1. Introduction
The reduction in fossil fuel resources and the environmental impacts that are associated with the use of these fuels influence the sustainability of future energy systems. Therefore, alternative energy sources and their more efficient utilization has been a major concern for researchers in recent years. Many consider fuel cells to be promising in this regard, as they use clean energy and have reduced greenhouse gas emission. In addition, the Carnot cycle efficiency does not limit their efficiency and they do not emit pollutants, such as sulfur dioxide and nitrous oxides [
1]. Proton exchange membrane fuel cells (PEMFCs), among other types, are expected to play a major role in the future energy sector due to their high power density, quick starting, low operating temperatures, and simple stack design. However, reducing the capital costs, weight, and volume of these fuel cells is a challenge in making them competitive with traditional power producing systems like internal combustion engines [
2]. In addition, it is necessary to have an appropriate and optimum value of water content inside PEMFCs to retain the durability of these fuel cells and the efficient operation of them. This can be achieved by humidifying the reactant gases before they enter the cell [
3,
4].
Semi-empirical and numerical models have been introduced in the literature in order to predict the variation of voltage with current density of a PEMFC and to understand and improve the performance of PEMFC systems. These models are also capable of predicting the fuel cell performance for engineering systems applications [
5,
6].
This section briefly reviews a selection of previously reported works that are related to the semi-empirical models. Amphlett [
7] proposed a steady-state model for the Ballard Mark IV fuel cell produced by Ballard Power Systems Inc. when considering such operating parameters as the partial pressures at the electrodes, the operating temperature, and the fuel cell current. Some other researchers have used semi-empirical models for simulating PEM fuel cell performance [
8,
9,
10]. The waste heat from these fuel cells is in the range of 50 °C to 100 °C, so that its recovery for performance enhancement is considered to be a technical challenge [
11]. A few papers in the literature have dealt with the utilization of PEMFC waste heat [
12,
13]. Hwang et al. [
14] proposed a combined heat and power (CHP) system that is based on a PEMFC for cogenerating electrical power and hot water and reported a maximum efficiency of 81% for the combined system. Zhang et al. [
15] reported that such bottoming cycles as organic Rankine cycles (ORCs), Kalina cycles, and transcritical carbon dioxide cycles could conveniently use the waste heat from PEMFCs. Zhao et al. [
16] proposed a hybrid power system and concluded that there are optimum values of fuel cell operating pressure at which the efficiencies of both PEMFC and overall system are maximized while using an ORC to recover waste heat from a PEMFC. However, they only carried out energy assessments of the system’s performance. He et al. [
11] proposed an ORC/heat pump combined organic cycle for waste heat recovery from a PEMFC and concluded that an increase of approximately 4.7% in the thermal efficiency is possible for the recovery system. Ahmadi et al. [
10] proposed a hybrid system consisting of liquefied natural gas and transcritical carbon dioxide cycles for waste heat recovery from a PEMFC to improve its performance. They concluded that the system efficiency could increase by 9% when compared to the standalone fuel cell. However, the water management of the PEMFC and the performance of the humidifiers were not separately considered in their work. Additionally, they did not analyze the system performance from the viewpoints of exergy and exergoeconomics. Chang et al. [
17] designed a PEMFC–solar based residential combined cooling, heating, and power (CCHP) system and showed that the CCHP efficiency increases by up to 75.4% and 85.0% in the summer and winter times, respectively. However, the proposed system was not analyzed from the perspective of exergy and exergoeconomics. Chahartaghi et al. [
18] reported energy and exergy efficiencies of 81.6% and 54.6%, respectively, for a CCHP system, but they did not address economic aspects, while using part of the heat rejected from a fuel cell for cooling production. Romdhane et al. [
19] introduced a residential micro-CCHP system, including a low temperature PEMFC (LT-PEMFC) and a single effect lithium bromide-water (LiBr-H
2O) absorption cycle. They only investigated the system from an energy viewpoint, and reported a maximum fuel saving ratio of about 35%. Ebrahimi et al. [
20] carried out energy and exergy analyses for a micro-CCHP system that is based on a LT-PEMFC. A portion of the produced electric power is utilized to drive a thermoelectric cooler. Energy and exergy efficiencies of up to 76.9% and 53.9%, respectively, are calculated for the proposed system, but the economic aspects were not addressed. Arsalis [
21] reviewed micro-CHP systems that are based on fuel cells, and concluded that PEMFCs are the most promising technology for cogeneration configurations.
Exergy analysis accounts for the quantity of energy and its quality, distinguishing it from energy analysis. Although exergy analysis provides a true measure of evaluating thermodynamic inefficiencies of systems, the cost of inefficiencies is an important issue in analyzing system performance. Exergoeconomic analysis assesses this parameter, which is as an exergy-aided method that provides important information regarding a system’s cost formation process [
22]. In recent years, several studies have been carried out on the exergy analysis of the low temperature PEMFC (LT-PEMFC) [
23,
24]. Kazim [
25] conducted an exergy analysis on the PEM fuel cell operating at voltages of 0.5 and 0.6 V. He investigated the effect of fuel cell parameters on exergy efficiency. However, the exergy destruction of the fuel cell and other auxiliary components was not evaluated in their work. Barelli et al. [
26] conducted energy and exergy analyses for a CHP system that is based on a PEM fuel cell for residential applications and reported an exergy efficiency of 58% under optimum operating conditions. In another paper [
27], Barelli et al. compared the energy and exergy performances of two micro CHP units that are comprised of solid oxide and PEM fuel cells. They concluded that the PEMFC based CHP system has higher exergy efficiency, due to its operation at atmospheric pressure and low temperature. Hanapi et al. [
28] presented an exergy analysis for a PEM fuel cell that was employed in a mini urban car. Through a parametric study, they showed that an increase in operating temperature and pressure resulted in a higher value of exergy efficiency. However, they did not study the exergoeconomic performance of the system. Kalinci et al. [
29] proposed a hybrid power system based on a PEMFC while using renewable energies to produce hydrogen and electricity. They performed a thermodynamic analysis using the energy and exergy approaches.
As mentioned earlier, much effort has been devoted to the exergy analysis of small scale LT-PEMFCs. Parametric studies have been performed to investigate the effects of the exergetic efficiency of various operating conditions. However, little data are available in the literature regarding the details of exergy destruction occurring in the various components of PEM hybrid systems. In addition, limited information is available in the literature on the exergoeconomic analysis of these fuel cells. Kazim [
30] performed an exergoeconomic analysis for a 10 kW PEMFC and concluded that increasing the operating pressure and air stoichiometry can improve the fuel cell product exergy cost. In their work, the exergy destruction cost and exergoeconomic factors were not studied. Using a semi empirical model for simulating PEMFC performance, Mert et al. [
2] presented an exergoeconomic analysis of a vehicular PEM fuel cell system. They reported that the fuel cell stack made the highest contribution to the overall cost. These authors did not present the exergy destruction and exergy cost of other components, such as humidifiers and compressor, separately. Suleiman et al. [
31] carried out a thermoeconomic analysis for PEMFC systems that are fuelled with methanol and methane and showed that the system using methane exhibits comparatively lower capital cost. Sayadi et al. [
32] performed an exergoeconomic analysis to address the trade-off among the advantageous and disadvantageous effects of incorporating an expander as part of a PEMFC system, to assist in selecting a proper design concept.
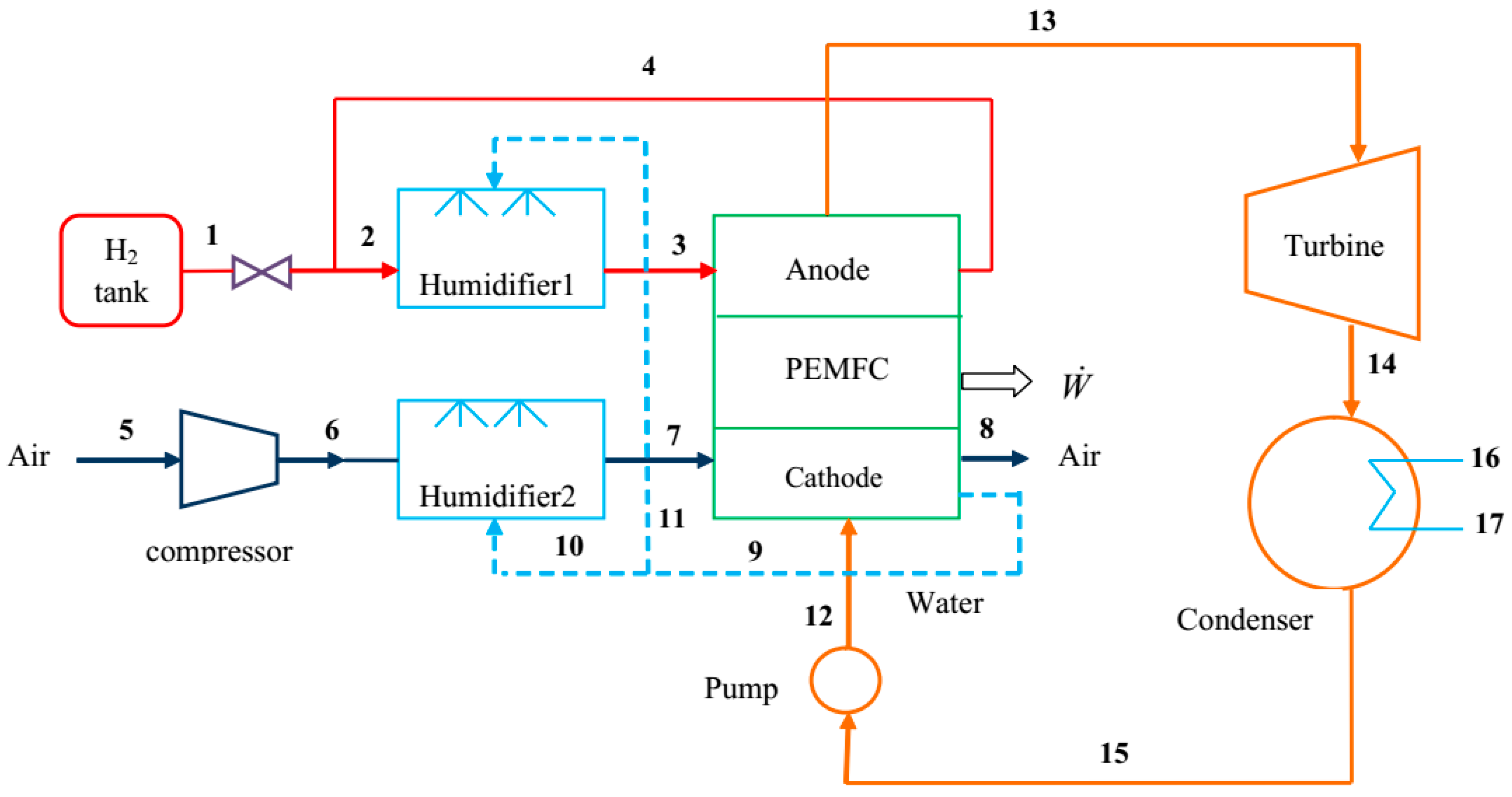
The use of ORC for the recovery of waste heat from energy conversion systems has been shown to be a promising technique in efficiency enhancement. As mentioned before, although the combination of an ORC and a LT-PEMFC has been reported in the literature, detailed exergy and exergoeconomic analyses of this combination are lacking, and inadequate attention has been paid to the modelling of the individual components. Water management in LT-PEMFCs is a crucial issue and it needs more attention. The present work addresses these gaps. A complete mathematical model is presented for simulating the overall system performance. In addition, the developed PEMFC model considers the multi-phase existence of water in the channels, thus more thoroughly investigating the fuel cell phenomena. A parametric analysis is conducted to evaluate the effects of several important parameters on the system’s thermodynamic and economic performances. The objectives are to understand the details of processes occurring in the proposed system and their impacts on its thermodynamic and economic performance.
5. Results and Discussion
Energy, exergy, and exergoeconomic analyses are performed for the proposed system using the input data that are presented in
Table 1 and Engineering Equation Solver (EES) software [
43]. The electrochemical modeling of the PEMFC is validated while using the theoretical and experimental data of Miansari et al. [
8]. The comparison is shown in
Figure 3 and it indicates good agreement between the results in present work and those that are reported in literature. The slight difference between the results from the present model and the experimental data can be attributed to parameters, such as fuel cell voltage loss due to of cell coupling and heat loss, which are neglected in the present work.
Parametric studies are performed to quantify the effects on the proposed system performance of decision parameters, such as the fuel cell current density, fuel cell operating pressure, and ORC turbine pressure.
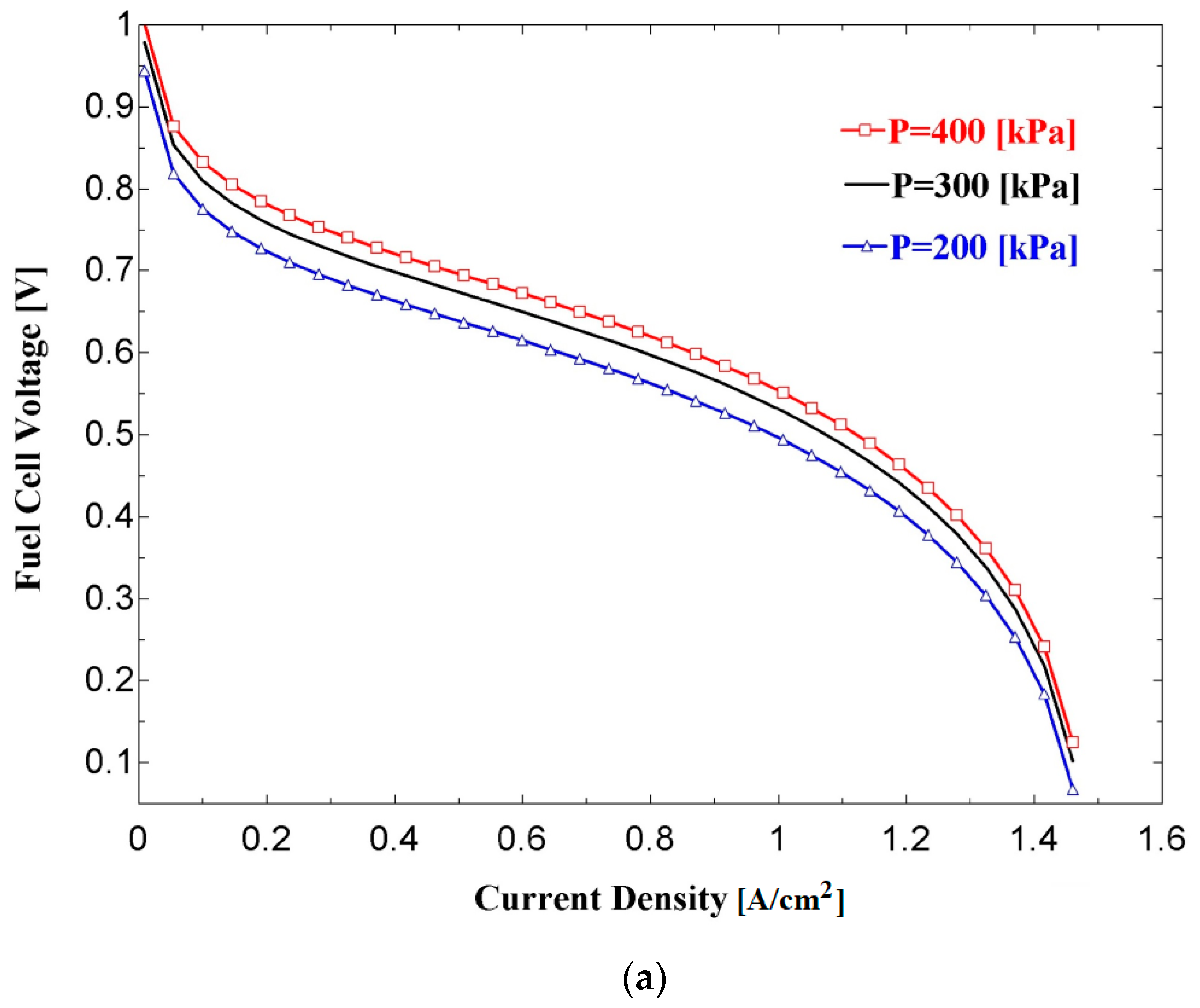
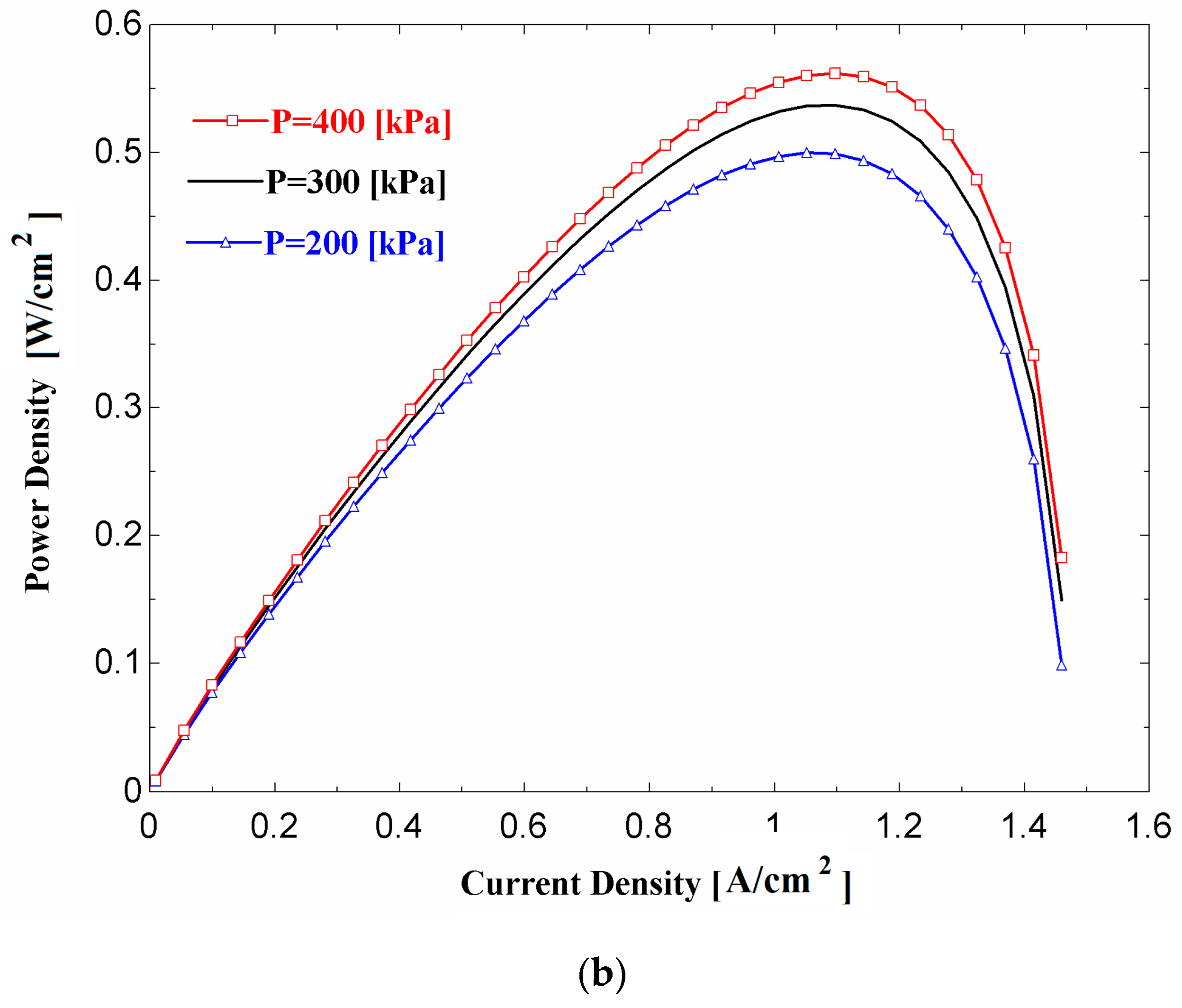
Figure 4a,b show the effects of current density on the fuel cell voltage and power density, respectively, for three values of fuel cell operating pressure. Referring to
Figure 4a, the cell voltage decreases with an increasing current density. However, the slope of the variation is higher at lower and higher values of current density. This voltage-current density characteristic curve results from the combined effects of irreversibilities that are caused by activation, ohmic, and concentration overpotentials.
Figure 4b indicates that, for a given value of cell operating pressure, the stack power density is a maximum at a particular value of current density, and the optimal value of current density is slightly higher at higher values of cell operating pressure.
Figure 4b does not suggest the operation of fuel cell beyond the point at which power is a maximum, as the same output power may be achieved with a lower current density and consequently a higher voltage (see
Figure 3).
Figure 4a,b also show that a higher voltage and power density are achieved with a higher operating pressure. This is due to the improvements in the reversible thermodynamic potential (E
Nernst) and the reductions in the activation losses at the electrodes, especially at the cathode.
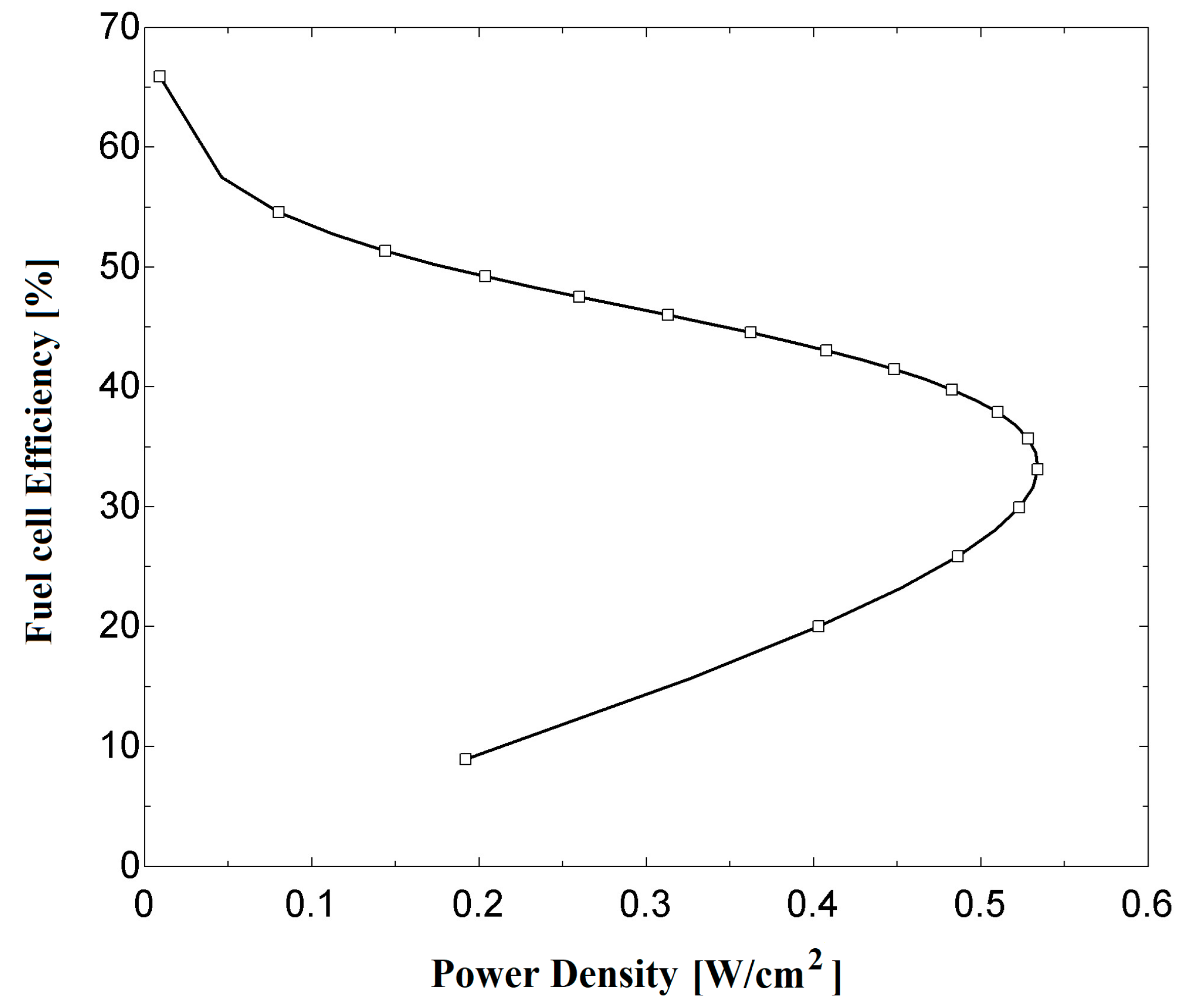
Figure 5 shows the relation between fuel cell (as a standalone system) efficiency and power density. When referring to
Figure 4b, the maximum power density is around 0.53 W/cm
2 for an operating pressure of 300 kPa. With this value of power density, an efficiency of 33.5% is obtained from
Figure 5. This efficiency is much lower than the maximum theoretical value, which is around 80%. Referring to
Figure 5, much higher efficiencies may be obtained at significantly lower power densities. This indicates that, for a given value output power, choosing an appropriate point on the polarization curve (
Figure 4a) could result in a larger fuel cell (with a larger active area) with higher efficiency or a compact fuel cell with less efficiency. In the literature, it has been reported that the maximum power density is not recommended in fuel cell sizing [
44]. An operating point corresponding to a cell potential of around 0.65–0.7 V is common practice. Therefore, this results in a power density of 0.4 W cm
−2 and an efficiency of around 44% for the proposed system at the operating condition.
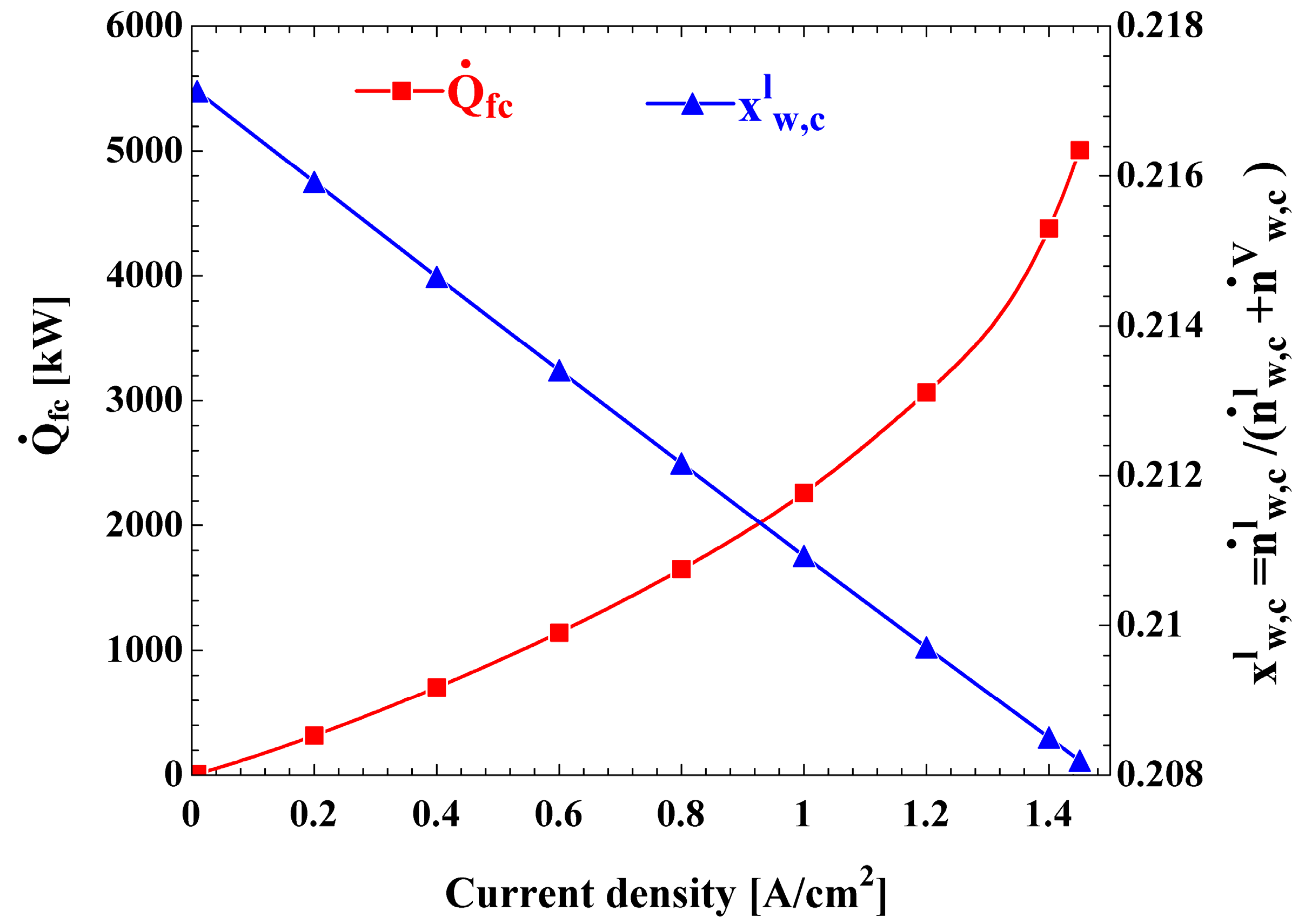
The effects on the heat rejected from the fuel cell
and the mole fraction of liquid water at the cathode outlet
are shown in
Figure 6 of the current density. It is observed that, with increasing current density, the
considerably increases and the
slightly decreases.
Figure 7 shows the effects of current density on exergy efficiency and net power output of the proposed system. Referring to
Figure 7, the exergy efficiency drops as the current density increases. This trend can be explained by noting that, as the current density increases, the heat that is generated by fuel cell rises, so that the heat provided to the bottom cycle increases as more power is produced by the ORC. Additionally, referring to
Figure 4b, there is a maximum value for the fuel cell output power as the current density changes. Therefore, as shown in
Figure 7, the net output power is maximized at a particular value of current density. However, the amount of fuel input to the system and, subsequently the input exergy rate (
), increases with increasing current density. In addition, the rate of increase in input exergy is higher than the rate of increase in system net power output at a current density less than the optimum value. Therefore, a decrease of exergy efficiency with current density is expected (see Equation (56)). For current densities that are higher than the optimum value, both
and
decrease, resulting in a reduction in exergy efficiency (see Equation (56)).
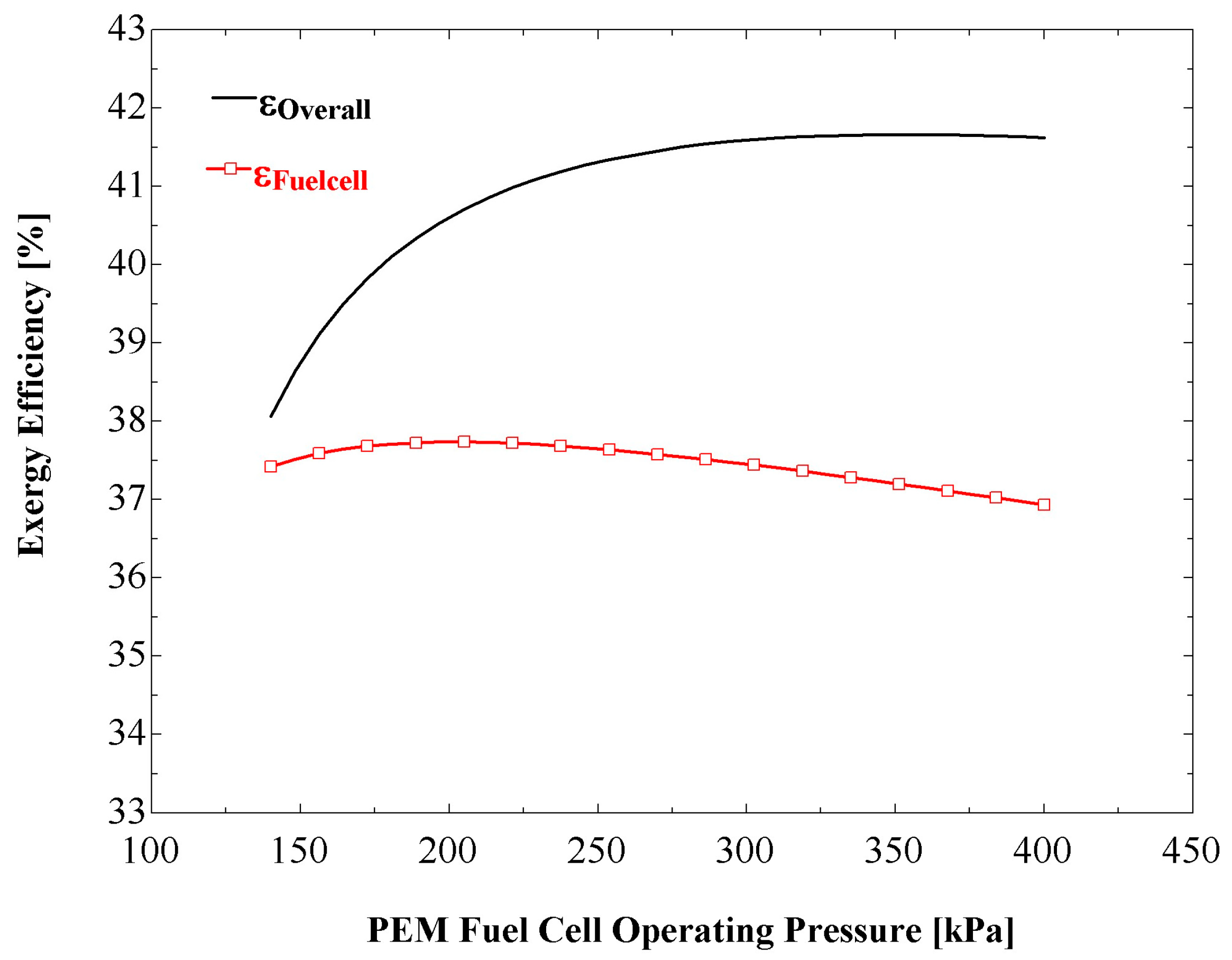
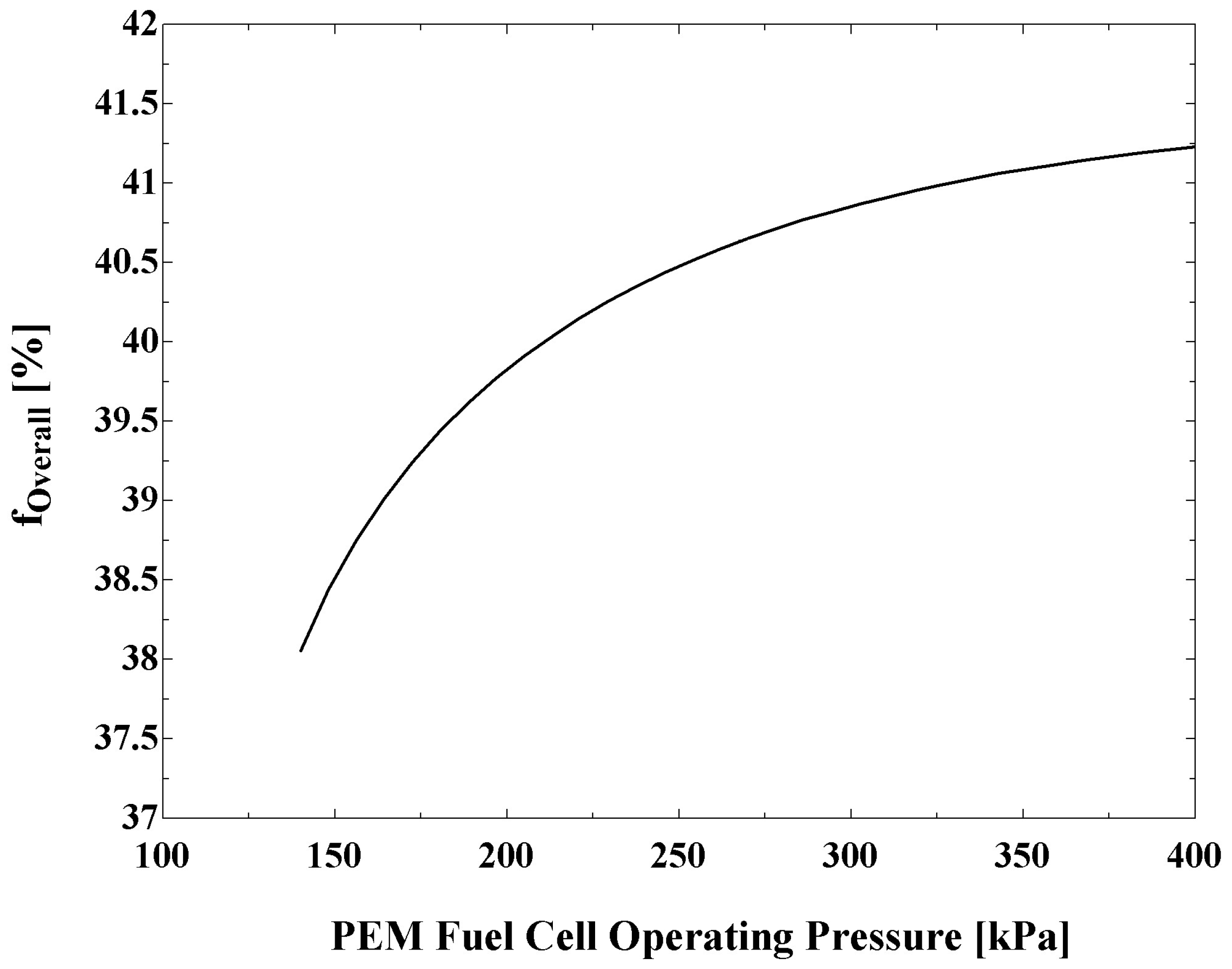
Figure 8 shows the influences on the overall and fuel cell exergy efficiencies of the fuel cell operating pressure. It is observed that an increase in the operating pressure increases the overall exergy efficiency. This is expected, as the increase in operating pressure results in an increased value of net output power.
Figure 8 also indicates that the fuel cell exergy efficiency attains a maximum value at a particular operating pressure. Note that, at a fuel cell operating pressure of 300 kPa, the exergy efficiency of overall system is about four percent points greater than for a standalone PEM fuel cell system.
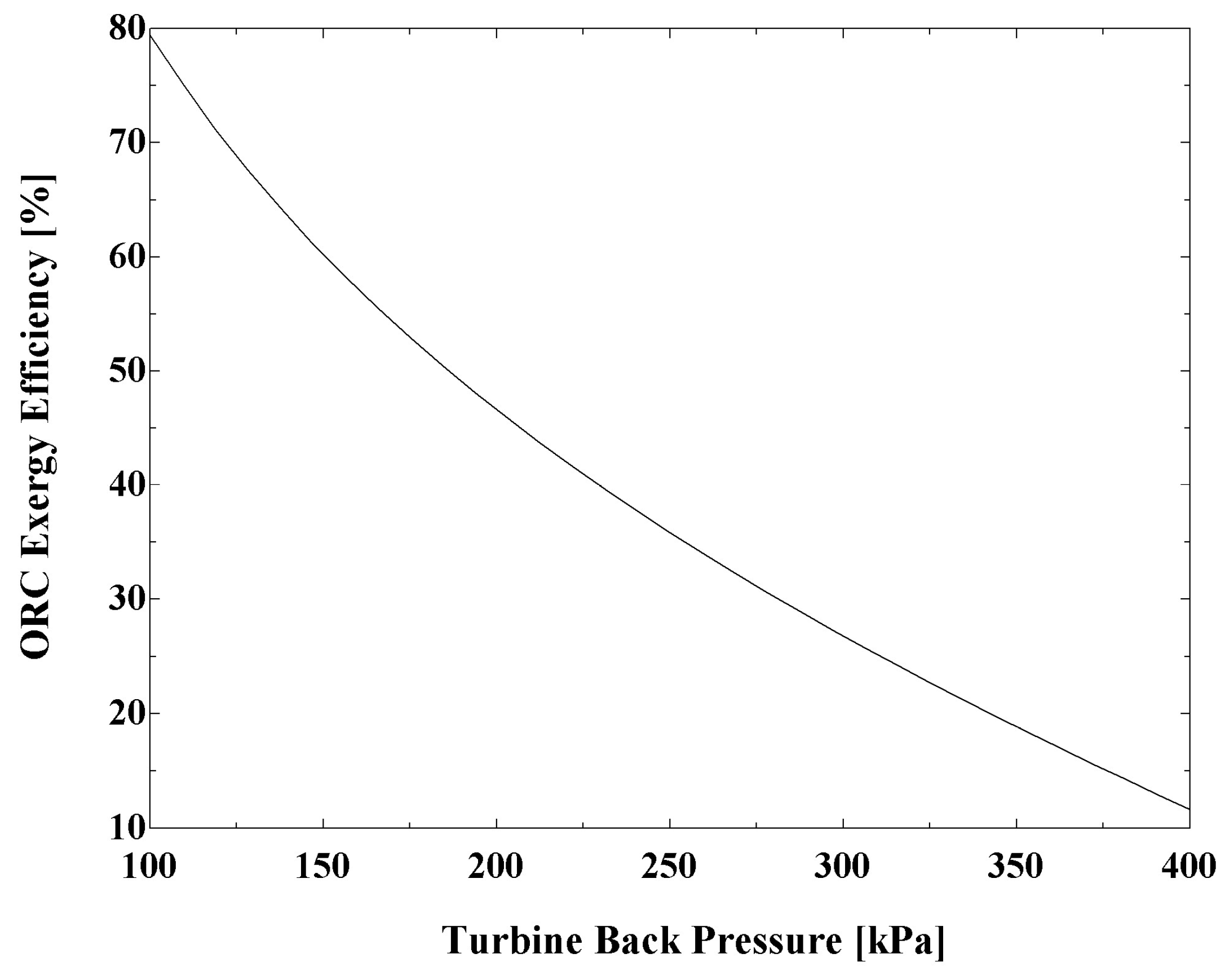
Figure 9 presents the effect of turbine back pressure on ORC exergy efficiency. It confirms the reduction of exergy efficiency with increasing turbine back pressure.
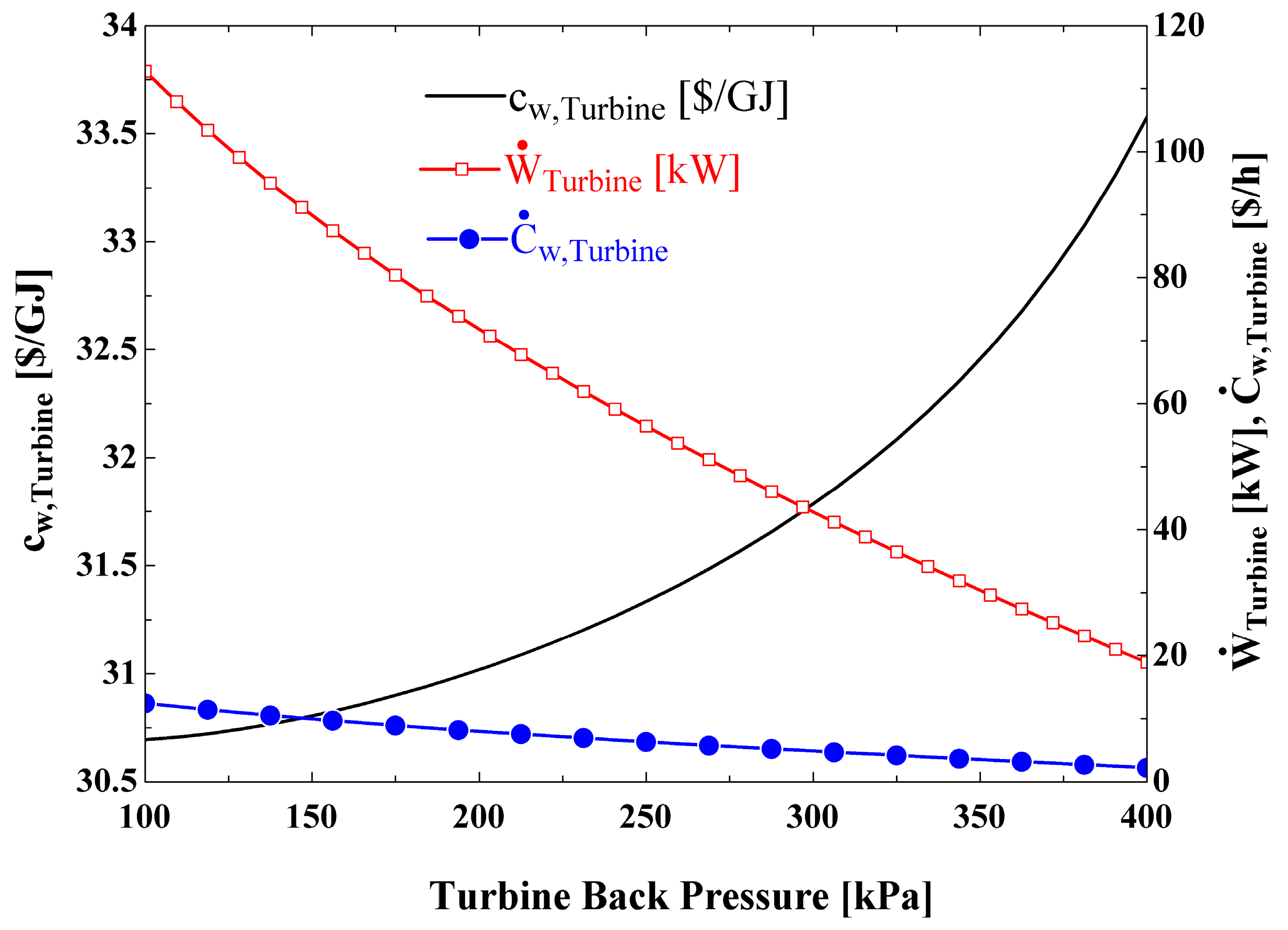
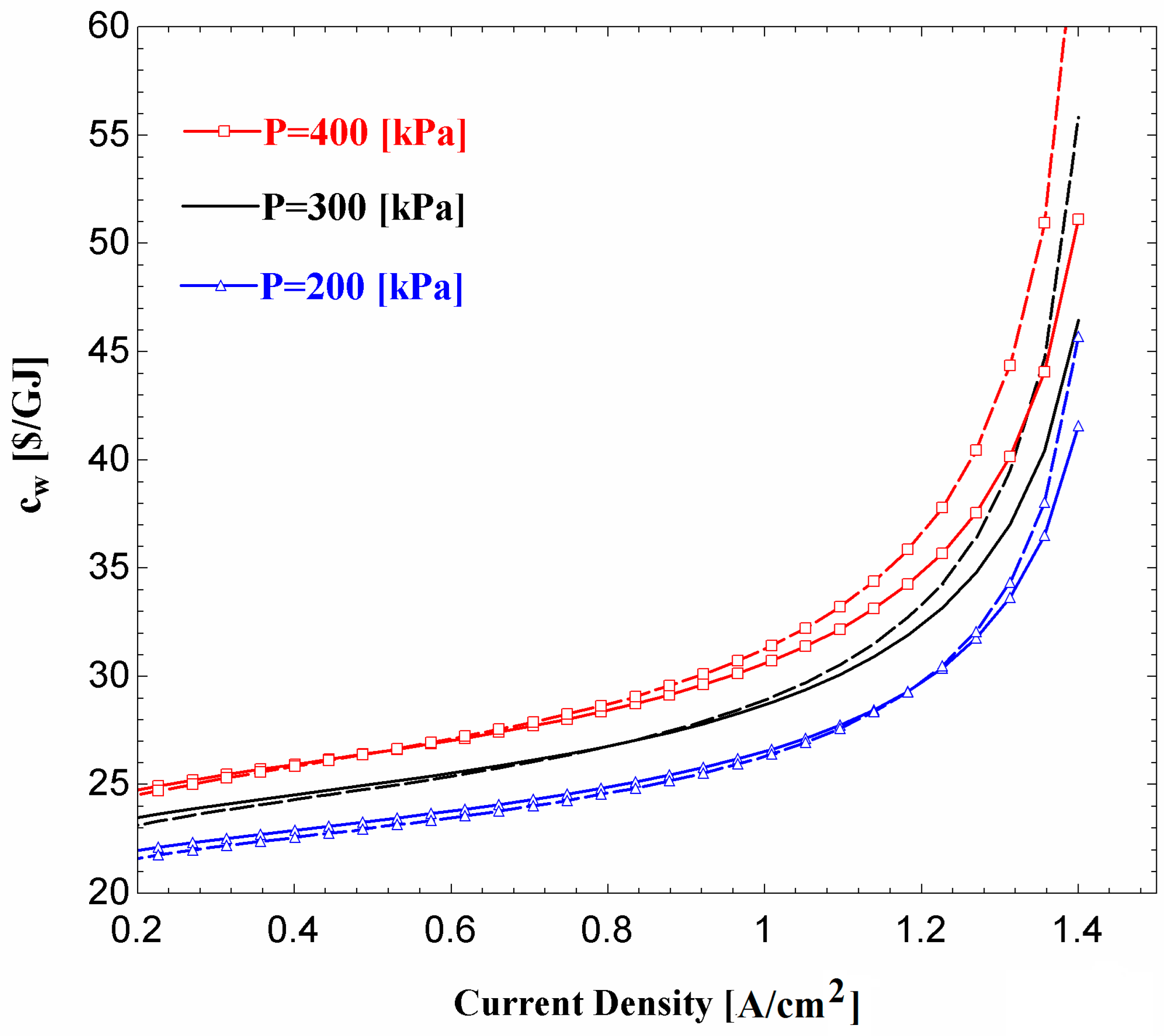
Figure 10 shows the variations in cost per unit of electrical power produced by the turbine (
), turbine power cost rate (
), and the turbine generated power as the turbine back pressure changes. It is observed that the turbine power output and
decrease as turbine back pressure increases. However, the decrease in
is dominant, so that the net effect is an increase in
as the pressure rises (
).
Figure 11 shows the effect of current density on the unit cost of fuel cell net output power (
) and unit cost of the overall system net output power (
), respectively, for three values of fuel cell operating pressure. It is observed that
and
increase with an increase in the current density. Referring to
Figure 11, regardless of the operating pressure, little difference is observed between
and
when the current density is less than 0.8 A/cm
2. At higher current densities, the addition of the ORC to the fuel cell results in a lower value of the product unit cost. The difference between the two product unit costs is more prominent at higher values of the fuel cell operating pressure. A comparison between the results in
Figure 7 and
Figure 10 shows that the operating pressure of 300 kPa is better than the other two pressure values that are indicated in
Figure 11. The reasoning is as follows: comparing the results for the operating pressures of 300 kPa and 400 kPa it is observed that
does not change much, but
is higher for 400 kPa. In addition, although a lower value of
is obtained for P = 200 kPa,
for this operating pressure is much less than that for P = 300 kPa.
Figure 12 depicts the variations in
and
with fuel cell current density, where it is observed that the
increases with an increase in the current density and
is maximized at a specific value of the current density. The increase in
is due to the increase in exergy destruction and its associated cost for all of the components, particularly for the PEMFC component, which contributes the most to the overall exergy destruction. The trend of
is similar to that of
, since the contribution of PEMFC is very high in the overall capital cost rate.
Figure 13 shows the variations in exergoeconomic factor,
, with PEMFC operating pressure. An increase in the operating pressure is seen to lead to a higher value of
, mainly due to the increase in owning and operating costs.
The solution to the system of cost balance and auxiliary equations determines the unit exergy cost at different points in the system.
Table 6 gives the values of important exergy and exergoeconomic factors for the system. Referring to
Table 6, the PEMFC stack, turbine, and air humidifier, among other system components, have the highest values of the sum
and are, therefore, the most important components from a thermoeconomic viewpoint. The air compressor, condenser, and air humidifier exhibit the lowest f values. Therefore, regarding these components, selecting more expensive components are beneficial for better exergoeconomic system performance. In addition, the exergoeconomic factor of 41% that was obtained for the PEMFC indicates that the cost associated with exergy destruction for this component greatly exceeds the cost of its ownership and operation. The high exergy destruction in the fuel cell is mainly due to the irreversible nature of chemical reactions and voltage losses occurring in this component. An increase in the PEMFC operating pressure and air stoichiometry rate causes a decrease in the exergy destruction of the PEMFC and also in the associated cost, i.e., a higher operating pressure brings about a higher exergoeconomic factor for the PEMFC. For the turbine, the exergy efficiency is 86.0% and the exergoeconomic factor 52.1%. Therefore, the exergy and exergoeconomic performances of this component are satisfactory. The overall value of exergoeconomic factor for the proposed system is determined to be 40.8%. Thus, approximately 59% of the total cost rate of the system is due to exergy destruction. Therefore, in general, better exergoeconomic system performance is achieved with more expensive components.