Experimental Study of Thermal and Catalytic Pyrolysis of Plastic Waste Components
Abstract
1. Introduction
2. Materials and Methods
2.1. Thermogravimetric Analysis
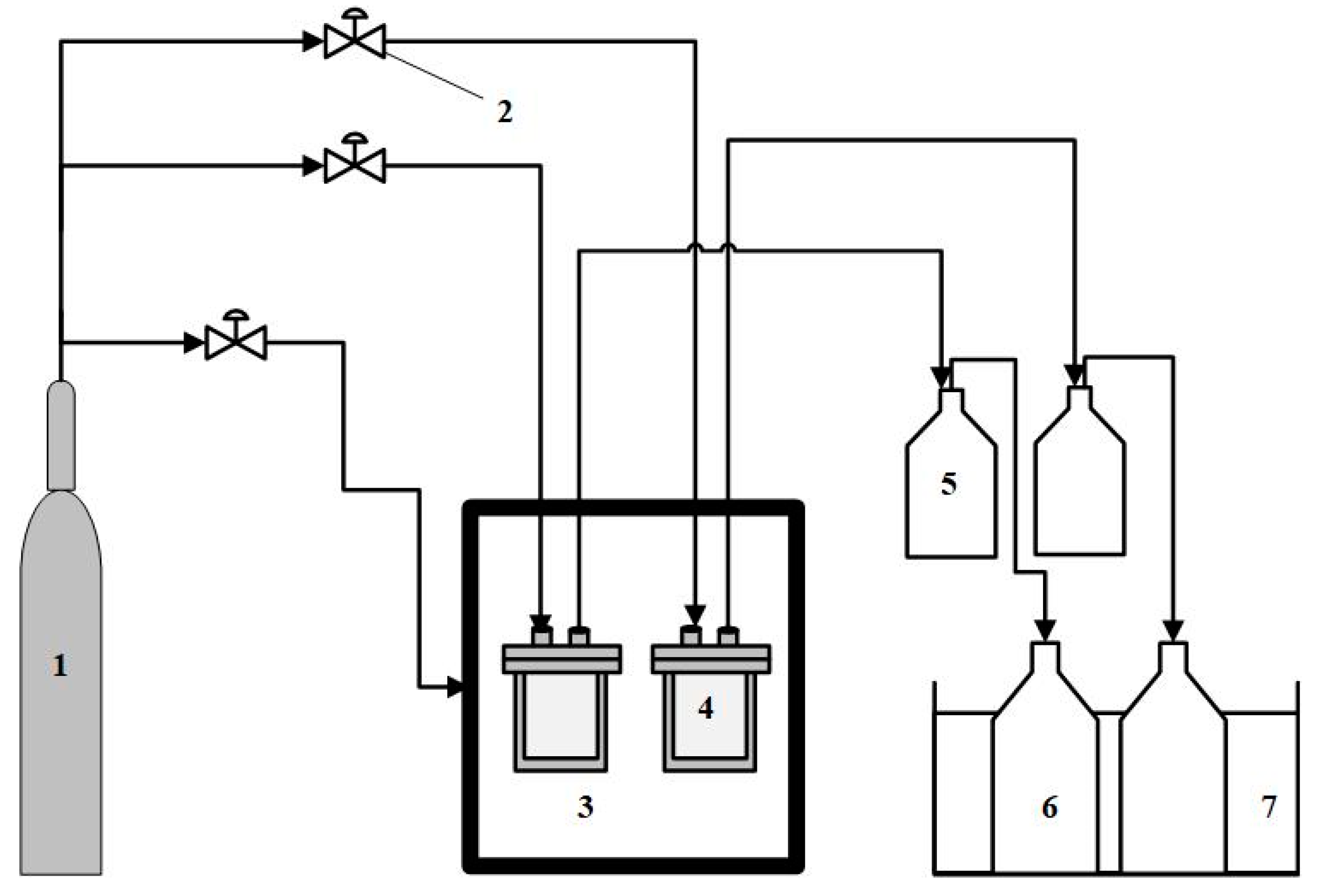
2.2. Pyrolysis Experiment and Procedure
- M1: Mass of the sample,
- M2: Mass of liquid product,
- M3: Mass of residue.
2.3. Pyrolysis Product Analysis and Characterisation
3. Results and Discussion
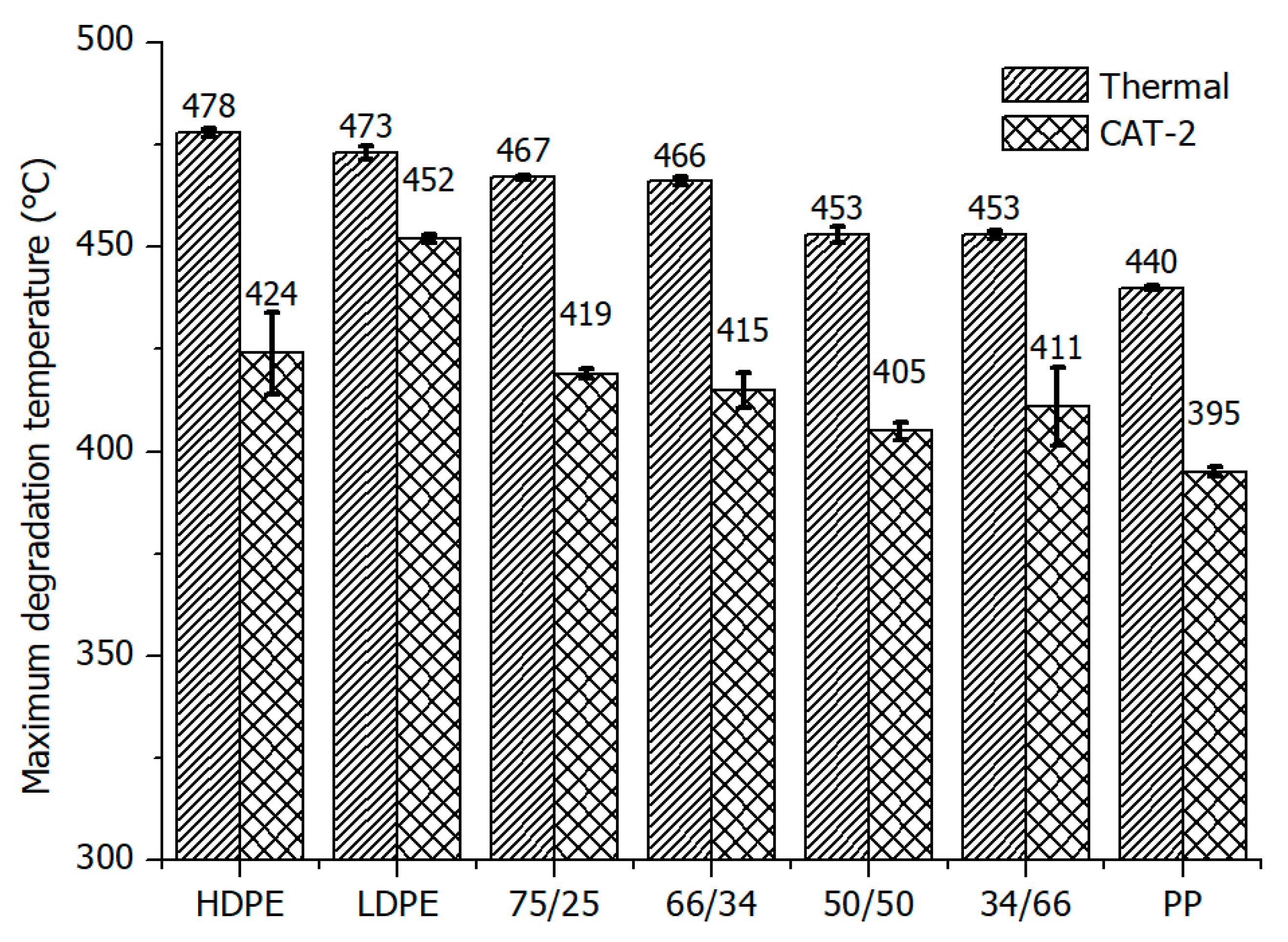
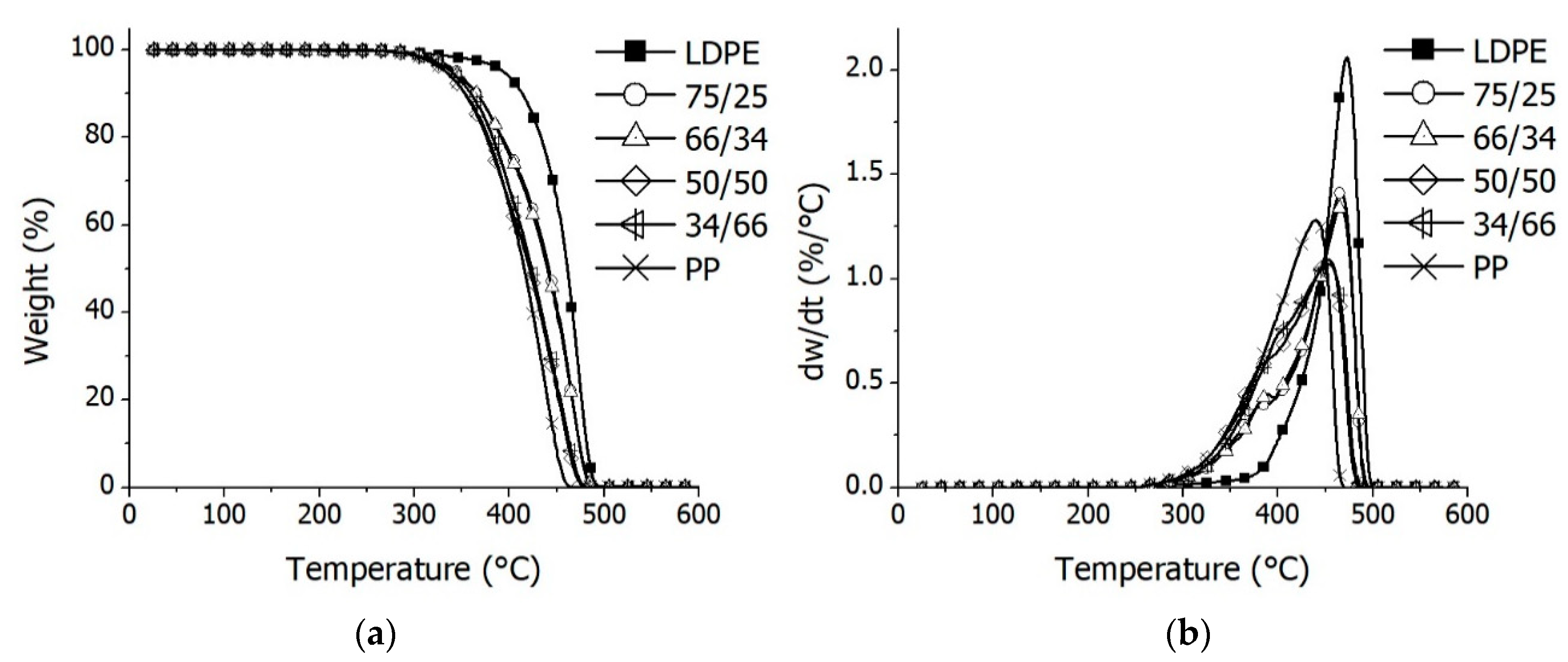
3.1. Thermal and Catalytic Degradation of Polymers and Their Mixtures
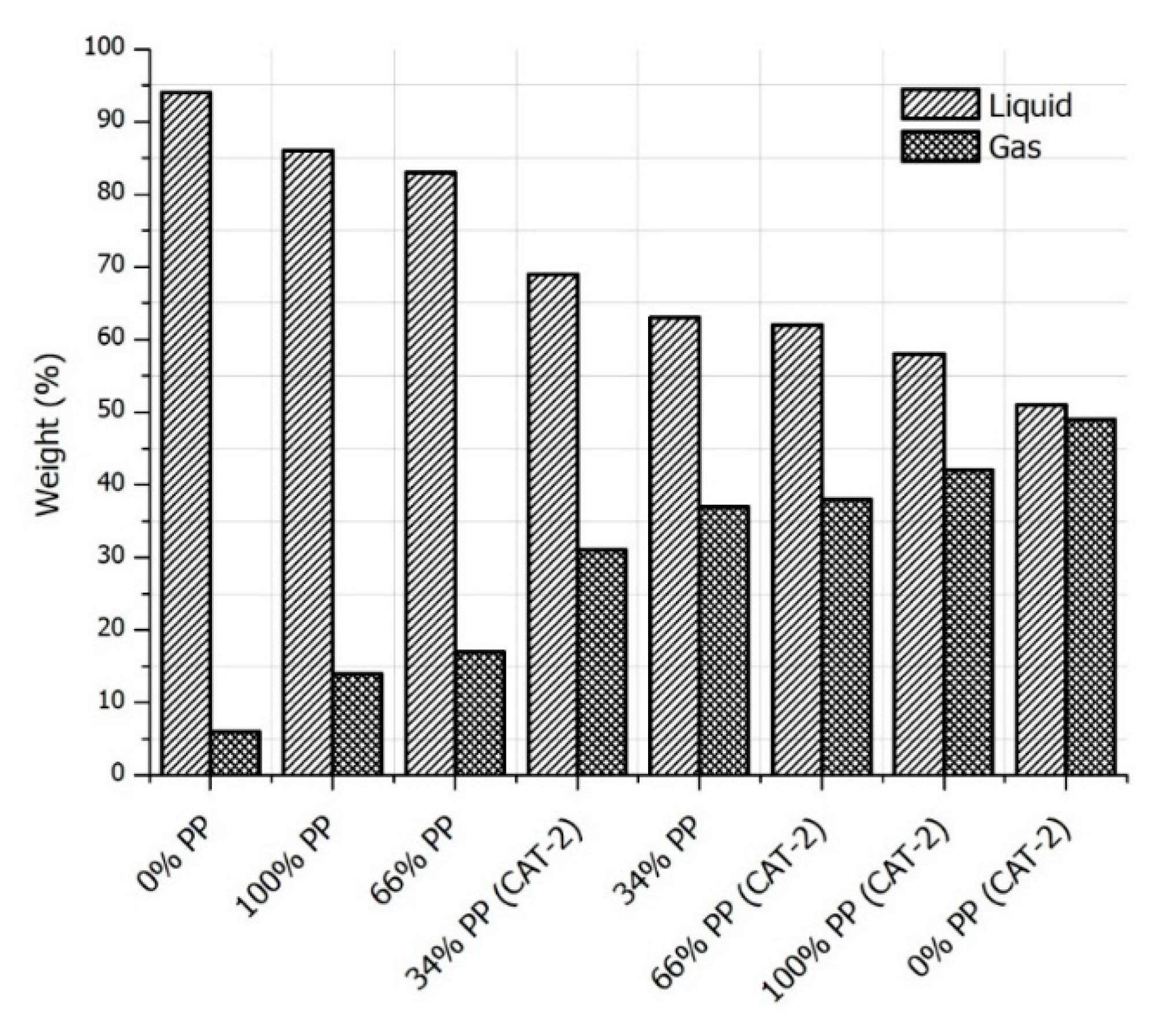
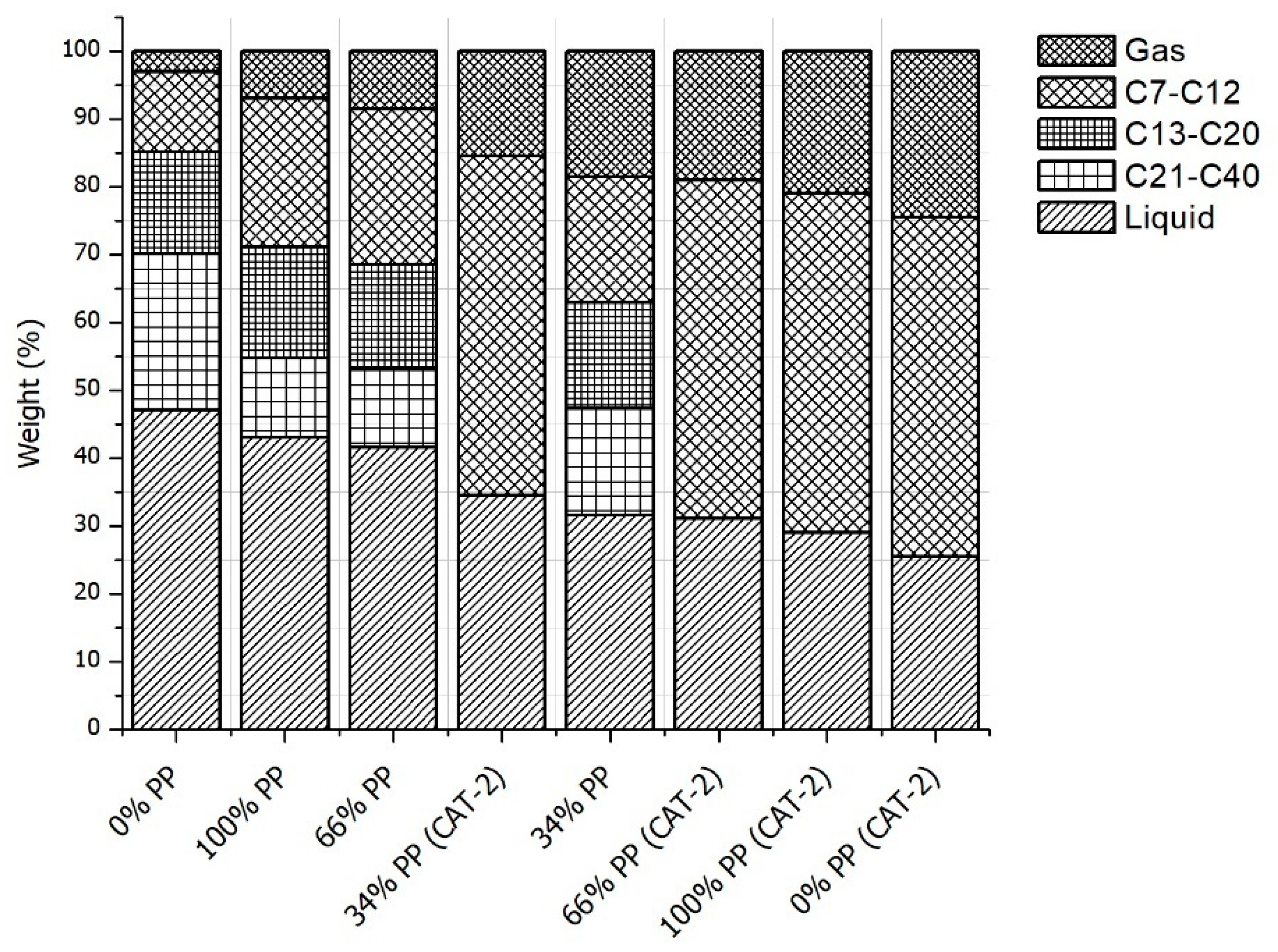
3.2. Pyrolysis Yields
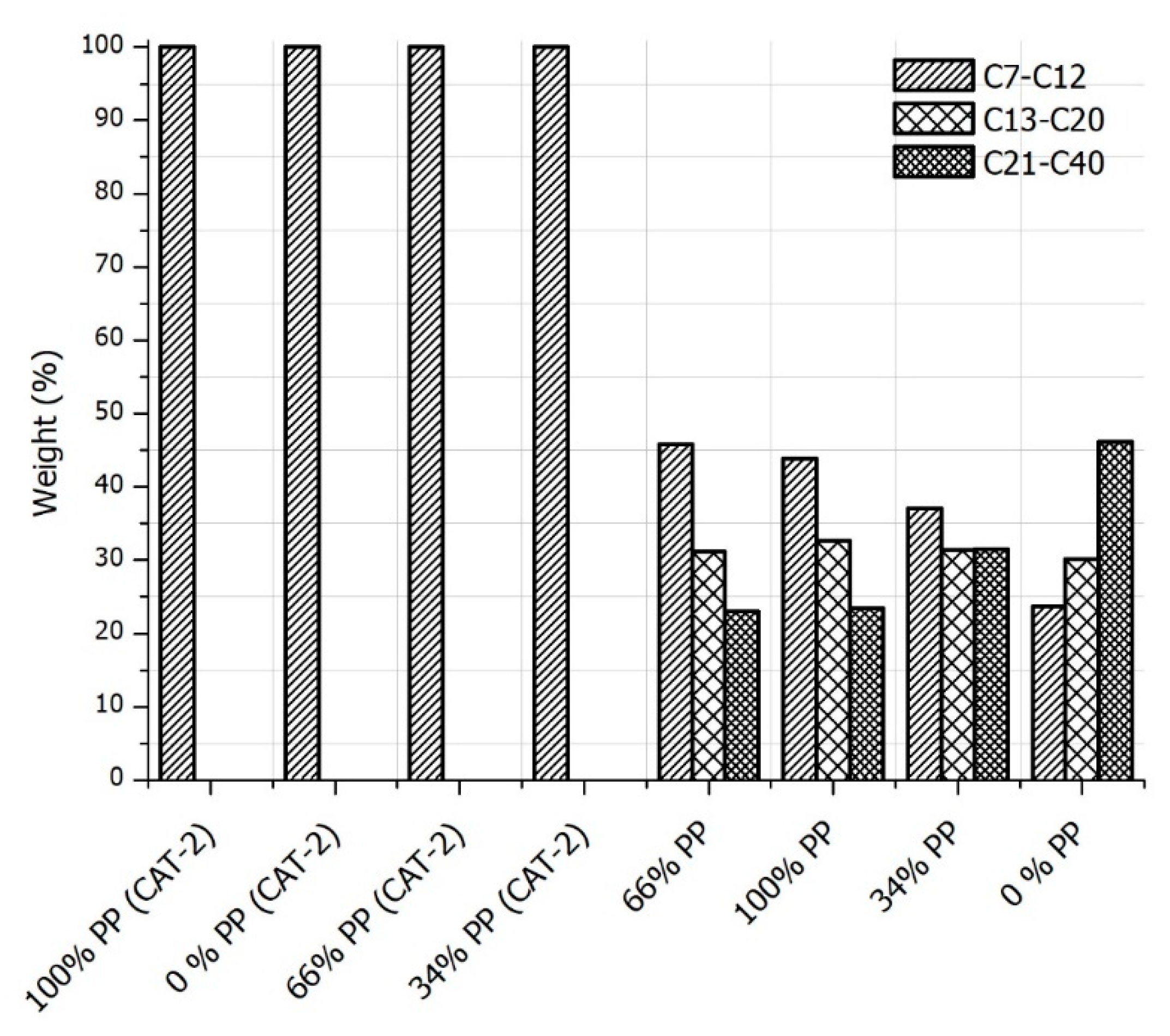
3.3. GC-FID Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Forum, W.E. The New Plastics Economy: Rethinking the Future of Plastics; Ellen MacArthur Foundation: Geneva, Switzerland, 2016. [Google Scholar]
- Ahmad, I.; Khan, M.I.; Khan, H.; Ishaq, M.; Tariq, R.; Gul, K.; Ahmad, W. Pyrolysis study of polypropylene and polyethylene into premium oil products. Int. J. Green Energy 2015, 12, 663–671. [Google Scholar] [CrossRef]
- Sharuddin, S.D.A.; Abnisa, F.; Daud, W.M.A.W.; Aroua, M.K. A review on pyrolysis of plastic wastes. Energy Convers. Manag. 2016, 115, 308–326. [Google Scholar] [CrossRef]
- European Commission. Plastic Waste—Strategy and Background. 2017. Available online: http://ec.europa.eu/environment/waste/plastic_waste.htm (accessed on 28 February 2017).
- Kunwar, B.; Cheng, H.N.; Chandrashekaran, S.R.; Sharma, B.K. Plastics to fuel: A review. Renew. Sustain. Energy Rev. 2016, 54, 421–428. [Google Scholar] [CrossRef]
- Kyaw, K.T.; Hmwe, C.S.S. Effect of various catalysts on fuel oil pyrolysis process of mixed plastic wastes. Int. J. Adv. Eng. Technol. 2015, 8, 794–802. [Google Scholar]
- Ayhan, D. Pyrolysis of municipal plastic wastes for recovery of gasoline-range hydrocarbons. J. Anal. Appl. Pyrolysis 2004, 72, 97–102. [Google Scholar]
- Scott, D.S.; Czernik, S.R.; Piskorz, J.; Radlein, D.S.A.G. Fast pyrolysis of plastic wastes. Energy Fuels 1990, 4, 407–411. [Google Scholar] [CrossRef]
- Panda, A.K. Studies on Process Optimization for Production of Liquid Fuels from Waste Plastics. Ph.D. Thesis, National Institute of Technology, Rourkela, India, 2011. [Google Scholar]
- Miandad, R.; Barakat, M.A.; Aburiazaiza, A.S.; Rehan, M.; Nizami, A.S. Catalytic pyrolysis of plastic waste: A review. Process Saf. Environ. Prot. 2016, 102, 822–838. [Google Scholar] [CrossRef]
- Miandad, R.; Barakat, M.A.; Aburiazaiza, A.S.; Rehan, M.; Ismail, I.M.I.; Nizami, A.S. Effect of plastic waste types on pyrolysis liquid oil. Int. Biodeterior. Biodegrad. 2017, 119, 239–252. [Google Scholar] [CrossRef]
- Breyer, S.; Mekhitarian, L.; Rimez, B.; Haut, B. Production of an alternative fuel by the co-pyrolysis of landfill recovered plastic wastes and used lubrication oils. Waste Manag. 2017, 60, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Almeida, D.; de Fatima Marques, M. Thermal and catalytic pyrolysis of plastic waste. Polímeros 2016, 26, 44–51. [Google Scholar] [CrossRef]
- López, A.; de Marco, I.; Caballero, B.M.; Laresgoiti, M.F.; Adrados, A.; Aranzabal, A. Catalytic pyrolysis of plastic wastes with two different types of catalysts: ZSM-5 zeolite and Red Mud. Appl. Catal. B Environ. 2011, 104, 211–219. [Google Scholar] [CrossRef]
- Marcilla, A.; García-Quesada, J.C.; Sánchez, S.; Ruiz, R. Study of the catalytic pyrolysis behaviour of polyethylene–polypropylene mixtures. J. Anal. Appl. Pyrolysis 2005, 74, 387–392. [Google Scholar] [CrossRef]
- Williams, P.T.; Slaney, E. Analysis of products from the pyrolysis and liquefaction of single plastics and waste plastic mixtures. Resour. Conserv. Recycl. 2007, 51, 754–769. [Google Scholar] [CrossRef]
- Yan, G.; Jing, X.; Wen, H.; Xiang, S. Thermal Cracking of Virgin and Waste Plastics of PP and LDPE in a Semibatch Reactor under Atmospheric Pressure. Energy Fuels 2015, 29, 2289–2298. [Google Scholar] [CrossRef]
- Achilias, D.S.; Roupakias, C.; Megalokonomos, P.; Lappas, A.A.; Antonakou, E.V. Chemical recycling of plastic wastes made from polyethylene (LDPE and HDPE) and polypropylene (PP). J. Hazard. Mater. 2007, 149, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Chandrasekaran, S.R.; Kunwar, B.; Moser, B.R.; Rajagopalan, N.; Sharma, B.K. Catalytic Thermal Cracking of Postconsumer Waste Plastics to Fuels. 1. Kinetics and Optimization. Energy Fuels 2015, 29, 6068–6077. [Google Scholar] [CrossRef]
- Marcilla, A.; Beltrán, M.I.; Navarro, R. Thermal and catalytic pyrolysis of polyethylene over HZSM5 and HUSY zeolites in a batch reactor under dynamic conditions. Appl. Catal. B Environ. 2009, 86, 78–86. [Google Scholar] [CrossRef]
- Seo, Y.-H.; Lee, K.-H.; Shin, D.-H. Investigation of catalytic degradation of high-density polyethylene by hydrocarbon group type analysis. J. Anal. Appl. Pyrolysis 2003, 70, 383–398. [Google Scholar] [CrossRef]
- Li, K.; Lee, S.W.; Yuan, G.; Lei, J.; Lin, S.; Weerachanchai, P.; Yang, Y.; Wang, J.-Y. Investigation into the Catalytic Activity of Microporous and Mesoporous Catalysts in the Pyrolysis of Waste Polyethylene and Polypropylene Mixture. Energies 2016, 9, 431. [Google Scholar] [CrossRef]
- Sundararajan, N.K.; Bhagavathi, A.R. Experimental Investigation on Thermocatalytic Pyrolysis of HDPE Plastic Waste and the Effects of Its Liquid Yield over the Performance, Emission, and Combustion Characteristics of CI Engine. Energy Fuels 2016, 30, 5379–5390. [Google Scholar] [CrossRef]
- Aboulkas, A.; El Bouadili, A. Thermal degradation behaviors of polyethylene and polypropylene. Part I: Pyrolysis kinetics and mechanisms. Energy Convers. Manag. 2010, 51, 1363–1369. [Google Scholar] [CrossRef]
- Jing, X.; Yan, G.; Zhao, Y.; Wen, H.; Xu, Z. Study on mild cracking of polyolefins to liquid hydrocarbons in a closed batch reactor for subsequent olefin recovery. Polym. Degrad. Stab. 2014, 109, 79–91. [Google Scholar] [CrossRef]
- Ciliz, N.K.; Ekinci, E.; Snape, C.E. Pyrolysis of virgin and waste polypropylene and its mixtures with waste polyethylene and polystyrene. Waste Manag. 2004, 24, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Jing, X.; Zhao, Y.; Wen, H.; Xu, Z. Interactions between low-density polyethylene (LDPE) and polypropylene (PP) during the mild cracking of polyolefin mixtures in a closed-batch reactor. Energy Fuels 2013, 27, 5841–5851. [Google Scholar] [CrossRef]
| Plastic | Type | Density [kg/m³]—ISO 1183 | Melt Flow Rate [g/10 min]—ISO 1133 | Melt Temperature [°C] ª |
|---|---|---|---|---|
| HDPE | PE copolymer | 945 | 0.3 (190 °C, 2.16 kg) | 129 |
| LDPE | PE copolymer | 923 | 0.2 (190 °C, 2.16 kg) | 124 |
| PP | PP homopolymer | 905 | 12 (230 °C, 2.16 kg) | 220–260 |
| CAT-2 | HDPE | LDPE | PP |
|---|---|---|---|
| NO | 100 | - | - |
| NO | - | 100 | - |
| NO | - | 75 | 25 |
| NO | - | 66 | 34 |
| NO | - | 50 | 50 |
| NO | - | 34 | 66 |
| NO | - | - | 100 |
| YES | 100 | - | - |
| YES | - | 100 | - |
| YES | - | 75 | 25 |
| YES | - | 66 | 34 |
| YES | - | 50 | 50 |
| YES | - | 34 | 66 |
| YES | - | - | 100 |
| CAT-2 | LDPE | PP |
|---|---|---|
| No | - | 100 |
| No | 34 | 66 |
| No | 66 | 34 |
| No | 100 | - |
| Yes | - | 100 |
| Yes | 34 | 66 |
| Yes | 66 | 34 |
| Yes | 100 | - |
| GC | HP 6890 Series |
|---|---|
| Column | DB-1 HT |
| Column length | 15 |
| Column ID (mm) | 0.32 |
| Stationary Phase thickness | 0.1 |
| Carrier gas | He/H2 |
| Total flow rate (mL/min) | 68.9 |
| Initial column temperature (°C) | 40 |
| Final column temperature (°C) | 350 |
| Detector | FID |
| Injection temperature (°C) | 340 |
| Injection volume (µL) | 1 |
| Detector temperature (°C) | 360 |
| Heating rate (°C/min) | 10 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anene, A.F.; Fredriksen, S.B.; Sætre, K.A.; Tokheim, L.-A. Experimental Study of Thermal and Catalytic Pyrolysis of Plastic Waste Components. Sustainability 2018, 10, 3979. https://doi.org/10.3390/su10113979
Anene AF, Fredriksen SB, Sætre KA, Tokheim L-A. Experimental Study of Thermal and Catalytic Pyrolysis of Plastic Waste Components. Sustainability. 2018; 10(11):3979. https://doi.org/10.3390/su10113979
Chicago/Turabian StyleAnene, Azubuike Francis, Siw Bodil Fredriksen, Kai Arne Sætre, and Lars-Andre Tokheim. 2018. "Experimental Study of Thermal and Catalytic Pyrolysis of Plastic Waste Components" Sustainability 10, no. 11: 3979. https://doi.org/10.3390/su10113979
APA StyleAnene, A. F., Fredriksen, S. B., Sætre, K. A., & Tokheim, L.-A. (2018). Experimental Study of Thermal and Catalytic Pyrolysis of Plastic Waste Components. Sustainability, 10(11), 3979. https://doi.org/10.3390/su10113979





