The Development of Sarcoidosis in an Ulcerative Colitis Patient Treated with Vedolizumab: A Case Report and Review of the Literature
Abstract
1. Introduction
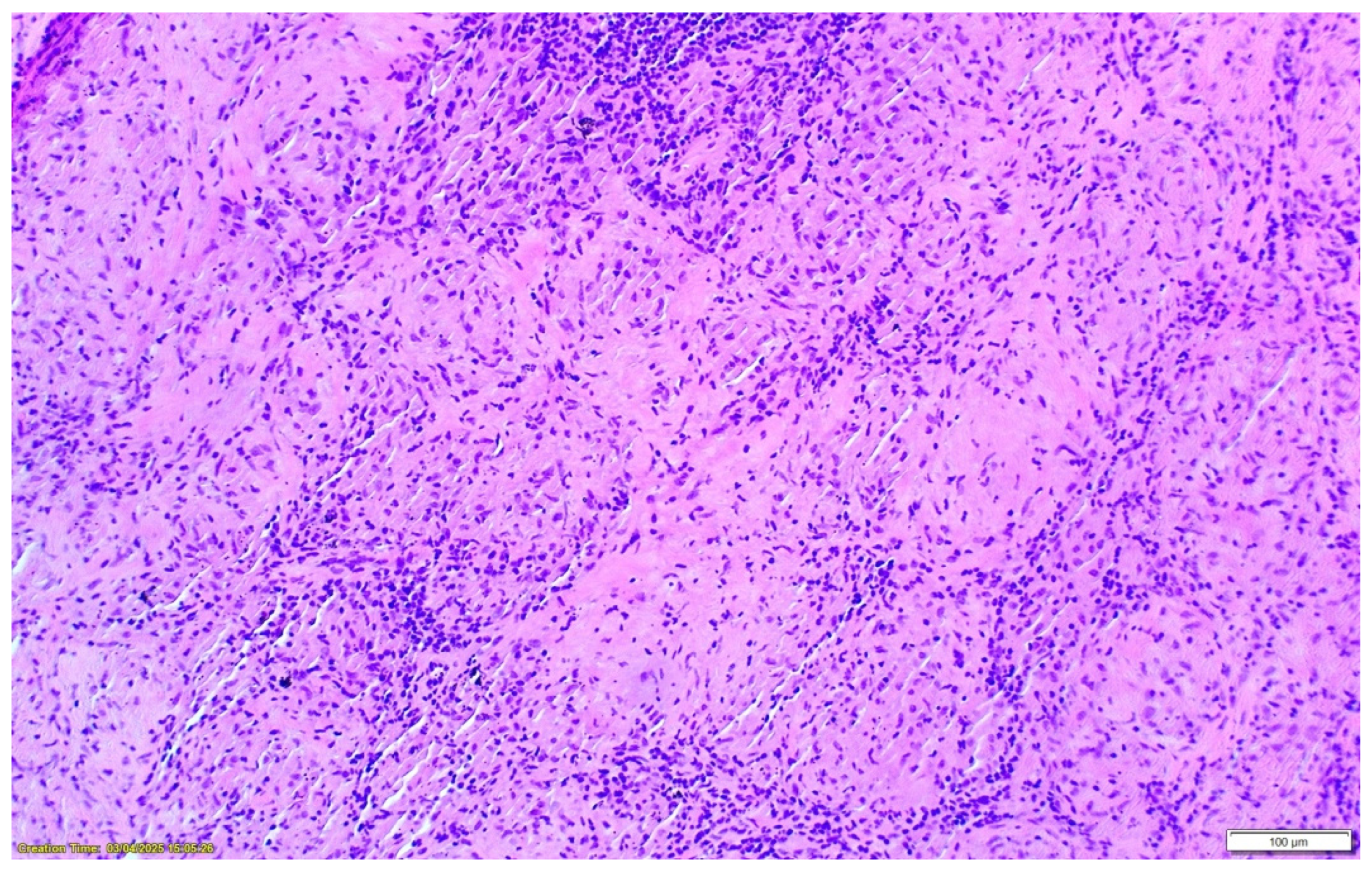
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Correction Statement
Abbreviations
| ACE | Angiotensin-converting enzyme |
| AZA | Azathioprine |
| CS | Corticosteroids |
| CT | Computed tomography |
| FDG | Fluorodeoxyglucose |
| IBD | Inflammatory bowel disease |
| IL | Interleukin |
| JAK | Janus kinase |
| PET-CT | Positron emission tomography–computed tomography |
| TNF-α | Tumor necrosis factor alpha |
| UC | Ulcerative colitis |
| VDZ | Vedolizumab |
References
- Jeny, F.; Valeyre, D.; Lower, E.E.; Baughman, R.P. Advanced pulmonary sarcoidosis. J. Autoimmun. 2025, 152, 103397. [Google Scholar] [CrossRef] [PubMed]
- Kahlmann, V.; Janssen Bonás, M.; Moor, C.C.; Grutters, J.C.; Mostard, R.L.M.; van Rijswijk, H.N.A.J.; van der Maten, J.; Marges, E.R.; Moonen, L.A.A.; Overbeek, M.J.; et al. First-Line Treatment of Pulmonary Sarcoidosis with Prednisone or Methotrexate. N. Engl. J. Med. 2025, 393, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Triantafillidis, J.K.; Stanciu, C. (Eds.) Inflammatory Bowel Disease: Pathogenesis, Clinical Manifestations, Diagnosis, Treatment; Technogramma: Athens, Greece, 2012; pp. 126–236. [Google Scholar]
- Triantafillidis, J.K.; Zografos, C.G.; Konstadoulakis, M.M.; Papalois, A.E. Combination treatment of inflammatory bowel disease: Present status and future perspectives. World J. Gastroenterol. 2024, 30, 2068–2080. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sands, B.E.; Peyrin-Biroulet, L.; Schreiber, S.; Danese, S.; Armuzzi, A.; Buisson, A.; Fumery, M.; Dignass, A.; Powell, N.; Kennedy, N.A.; et al. Efficacy and Safety of Infliximab and Vedolizumab Maintenance Therapy in Patients with Crohn’s Disease and Ulcerative Colitis: A Systematic Review and Meta-Analysis. J. Clin. Med. 2025, 14, 4419. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, Z.; Li, H.; Li, Z.; Ouyang, R.; Huang, S.; Qin, S.; Qin, J.; Huang, J. The efficacy, safety, and persistence of vedolizumab versus adalimumab in patients with inflammatory bowel disease: A systematic review and meta-analysis. Inflammopharmacology 2025, 33, 1761–1773. [Google Scholar] [CrossRef] [PubMed]
- Arenas Aravena, A.F.; Ruedi, D.; Sanhueza, M.; Ibáñez, S.; Carrasco-Avino, G.; Ibáñez, P. Sarcoidosis and ulcerative colitis: Overlap or coexistence. Rev. Esp. Enferm. Dig. 2025, 117, 286–287. [Google Scholar] [CrossRef] [PubMed]
- Theodoropoulos, G.; Archimandritis, A.; Davaris, P.; Plataris, J.; Melissinos, K. Ulcerative colitis and sarcoidosis: A curious association-report of a case. Dis. Colon Rectum 1981, 24, 308–310. [Google Scholar] [PubMed]
- Jarrot, P.A.; Dury, S.; Rakotomalala, A.; Vella-Boucaud, J.; Patey, M.; Deslee, G.; Lebargy, F. Association of sarcoidosis and ulcerative colitis: A review of 20 cases. Sarcoidosis Vasc. Diffus. Lung Dis. 2013, 30, 212–216. [Google Scholar] [PubMed]
- Nilubol, N.; Taub, P.J.; Venturero, M.; Lichtiger, S.; Bauer, J.J. Ulcerative colitis and sarcoidosis. Mt. Sinai J. Med. 2001, 68, 400–402. [Google Scholar] [PubMed]
- Vaiphei, K.; Gupta, N.; Sinha, S.K.; Nagi, B.; Singh, K. Association of ulcerative colitis with pulmonary sarcoidosis, subcutaneous lipomatosis and appendiceal adenocarcinoma. Indian J. Gastroenterol. 2003, 22, 193–194. [Google Scholar] [PubMed]
- Yoshioka, K.; Nishimura, S.; Kitai, S.; Kondo, M. Association of sarcoidosis, insulin-dependent diabetes mellitus, and ulcerative colitis. Arch. Intern. Med. 1997, 157, 465–467. [Google Scholar] [CrossRef] [PubMed]
- Sumi, T.; Yamada, G.; Yorozuya, T.; Tanaka, Y.; Tanaka, Y.; Sakuma, Y.; Takahashi, H. Sarcoidosis development during ulcerative colitis remission in a patient with a susceptible human leukocyte antigen serotype. Sarcoidosis Vasc. Diffus. Lung Dis. 2021, 38, e2021010. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yi, J.; Wu, S.; He, H. Causal association of inflammatory bowel disease with sarcoidosis and the mediating role of primary biliary cholangitis. Front. Immunol. 2024, 15, 1448724. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Terwiel, M.; Grutters, J.C.; van Moorsel, C.H.M. Clustering of immune-mediated diseases in sarcoidosis. Curr. Opin. Pulm. Med. 2019, 25, 539–553. [Google Scholar] [CrossRef] [PubMed]
- Burisch, J.; Jess, T.; Egeberg, A. Incidence of immune-mediated inflammatory diseases among patients with inflammatory bowel diseases in Denmark. Clin. Gastroenterol. Hepatol. 2019, 17, 2704–2712.e3. [Google Scholar] [CrossRef] [PubMed]
- Halling, M.L.; Kjeldsen, J.; Knudsen, T.; Nielsen, J.; Hansen, L.K. Patients with inflammatory bowel disease have increased risk of autoimmune and inflammatory diseases. World J. Gastroenterol. 2017, 23, 6137–6146. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ju, H.J.; Lee, J.Y.; Han, J.H.; Lee, J.H.; Bae, J.M.; Lee, S. Risk of autoimmune skin and connective tissue disorders after mRNA-based COVID-19 vaccination. J. Am. Acad. Dermatol. 2023, 89, 685–693. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sun, Y.; Zhou, Q.; Onzere, L.E.; Dian, Y.; Meng, Y.; Li, D.; Zeng, F.; Lei, S.; Deng, G. Evaluating the causal effect of using glucagon-like peptide-1 receptor agonists on the risk of autoimmune diseases. Diabetes Metab. Syndr. 2025, 19, 103186. [Google Scholar] [CrossRef] [PubMed]
- Sakhamuru, S.; Kambampati, S.; Wasim, S.; Kukkar, V.; Malik, B.H. The role of propionibacterium acnes in the pathogenesis of sarcoidosis and ulcerative colitis: How this connection may inspire novel management of these conditions. Cureus 2020, 12, e10812. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gîlcă, G.E.; Diaconescu, S.; Bălan, G.G.; Timofte, O.; Ştefănescu, G. Sarcoidosis associated with infliximab therapy in ulcerative colitis: A case report. Medicine 2017, 96, e6156. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, T.K.; Kang, S.H.; Moon, H.S.; Sung, J.K.; Jeong, H.Y.; Eun, H.S. Pulmonary sarcoidosis that developed during the treatment of a patient with crohn disease by using infliximab. Ann. Coloproctol. 2017, 33, 74–77. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Donzella, D.; Bellis, E.; Campisi, P.; Crepaldi, G.; Data, V.; Dapavo, P.; Lomater, C.; Marucco, E.; Saracco, M.; Gatto, M.; et al. New onset sarcoidosis following biologic treatment in patients with seronegative inflammatory arthritis: A case series and systematic literature review. Autoimmun. Rev. 2024, 23, 103481. [Google Scholar] [CrossRef] [PubMed]
- Kopylov, U.; Verstockt, B.; Biedermann, L.; Sebastian, S.; Pugliese, D.; Sonnenberg, E.; Steinhagen, P.; Arebi, N.; Ron, Y.; Kucharzik, T.; et al. Effectiveness and Safety of Vedolizumab in Anti-TNF-Naïve Patients with Inflammatory Bowel Disease-A Multicenter Retrospective European Study. Inflamm. Bowel Dis. 2018, 24, 2442–2451. [Google Scholar] [CrossRef] [PubMed]
- Ben-Horin, S.; Ungar, B.; Kopylov, U.; Lahat, A.; Yavzori, M.; Fudim, E.; Picard, O.; Peled, Y.; Eliakim, R.; Del Tedesco, E.; et al. Safety, efficacy and pharmacokinetics of vedolizumab in patients with simultaneous exposure to an anti-tumour necrosis factor. Aliment. Pharmacol. Ther. 2018, 47, 1117–1125. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Triantafillidis, J.K.; Malgarinos, K.; Kaklamanis, L.; Kritsotakis, E.; Polydorou, V.; Pantos, K.; Sfakianoudis, K.; Pantou, A.; Bramis, K.; Konstantoulakis, M.M.; et al. The Development of Sarcoidosis in an Ulcerative Colitis Patient Treated with Vedolizumab: A Case Report and Review of the Literature. Clin. Pract. 2026, 16, 44. https://doi.org/10.3390/clinpract16020044
Triantafillidis JK, Malgarinos K, Kaklamanis L, Kritsotakis E, Polydorou V, Pantos K, Sfakianoudis K, Pantou A, Bramis K, Konstantoulakis MM, et al. The Development of Sarcoidosis in an Ulcerative Colitis Patient Treated with Vedolizumab: A Case Report and Review of the Literature. Clinics and Practice. 2026; 16(2):44. https://doi.org/10.3390/clinpract16020044
Chicago/Turabian StyleTriantafillidis, John K., Konstantinos Malgarinos, Loukas Kaklamanis, Emmanouil Kritsotakis, Victoria Polydorou, Konstantinos Pantos, Konstantinos Sfakianoudis, Agni Pantou, Konstantinos Bramis, Manousos M. Konstantoulakis, and et al. 2026. "The Development of Sarcoidosis in an Ulcerative Colitis Patient Treated with Vedolizumab: A Case Report and Review of the Literature" Clinics and Practice 16, no. 2: 44. https://doi.org/10.3390/clinpract16020044
APA StyleTriantafillidis, J. K., Malgarinos, K., Kaklamanis, L., Kritsotakis, E., Polydorou, V., Pantos, K., Sfakianoudis, K., Pantou, A., Bramis, K., Konstantoulakis, M. M., & Papalois, A. E. (2026). The Development of Sarcoidosis in an Ulcerative Colitis Patient Treated with Vedolizumab: A Case Report and Review of the Literature. Clinics and Practice, 16(2), 44. https://doi.org/10.3390/clinpract16020044







