Pre-Infection Nutritional Status, Oxidative Stress, and One-Year-Long COVID Persistence in Patients Undergoing Hemodialysis: A Prospective Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Cohort Selection
2.2. Sample Size
2.3. Data Collection and Follow-Up
2.4. Routine Clinical Evaluations
2.5. CONUT Score Calculation
2.6. Oxidative Stress Markers Determination
2.7. Statistical Analysis
3. Results
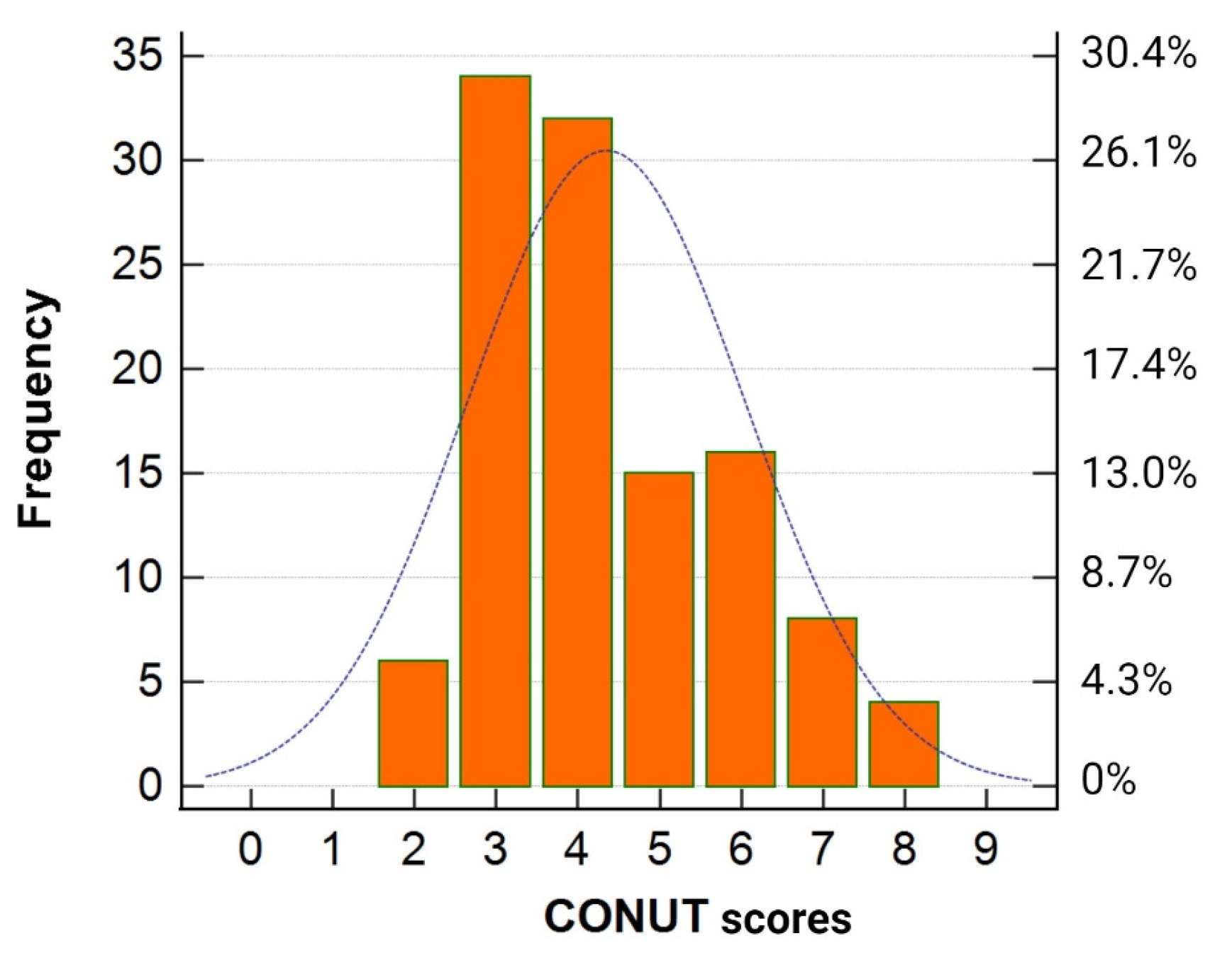
3.1. Baseline Patient Characteristics
3.2. Pre-Infection CONUT Scores and Post-COVID Oxidative Damage
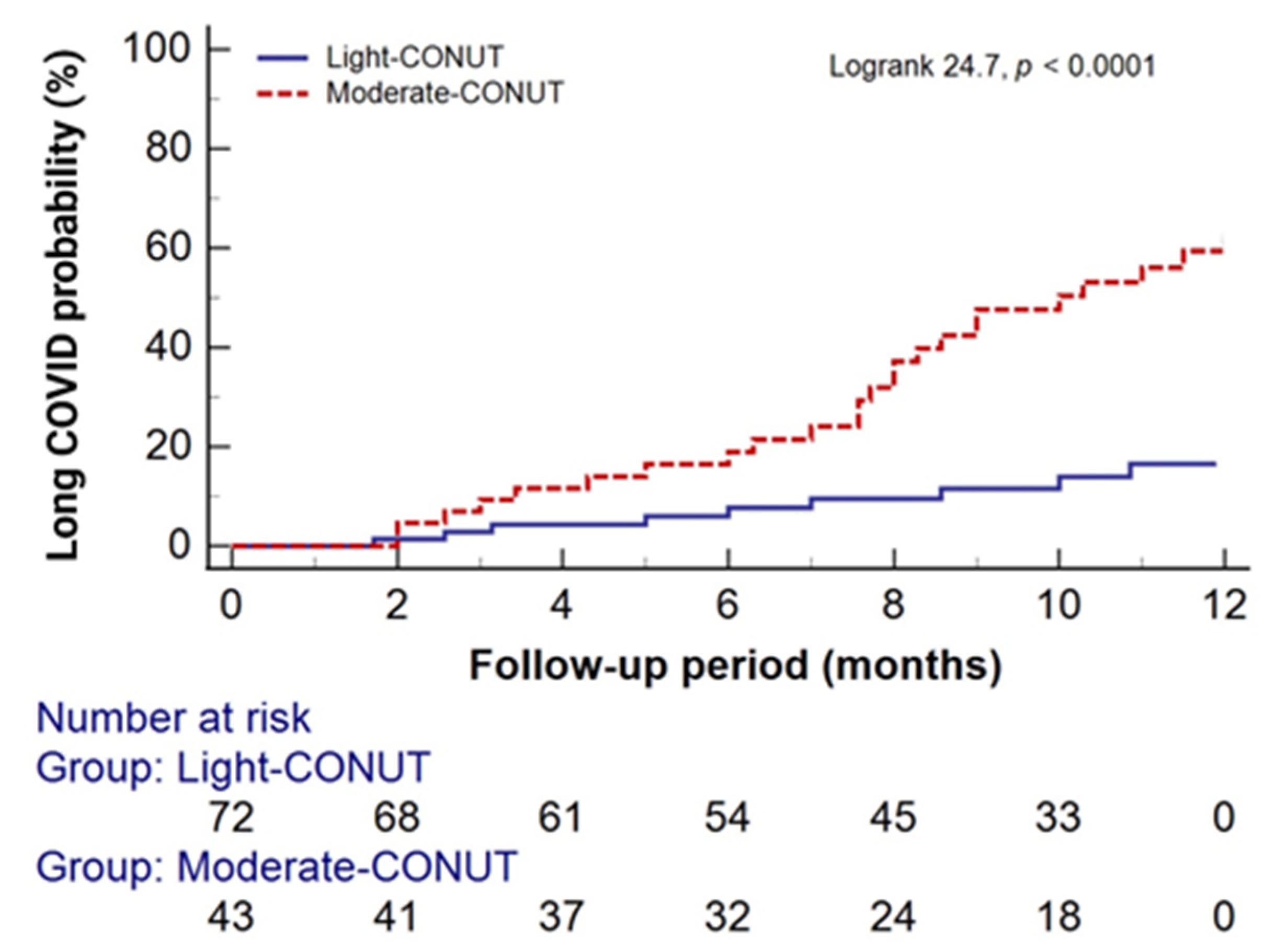
3.3. Pre-Infection CONUT Scores and One-Year-Long COVID Persistence
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prashar, J. Long COVID: Conceptualizing the Challenges for Public Health. J. Public Health 2023, 45, 771–779. [Google Scholar] [CrossRef]
- Davis, H.E.; McCorkell, L.; Vogel, J.M.; Topol, E.J. Long COVID: Major Findings, Mechanisms and Recommendations. Nat. Rev. Microbiol. 2023, 21, 408. [Google Scholar] [CrossRef]
- Jacobs, E.T.; Catalfamo, C.J.; Colombo, P.M.; Khan, S.M.; Austhof, E.; Cordova-Marks, F.; Ernst, K.C.; Farland, L.V.; Pogreba-Brown, K. Pre-Existing Conditions Associated with Post-Acute Sequelae of COVID-19. J. Autoimmun. 2023, 135, 102991. [Google Scholar] [CrossRef]
- Salerno, S.; Messana, J.M.; Gremel, G.W.; Dahlerus, C.; Hirth, R.A.; Han, P.; Segal, J.H.; Xu, T.; Shaffer, D.; Jiao, A.; et al. COVID-19 Risk Factors and Mortality Outcomes Among Medicare Patients Receiving Long-Term Dialysis. JAMA Netw. Open 2021, 4, e2135379. [Google Scholar] [CrossRef]
- Demiray, A.; Kanbay, A.; Kanbay, M. Long-Term Effect of COVID-19 Infection on Hemodialysis Patients: Should We Follow Hemodialysis Patients More Closely? Clin. Kidney J. 2022, 15, 369–371. [Google Scholar] [CrossRef]
- Subramanian, A.; Nirantharakumar, K.; Hughes, S.; Myles, P.; Williams, T.; Gokhale, K.M.; Taverner, T.; Chandan, J.S.; Brown, K.; Simms-Williams, N.; et al. Symptoms and Risk Factors for Long COVID in Non-Hospitalized Adults. Nat. Med. 2022, 28, 1706–1714. [Google Scholar] [CrossRef]
- Asadi-Pooya, A.A.; Akbari, A.; Emami, A.; Lotfi, M.; Rostamihosseinkhani, M.; Nemati, H.; Barzegar, Z.; Kabiri, M.; Zeraatpisheh, Z.; Farjoud-Kouhanjani, M.; et al. Risk Factors Associated with Long COVID Syndrome: A Retrospective Study. Iran. J. Med. Sci. 2021, 46, 428–436. [Google Scholar] [CrossRef]
- Tsampasian, V.; Elghazaly, H.; Chattopadhyay, R.; Debski, M.; Naing, T.K.P.; Garg, P.; Clark, A.; Ntatsaki, E.; Vassiliou, V.S. Risk Factors Associated With Post−COVID-19 Condition: A Systematic Review and Meta-Analysis. JAMA Intern. Med. 2023, 183, 566–580. [Google Scholar] [CrossRef]
- Sahathevan, S.; Khor, B.H.; Ng, H.M.; Gafor, A.H.A.; Daud, Z.A.M.; Mafra, D.; Karupaiah, T. Understanding Development of Malnutrition in Hemodialysis Patients: A Narrative Review. Nutrients 2020, 12, 3147. [Google Scholar] [CrossRef]
- Médica, G.A.; Ulibarri, D.; González-Madroño, J.I.; De Villar, A.; González, N.G.; González, P.; Mancha, B.; Rodríguez, A.; Fernández, F.; Hospitalaria, N.; et al. CONUT: A Tool for Controlling Nutritional Status. First Validation in a Hospital Population. Nutr. Hosp. 2005, 20, 38–45. [Google Scholar]
- Takagi, K.; Takahashi, H.; Miura, T.; Yamagiwa, K.; Kawase, K.; Muramatsu-Maekawa, Y.; Koie, T.; Mizuno, M. Prognostic Value of the Controlling Nutritional Status (CONUT) Score in Patients at Dialysis Initiation. Nutrients 2022, 14, 2317. [Google Scholar] [CrossRef] [PubMed]
- Bengelloun, A.K.; Ortega, G.J.; Ancochea, J.; Sanz-Garcia, A.; Rodríguez-Serrano, D.A.; Fernández-Jiménez, G.; Girón, R.; Ávalos, E.; Soriano, J.B.; de Ulíbarri, J.I. Usefulness of the CONUT Index upon Hospital Admission as a Potential Prognostic Indicator of COVID-19 Health Outcomes. Chin. Med. J. 2022, 135, 187–193. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.W.; Chen, Q.; Zhang, X.T.; Luo, Y.K. The CONUT Score Predicts the Length of Hospital Stay and the Risk of Long COVID. Nutr. Hosp. 2024, 41, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Deng, W.; Zhao, J.; Guo, Y.; Lai, H.; G Hu, Y.; Kang, W.; Li, Y.; Zuo, J. Improving Nutritional Status Was Associated with Decreasing Disease Severity and Shortening of Negative Conversion Time of PCR Test in Non-ICU Patients with COVID-19. Infect. Drug. Resist. 2023, 16, 4443–4452. [Google Scholar] [CrossRef] [PubMed]
- Golabi, S.; Ghasemi, S.; Adelipour, M.; Bagheri, R.; Suzuki, K.; Wong, A.; Seyedtabib, M.; Naghashpour, M. Oxidative Stress and Inflammatory Status in COVID-19 Outpatients: A Health Center-Based Analytical Cross-Sectional Study. Antioxidants 2022, 11, 606. [Google Scholar] [CrossRef] [PubMed]
- Vollbracht, C.; Kraft, K. Oxidative Stress and Hyper-Inflammation as Major Drivers of Severe COVID-19 and Long COVID: Implications for the Benefit of High-Dose Intravenous Vitamin C. Front. Pharmacol. 2022, 13, 899198. [Google Scholar] [CrossRef] [PubMed]
- Al-Hakeim, H.K.; Al-Rubaye, H.T.; Al-Hadrawi, D.S.; Almulla, A.F.; Maes, M. Long-COVID Post-Viral Chronic Fatigue and Affective Symptoms are Associated with Oxidative Damage, Lowered Antioxidant Defenses and Inflammation: A Proof of Concept and Mechanism Study. Mol. Psychiatry 2022, 28, 564–578. [Google Scholar] [CrossRef] [PubMed]
- Georgieva, E.; Ananiev, J.; Yovchev, Y.; Arabadzhiev, G.; Abrashev, H.; Abrasheva, D.; Atanasov, V.; Kostandieva, R.; Mitev, M.; Petkova-Parlapanska, K.; et al. COVID-19 Complications: Oxidative Stress, Inflammation, and Mitochondrial and Endothelial Dysfunction. Int. J. Mol. Sci. 2023, 24, 14876. [Google Scholar] [CrossRef]
- Korol, L.V.; Mygal, L.Y.; Stepanova, N.M. Intensity of Oxidative Stress and Activity of Angiotensin Converting Enzyme in Blood of Patients with Uncomplicated Pyelonephritis. Ukr. Biochem. J. 2017, 89, 99–105. [Google Scholar] [CrossRef][Green Version]
- Dufrusine, B.; Valentinuzzi, S.; Bibbò, S.; Damiani, V.; Lanuti, P.; Pieragostino, D.; Del Boccio, P.; D’Alessandro, E.; Rabottini, A.; Berghella, A.; et al. Iron Dyshomeostasis in COVID-19: Biomarkers Reveal a Functional Link to 5-Lipoxygenase Activation. Int. J. Mol. Sci. 2023, 24, 15. [Google Scholar] [CrossRef]
- Bharadwaj, S.; Ginoya, S.; Tandon, P.; Gohel, T.D.; Guirguis, J.; Vallabh, H.; Jevenn, A.; Hanouneh, I. Malnutrition: Laboratory Markers vs Nutritional Assessment. Gastroenterol. Rep. 2016, 4, 272–280. [Google Scholar] [CrossRef] [PubMed]
- McLaughlin, K.M.; Bechtel, M.; Bojkova, D.; Münch, C.; Ciesek, S.; Wass, M.N.; Michaelis, M.; Cinatl, J. COVID-19-Related Coagulopathy—Is Transferrin a Missing Link? Diagnostics 2020, 10, 539. [Google Scholar] [CrossRef] [PubMed]
- Duran, E.; Taşkın, A.; Pehlivan, B.; Çelik, H.; Pehlivan, V.F.; Taşkın, S. Dynamic Thiol Disulphide Homeostasis in the Follow-Up of the Prognosis of Patients Treated for COVID-19 in the Intensive Care Unit. Cureus 2022, 14, e27542. [Google Scholar] [CrossRef] [PubMed]
- Stepanova, N.; Rysyev, A.; Rusyn, O.; Ostapenko, T.; Snisar, L.; Kompaniets, O.; Kolesnyk, M. High-Density Lipoproteins and Clinical Outcomes of COVID-19 in Hemodialysis Patients: A Multicenter, Propensity-Score Matched Case-Control Study. Ukr. J. Nephrol. Dial. 2022, 1, 22–30. [Google Scholar] [CrossRef]
- Tosato, M.; Calvani, R.; Ciciarello, F.; Galluzzo, V.; Martone, A.M.; Zazzara, M.B.; Pais, C.; Savera, G.; Robles, M.C.; Ramirez, M.; et al. Malnutrition in COVID-19 Survivors: Prevalence and Risk Factors. Aging Clin. Exp. Res. 2023, 35, 2257–2265. [Google Scholar] [CrossRef] [PubMed]
- Deer, R.R.; Hosein, E.; Harvey, M.; Nguyen, T.; Givan, A.; Hamilton, M.; Turner, K.; Kretzmer, R.; Rock, M.; Swartz, M.C.; et al. Impact of COVID-19 Infection and Persistent Lingering Symptoms on Patient Reported Indicators of Nutritional Risk and Malnutrition. Nutrients 2022, 14, 642. [Google Scholar] [CrossRef] [PubMed]
- Mortaz, E.; Bezemer, G.; Alipoor, S.D.; Varahram, M.; Mumby, S.; Folkerts, G.; Garssen, J.; Adcock, I.M. Nutritional Impact and Its Potential Consequences on COVID-19 Severity. Front. Nutr. 2021, 8, 698617. [Google Scholar] [CrossRef]
- Schloss, J.V. Nutritional Deficiencies That May Predispose to Long COVID. Inflammopharmacology 2023, 31, 573–583. [Google Scholar] [CrossRef] [PubMed]
- De Araújo Morais, A.H.; Aquino, J.D.S.; Da Silva-Maia, J.K.; Vale, S.H.D.L.; MacIel, B.L.L.; Passos, T.S. Nutritional Status, Diet and Viral Respiratory Infections: Perspectives for Severe Acute Respiratory Syndrome Coronavirus 2. Br. J. Nutr. 2021, 125, 851–862. [Google Scholar] [CrossRef]
- Rodriguez-leyva, D.; Pierce, G.N. The Impact of Nutrition on the COVID-19 Pandemic and the Impact of the COVID-19 Pandemic on Nutrition. Nutrients 2021, 13, 1752. [Google Scholar] [CrossRef]
- Shifris, I.; Korol, L.; Krasiuk, E.; Dudar, S. Activation of Oxidative Stress, Comorbidity and Survival of End-Stage Renal Disease Patients Treated with Hemodialysis. Ukr. J. Nephrol. Dial. 2021, 4, 67–77. [Google Scholar] [CrossRef]
- Lin, T.Y.; Hung, N.K.; Hung, S.C. Association of Malnutrition with SARS-CoV-2 Vaccine Response in Patients Undergoing Hemodialysis. Clin. Nutr. 2022, 41, 2683–2690. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, H.; Önder, A.; Becker, J.; Gröger, M.; Müller, M.M.; Zink, F.; Stein, B.; Radermacher, P.; Waller, C. Markers of Oxidative Stress during Post-COVID-19 Fatigue: A Hypothesis-Generating, Exploratory Pilot Study on Hospital Employees. Front. Med. 2023, 10, 1305009. [Google Scholar] [CrossRef] [PubMed]
- Stepanova, N.; Korol, L.; Snisar, L.; Rysyev, A.; Ostapenko, T.; Marchenko, V.; Belousova, O.; Popova, O.; Malashevska, N.; Kolesnyk, M. Long-COVID Sequelae Are Associated with Oxidative Stress in Hemodialysis Patients. Ukr. J. Nephrol. Dial. 2023, 1, 31–39. [Google Scholar] [CrossRef]
- Stufano, A.; Isgrò, C.; Palese, L.L.; Caretta, P.; De Maria, L.; Lovreglio, P.; Sardanelli, A.M. Oxidative Damage and Post-COVID Syndrome: A Cross-Sectional Study in a Cohort of Italian Workers. Int. J. Mol. Sci. 2023, 24, 7445. [Google Scholar] [CrossRef] [PubMed]
- Reștea, P.A.; Țigan, Ș.; Vicaș, L.G.; Fritea, L.; Marian, E.; Jurca, T.; Pallag, A.; Mureșan, I.L.; Moisa, C.; Micle, O.; et al. Serum Level of Ceruloplasmin, Angiotensin-Converting Enzyme and Transferrin as Markers of Severity in SARS-CoV-2 Infection in Patients with Type 2 Diabetes. Microbiol. Res. 2023, 14, 1670–1686. [Google Scholar] [CrossRef]
- Iddir, M.; Brito, A.; Dingeo, G.; Del Campo, S.S.F.; Samouda, H.; La Frano, M.R.; Bohn, T. Strengthening the Immune System and Reducing Inflammation and Oxidative Stress through Diet and Nutrition: Considerations during the COVID-19 Crisis. Nutrients 2020, 12, 1562. [Google Scholar] [CrossRef] [PubMed]
- Arruda de Souza Monnerat, J.; Ribeiro de Souza, P.; Monteiro da Fonseca Cardoso, L.; Dario Mattos, J.; de Souza Rocha, G.; Frauches Medeiros, R. Micronutrients and Bioactive Compounds in the Immunological Pathways Related to SARS-CoV-2 (Adults and Elderly). Eur. J. Nutr. 2020, 60, 559–579. [Google Scholar] [CrossRef] [PubMed]
- Ye, H.; Liu, Q.; Wang, Y.; Zhen, X.; Yan, N. The Effect of Cholesterol Efflux on Endothelial Dysfunction Caused by Oxidative Stress. Int. J. Mol. Sci. 2023, 24, 5939. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Gevezova, M.; Sarafian, V.; Maes, M. Redox Regulation of the Immune Response. Cell. Mol. Immunol. 2022, 19, 1079–1101. [Google Scholar] [CrossRef]
- Liakopoulos, V.; Roumeliotis, S.; Zarogiannis, S.; Eleftheriadis, T.; Mertens, P.R. Oxidative Stress in Hemodialysis: Causative Mechanisms, Clinical Implications, and Possible Therapeutic Interventions. Semin. Dial. 2019, 32, 58–71. [Google Scholar] [CrossRef] [PubMed]
- Trujillo-Mayol, I.; Guerra-Valle, M.; Casas-Forero, N.; Sobral, M.M.C.; Viegas, O.; Alarcón-Enos, J.; Ferreira, I.M.; Pinho, O. Western Dietary Pattern Antioxidant Intakes and Oxidative Stress: Importance During the SARS-CoV-2/COVID-19 Pandemic. Adv. Nutr. 2021, 12, 670–681. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, P.; Janmeda, P.; Docea, A.O.; Yeskaliyeva, B.; Abdull Razis, A.F.; Modu, B.; Calina, D.; Sharifi-Rad, J. Oxidative Stress, Free Radicals and Antioxidants: Potential Crosstalk in the Pathophysiology of Human Diseases. Front. Chem. 2023, 11, 1158198. [Google Scholar] [CrossRef] [PubMed]
- Voelkle, M.; Gregoriano, C.; Neyer, P.; Koch, D.; Kutz, A.; Bernasconi, L.; Conen, A.; Mueller, B.; Schuetz, P. Prevalence of Micronutrient Deficiencies in Patients Hospitalized with COVID-19: An Observational Cohort Study. Nutrients 2022, 14, 1862. [Google Scholar] [CrossRef] [PubMed]
- Bakadia, B.M.; Boni, B.O.O.; Ahmed, A.A.Q.; Yang, G. The Impact of Oxidative Stress Damage Induced by the Environmental Stressors on COVID-19. Life Sci. 2021, 264, 118653. [Google Scholar] [CrossRef] [PubMed]
- Muhammad, Y.; Kani, Y.A.; Iliya, S.; Muhammad, J.B.; Binji, A.; El-Fulaty Ahmad, A.; Kabir, M.B.; Umar Bindawa, K.; Ahmed, A. Deficiency of Antioxidants and Increased Oxidative Stress in COVID-19: A Cross-Sectional Comparative Study in Jigawa, Northwestern. SAGE Open Med. 2021, 9, 2050312121991246. [Google Scholar] [CrossRef]
- Naidu, A.S.; Wang, C.K.; Rao, P.; Mancini, F.; Clemens, R.A.; Wirakartakusumah, A.; Chiu, H.F.; Yen, C.H.; Porretta, S.; Mathai, I.; et al. Precision Nutrition to Reset Virus-Induced Human Metabolic Reprogramming and Dysregulation (HMRD) in Long-COVID. NPJ Sci. Food 2024, 8, 19. [Google Scholar] [CrossRef]


| Parameter | Undernutrition Degree | |||
|---|---|---|---|---|
| Normal | Light | Moderate | Severe | |
| Serum albumin, g/L | 30.5–40.5 | 30.0–30.49 | 20.5–20.9 | <20.5 |
| Score | 0 | 2 | 4 | 6 |
| TLC, /mm3 | >1600 | 1200–1599 | 800–1199 | <800 |
| Score | 0 | 1 | 2 | 3 |
| Cholesterol, mmol/L | >4.65 | 3.62–4.65 | 2.59–3.61 | <2.59 |
| Score | 0 | 1 | 2 | 3 |
| Screening total score | 0–1 | 2–4 | 5–8 | 9–12 |
| Clinical Parameters | All Patients (n = 115) | Light CONUT Group (n = 72) | Moderate CONUT Group (n = 43) | p-Value |
|---|---|---|---|---|
| Demographic and clinical data | ||||
| Male sex, n (%) | 50 (43.5%) | 34 (47.2%) | 16 (37.2%) | 0.29 |
| Age, years | 52.2 ± 11.6 | 49.8 ± 10.6 | 53.7 ± 12.1 | 0.08 |
| Diabetes | 17 (14.8%) | 10 (13.9%) | 7 (16.3%) | 0.73 |
| Systolic blood pressure, mm Hg | 130 (120–140) | 130 (120–140) | 130 (130–140) | 0.14 |
| Diastolic blood pressure, mm Hg | 80 (70–90) | 80 (70–85) | 80 (72–90) | 0.32 |
| BMI, kg/m2 | 25.0 (22.1–30.1) | 25.3 (22.4–30.3) | 24.3 (21.1–29.4) | 0.36 |
| Time on HD, months | 72.0 (30.3–138.2) | 69.0 (29.5–126.0) | 72.0 (36.0–163.7) | 0.36 |
| spKt/V | 1.4 (1.3–1.6) | 1.4 (1.3–1.7) | 1.3 (1.2–1.6) | 0.16 |
| Anuric patients, n (%) | 58 (50.4%) | 31 (31.9%) | 27 (62.8%) | 0.001 |
| Serum albumin, g/L | 35.2 (34.2–40.6) | 36.5 (34.1–41.6) | 34.7 (31.5–38.7) | 0.003 |
| TLC, mm3 | 1200 (824–3760) | 1200 (780–3750) | 981 (560–2350) | 0.04 |
| CRP, mg/L | 5.9 (4.6–10.7) | 4.6 (3.3–9.4) | 6.8 (5.1–11.2) | 0.02 |
| Hb, g/L | 102 (95–108) | 103 (95–107.5) | 102 (95–108) | 0.65 |
| Potassium, mmol/L | 4.42 (3.9–5.1) | 4.7 (3.9–5.7) | 4.3 (3.8–4.9) | 0.76 |
| Calcium, mmol/L | 2.25 (2.1–2.4) | 2.28 (2.1–2.4) | 2.16 (2.1–2.3) | 0.62 |
| Phosphorus, mmol/L | 1.75 (1.5–2.1) | 1.6 (1.4–1.9) | 1.8 (1.5–2.1) | 0.14 |
| PTH, ng/L | 259 (164–381) | 222.3 (125.2–352.6) | 303.6 (192.5–476.8) | 0.06 |
| Total cholesterol, mmol/L | 4.9 ± 1.18 | 5.2 ± 1.17 | 4.5 ± 1.07 | 0.002 |
| CONUT score | 4 (4–6) | 4 (3–4) | 6 (5–7) | <0.0001 |
| Medications | ||||
| ACE inhibitors/RAAS blockers, n (%) | 64 (5.6%) | 42 (58.3%) | 22 (51.2%) | 0.46 |
| Beta blockers, n (%) | 81 (70.4%) | 55 (76.4%) | 26 (60.4%) | 0.07 |
| Calcium channel blockers, n (%) | 76 (66.1%) | 49 (68.1%) | 27 (62.8%) | 0.56 |
| Alpha blockers, n (%) | 19 (16.5%) | 11 (15.3%) | 8 (18.6%) | 0.64 |
| Iron supplementation, n (%) | 63 (54.8%) | 39 (54.2%) | 24 (55.8%) | 0.87 |
| Erythropoietins, n (%) | 96 (83.5%) | 61 (84.7%) | 35 (81.4%) | 0.65 |
| Non-calcium phosphate binders, n (%) | 45 (39.1%) | 28 (38.9%) | 17 (39.5%) | 0.95 |
| Vaccination status and acute COVID-19 severity | ||||
| Vaccinated status for COVID-19, n (%) | 40 (34.8%) | 32 (44.4%) | 8 (18.8%) | 0.005 |
| Asymptomatic COVID-19, n (%) | 24 (20.9%) | 21 (29.2%) | 5 (11.6%) | 0.03 |
| Mild to moderate COVID-19, n (%) | 73 (63.5%) | 41 (56.9%) | 26 (60.5%) | 0.71 |
| Hospitalization with oxygen supply, n (%) | 18 (15.6%) | 10 (13.9%) | 12 (27.9%) | 0.07 |
| Markers | All Patients (n = 115) | Light CONUT Group (n = 72) | Moderate CONUT Group (n = 43) | p-Value |
|---|---|---|---|---|
| MDAs, μmol/L | 269.0 (218.0–368.7) | 240.3 (205.1–274.5) | 403.6 (258.2–512.7) | <0.0001 |
| MDAe, μmol/L | 769.0 (641.3–945.5) | 750.4 (728.1–936.3) | 859.3 (746.4–948.8) | 0.051 |
| Transferrin, g/L | 2.5 (1.7–5.0) | 3.0 (1.7–5.2) | 2.3 (1.6–5.1) | 0.49 |
| Ceruloplasmin, g/L | 0.19 (0.12–0.24) | 0.21 (0.14–0.31) | 0.14 (0.11–0.21) | 0.0009 |
| SH groups, mmol/L | 1.56 (1.34–1.76) | 1.56 (1.42–1.75) | 1.54 (1.34–1.81) | 0.89 |
| Variable | b | SE | Wald χ2 | p-Values | HR (95% CI) |
|---|---|---|---|---|---|
| Unadjusted | 1.3 | 0.31 | 18.4 | <0.0001 | 3.7 (2.1; 6.8) |
| Model 1 | 1.9 | 0.38 | 4.85 | <0.0001 | 6.8 (3.2; 9.4) |
| Model 2 | 1.5 | 0.61 | 6.5 | 0.01 | 4.6 (1.4; 9.9) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stepanova, N.; Korol, L.; Ostapenko, T.; Marchenko, V.; Belousova, O.; Snisar, L.; Shifris, I.; Kolesnyk, M. Pre-Infection Nutritional Status, Oxidative Stress, and One-Year-Long COVID Persistence in Patients Undergoing Hemodialysis: A Prospective Cohort Study. Clin. Pract. 2024, 14, 892-905. https://doi.org/10.3390/clinpract14030070
Stepanova N, Korol L, Ostapenko T, Marchenko V, Belousova O, Snisar L, Shifris I, Kolesnyk M. Pre-Infection Nutritional Status, Oxidative Stress, and One-Year-Long COVID Persistence in Patients Undergoing Hemodialysis: A Prospective Cohort Study. Clinics and Practice. 2024; 14(3):892-905. https://doi.org/10.3390/clinpract14030070
Chicago/Turabian StyleStepanova, Natalia, Lesya Korol, Tetyana Ostapenko, Valeriia Marchenko, Olga Belousova, Lyudmyla Snisar, Iryna Shifris, and Mykola Kolesnyk. 2024. "Pre-Infection Nutritional Status, Oxidative Stress, and One-Year-Long COVID Persistence in Patients Undergoing Hemodialysis: A Prospective Cohort Study" Clinics and Practice 14, no. 3: 892-905. https://doi.org/10.3390/clinpract14030070
APA StyleStepanova, N., Korol, L., Ostapenko, T., Marchenko, V., Belousova, O., Snisar, L., Shifris, I., & Kolesnyk, M. (2024). Pre-Infection Nutritional Status, Oxidative Stress, and One-Year-Long COVID Persistence in Patients Undergoing Hemodialysis: A Prospective Cohort Study. Clinics and Practice, 14(3), 892-905. https://doi.org/10.3390/clinpract14030070








