol. 2005, 7, 207–209. [Google Scholar]
- Shaharooma, B.; Naveed, M.; Arshad, M.; Zahir, Z.A. Fertilizer dependent efficiency of Pseudomonads for improving growth, yield, and nutrient use efficiency of wheat (Triticum aestivum L.). Appl. Microbiol. Biotechnol. 2008, 79, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Shaharoona, B.; Arshad, M.; Zahir, Z.A. Effect of plant growth promoting rhizobacteria containing ACC-deaminase on maize (Zea mays L.) growth under axenic conditions and on nodulation in mung bean (Vigna radiata L.). Lett. Appl. Microbiol. 2006, 42, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Saber, Z.; Pirdashti, H.; Esmaeili, M.; Abbasian, A.; Heidarzadeh, A. Response of Wheat Growth Parameters to Co-Inoculation of Plant Growth Promoting Rhizobacteria (PGPR) and Different Levels of Inorganic Nitrogen and Phosphorus. World Appl. Sci. J. 2012, 16, 213–219. [Google Scholar]
- Akhtar, J.M.; Asghar, N.H.; Shahzad, K.; Arshad, M. Role of plant gra applied in combination with compost and mineral fertilizers to improve growth and yield of wheat (Triticum aestivum L.). Pak. J. Bot. 2009, 41, 381–390. [Google Scholar]
- Duffy, B.K.; Ownley, B.H.; Weller, D.M. Soil chemical and physical properties associated with suppression of take-all of wheat by Trichoderma koningii. Phytopathology 1997, 87, 1118–1124. [Google Scholar] [CrossRef] [PubMed]
- Khalid, A.; Arshad, M.; Zahir, Z.A. Screening plant growth promoting rhizobacteria for improving growth promoting rhizobacteria for improving growth and yield of wheat. J. Appl. Microbiol. 2004, 96, 473–480. [Google Scholar] [CrossRef]
- Dey, R.; Pal, K.; Bhatt, D.M.; Chauhan, S.M. Growth promotion and yield enhancement of peanut (Arachis hypogea L.) by application of plant growth promoting rhizobacteria. Microbiol. Res. 2004, 159, 371–394. [Google Scholar] [CrossRef]
- Mishra, M.; Kumar, U.; Mishra, P.K.; Prakash, V. Efficiency of plant growth promoting rhizobacteria for the enhancement of Cicer arietinum L. growth and germination under salinity. Adv. Biol. Res. 2010, 4, 92–96. [Google Scholar]
- Pathma, J.; Kennedy, R.K.; Sakthivel, N. Mechanisms of fluorescent pseudomonads that mediate biological control of phytopathogens and plant growth promotion of crop plants. In Bacteria in Agrobiology: Plant Growth Responses; Maheshwari, D.K., Ed.; Springer: Berlin, Germany, 2011; pp. 77–105. [Google Scholar]
- Li, Y.S.; Gao, Y.; Tian, Q.Y.; Shi, F.L.; Li, L.H.; Zhang, W.H. Stimulation of root acid phosphatase by phosphorus deficiency is regulated by ethylene in Medicago falcata. Environ. Exp. Bot. 2011, 71, 114–120. [Google Scholar] [CrossRef]
- Vansuy, G.; Robin, A.; Briat, J.F.; Curie, C.; Lemanceau, P. Iron acquisition from Fe-pyoverdine by Arabidopsis thaliana. Mol. Plant Microbe Interact. 2007, 20, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Chincholkar, S.B.; Chaudhari, B.L.; Rane, M.R.; Sarode, P.D. Fungal phytopathogen suppression using siderophoregenic bio-inoculants. In Biological Control of Plant Diseases: Current Concepts; Chincholkar, S.B., Mukerji, K.G., Eds.; Haworth Press: Binghamton, NY, USA, 2007; pp. 401–417. [Google Scholar]
- Bholay, A.D.; Borkhataria, B.V.; Jadhav, P.U.; Palekar, K.S.; Dhalkari, M.V.; Nalawade, P.M. Bacterial Lignin Peroxidase: A Tool for Biobleaching and Biodegradation of Industrial Effluents. Univers. J. Environ. Res. Technol. 2012, 2, 58–64. [Google Scholar]
- Swain, M.R.; Naskar, S.K.; Ray, R.C. Indole-3-acetic acid production and effect on sprouting of yam (Dioscorea rotundata L.) minisetts by Bacillus subtilis isolated from culturable cowdung microflora. Pol. J. Microbiol. 2007, 56, 103–110. [Google Scholar] [PubMed]
- Brick, J.M.; Bostock, R.M.; Silverstone, S.E. Rapid in situ assay for indole acetic acid production by bacteria immobilized on nitrocellulose membrane. Appl. Environ. Microbiol. 1991, 57, 535–538. [Google Scholar]
- Pikovskaya, R. Mobilization of phosphorus in soil in connection with vital activity of some microbial species. Mikrobiologiya 1948, 17, 362–370. [Google Scholar]
- Watanabe, F.; Olsen, S. Test of an Ascorbic Acid Method for Determining Phosphorus in Water and NaHCO3 Extracts from Soil 1. Soil Sci. 1965, 29, 677–678. [Google Scholar] [CrossRef]
- Payne, S.M. Detection, isolation and characterization of siderophores. Methods Enzymol. 1994, 235, 329–344. [Google Scholar]
- Penrose, D.M.; Glick, B.R. Methods for isolating and characterizing of ACC deaminase-containing plant growth promoting rhizobacteria. Plant Physiol. 2003, 118, 10–15. [Google Scholar] [CrossRef]
- Bradford, M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Yamamoto, S.; Harayama, S. PCR amplification and direct sequencing of gyrB genes with universal primers and their application to the detection and taxonomic analysis of Pseudomonas putida strains. Appl. Environ. Microbiol. 1995, 61, 1104–1109. [Google Scholar] [PubMed]
- Kim, O.S.; Cho, Y.J.; Lee, K.; Yoon, S.H.; Kim, M.; Na, H.; Park, S.C.; Jeon, Y.S.; Lee, J.H.; Yi, H.; et al. Introducing EzTaxon-e: A prokaryotic 16S rRNA gene sequence database with phylotypes that represent uncultured species. Int. J. Syst. Evol. Microbiol. 2012, 62, 716–721. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Dudley, J.; Nei, M.; Kumar, S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol. Biol. Evol. 2007, 24, 1596–1599. [Google Scholar] [CrossRef] [PubMed]
- Sasser, M. Identification of Bacteria by Gas Chromatography of Cellular Fatty Acids; MIDI Technical Note 101; MIDI Inc.: Newark, DE, USA, 1990. [Google Scholar]
| P-Solubilization | ACC-Deaminase Activity (nmol h−1) | Siderophore Activity Level | A/Ar | IAA mg L−1 with Tryptophan | IAA mg L−1 without Tryptophan | ||
|---|---|---|---|---|---|---|---|
| (µgmL−1) ± S.E | pH (7.0) | (µg mL−1) ± S.E | (µg mL−1) ± S.E | ||||
| RA-1 | 84.41 ± 1.66 | 5.74 | 654 ± 54 | +++ | 0.478 ± 0.025 | 2.08 ± 0.085 | 1.36 ± 0.13 |
| RA-2 | 117.73 ± 2.41 | 5.12 | 754 ± 121 | +++ | 0.514 ± 0.021 | 1.84 ± 0.060 | 1.24 ± 0.091 |
| RA-3 | 93.34 ± 1.80 | 4.82 | 541 ± 47 | +++ | 0.361 ± 0.017 | 2.04 ± 0.11 | 2.42 ± 0.40 |
| RA-4 | 88.18 ± 1.77 | 5.02 | 475 ± 69 | ++ | 0.723 ± 0.029 | 3.50 ± 0.27 | 1.30 ± 0.17 |
| RA-5 | 162.16 ± 1.46 | 4.58 | 589 ± 79 | ++ | 0.651 ± 0.031 | 2.3 ± 0.098 | 1.06 ± 0.067 |
| RA-6 | 127.84 ± 1.59 | 4.75 | 671 ± 56 | ++ | 0.715 ± 0.042 | 2.61 ± 0.28 | 1.097 ± 0.035 |
| RA-7 | 212.47 ± 2.72 | 4.38 | 621 ± 98 | +++ | 0.586 ± 0.015 | 12.02 ± 0.61 | 2.408 ± 0.31 |
| RA-8 | 104.34 ± 0.98 | 4.55 | 782 ± 79 | +++ | 0.681 ± 0.034 | 9.51 ± 0.73 | 2.17 ± 0.28 |
| RA-9 | 105.81 ± 1.80 | 5.12 | 480 ± 56 | ++ | 0.814 ± 0.044 | 1.91 ± 0.13 | 1.32 ± 0.32 |
| RA-10 | 110.73 ± 2.82 | 4.98 | 590 ± 74 | + | 0.976 ± 0.036 | 2.12 ± 0.086 | 1.36 ± 0.16 |
| 16S rRNA Gene (bp) | DDBJ Accession Number for the 16S rRNA Gene Sequence | Closely Related Taxa (Species) | Type Strain (Gene Bank ID) | DDBJ ACCESSION of the 16S rRNA Gene Sequence | Similarity (%) | |
|---|---|---|---|---|---|---|
| RA-1 | 1330 | KF848983 | Pseudomonas fragi | ATCC 4973(T) | AF094733 | 99.47 |
| RA-2 | 1332 | KF848984 | Alcaligenes faecalis subsp. faecalis | IAM12369(T) | D88008 | 99.1 |
| RA-3 | 1321 | KF848985 | Bacillus safensis | FO-036b(T) | AF234854 | 100 |
| RA-4 | 1467 | KF848986 | Pseudomonas corrugate | ATCC 29736(T) | D84012 | 99.23 |
| RA-5 | 1335 | KF848987 | Bacillus cereus | ATCC 14579(T) | AE016877 | 100 |
| RA-6 | 1302 | KF848988 | Pseudomonas arsenicoxydans | VC-1(T) | FN645213 | 99.31 |
| RA-7 | 1323 | KF848989 | Bacillus aryabhattai | B8W22(T) | EF114313 | 100 |
| RA-8 | 1280 | KF848990 | Pseudomonas brassicacearum subsp. neoaurantiaca | ATCC 49054(T) | EU391388 | 99.92 |
| RA-9 | 1327 | KF848991 | Bacillus thuringiensis | ATCC 10792(T) | ACNF01000156 | 100 |
| RA-10 | 1311 | KF848992 | Pseudomonas azotoformans | IAM1603(T) | D84009 | 99.62 |
| Shoot Length | Root Length | Fresh Weight (gm) | Dry Weight (gm) | |||
|---|---|---|---|---|---|---|
| (cm) | (%) | (cm) | (%) | |||
| Control | 15.9 ± 2.23 D | 100 | 5.13 ± 0.93 F | 100 | 1.47 ± 0.46 D | 0.76 ± 0.15 E |
| RA-1 | 22.8 ± 3.18 BC | 143 | 7.44 ± 1.01 DEF | 145 | 2.96 ± 1.70 CD | 1.24 ± 0.44 CDE |
| RA-2 | 27.9 ± 3.16 A | 175 | 10.69 ± 2.63 ABCD | 208 | 5.69 ± 1.94 AB | 2.79 ± 1.39 AB |
| RA-3 | 22.1 ± 3.09 C | 139 | 6.85 ± 1.21 EF | 134 | 2.75 ± 1.34 CD | 1.14 ± 0.81 DE |
| RA-4 | 26.7 ± 3.41 AB | 168 | 8.81 ± 2.06 CDE | 172 | 2.99 ± 1.14 CD | 1.68 ± 1.34 BCDE |
| RA-5 | 22.3 ± 3.06 C | 140 | 7.96 ± 1.04 CDEF | 155 | 2.64 ± 1.19 D | 1.37 ± 1.22 CDE |
| RA-6 | 28.2 ± 3.45 A | 177 | 13.37 ± 3.17 A | 261 | 6.01 ± 1.54 AB | 3.98 ± 1.06 A |
| 7-RA | 28.9 ± 3.55 A | 182 | 12.34 ± 2.03 AB | 241 | 6.40 ± 1.09 A | 3.96 ± 1.26 A |
| RA-8 | 25.7 ± 3.97 ABC | 162 | 11.25 ± 2.45 ABC | 219 | 4.53 ± 1.57 BC | 2.35 ± 1.17 BC |
| RA-9 | 22.9 ± 3.19 BC | 144 | 7.68 ± 1.24 DEF | 150 | 3.13 ± 1.36 CD | 1.71 ± 1.31 BCDE |
| RA-10 | 27.7 ± 4.09 A | 174 | 9.91 ± 2.19 BCDE | 193 | 5.26 ± 2.19 AB | 2.39 ± 1.09 BCD |
| CV | 10.44 | 21.42 | 26.40 | 37.16 | ||
| p-value | 0.0001 | 0.001 | 0.0000 | 0.0004 | ||
| Shoot Length | Root Length | Number of Tillers | Fresh Weight (gm/plant) | Dry Weight (gm/plant) | Spike Length (cm) | Grain Yield | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| (cm) | (%) | (cm) | (%) | (kg ha−1) | (%) | |||||
| Control | 66.61 ± 5.16 F | 100 | 19.56 ± 3.35 F | 100 | 4 D | 17.72 ± 2.54 GE | 6.64 ± 1.23 E | 6.70 ± 0.99 B | 2900.43 ± 203 E | 100 |
| RA-2 | 88.39 ± 8.1 C | 133 | 27.82 ± 3.45 BC | 142 | 8 C | 26.98 ± 3.19 DE | 8.63 ± 2.65 DE | 7.55 ± 1.27 B | 4119.82 ± 249 C | 142 |
| RA-4 | 84.01 ± 8.6 E | 126 | 24.97 ± 4.15 CDE | 128 | 6 CD | 21.87 ± 2.15 FG | 7.92 ± 1.19 DE | 6.99 ± 1.09 B | 4158.98 ± 293 C | 143 |
| RA-6 | 6.96 ± 8.4 DE | 131 | 25.87 ± 4.58 CD | 132 | 6 CD | 22.52 ± 2.19 EF | 8.32 ± 2.19 DE | 7.19 ± 2.14 B | 3612.36 ± 353 D | 125 |
| RA-7 | 90.02 ± 8.9 C | 135 | 21.56 ± 4.19 EF | 110 | 9 BC | 28.55 ± 2.48 CD | 8.36 ± 2.22 DE | 8.51 ± 2.06 B | 4206.76 ± 393 C | 145 |
| RA-8 | 82.83 ± 9.5 E | 124 | 22.21 ± 5.16 DEF | 114 | 8 C | 24.27 ± 3.76 DEF | 8.66 ± 2.14 DE | 7.59 ± 2.58 B | 4103.22 ± 416 C | 141 |
| RA-10 | 86.44 ± 8.4 DE | 130 | 21.57 ± 4.78 EF | 110 | 7 CD | 25.48 ± 2.93 DEF | 9.87 ± 3.09 CD | 7.85 ± 2.77 B | 3805.54 ± 347 D | 131 |
| Full dose | 94.42 ± 9.8 A | 142 | 32.72 ± 5.9 A | 167 | 12 AB | 36.41 ± 2.41 B | 12.92 ± 3.44 AB | 10.9 ± 3.34 A | 4812.96 ± 389 A | 166 |
| Half dose | 86.42 ± 8.1 D | 130 | 24.52 ± 4.9 CDE | 125 | 8 C | 28.55 ± 2.55 CD | 9.96 ± 2.34 CD | 7.41 ± 2.06 B | 3706.56 ± 338 D | 128 |
| Consortium + half dose | 97.83 ± 8.7 A | 147 | 31.18 ± 5.5 AB | 159 | 15 A | 43.27 ± 5.97 A | 14.66 ± 4.54 A | 12.9 ± 2.67 A | 4903.92 ± 416 A | 169 |
| Consortium + full dose | 94.56 ± 8.9 B | 142 | 28.12 ± 5.1 BC | 144 | 14 A | 32.48 ± 4.18 BC | 11.87 ± 4.15 BC | 10.8 ± 2.97 A | 4605.84 ± 347B | 159 |
| CV | 2.84 | 9.14 | 2.99 | 9.84 | 14.24 | 14.86 | 21.64 | |||
| p-value | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | 0.0000 | |||
| Treatments | Plant Height | 1000 Grain wt. | Grain Yield | |||
|---|---|---|---|---|---|---|
| (cm) | (%) | (g) | (%) | (kg ha−1) | (%) | |
| Control | 57.8 I | 100 | 32.53 J | 100 | 3902 I | 100 |
| Half dose | 61.7 I | 108 | 37.09 I | 114 | 4700 G | 120 |
| Full dose | 100.8 BC | 177 | 61.08 D | 188 | 5709 C | 146 |
| RA-2 | 79.3 E | 139 | 55.86 E | 172 | 4977 F | 128 |
| RA-4 | 73.8 FG | 129 | 48.78 H | 150 | 4439 H | 114 |
| RA-6 | 76.9 EF | 135 | 54.22 EF | 167 | 5206 E | 133 |
| RA-7 | 92.9 D | 163 | 53.54 F | 165 | 5452 D | 140 |
| RA-8 | 84.5 EF | 148 | 56.28 G | 173 | 5657 C | 145 |
| RA-10 | 86.1 E | 151 | 48.92 H | 150 | 4728 G | 121 |
| RA-2 + RA-4 + RA-6 | 73.0 H | 128 | 54.91 F | 169 | 5157 E | 132 |
| RA-7, RA-8, RA-10 | 77.8 G | 136 | 53.43 F | 164 | 5127 E | 131 |
| Consortium 1 + half dose | 103.8 AB | 182 | 69.76 BC | 214 | 6337 B | 162 |
| Consortium 1 + Full dose | 97.1 CD | 170 | 68.58 C | 211 | 6256 B | 160 |
| Consortium 2 + half dose | 107.1 A | 188 | 75.94 A | 233 | 6597 A | 169 |
| Consortium 2 + Full dose | 99.7 BC | 175 | 70.64 B | 217 | 6283 B | 161 |
| CV | 1.36 | 1.80 | 1.26 | |||
| p-value | 0.0000 | 0.0000 | 0.0000 | |||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sheirdil, R.A.; Hayat, R.; Zhang, X.-X.; Abbasi, N.A.; Ali, S.; Ahmed, M.; Khattak, J.Z.K.; Ahmad, S. Exploring Potential Soil Bacteria for Sustainable Wheat (Triticum aestivum L.) Production. Sustainability 2019, 11, 3361. https://doi.org/10.3390/su11123361
Sheirdil RA, Hayat R, Zhang X-X, Abbasi NA, Ali S, Ahmed M, Khattak JZK, Ahmad S. Exploring Potential Soil Bacteria for Sustainable Wheat (Triticum aestivum L.) Production. Sustainability. 2019; 11(12):3361. https://doi.org/10.3390/su11123361
Chicago/Turabian StyleSheirdil, Rizwan Ali, Rifat Hayat, Xiao-Xia Zhang, Nadeem Akhtar Abbasi, Safdar Ali, Mukhtar Ahmed, Jabar Zaman Khan Khattak, and Shakeel Ahmad. 2019. "Exploring Potential Soil Bacteria for Sustainable Wheat (Triticum aestivum L.) Production" Sustainability 11, no. 12: 3361. https://doi.org/10.3390/su11123361
APA StyleSheirdil, R. A., Hayat, R., Zhang, X.-X., Abbasi, N. A., Ali, S., Ahmed, M., Khattak, J. Z. K., & Ahmad, S. (2019). Exploring Potential Soil Bacteria for Sustainable Wheat (Triticum aestivum L.) Production. Sustainability, 11(12), 3361. https://doi.org/10.3390/su11123361







