Phytotherapy Might Have a Role in Reducing Unnecessary Prostate Biopsies: Results from an Exploratory, Randomized Controlled Trial of Two Different Phytotherapeutic Agents
Abstract
1. Introduction
2. Materials and Methods
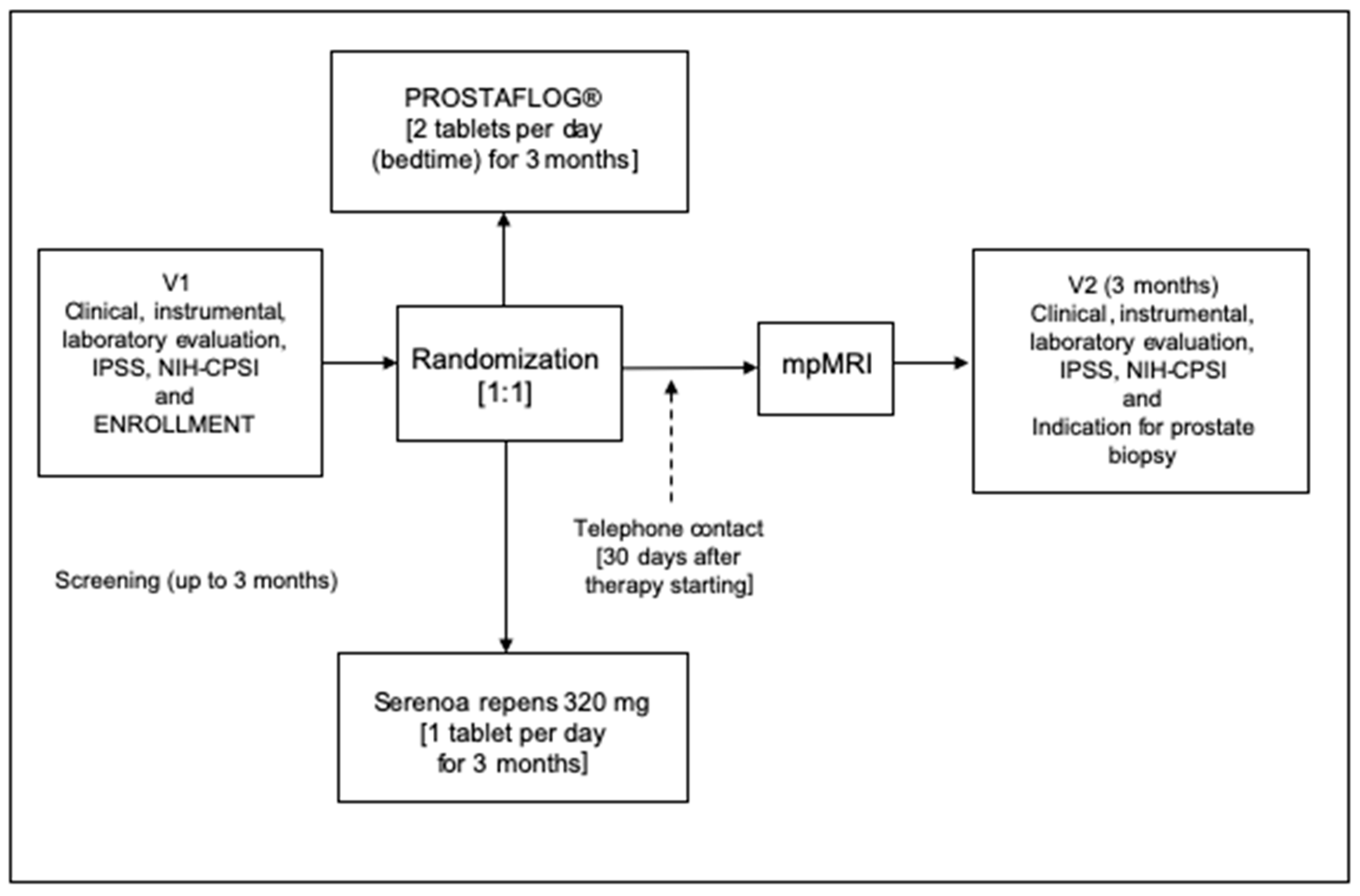
2.1. Study Design and Schedule
2.2. Inclusion and Exclusion Criteria
2.3. Laboratory Considerations
2.4. Instrumental Considerations
2.5. Questionnaires
2.6. Composition and Characterization of the Extracts Used
2.7. Ethical and Statistical Considerations
3. Results
3.1. Patient Populations
3.2. mpMRI Data
3.3. PSA Values at the Follow-Up Visit
3.4. Decisions on Prostate Biopsy and Findings of Prostate Cancer
4. Discussion
4.1. Major Findings
4.2. Understanding Study Findings
4.3. Results in Comparison with Other Studies
4.4. Clinical Implications
4.5. Strengths and Limitations of the Present Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bergengren, O.; Pekala, K.R.; Matsoukas, K.; Fainberg, J.; Mungovan, S.F.; Bratt, O.; Bray, F.; Brawley, O.; Luckenbaugh, A.N.; Mucci, L.; et al. 2022 Update on Prostate Cancer Epidemiology and Risk Factors-A Systematic Review. Eur. Urol. 2023, 84, 191–206. [Google Scholar] [CrossRef]
- Van Poppel, H.; Albreht, T.; Basu, P.; Hogenhout, R.; Collen, S.; Roobol, M. Serum PSA-based early detection of prostate cancer in Europe and globally: Past, present and future. Nat. Rev. Urol. 2022, 19, 562–572. [Google Scholar] [CrossRef]
- Mottet, N.; van den Bergh, R.C.N.; Briers, E.; Van den Broeck, T.; Cumberbatch, M.G.; De Santis, M.; Fanti, S.; Fossati, N.; Gandaglia, G.; Gillessen, S.; et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on Prostate Cancer-2020 Update. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2021, 79, 243–262. [Google Scholar] [CrossRef]
- Thompson, J.; Lawrentschuk, N.; Frydenberg, M.; Thompson, L.; Stricker, P.; USANZ. The role of magnetic resonance imaging in the diagnosis and management of prostate cancer. BJU Int. 2013, 112 (Suppl. 2), 6–20. [Google Scholar] [CrossRef]
- Barret, E.; Turkbey, B.; Puech, P.; Durand, M.; Panebianco, V.; Fütterer, J.J.; Renard-Penna, R.; Rouvière, O. Update on the ICUD-SIU consultation on multi-parametric magnetic resonance imaging in localised prostate cancer. World J. Urol. 2019, 37, 429–436. [Google Scholar] [CrossRef]
- Alidjanov, J.F.; Cai, T.; Bartoletti, R.; Bonkat, G.; Bruyère, F.; Köves, B.; Kulchavenya, E.; Medina-Polo, J.; Naber, K.; Perepanova, T.; et al. The negative aftermath of prostate biopsy: Prophylaxis, complications and antimicrobial stewardship: Results of the global prevalence study of infections in urology 2010–2019. World J. Urol. 2021, 39, 3423–3432. [Google Scholar] [CrossRef]
- Kayalı, Y.; Balbay, M.D.; İlktaç, A.; Ersöz, C.; Toprak, H.; Tarım, K.; Eden, A.B.; Akçay, M.; Doğan, B. PSA change after antibiotic treatment should not affect decisionmaking on performing a prostate biopsy. Turk. J. Med. Sci. 2023, 53, 183–192. [Google Scholar] [CrossRef]
- Muratore, E.; Baccelli, F.; Leardini, D.; Campoli, C.; Belotti, T.; Viale, P.; Prete, A.; Pession, A.; Masetti, R.; Zama, D. Antimicrobial Stewardship Interventions in Pediatric Oncology: A Systematic Review. J. Clin. Med. 2022, 11, 4545. [Google Scholar] [CrossRef]
- Cicero, A.F.G.; Allkanjari, O.; Busetto, G.M.; Cai, T.; Larganà, G.; Magri, V.; Perletti, G.; Robustelli Della Cuna, F.S.; Russo, G.I.; Stamatiou, K.; et al. Nutraceutical treatment and prevention of benign prostatic hyperplasia and prostate cancer. Arch. Ital. Urol. Androl. 2019, 91. [Google Scholar] [CrossRef]
- Milanese, G.; Agostini, E.; De Angelis, M.V.; Pretore, E.; Galosi, A.B.; Castellani, D. Efficacy of 1-Year Cavacurmin® Therapy in Reducing Prostate Growth in Men Suffering from Lower Urinary Tract Symptoms. J. Clin. Med. 2023, 12, 1689. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.-P.; Liang, S.-B.; Liang, N.; Bu, F.-L.; Lai, B.-Y.; Zhang, Y.-P.; Cao, H.-J.; Fei, Y.-T.; Robinson, N. The potential effects and use of Chinese herbal medicine pine pollen (Pinus pollen): A bibliometric analysis of pharmacological and clinical studies. World J. Tradit. Chin. Med. 2020, 6, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Leslie, S.W.; Soon-Sutton, T.L.; Anu, R.I.; Hussain, S.; Larry, E. Siref. Prostate Cancer. [Updated 2023 May 30]. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK470550/ (accessed on 19 September 2023).
- De Nunzio, C.; Lombardo, R.; Nacchia, A.; Tema, G.; Tubaro, A. Repeat prostate-specific antigen (PSA) test before prostate biopsy: A 20% decrease in PSA values is associated with a reduced risk of cancer and particularly of high-grade cancer. BJU Int. 2018, 122, 83–88. [Google Scholar] [CrossRef]
- Ferraro, S.; Biganzoli, E.M. The clinical value of assessing the inter-method bias: The lesson from prostate specific antigen measurement. Clin. Chem. Lab. Med. 2021, 60, 149–151. [Google Scholar] [CrossRef] [PubMed]
- Heidegger, I.; Fritz, J.; Klocker, H.; Pichler, R.; Bektic, J.; Horninger, W. Age-Adjusted PSA Levels in Prostate Cancer Prediction: Updated Results of the Tyrol Prostate Cancer Early Detection Program. PLoS ONE 2015, 10, e0134134. [Google Scholar] [CrossRef] [PubMed]
- Turkbey, B.; Rosenkrantz, A.B.; Haider, M.A.; Padhani, A.R.; Villeirs, G.; Macura, K.J.; Tempany, C.M.; Choyke, P.L.; Cornud, F.; Margolis, D.J.; et al. Prostate Imaging Reporting and Data System Version 2.1: 2019 Update of Prostate Imaging Reporting and Data System Version 2. Eur. Urol. 2019, 76, 340–351. [Google Scholar] [CrossRef]
- Badia, X.; Garcia-Losa, M.; Dal-Re, R. Ten-language translation and harmonization of the International Prostate Symptom Score: Developing a methodology for multinational clinical trials. Eur. Urol. 1997, 31, 129–140. [Google Scholar] [CrossRef]
- Giubilei, G.; Mondaini, N.; Crisci, A.; Raugei, A.; Lombardi, G.; Travaglini, F.; Del Popolo, G.; Bartoletti, R. The Italian version of the National Institutes of Health Chronic Prostatitis Symptom Index. Eur. Urol. 2005, 47, 805–811. [Google Scholar] [CrossRef]
- Engelhardt, P.F.; Riedl, C.R. Effects of one-year treatment with isoflavone extract from red clover on prostate, liver function, sexual function, and quality of life in men with elevated PSA levels and negative prostate biopsy findings. Urology 2008, 71, 185–190, discussion 190. [Google Scholar] [CrossRef]
- Cai, T.; Anceschi, U.; Tamanini, I.; Verze, P.; Palmieri, A. Soybean Extracts (Glycine Max) with Curcuma, Boswellia, Pinus and Urtica Are Able to Improve Quality of Life in Patients Affected by CP/CPPS: Is the Pro-Inflammatory Cytokine IL-8 Level Decreasing the Physiopathological Link? Uro 2022, 2, 40–48. [Google Scholar] [CrossRef]
- Sibona, M.; Destefanis, P.; Agnello, M.; Lillaz, B.; Giuliano, M.; Cai, T.; Gontero, P. The association of Boswellia resin extract and propolis derived polyphenols can improve quality of life in patients affected by prostatitis-like symptoms. Arch. Ital. Urol. Androl. 2020, 91, 251–255. [Google Scholar] [CrossRef]
- Poeckel, D.; Werz, O. Boswellic acids: Biological actions and molecular targets. Curr. Med. Chem. 2006, 13, 3359–3369. [Google Scholar] [CrossRef] [PubMed]
- Namazi, F.; Bordbar, E.; Bakhshaei, F.; Nazifi, S. The effect of Urtica dioica extract on oxidative stress, heat shock proteins, and brain histopathology in multiple sclerosis model. Physiol. Rep. 2022, 10, e15404. [Google Scholar] [CrossRef] [PubMed]
- Kryvenko, O.N.; Wang, Y.; Sadasivan, S.; Gupta, N.S.; Rogers, C.; Bobbitt, K.; Chitale, D.A.; Rundle, A.; Tang, D.; Rybicki, B.A. Potential effect of anti-inflammatory drug use on PSA kinetics and subsequent prostate cancer diagnosis: Risk stratification in black and white men with benign prostate biopsy. Prostate 2019, 79, 1090–1098. [Google Scholar] [CrossRef] [PubMed]
- Greiman, A.; Shah, J.; Bhavsar, R.; Armeson, K.; Caulder, S.; Jones, R.; Keane, T.E.; Clarke, H.S.; Savage, S.J. Six Weeks of Fluoroquinolone Antibiotic Therapy for Patients with Elevated Serum Prostate-specific Antigen Is Not Clinically Beneficial: A Randomized Controlled Clinical Trial. Urology 2016, 90, 32–37. [Google Scholar] [CrossRef]
- Eggener, S.E.; Large, M.C.; Gerber, G.S.; Pettus, J.; Yossepowitch, O.; Smith, N.D.; Kundu, S.; Kunnavakkam, R.; Zorn, K.; Raman, J.D. Empiric antibiotics for an elevated prostate-specific antigen (PSA) level: A randomised, prospective, controlled multi-institutional trial. BJU Int. 2013, 112, 925–929. [Google Scholar] [CrossRef]
- Busato, W.F.; Almeida, G.L.; Geraldo, J.; Busato, F.S. Does PSA reduction after antibiotic therapy permits postpone prostate biopsy in asymptomatic men with PSA levels between 4 and 10 ng/mL? Int. Braz. J. Urol. 2015, 41, 329–336. [Google Scholar] [CrossRef]
- Baltaci, S.; Aksoy, H.; Türkölmez, K.; Elhan, A.H.; Ozden, E.; Göğüş, O. Use of percent free prostate-specific antigen density to improve the specificity for detecting prostate cancer in patients with normal rectal examinations and intermediate prostate-specific antigen levels. Urol. Int. 2003, 70, 36–41. [Google Scholar] [CrossRef]
- Fabiani, A.; Morosetti, C.; Filosa, A.; Principi, E.; Lepri, L.; Maurelli, V.; Fioretti, F.; Servi, L. Effect on prostatic specific antigen by a short time treatment with a Curcuma extract: A real life experience and implications for prostate biopsy. Arch. Ital. Urol. Androl. 2018, 90, 107–111. [Google Scholar] [CrossRef]
- Cindolo, L.; Fabiani, A.; Vitelli, D.; Cianci, F.; Gatti, L.; Ghidini, N.; Nyek Ntep, N.; Piazza, R.C.; Filosa, A.; Ferrari, G. Variation of inflammatory indexes in patients with chronic abacterial prostatitis treated with an herbal compound/extract. Arch. Ital. Urol. Androl. 2023, 95, 11441. [Google Scholar] [CrossRef]
- Corsi, A.; De Bernardi, E.; Bonaffini, P.A.; Franco, P.N.; Nicoletta, D.; Simonini, R.; Ippolito, D.; Perugini, G.; Occhipinti, M.; Da Pozzo, L.F.; et al. Radiomics in PI-RADS 3 Multiparametric MRI for Prostate Cancer Identification: Literature Models Re-Implementation and Proposal of a Clinical-Radiological Model. J. Clin. Med. 2022, 11, 6304. [Google Scholar] [CrossRef]
- Krimphove, M.J.; Fletcher, S.A.; Trinh, Q.D. Multiparametric magnetic resonance imaging for prostate cancer detection: Do clinical trial findings reflect real-world practice? BJU Int. 2019, 123, 197–198. [Google Scholar] [CrossRef] [PubMed]

| Treatment Group A | Control Group B | p | |
|---|---|---|---|
| No. of patients | 66 | 76 | |
| Age (years) | 0.97 | ||
| Median (IQR †) | 65 (56–76) | 66 (54–77) | |
| BMI (Kg/m2) | 0.71 | ||
| Median (IQR †) | 27 (25–31) | 28 (26–30) | |
| Prostate volume (cc) | 0.11 | ||
| Median (IQR †) | 52 (41–87) | 54 (42–86) | |
| Charlson Comorbidity Index (CCI) | 0.08 | ||
| 0 | 51 (77.2) | 57 (75.0) | |
| 1 | 15 (22.8) | 19 (25.0) | |
| 2 | 0 (-) | 0 (-) | |
| Urinary symptoms | 0.10 | ||
| Voiding | 12 (18.2) | 15 (19.8) | |
| Storage | 4 (6.0) | 5 (6.5) | |
| Mixed | 15 (22.7) | 17 (22.3) | |
| Absence of symptoms | 35 (53.1) | 39 (51.4) | |
| PSA total (ng/mL) | 0.52 | ||
| Median (IQR †) | 6.7 (4.1–8.9) | 6.5 (4.3–9.0) | |
| PSA free/total (%) | 0.90 | ||
| Median (IQR †) | 12 (8–14) | 11 (8–13) |
| Treatment Group A | Control Group B | |
|---|---|---|
| No. of patients | 66 | 76 |
| mpMRI | ||
| PI-RADS I | 10 (15.1) | 12 (15.8) |
| PI-RADS II | 44 (66.6) | 50 (65.7) |
| PI-RADS III | 9 (13.6) | 9 (11.9) |
| PI-RADS IV | 2 (3.1) | 4 (5.3) |
| PI-RADS V | 1 (1.6) | 1 (1.3) |
| PSA total (ng/mL) | ||
| Median (IQR †) | 3.1 (1.8–5.1) | 4.2 (2.1–8.4) |
| PSA free/total (%) | ||
| Median (IQR †) | 14 (10–17) | 12 (9–15) |
| PSA density (ng/mL2) | ||
| Median (IQR †) | 0.13 (0.11–0.16) | 0.13 (0.12–0.16) |
| Treatment Group A | Control Group B | |||
|---|---|---|---|---|
| No. of patients | 66 | 76 | ||
| ΔPSA (−) | ΔPSA (+) | ΔPSA (−) | ΔPSA (+) | |
| mpMRI (PI-RADS III or higher) | 3 | 9 | 9 | 5 |
| mpMRI (PI-RADS I or II) | 47 | 7 | 40 | 22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, T.; Tamanini, I.; Puglisi, M.; Bizzotto, L.; Rizzo, M.; Liguori, G.; Gallelli, L.; Palmieri, A.; Bjerklund Johansen, T.E. Phytotherapy Might Have a Role in Reducing Unnecessary Prostate Biopsies: Results from an Exploratory, Randomized Controlled Trial of Two Different Phytotherapeutic Agents. Clin. Pract. 2024, 14, 188-197. https://doi.org/10.3390/clinpract14010016
Cai T, Tamanini I, Puglisi M, Bizzotto L, Rizzo M, Liguori G, Gallelli L, Palmieri A, Bjerklund Johansen TE. Phytotherapy Might Have a Role in Reducing Unnecessary Prostate Biopsies: Results from an Exploratory, Randomized Controlled Trial of Two Different Phytotherapeutic Agents. Clinics and Practice. 2024; 14(1):188-197. https://doi.org/10.3390/clinpract14010016
Chicago/Turabian StyleCai, Tommaso, Irene Tamanini, Marco Puglisi, Leonardo Bizzotto, Michele Rizzo, Giovanni Liguori, Luca Gallelli, Alessandro Palmieri, and Truls E. Bjerklund Johansen. 2024. "Phytotherapy Might Have a Role in Reducing Unnecessary Prostate Biopsies: Results from an Exploratory, Randomized Controlled Trial of Two Different Phytotherapeutic Agents" Clinics and Practice 14, no. 1: 188-197. https://doi.org/10.3390/clinpract14010016
APA StyleCai, T., Tamanini, I., Puglisi, M., Bizzotto, L., Rizzo, M., Liguori, G., Gallelli, L., Palmieri, A., & Bjerklund Johansen, T. E. (2024). Phytotherapy Might Have a Role in Reducing Unnecessary Prostate Biopsies: Results from an Exploratory, Randomized Controlled Trial of Two Different Phytotherapeutic Agents. Clinics and Practice, 14(1), 188-197. https://doi.org/10.3390/clinpract14010016








