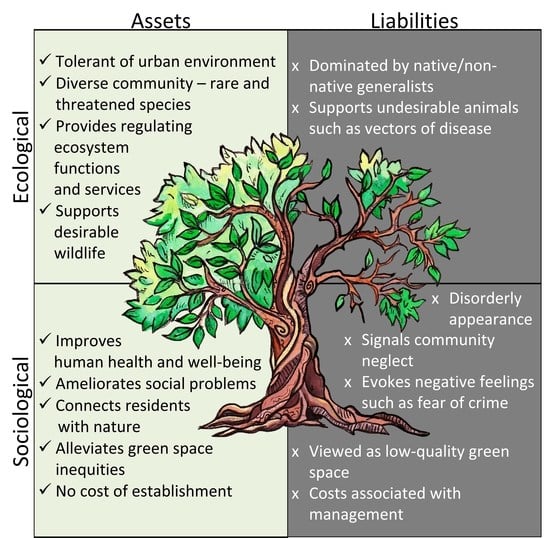
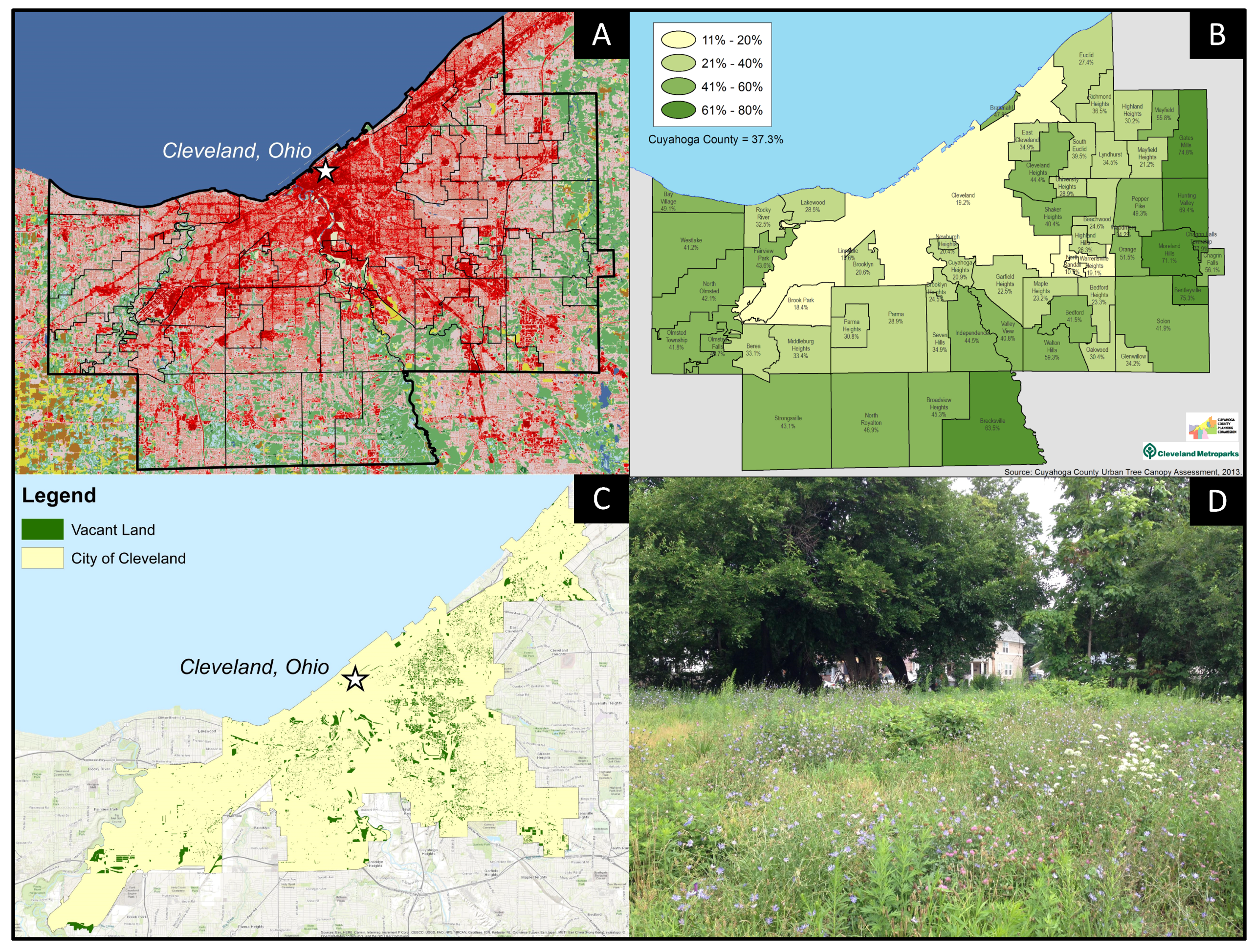
Asset or Liability? Ecological and Sociological Tradeoffs of Urban Spontaneous Vegetation on Vacant Land in Shrinking Cities
Abstract
1. Introduction
2. Ecological Considerations of USV
3. Sociological Considerations of USV
4. Conclusions and Future Research Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bernt, M.; Cocks, M.; Couch, C.; Grossmann, K.; Haase, A.; Dieter, R. Policy Response, Governance and Future Directions. 2012, pp. 1–17. Available online: https://www.ufz.de/export/data/400/39031_ResearchBrief2_.pdf (accessed on 4 April 2018).
- Buhnik, S. From shrinking cities to Toshi no Shukushō: Identifying patterns of urban shrinkage in the Osaka Metropolitan Area. Berkeley Plan. J. 2010, 23, 132–155. [Google Scholar] [CrossRef]
- Wiechmann, T. Errors expected—Aligning urban strategy with demographic uncertainty in shrinking cities. Int. Plan. Stud. 2008, 13, 431–446. [Google Scholar] [CrossRef]
- Schatz, L.K. What Helps or Hinders the Adoption of “Good Planning” Principles in Shrinking Cities? A Comparison of Recent Planning Exercises in Sudbury, Ontario and Youngstown, Ohio. Ph.D. Thesis, University of Waterloo, Waterloo, ON, Canada, 2010. [Google Scholar]
- Haase, D. Chapter 12: Shrinking cities, biodiversity and ecosystem services. In Urbanization, Biodiversity and Ecosystem Services: Challenges and Opportunities: A Global Assessment; Springer: Dordrecht, The Netherlands, 2013. [Google Scholar]
- Herrmann, D.L.; Shuster, W.D.; Mayer, A.L.; Garmestani, A.S. Sustainability for shrinking cities. Sustainability 2016, 8, 911. [Google Scholar] [CrossRef]
- Haase, D.; Haase, A.; Rink, D. Conceptualizing the nexus between urban shrinkage and ecosystem services. Landsc. Urban Plan. 2014, 132, 159–169. [Google Scholar] [CrossRef]
- Hollander, J.B.; Pallagst, K.; Schwarz, T.; Popper, F.J.; Hollander, J.B. Planning shrinking cities. Prog. Plan. 2009, 72, 223–232. [Google Scholar]
- Martinez-Fernandez, C.; Audirac, I.; Fol, S.; Cunningham-Sabot, E. Shrinking cities: Urban challenges of globalization. Int. J. Urban Reg. Res. 2012, 36, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, D.L.; Schwarz, K.; Shuster, W.D.; Berland, A.; Chaffin, B.C.; Garmestani, A.S.; Hopton, M.E. Ecology for the shrinking city. Bioscience 2016, 66, 965–973. [Google Scholar] [CrossRef]
- Rupprecht, C. Informal urban green space: Residents’ perception, use, and management preferences across four major Japanese shrinking cities. Land 2017, 6, 59. [Google Scholar] [CrossRef]
- Kattwinkel, M.; Biedermann, R.; Kleyer, M. Temporary conservation for urban biodiversity. Biol. Conserv. 2011, 144, 2335–2343. [Google Scholar] [CrossRef]
- Robinson, S.L.; Lundholm, J.T. Ecosystem services provided by urban spontaneous vegetation. Urban Ecosyst. 2012, 15, 545–557. [Google Scholar] [CrossRef]
- Kühn, N. Intentions for the unintentional: Spontaneous vegetation as the basis for innovative planting design in urban areas. J. Landsc. Archit. 2006, 1, 46–53. [Google Scholar] [CrossRef]
- Kim, G.; Miller, P.A.; Nowak, D.J. Urban vacant land typology: A tool for managing urban vacant land. Sustain. Cities Soc. 2018, 36, 144–156. [Google Scholar] [CrossRef]
- Kim, G. The public value of urban vacant land: Social responses and ecological value. Sustainability 2016, 8, 486. [Google Scholar] [CrossRef]
- Gardiner, M.M.; Burkman, C.E.; Prajzner, S.P. The value of urban vacant land to support arthropod biodiversity and ecosystem services. Environ. Entomol. 2013, 42, 1123–1136. [Google Scholar] [CrossRef] [PubMed]
- Bonthoux, S.; Brun, M.; Di Pietro, F.; Greulich, S.; Bouche-Pillon, S. How can wastelands promote biodiversity in cities? A review. Landsc. Urban Plan. 2014, 132, 79–88. [Google Scholar] [CrossRef]
- Kowarik, I. Novel urban ecosystems, biodiversity, and conservation. Environ. Pollut. 2011, 159, 1974–1983. [Google Scholar] [CrossRef] [PubMed]
- Rupprecht, C.D.D.; Byrne, J.A.; Garden, J.G.; Hero, J.M. Informal urban green space: A trilingual systematic review of its role for biodiversity and trends in the literature. Urban For. Urban Green. 2015, 14, 883–908. [Google Scholar] [CrossRef]
- Rupprecht, C.D.D.; Byrne, J.A. Informal urban greenspace: A typology and trilingual systematic review of its role for urban residents and trends in the literature. Urban For. Urban Green. 2014, 13, 597–611. [Google Scholar] [CrossRef]
- Threlfall, C.G.; Kendal, D. The distinct ecological and social roles that wild spaces play in urban ecosystems. Urban For. Urban Green. 2018, 29, 348–356. [Google Scholar] [CrossRef]
- Hobbs, R.J.; Higgs, E.; Hall, C.M.; Bridgewater, P.; Chapin, F.S.; Ellis, E.C.; Ewel, J.J.; Hallett, L.M.; Harris, J.; Hulvey, K.B.; et al. Managing the whole landscape: Historical, hybrid, and novel ecosystems. Front. Ecol. Environ. 2014, 12, 557–564. [Google Scholar] [CrossRef]
- Hobbs, R.J.; Arico, S.; Aronson, J.; Baron, J.S.; Bridgewater, P.; Cramer, V.A.; Epstein, P.R.; Ewel, J.J.; Klink, C.A.; Lugo, A.E.; et al. Novel ecosystems: Theoretical and management aspects of the new ecological world order. Glob. Ecol. Biogeogr. 2006, 15, 1–7. [Google Scholar] [CrossRef]
- Kowarik, I. Wild urban woodlands: Towards a conceptual framework. In Wild Urban Woodlands; Springer: Berlin/Heidelberg, Germany, 2005; Chapter 1. [Google Scholar]
- Kowarik, I. Urban wilderness: Supply, demand, and access. Urban For. Urban Green. 2018, 29, 336–347. [Google Scholar] [CrossRef]
- Jorgensen, A.; Keenen, R. (Eds.) Urban Wildscapes; Routledge: Abingdon, UK, 2012. [Google Scholar]
- Konijnendijk, C.C. Between fascination and fear—The impacts of urban wilderness on human health and wellbeing. Socialmed. Tidskr. 2012, 89, 289–295. [Google Scholar]
- Bowman, A.O. Terra incognita: Vacant Land and Urban Strategies; Brookings Instituion, Center on Urban and Metropolitan Policy: Washington, DC, USA, 2004. [Google Scholar]
- Pagano, M.A.; Bowman, A.O. Vacant Land in Cities: An Urban Resource; Brookings Institution: Washington, DC, USA, 2000. [Google Scholar]
- Grădinaru, S.R.; Iojă, C.I.; Onose, D.A.; Gavrilidis, A.A.; Pătru-Stupariu, I.; Kienast, F.; Hersperger, A.M. Land abandonment as a precursor of built-up development at the sprawling periphery of former socialist cities. Ecol. Indic. 2015, 57, 305–313. [Google Scholar] [CrossRef]
- Millard, A. The potential role of natural colonisation as a design tool for urban forestry a pilot study. Landsc. Urban Plan. 2000, 52, 173–179. [Google Scholar] [CrossRef]
- Grimm, N.B.; Faeth, S.H.; Golubiewski, N.E.; Redman, C.L.; Wu, J.; Bai, X.; Briggs, J.M. Global change and the ecology of cities. Science 2008, 319, 756–760. [Google Scholar] [CrossRef] [PubMed]
- Sukopp, H. Human-caused impact on preserved vegetation. Landsc. Urban Plan. 2004, 68, 347–355. [Google Scholar] [CrossRef]
- Hwang, Y.H.; Yue, Z.E.J.; Tan, Y.C. Observation of floristic succession and biodiversity on rewilded lawns in a tropical city. Landsc. Res. 2017, 42, 106–119. [Google Scholar] [CrossRef]
- Albrecht, H.; Eder, E.; Langbehn, T.; Tschiersch, C. The soil seed bank and its relationship to the established vegetation in urban wastelands. Landsc. Urban Plan. 2011, 100, 87–97. [Google Scholar] [CrossRef]
- Schröder, R.; Glandorf, S.; Kiehl, K. Temporal revegetation of a demolition site—A contribution to urban restoration? J. Urban Ecol. 2018, 4, 1–10. [Google Scholar] [CrossRef]
- Rebele, F.; Lehmann, C. Twenty years of woodland establishment through natural succession on a sandy landfill site in Berlin, Germany. Urban For. Urban Green. 2016, 18, 182–189. [Google Scholar] [CrossRef]
- Muratet, A.; Machon, N.; Jiguet, F.; Moret, J.; Porcher, E. The role of urban structures in the distribution of wasteland flora in the Greater Paris area, France. Ecosystems 2007, 10, 661–671. [Google Scholar] [CrossRef]
- Prach, K.; Pyšek, P. Using spontaneous succession for restoration of human-disturbed habitats: Experience from Central Europe. Ecol. Eng. 2001, 17, 55–62. [Google Scholar] [CrossRef]
- Meffert, P.J. Birds on urban wastelands. In Ecology and Conservation of Birds in Urban Environments; Springer: Berlin/Heidelberg, Germany, 2017; Chapter 19; pp. 399–411. ISBN 9783319433141. [Google Scholar]
- Eyre, M.D.; Luff, M.L.; Woodward, J.C. Beetles (Coleoptera) on brownfield sites in England: An important conservation resource? J. Insect Conserv. 2003, 7, 223–231. [Google Scholar] [CrossRef]
- Small, E.C.; Sadler, J.P.; Telfer, M.G. Carabid beetle assemblages on urban derelict sites in Birmingham, UK. J. Insect Conserv. 2003, 6, 233–246. [Google Scholar] [CrossRef]
- Delgado de la Flor, Y.A.; Burkman, C.E.; Eldredge, T.K.; Gardiner, M.M. Patch and landscape-scale variables influence the taxonomic and functional composition of beetles in urban greenspaces. Ecosphere 2017, 8, 1–17. [Google Scholar] [CrossRef]
- Riley, C.B.; Herms, D.A.; Gardiner, M.M. Exotic trees contribute to urban forest diversity and ecosystem services in inner-city Cleveland, OH. Urban For. Urban Green. 2017, 1–10. [Google Scholar] [CrossRef]
- Tredici, P. Del Spontaneous Urban Vegetation: Reflections of Change in a Globalized World. Nat. Cult. 2010, 5, 299–315. [Google Scholar] [CrossRef]
- Jennings, V.; Larson, L.; Yun, J. Advancing sustainability through urban green space: Cultural ecosystem services, equity, and social determinants of health. Int. J. Environ. Res. Public Health 2016, 13, 196. [Google Scholar] [CrossRef] [PubMed]
- Hartig, T.; Mitchell, R.; de Vries, S.; Frumkin, H. Nature and Health. Annu. Rev. Public Health 2014, 35, 207–228. [Google Scholar] [CrossRef] [PubMed]
- De Vries, S.; Verheij, R.A.; Groenewegen, P.P.; Spreeuwenberg, P. Natural environments—Healthy environments? An exploratory analysis of the relationship between greenspace and health. Environ. Plan. A 2003, 35, 1717–1731. [Google Scholar] [CrossRef]
- Wolch, J.R.; Byrne, J.; Newell, J.P. Urban green space, public health, and environmental justice: The challenge of making cities “just green enough”. Landsc. Urban Plan. 2014, 125, 234–244. [Google Scholar] [CrossRef]
- Brown, C.; Grant, M. Biodiversity and human health: What role for nature in healthy urban planning? Built Environ. 2005, 31, 326–338. [Google Scholar] [CrossRef]
- Sandifer, P.A.; Sutton-Grier, A.E.; Ward, B.P. Exploring connections among nature, biodiversity, ecosystem services, and human health and well-being: Opportunities to enhance health and biodiversity conservation. Ecosyst. Serv. 2015, 12, 1–15. [Google Scholar] [CrossRef]
- Kamvasinou, K. The public value of vacant urban land. Munic. Eng. 2011, 164, 157–166. [Google Scholar] [CrossRef]
- Pyle, R.M. Eden in a vacant lot: Special places, species, and kids in the neighborhood of life. In Children and Nature: Psychological, Sociocultural, and Evolutionary Investigations; MIT Press: Cambridge, MA, USA, 2002; Chapter 12; pp. 305–327. [Google Scholar]
- Schwarz, K.; Berland, A.; Herrmann, D.L. Green, but not just? Rethinking environmental justice indicators in shrinking cities. Sustain. Cities Soc. 2018, in press. [Google Scholar]
- Dunnett, N.; Nagase, A.; Hallam, A. The dynamics of planted and colonising species on a green roof over six growing seasons 2001–2006: Influence of substrate depth. Urban Ecosyst. 2008, 11, 373–384. [Google Scholar] [CrossRef]
- Hwang, Y.H.; Yue, Z.E.J. Observation of biodiversity on minimally managed green roofs in a tropical city. J. Living Archit. 2015, 2, 9–26. [Google Scholar] [CrossRef]
- Dickie, I.A.; Bennett, B.M.; Burrows, L.E.; Nuñez, M.A.; Peltzer, D.A.; Porté, A.; Richardson, D.M.; Rejmánek, M.; Rundel, P.W.; van Wilgen, B.W. Conflicting values: Ecosystem services and invasive tree management. Biol. Invasions 2014, 16, 705–719. [Google Scholar] [CrossRef]
- Lyytimäki, J.; Sipilä, M. Hopping on one leg—The challenge of ecosystem disservices for urban green management. Urban For. Urban Green. 2009, 8, 309–315. [Google Scholar] [CrossRef]
- Goldstein, J.; Jensen, M.; Reiskin, E. Urban Vacant Land Redevelopment: Challenges and Progress; Lincoln Institute of Land Policy: Cambridge, MA, USA, 2001; Volume 37. [Google Scholar]
- Draus, P.J.; Roddy, J.; McDuffie, A. ‘We don’t have no neighbourhood’: Advanced marginality and urban agriculture in Detroit. Urban Stud. 2014, 51, 2523–2538. [Google Scholar] [CrossRef]
- Lafortezza, R.; Corry, R.C.; Sanesi, G.; Brown, R.D. Visual preference and ecological assessments for designed alternative brownfield rehabilitations. J. Environ. Manag. 2008, 89, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Mathey, J.; Arndt, T.; Banse, J.; Rink, D. Public perception of spontaneous vegetation on brownfields in urban areas—Results from surveys in Dresden and Leipzig (Germany). Urban For. Urban Green. 2018, 29, 384–392. [Google Scholar] [CrossRef]
- Brun, M.; Di, F.; Bonthoux, S. Residents’ perceptions and valuations of urban wastelands are influenced by vegetation structure. Urban For. Urban Green. 2018, 29, 393–403. [Google Scholar] [CrossRef]
- Pickett, S.T.A.; Cadenasso, M.L.; Childers, D.L.; McDonnell, M.J.; Zhou, W. Evolution and future of urban ecological science: Ecology in, of, and for the city. Ecosyst. Health Sustain. 2016, 2, e01229. [Google Scholar] [CrossRef]
- Williams, N.S.G.; Schwartz, M.W.; Vesk, P.A.; McCarthy, M.A.; Hahs, A.K.; Clemants, S.E.; Corlett, R.T.; Duncan, R.P.; Norton, B.A.; Thompson, K.; et al. A conceptual framework for predicting the effects of urban environments on floras. J. Ecol. 2009, 97, 4–9. [Google Scholar] [CrossRef]
- Pickett, S.T.A.; Cadenasso, M.L.; Grove, J.M.; Boone, C.G.; Groffman, P.M.; Irwin, E.; Kaushal, S.S.; Marshall, V.; McGrath, B.P.; Nilon, C.H.; et al. Urban ecological systems: Scientific foundations and a decade of progress. J. Environ. Manag. 2011, 92, 331–362. [Google Scholar] [CrossRef] [PubMed]
- George, K.; Ziska, L.H.; Bunce, J.A.; Quebedeaux, B.; Hom, J.L.; Wolf, J.; Teasdale, J.R. Macroclimate associated with urbanization increases the rate of secondary succession from fallow soil. Oecologia 2009, 159, 637–647. [Google Scholar] [CrossRef] [PubMed]
- Gregg, J.W.; Jones, C.G.; Dawson, T.E. Urbanization effects on tree growth in the vicinity of New York City. Earth 2003, 424, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Ziska, L.H.; Gebhard, D.E.; Frenz, D.A.; Faulkner, S.; Singer, B.D.; Straka, J.G. Cities as harbingers of climate change: Common ragweed, urbanization, and public health. J. Allergy Clin. Immunol. 2003, 111, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Williams, N.S.G.; Hahs, A.K.; Vesk, P.A. Urbanisation, plant traits and the composition of urban floras. Perspect. Plant Ecol. Evol. Syst. 2015, 17, 78–86. [Google Scholar] [CrossRef]
- Lososová, Z.; Chytrý, M.; Kühn, I.; Hájek, O.; Horáková, V.; Pyšek, P.; Tichý, L. Patterns of plant traits in annual vegetation of man-made habitats in central Europe. Perspect. Plant Ecol. Evol. Syst. 2006, 8, 69–81. [Google Scholar] [CrossRef]
- Aronson, M.F.; Nilon, C.H.; Lepczyk, C.A.; Parker, T.S.; Warren, P.S.; Cilliers, S.S.; Goddard, M.A.; Hahs, A.K.; Herzog, C.; Katti, M.; et al. Hieracrhical filters determine community assembly of urban species pools. Ecology 2016, 86, 4–19. [Google Scholar] [CrossRef]
- Gilbert, O. The Ecology of Urban Habitats; Springer Science & Business Media: Berlin, Germany, 2012. [Google Scholar]
- Deng, H.; Jim, C.Y. Spontaneous plant colonization and bird visits of tropical extensive green roof. Urban Ecosyst. 2017, 20, 337–352. [Google Scholar] [CrossRef]
- Li, X.; Yin, X.; Wang, Y. Diversity and ecology of vascular plants established on the extant world-longest ancient city wall of Nanjing, China. Urban For. Urban Green. 2016, 18, 41–52. [Google Scholar] [CrossRef]
- El-Ghani, M.A.; Bornkamm, R.; El-Sawaf, N.; Turky, H. Plant species distribution and spatial habitat heterogeneity in the landscape of urbanizing desert ecosystems in Egypt. Urban Ecosyst. 2011, 14, 585–616. [Google Scholar] [CrossRef]
- Schwoertzig, E.; Ertlen, D.; Trémolières, M. Are plant communities mainly determined by anthropogenic land cover along urban riparian corridors? Urban Ecosyst. 2016, 19, 1767–1786. [Google Scholar] [CrossRef]
- Cilliers, S.S.; Bredenkamp, G.J. Analysis of the spontaneous vegetation of intensively managed urban open spaces in the Potchefstroom Municipal Area, North West Province, South Africa. S. Afr. J. Bot. 1999, 65, 59–68. [Google Scholar] [CrossRef]
- Müller, A.; Bøcher, P.K.; Fischer, C.; Svenning, J.C. ‘Wild’ in the city context: Do relative wild areas offer opportunities for urban biodiversity? Landsc. Urban Plan. 2018, 170, 256–265. [Google Scholar] [CrossRef]
- Maurer, U.; Peschel, T.; Schmitz, S. The flora of selected urban land-use types in Berlin and Potsdam with regard to nature conservation in cities. Landsc. Urban Plan. 2000, 46, 209–215. [Google Scholar] [CrossRef]
- McKinney, M.L. Effects of urbanization on species richness: A review of plants and animals. Urban Ecosyst. 2008, 11, 161–176. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Mooney, H.A.; Lubchenco, J.; Melillo, J.M. Human domination of Earth’s ecosystems. Science 1997, 277, 494–499. [Google Scholar] [CrossRef]
- Pyšek, P. Alien and native species in Central European urban floras: A quantitative comparison. J. Biogeogr. 1998, 25, 155–163. [Google Scholar] [CrossRef]
- Aronson, M.F.J.; La Sorte, F.A.; Nilon, C.H.; Katti, M.; Goddard, M.A.; Lepczyk, C.A.; Warren, P.S.; Williams, N.S.G.; Cilliers, S.; Clarkson, B.; et al. A global analysis of the impacts of urbanization on bird and plant diversity reveals key anthropogenic drivers. Proc. R. Soc. B Biol. Sci. 2014, 281, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Bornkamm, R. Spontaneous development of urban woody vegetation on differing soils. Flora Morphol. Distrib. Funct. Ecol. Plants 2007, 202, 695–704. [Google Scholar] [CrossRef]
- Jim, C.Y. Urban Biogeographical Analysis of Spontaneous Tree Growth on Stone Retaining Walls. Phys. Geogr. 2008, 29, 351–373. [Google Scholar] [CrossRef]
- Zipperer, W. Species composition and structure of regenerated and remnant forest patches within an urban landscape. Urban Ecosyst. 2002, 6, 271–290. [Google Scholar] [CrossRef]
- Pimentel, D. Biological Invasions: Economic and Environmental Costs of Alien Plant, Animal, and Microbe Species; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Hejda, M.; Pyšek, P.; Jarošík, V. Impact of invasive plants on the species richness, diversity and composition of invaded communities. J. Ecol. 2009, 97, 393–403. [Google Scholar] [CrossRef]
- Vilà, M.; Espinar, J.L.; Hejda, M.; Hulme, P.E.; Jarošík, V.; Maron, J.L.; Pergl, J.; Schaffner, U.; Sun, Y.; Pyšek, P. Ecological impacts of invasive alien plants: A meta-analysis of their effects on species, communities and ecosystems. Ecol. Lett. 2011, 14, 702–708. [Google Scholar] [CrossRef] [PubMed]
- Knight, K.S.; Kurylo, J.S.; Endress, A.G.; Stewart, J.R.; Reich, P.B. Ecology and ecosystem impacts of common buckthorn (Rhamnus cathartica): A review. Biol. Invasions 2007, 9, 925–937. [Google Scholar] [CrossRef]
- Sharma, G.P.; Raghubanshi, A.S.; Singh, J.S. Lantana invasion: An overview. Weed Biol. Manag. 2005, 5, 157–165. [Google Scholar] [CrossRef]
- Kull, C.A.; Rangan, H. The political ecology of weeds: A scalar approach to landscape transformations. In The International Handbook of Political Ecology; Edward Elgar Publishing: Cheltenham, UK, 2015; pp. 487–500. ISBN 9780857936165. [Google Scholar]
- Kohli, R.K.; Batish, D.R.; Singh, J.S.; Singh, H.P.; Bhatt, J.R. Plant invasion in India: An overview. In Invasive Alien Plants: An Ecological Appraisal for the Indian Subcontinent; Centre for Agriculture and Bioscience International: Wallingford, UK, 2011; Chapter 1; pp. 1–9. [Google Scholar]
- Nowak, D.J.; Hoehn, R.E.; Bodine, A.R.; Greenfield, E.J.; O’Neil-Dunne, J. Urban forest structure, ecosystem services and change in Syracuse, NY. Urban Ecosyst. 2016, 19, 1455–1477. [Google Scholar] [CrossRef]
- Sjöman, H.; Morgenroth, J.; Deak, J.; Sæbø, A. Diversification of the urban forest—Can we afford to exclude exotic tree species? Urban For. Urban Green. 2016, 18, 237–241. [Google Scholar] [CrossRef]
- Corlett, R.T. Interactions between birds, fruit bats and exotic plants in urban Hong Kong, South China. Urban Ecosyst. 2005, 8, 275–283. [Google Scholar] [CrossRef]
- Dearborn, D.C.; Kark, S. Motivations for conserving urban biodiversity. Conserv. Biol. 2010, 24, 432–440. [Google Scholar] [CrossRef] [PubMed]
- Beninde, J.; Veith, M.; Hochkirch, A. Biodiversity in cities needs space: A meta-analysis of factors determining intra-urban biodiversity variation. Ecol. Lett. 2015, 18, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, R.; Philpott, S.M. Nest-site limitation and nesting resources of ants (Hymenoptera: Formicidae) in urban green spaces. Environ. Entomol. 2009, 38, 600–607. [Google Scholar] [CrossRef] [PubMed]
- Philpott, S.M.; Cotton, J.; Bichier, P.; Friedrich, R.L.; Moorhead, L.C.; Uno, S.; Valdez, M. Local and landscape drivers of arthropod abundance, richness, and trophic composition in urban habitats. Urban Ecosyst. 2014, 17, 513–532. [Google Scholar] [CrossRef]
- Strauss, B.; Biedermann, R. Urban brownfields as temporary habitats: Forces for the driving of phytophagous insects diversity. Ecography 2006, 29, 928–940. [Google Scholar] [CrossRef]
- Angold, P.G.; Sadler, J.P.; Hill, M.O.; Pullin, A.; Rushton, S.; Austin, K.; Small, E.; Wood, B.; Wadsworth, R.; Sanderson, R.; et al. Biodiversity in urban habitat patches. Sci. Total Environ. 2006, 360, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Öckinger, E.; Dannestam, Å.; Smith, H.G. The importance of fragmentation and habitat quality of urban grasslands for butterfly diversity. Landsc. Urban Plan. 2009, 93, 31–37. [Google Scholar] [CrossRef]
- Yang, L. From Molecules to Ecosystems: How Do Mosquitoes Respond to Changing Environments? Ph.D Thesis, The Ohio State University, Columbus, OH, United States, 2017. [Google Scholar]
- Sivakoff, F.S.; Prajzner, S.P.; Gardiner, M.M. Unique bee community assembly within vacant lots and urban farms results from variation in surrounding landscape urbanization intensity. Sustainability 2018, 6, 1926. [Google Scholar] [CrossRef]
- Moorhead, L.C.; Philpott, S.M. Richness and composition of spiders in urban green spaces in Toledo Ohio. J. Arachnol. 2013, 41, 356–363. [Google Scholar] [CrossRef]
- Burkman, C.E.; Gardiner, M.M. Spider assemblages within greenspaces of a deindustrialized urban landscape. Urban Ecosyst. 2015, 18, 793–818. [Google Scholar] [CrossRef]
- Uno, S.; Cotton, J.; Philpott, S.M. Diversity, abundance, and species composition of ants in urban green spaces. Urban Ecosyst. 2010, 13, 425–441. [Google Scholar] [CrossRef]
- Gardiner, M.M.; Prajzner, S.P.; Burkman, C.E.; Albro, S.; Grewal, P.S. Vacant land conversion to community gardens: Influences on generalist arthropod predators and biocontrol services in urban greenspaces. Urban Ecosyst. 2014, 17, 101–122. [Google Scholar] [CrossRef]
- McIntyre, N.E.; Rango, J.; Fagan, W.F.; Faeth, S.H. Ground arthropod community structure in a heterogeneous urban environment. Landsc. Urban Plan. 2001, 52, 257–274. [Google Scholar] [CrossRef]
- Isaacs, R.; Tuell, J.; Fiedler, A.; Gardiner, M.; Landis, D. Maximizing arthropod-mediated ecosystem services in agricultural landscapes: The role of native plants. Front. Ecol. Environ. 2009, 7, 196–203. [Google Scholar] [CrossRef]
- McIntyre, N.E. Ecology of Urban Arthropods: A Review and a call to action. Ann. Entomol. Soc. Am. 2000, 93, 825–835. [Google Scholar] [CrossRef]
- Šálek, M.; Marhoul, P.; Pintíř, J.; Kopecký, T.; Slabý, L. Importance of unmanaged wasteland patches for the grey partridge Perdix perdix in suburban habitats. Acta Oecol. 2004, 25, 23–33. [Google Scholar] [CrossRef]
- Meffert, P.J.; Marzluff, J.M.; Dziock, F. Unintentional habitats: Value of a city for the wheatear (Oenanthe oenanthe). Landsc. Urban Plan. 2012, 108, 49–56. [Google Scholar] [CrossRef]
- Hofer, C.; Gallagher, F.J.; Holzapfel, C. Metal accumulation and performance of nestlings of passerine bird species at an urban brownfield site. Environ. Pollut. 2010, 158, 1207–1213. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, D.E.; Toms, M.P.; Cleary-McHarg, R.; Banks, A.N. House sparrow (Passer domesticus) habitat use in urbanized landscapes. J. Ornithol. 2007, 148, 453–462. [Google Scholar] [CrossRef]
- Murgui, E. Seasonal patterns of habitat selection of the house sparrow Passer domesticus in the urban landscape of Valencia (Spain). J. Ornithol. 2009, 150, 85–94. [Google Scholar] [CrossRef]
- Rega-Brodsky, C.C.; Nilon, C.H. Vacant lots as a habitat resource: Nesting success and body condition of songbirds. Ecosphere 2016, 7, 1–11. [Google Scholar] [CrossRef]
- Meffert, P.J.; Dziock, F. The influence of urbanisation on diversity and trait composition of birds. Landsc. Ecol. 2013, 28, 943–957. [Google Scholar] [CrossRef]
- Meffert, P.J.; Dziock, F. What determines occurrence of threatened bird species on urban wastelands? Biol. Conserv. 2012, 153, 87–96. [Google Scholar] [CrossRef]
- Foster, J. Hiding in plain view: Vacancy and prospect in Paris’ Petite Ceinture. Cities 2014, 40, 124–132. [Google Scholar] [CrossRef]
- Grinder, M.I.; Krausman, P.R. Home range, habitat use, and nocturnal activity of coyotes in an urban environment. J. Wildl. Manag. 2001, 65, 887–898. [Google Scholar] [CrossRef]
- Parris, K.M. Urban amphibian assemblages as metacommunities. J. Anim. Ecol. 2006, 75, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Riley, S.P.D.; Busteed, G.T.; Kats, L.B.; Vandergon, T.L.; Lee, L.F.S.; Dagit, R.G.; Kerby, J.L.; Fisher, R.N.; Sauvajot, R.M. Effects of urbanization on the distribution and abundance of amphibians and invasive species in Southern California streams. Conserv. Biol. 2005, 19, 1894–1907. [Google Scholar] [CrossRef]
- Germaine, S.S.; Wakeling, B.F. Lizard species distributions and habitat occupation along an urban gradient in Tucson, Arizona, USA. Biol. Conserv. 2001, 97, 229–237. [Google Scholar] [CrossRef]
- Little, E.; Bajwa, W.; Shaman, J. Local environmental and meteorological conditions influencing the invasive mosquito Ae. albopictus and arbovirus transmission risk in New York City. PLoS Negl. Trop. Dis. 2017, 11, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Little, E.; Biehler, D.; Leisnham, P.T.; Jordan, R.; Wilson, S.; LaDeau, S.L. Socio-ecological mechanisms supporting high densities of Aedes albopictus (Diptera: Culicidae) in Baltimore, MD. J. Med. Entomol. 2017, 54, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- LaDeau, S.L.; Leisnham, P.T.; Biehler, D.; Bodner, D. Higher mosquito production in low-income neighborhoods of baltimore and washington, DC: Understanding ecological drivers and mosquito-borne disease risk in temperate cities. Int. J. Environ. Res. Public Health 2013, 10, 1505–1526. [Google Scholar] [CrossRef] [PubMed]
- Soulsbury, C.D.; White, P.C.L. Human-wildlife interactions in urban areas: A review of conflicts, benefits and opportunities. Wildl. Res. 2015, 42, 541–553. [Google Scholar] [CrossRef]
- Bakken, A.J.; Schoof, S.C.; Bickerton, M.; Kamminga, K.L.; Jenrette, J.C.; Malone, S.; Abney, M.A.; Herbert, D.A.; Reisig, D.; Kuhar, T.P.; et al. Occurrence of brown marmorated stink bug (Hemiptera: Pentatomidae) on wild hosts in Nonmanaged Woodlands and soybean fields in North Carolina and Virginia. Environ. Entomol. 2015, 44, 1011–1021. [Google Scholar] [CrossRef] [PubMed]
- Barringer, L.E.; Donovall, L.R.; Spichiger, S.-E.; Lynch, D.; Henry, D. The First New World Record of Lycorma delicatula (Insecta: Hemiptera: Fulgoridae). Entomol. News 2015, 125, 20–23. [Google Scholar] [CrossRef]
- Bolund, P.; Hunhammar, S. Ecosystem services in urban areas. Ecol. Econ. 1999, 29, 293–301. [Google Scholar] [CrossRef]
- De Groot, R.S.; Wilson, M.A.; Boumans, R.M.J. A typology for the classification, description and valuation of ecosystem functions, goods and services. Ecol. Econ. 2002, 41, 393–408. [Google Scholar] [CrossRef]
- Elmqvist, T.; Fragkias, M.; Goodness, J.; Güneralp, B.; Marcotullio, P.J.; McDonald, R.I.; Parnell, S.; Schewenius, M.; Sendstad, M.; Seto, K.C.; et al. Urbanization, Biodiversity and Ecosystem Services: Challenges and Opportunities; Springer: Berlin/Heidelberg, Germany, 2013; ISBN 9789400770881. [Google Scholar]
- Reid, W.V. Millennium Ecosystem Assessment. In Ecosystems and Human Well-Being: Synthesis; Island Press: Washington, DC, USA, 2005. [Google Scholar]
- Gomez-Baggethunm, E.; Gren, Å.; Barton, D.N.; Langemeyer, J.; McPhearson, T.; O’Farrell, P.; Andersson, E.; Hamstead, Z.A.; Kremer, P. Urban ecosystem services. In Urbanization, Biodiversity and Ecosystem Services: Challenges and Opportunities; Springer: Berlin, Germany, 2013; Chapter 11; pp. 175–252. [Google Scholar]
- Kim, G.; Miller, P.A.; Nowak, D.J. Assessing urban vacant land ecosystem services: Urban vacant land as green infrastructure in the City of Roanoke, Virginia. Urban For. Urban Green. 2015, 14, 519–526. [Google Scholar] [CrossRef]
- Miller, J.R. Biodiversity conservation and the extinction of experience. Trends Ecol. Evol. 2005, 20, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Soga, M.; Gaston, K.J. Extinction of experience: The loss of human-nature interactions. Front. Ecol. Environ. 2016, 14, 94–101. [Google Scholar] [CrossRef]
- Lee, A.C.K.; Maheswaran, R. The health benefits of urban green spaces: A review of the evidence. J. Public Health 2011, 33, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Jim, C.Y.; Chen, W.Y. Bioreceptivity of buildings for spontaneous arboreal flora in compact city environment. Urban For. Urban Green. 2011, 10, 19–28. [Google Scholar] [CrossRef]
- Millard, A. Indigenous and spontaneous vegetation: Their relationship to urban development in the city of Leeds, UK. Urban For. Urban Green. 2004, 3, 39–47. [Google Scholar] [CrossRef]
- Rupprecht, C.D.D.; Byrne, J.A.; Ueda, H.; Lo, A.Y. “It’s real, not fake like a park”: Residents’ perception and use of informal urban green-space in Brisbane, Australia and Sapporo, Japan. Landsc. Urban Plan. 2015, 143, 205–218. [Google Scholar] [CrossRef]
- Heynen, N.; Perkins, H.A.; Parama, R. The political ecology of uneven urban green space. Urban Aff. Rev. 2006, 42, 3–25. [Google Scholar] [CrossRef]
- Schwarz, K.; Fragkias, M.; Boone, C.G.; Zhou, W.; McHale, M.; Grove, J.M.; O’Neil-Dunne, J.; McFadden, J.P.; Buckley, G.L.; Childers, D.; et al. Trees grow on money: Urban tree canopy cover and environmental justice. PLoS ONE 2015, 10, e0122051. [Google Scholar] [CrossRef] [PubMed]
- Greene, C.S.; Robinson, P.J.; Millward, A.A. Canopy of advantage: Who benefits most from city trees? J. Environ. Manag. 2018, 208, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Astell-Burt, T.; Feng, X.; Mavoa, S.; Badland, H.M.; Giles-Corti, B. Do low-income neighbourhoods have the least green space? A cross-sectional study of Australia’s most populous cities. BMC Public Health 2014, 14, 1–11. [Google Scholar] [CrossRef] [PubMed]
- De Vries, S.; van Dillen, S.M.E.; Groenewegen, P.P.; Spreeuwenberg, P. Streetscape greenery and health: Stress, social cohesion and physical activity as mediators. Soc. Sci. Med. 2013, 94, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Kuo, F.E.; Bacaicoa, M.; Sullivan, W.C. Transforming inner city landscapes: Trees, sence of safety, and preference. Environ. Behav. 1998, 30, 28–59. [Google Scholar] [CrossRef]
- Ard, K.; Colen, C.; Becerra, M.; Velez, T. Two mechanisms: The role of social capital and industrial pollution exposure in explaining racial disparities in self-rated health. Int. J. Environ. Res. Public Health 2016, 13, 1025. [Google Scholar] [CrossRef] [PubMed]
- Nassauer, J.I.; Raskin, J. Urban vacancy and land use legacies: A frontier for urban ecological research, design, and planning. Landsc. Urban Plan. 2014, 125, 245–253. [Google Scholar] [CrossRef]
- Rupprecht, C.D.D.; Byrne, J.A.; Lo, A.Y. Memories of vacant lots: How and why residents used informal urban green space as children and teenagers in Brisbane, Australia, and Sapporo, Japan. Child. Geogr. 2016, 14, 340–355. [Google Scholar] [CrossRef]
- Kahn, P.H. Children’s affiliations with nature: Structure, development, and the problem of environmental generational amnesia. In Children and Nature: Psychological, Sociocultural, and Evolutionary Investigations; MIT Press: Cambridge, MA, USA, 2002; pp. 92–115. ISBN 0-262-11267-1. [Google Scholar]
- The Cuyahoga County Planning Commission; Cleveland Metroparks. Cuyahoga County Urban Tree Canopy Assessment, 2013; The Cuyahoga County Planning Commission: Cleveland, OH, USA, 2013. [Google Scholar]
- Homer, C.G.; Dewitz, J.A.; Yang, L.; Jin, S.; Danielson, P.; Xian, G.; Coulston, J.; Herold, N.D.; Wickham, J.D.; Megown, K. Completion of the 2011 National Land Cover Database for the conterminous United States-Representing a decade of land cover change information. Photogramm. Eng. Remote Sens. 2015, 81, 345–354. [Google Scholar]
- Green, O.O.; Garmestani, A.S.; Albro, S.; Ban, N.C.; Berland, A.; Burkman, C.E.; Gardiner, M.M.; Gunderson, L.; Hopton, M.E.; Schoon, M.L.; et al. Adaptive governance to promote ecosystem services in urban green spaces. Urban Ecosyst. 2015, 19, 77–93. [Google Scholar] [CrossRef]
- Hofmann, M.; Westermann, J.R.; Kowarik, I.; Van der Meer, E. Perceptions of parks and urban derelict land by landscape planners and residents. Urban For. Urban Green. 2012, 11, 303–312. [Google Scholar] [CrossRef]
- Németh, J.; Langhorst, J. Rethinking urban transformation: Temporary uses for vacant land. Cities 2014, 40, 143–150. [Google Scholar] [CrossRef]
- Kremer, P.; Hamstead, Z.A.; McPhearson, T. A social-ecological assessment of vacant lots in New York City. Landsc. Urban Plan. 2013, 120, 218–233. [Google Scholar] [CrossRef]
- Nassauer, J.I. Care and stewardship: From home to planet. Landsc. Urban Plan. 2011, 100, 321–323. [Google Scholar] [CrossRef]
- Nassauer, J.I. Messy Ecosystems, Orderly frames. Landsc. J. 1995, 14, 161–170. [Google Scholar] [CrossRef]
- Mair, C.; Kaplan, G.A.; Everson-Rose, S.A. Are there hopeless neighborhoods? An exploration of environmental associations between individual-level feelings of hopelessness and neighborhood characteristics. Health Place 2012, 18, 434–439. [Google Scholar] [CrossRef] [PubMed]
- Curley, A.M. Neighborhood institution, facilities, and public space: A missing link for HOPE VI residents’ development of social capital? Cityscape J. Policy Dev. Res. 2010, 12, 33–64. [Google Scholar] [CrossRef]
- Wen, M.; Zhang, X.; Harris, C.D.; Holt, J.B.; Croft, J.B. Spatial disparities in the distribution of parks and green spaces in the USA. Ann. Behav. Med. 2013, 45, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Durant, N.; Kerr, J.; Harris, S.K.; Saelens, B.E.; Norman, G.J.; Sallis, J.F. Environmental and safety barriers to youth physical activity in neighborhood parks and streets: Reliability and validity. Pediatr. Exerc. Sci. 2009, 21, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Kuo, F.E.; Sullivan, W.C. Environment and crime in the inner city: Does vegetation reduce crime? Environ. Behav. 2001, 33, 343–367. [Google Scholar] [CrossRef]
- Wolfe, M.K.; Mennis, J. Does vegetation encourage or suppress urban crime? Evidence from Philadelphia, PA. Landsc. Urban Plan. 2012, 108, 112–122. [Google Scholar] [CrossRef]
- Branas, C.C.; Cheney, R.A.; MacDonald, J.M.; Tam, V.W.; Jackson, T.D.; Ten Havey, T.R. A difference-in-differences analysis of health, safety, and greening vacant urban space. Am. J. Epidemiol. 2011, 174, 1296–1306. [Google Scholar] [CrossRef] [PubMed]
- Garvin, E.C.; Cannuscio, C.C.; Branas, C.C. Greening vacant lots to reduce violent crime: A randomised controlled trial. Inj. Prev. 2013, 19, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Troy, A.; Morgan Grove, J.; O’Neil-Dunne, J. The relationship between tree canopy and crime rates across an urban-rural gradient in the greater Baltimore region. Landsc. Urban Plan. 2012, 106, 262–270. [Google Scholar] [CrossRef]
- Locke, D.H.; Han, S.H.; Kondo, M.C.; Murphy-Dunning, C.; Cox, M. Did community greening reduce crime? Evidence from New Haven, CT, 1996–2007. Landsc. Urban Plan. 2017, 161, 72–79. [Google Scholar] [CrossRef]
- Palmer, M.A. Socioenvironmental sustainability and actionable ccience. Bioscience 2012, 62, 5–6. [Google Scholar] [CrossRef]
- Shwartz, A.; Turbé, A.; Julliard, R.; Simon, L.; Prévot, A.C. Outstanding challenges for urban conservation research and action. Glob. Environ. Chang. 2014, 28, 39–49. [Google Scholar] [CrossRef]





© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Riley, C.B.; Perry, K.I.; Ard, K.; Gardiner, M.M. Asset or Liability? Ecological and Sociological Tradeoffs of Urban Spontaneous Vegetation on Vacant Land in Shrinking Cities. Sustainability 2018, 10, 2139. https://doi.org/10.3390/su10072139
Riley CB, Perry KI, Ard K, Gardiner MM. Asset or Liability? Ecological and Sociological Tradeoffs of Urban Spontaneous Vegetation on Vacant Land in Shrinking Cities. Sustainability. 2018; 10(7):2139. https://doi.org/10.3390/su10072139
Chicago/Turabian StyleRiley, Christopher B., Kayla I. Perry, Kerry Ard, and Mary M. Gardiner. 2018. "Asset or Liability? Ecological and Sociological Tradeoffs of Urban Spontaneous Vegetation on Vacant Land in Shrinking Cities" Sustainability 10, no. 7: 2139. https://doi.org/10.3390/su10072139
APA StyleRiley, C. B., Perry, K. I., Ard, K., & Gardiner, M. M. (2018). Asset or Liability? Ecological and Sociological Tradeoffs of Urban Spontaneous Vegetation on Vacant Land in Shrinking Cities. Sustainability, 10(7), 2139. https://doi.org/10.3390/su10072139