Abstract
End-stage renal disease (ESRD) patients have a high prevalence of coronary artery disease, and coronary artery bypass graft (CABG) is one of the essential treatments. ESRD patients undergoing CABG surgery have an increased risk of postoperative complications, including acute pancreatitis. Here, we present the unique case of an exceptionally large pancreatic pseudocyst caused by pancreatitis in an ESRD patient after CABG surgery. A 45-year-old male with ESRD under maintenance hemodialysis received CABG surgery for significant coronary artery disease. Two weeks later, he experienced worsening abdominal pain and a palpable mass was noticed in the epigastric region. Computer tomography revealed an unusually large pseudocyst measuring 21 × 17 cm in the retroperitoneum due to necrotizing pancreatitis. The patient underwent percutaneous cystic drainage, and the symptoms were significantly improved without surgical intervention. Factors such as prolonged cardiopulmonary bypass time, postoperative hypotension, and intradialytic hypotension appeared to have contributed to the development of severe pancreatitis in this case. This report highlights the rarity of a giant pancreatic pseudocyst in an ESRD patient after CABG surgery and emphasizes the importance of vigilant postoperative care.
1. Introduction
End-stage renal disease (ESRD) patients have a high prevalence of coronary artery disease, and their cardiovascular mortality rates are almost 20 times higher than those in the general population [1]. The high prevalence of coronary artery disease in ESRD patients is significantly influenced by the presence of comorbidities such as hypertension, diabetes mellitus, dyslipidemia, and tobacco use [2]. Additionally, in ESRD patients, several factors including uremia, inflammatory responses, elevated oxidative stress, neurohormonal activation, and endothelial dysfunction can contribute to the progression of coronary artery disease and the formation of atherosclerosis [2]. Although the optimal modality of revascularization therapy remains controversial in ESRD patients, coronary artery bypass graft (CABG) has been recommended as a treatment for significant coronary artery disease in these patients [3]. However, ESRD patients undergoing CABG are at an increased risk of postoperative mortality and complications compared to patients without kidney disease [4]. In a regional prospective cohort study of 15,500 consecutive patients who underwent CABG in northern New England from 1992 to 1997, individuals with end-stage renal disease (ESRD) were found to be 3.1 times more likely to experience post-CABG mortality than those with normal renal function (95% confidence interval 2.1 to 4.7) [4]. Additionally, the study also reported that ESRD patients had a higher risk of postoperative mediastinitis and postoperative stroke. Moreover, CABG causes a significant financial burden on ESRD patients, ranking among the top five non-dialytic medical expenditures [5]. Hence, optimizing perioperative care for ESRD patients undergoing CABG is crucial.
CABG is associated with several major complications, such as acute myocardial infarction, stroke, wound infection, prolonged mechanical ventilation, acute kidney injury, postoperative hemorrhage, and even death [6]. Among these complications, gastrointestinal complications are not commonly observed, but the mortality rate can be as high as 33% [7]. Gastrointestinal complications not only impact the gastrointestinal system but also increase the likelihood of encountering other adverse postoperative outcomes [7]. Patients with gastrointestinal complications have an increased risk of stroke, myocardial infarction, sternal wound infection, the need for reoperation due to bleeding, renal failure, and respiratory failure [7]. The gastrointestinal complications after CABG include abscess, ileus, and gastrointestinal ulcers with bleeding [8]. Of these, severe acute pancreatitis has been reported to develop in approximately one percent of post-CABG patients [9]. The primary risk factors for pancreatic cellular injury are preoperative renal dysfunction, perioperative calcium administration, and postoperative hypotension [9]. However, the diagnosis of acute pancreatitis in post-CABG patients with kidney dysfunction can be a challenge due to the falsely increased serum level of amylase [10].
Here, we present a case of necrotizing pancreatitis complicated with a giant pseudocyst in an ESRD patient after CABG surgery. Only a few cases of giant pancreatic pseudocysts in this population have been documented to date.
2. Case Report
A 45-year-old male had ESRD caused by chronic glomerulonephritis. He had been undergoing 4 h of hemodialysis with a blood flow of 250 mL/min through an arteriovenous fistula three times a week for 11 years. The hemodialysis was conducted using single-use hollow-fiber dialyzers with polysulfone membranes. Three years ago, he received a total parathyroidectomy with auto-transplantation over the left arm due to secondary hyperparathyroidism. The patient took a daily dosage of 6003 mg of calcium acetate in conjunction with meals to manage phosphate levels. He did not have the habits of smoking or alcohol consumption, but did have a history of dyslipidemia and coronary artery disease, which were adequately controlled with medication. However, he had recently visited our emergency department several times because of intermittent chest pain and diaphoresis. He underwent a series of studies, including cardiac catheterization. The coronary angiography revealed triple-vessel disease with left main coronary artery stenosis. Notably, there was a critical stenosis of 82% from the left main branch to the left anterior descending coronary artery ostium. Because of this, he underwent conventional CABG surgery two months ago.
During the CABG surgery, the patient was on cardiopulmonary bypass for a total of 286 min, with aortic clamping for 54 min. Intraoperatively, he received four units of packed red blood cell transfusion. During the operation, a bypass procedure was carried out, involving the left internal thoracic artery connecting to the left anterior descending artery, as well as a free graft using the right internal thoracic artery to the third obtuse marginal artery. After the surgery, the patient received norepinephrine infusion to stabilize blood pressure, with a maximum dosage of 0.11 mcg/kg/min. He was successfully weaned off the inotropic agent within one day of the operation. The postoperative ejection fraction of the left ventricle, measured by echocardiogram, was 58%.
Following his CABG surgery, the patient experienced intermittent epigastric pain but was discharged after recovering from the procedure. The pain was dull and occasionally radiated to his back. It was aggravated by eating and improved during fasting. The pain could be triggered occasionally during dialysis session, particularly when intradialytic hypotension occurred with a higher ultrafiltration rate (14.86 mL/kg/h). In addition to the clinical symptoms, a non-tender bulge had developed in his epigastric area with progressive enlargement over a period of about 2 weeks. He was readmitted due to these symptoms and the enlarged mass lesion. Physical examination disclosed a large and firm mass in the upper abdomen. The mass was round in shape, well-demarcated with the sensation of being fluid-filled. There were no apparent skin discolorations over the affected area. The laboratory data showed increased amylase and lipase (Table 1). A contrast-enhanced abdominal computed tomography (CT) scan revealed a giant multiloculated cystic lesion with a size of 21 × 17 cm in the retroperitoneum surrounding the pancreas and duodenum. This finding was consistent with necrotizing pancreatitis and pseudocyst formation (Figure 1). The CT scan did not reveal any biliary structure abnormality.

Table 1.
Patient’s laboratory examination result.
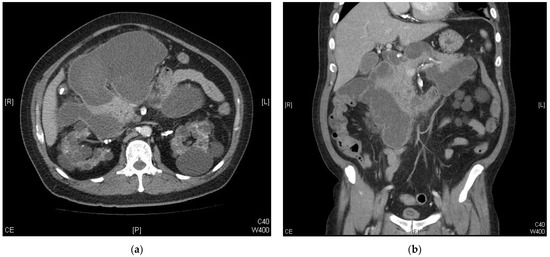
Figure 1.
Contrast-enhanced abdominal computed-tomography images of the patient: (a) the axial plane and (b) the coronal plane.
Following the diagnosis, CT-guided cystic drainage was performed successfully (Figure 2), and the amylase level in the cystic fluid was measured at 10,186 U/L. This elevated amylase level confirms the pancreas as the origin of the pseudocyst. The patient did not experience dyspnea during the following two days, and follow-up laboratory data are summarized in the Table 1. The clinical symptoms were significantly improved after cystic drainage and supportive treatment, and surgical intervention was not required. The patient was discharged from the hospital and has received maintenance hemodialysis in our dialysis units.
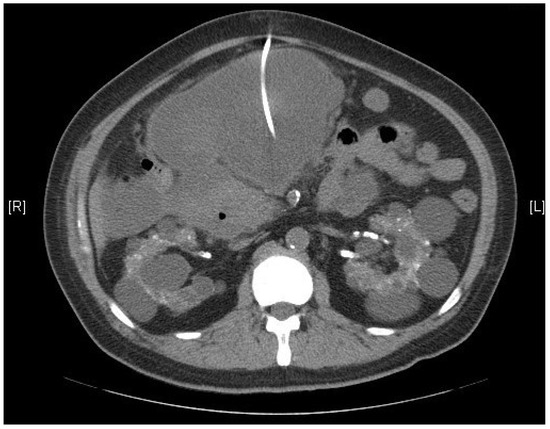
Figure 2.
Computed-tomography-guided drainage for pancreatic pseudocyst.
3. Discussion
There are many potential complications in patients receiving CABG surgery [11]. Among them, gastrointestinal complications are particularly critical due to their high mortality rate [7]. Acute pancreatitis after cardiac surgery has been reported in previous literature (Table 2). However, acute pancreatitis complicated with giant pseudocysts is rarely reported in patients after CABG surgery. In a study of 5621 patients receiving cardiopulmonary bypass (CPB), 25 patients developed acute pancreatitis [12]. Among them, 10 had pancreatic necrosis, resulting in 11 deaths, which equated to a mortality rate of 44% when acute pancreatitis occurred [12]. However, there were no reported cases of pancreatic pseudocyst development, highlighting the rarity of pseudocyst formation in this clinical context. The currently prevailing hypothesis regarding the cause of postoperative acute pancreatitis is related to splanchnic ischemia [9,13]. Low cardiac output in patients with cardiac surgery receiving CPB can lead to pancreatic injury due to splanchnic ischemia [13]. Elevated intracellular and intramitochondrial calcium concentrations are observed during ischemia [13]. Ischemia potentially increases intra-acinar calcium levels, triggering intracellular trypsinogen activation and the recruitment of neutrophils into the pancreatic circulation. These processes may ultimately lead to the development of acute pancreatitis [13]. The duration of CPB was observed to be correlated with pancreatic injury, as patients with pancreatic cellular injury tended to have longer CPB duration compared to those without [9]. Notably, our patient underwent a significantly prolonged CPB time of 286 min with aortic clamping, surpassing the average CPB time (124 ± 44.8 min) documented for individuals with pancreatic injury in the aforementioned study [9]. In addition to CPB-related ischemia, the blood lost during the operation and the occurrence of postoperative shock with the requirement of inotropic agents may further worsen splanchnic ischemia. In the porcine septic shock model, norepinephrine redirects blood flow away from the mesenteric circulation and decreases blood flow in the pancreas [14]. The combination of aforementioned factors may contribute to splanchnic ischemia, leading to the development of necrotizing pancreatitis complicated by the formation of an unusual giant pseudocyst in this patient.

Table 2.
Notable studies reporting acute pancreatitis after cardiac surgery.
In addition to perioperative factors, several other factors may play a role in the development of acute pancreatitis with the formation of a giant pseudocyst (Figure 3). Among these factors, individuals with ESRD have a higher incidence of acute pancreatitis and adverse events after CABG compared to those without ESRD [4,21]. There are several possible mechanisms proposed to explain why ESRD patients are more susceptible to acute pancreatitis. A higher level of uremic toxins is linked to an increased risk of pancreatitis [21]. The precise mechanism of uremia leading to pancreatitis is still under investigation. A study reported that elevated serum levels of cholecystokinin, glucagon, and gastric inhibitory polypeptide in patients with ESRD may lead to the overproduction of pancreatic enzymes, potentially resulting in impaired pancreatic function and pancreatic abnormalities [22]. Intradialytic hypotension and a higher ultrafiltration rate during hemodialysis may further compromise the already vulnerable splanchnic circulation [23]. A previous study of 91 hemodialysis patients demonstrated that decreased hepato-splanchnic circulation saturation was correlated with systemic systolic blood pressure [24]. Interestingly, this reduction in saturation was more pronounced among patients with intradialytic hypotension when compared to those without (−13.8 ± 9.3% vs. 0.4 ± 9.8%, p < 0.001). The decline in hepato-splanchnic circulation saturation was found to be associated with the percentage changes in systolic blood pressure and the ultrafiltration. In addition, there is evidence that ESRD patients undergoing peritoneal dialysis may have a higher risk of developing pancreatitis compared to those undergoing hemodialysis [25]. Peritoneal dialysis patients have a higher occurrence of abdominal anatomical anomalies, and the risk of pancreatic injury is further elevated due to the presence of toxic substances from the peritoneal dialysate, bags, or tubing [25].
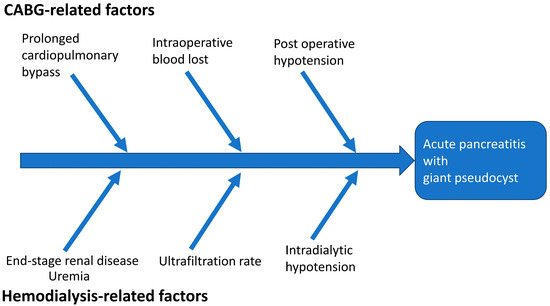
Figure 3.
Proposed contributing factors for acute pancreatitis with pseudocyst formation in our patient. Please note that the proportion of each contribution may not have been equal.
A higher serum calcium level and hyperparathyroidism may predispose patients to the deposition of calcium within the pancreatic duct and trigger the occurrence of pancreatitis [26]. Hypercalcemia can be induced by various factors, including hyperparathyroidism, certain medications, and even intravenous calcium administration, all of which have been documented as potential causes of acute pancreatitis [27]. Although the patient had received a parathyroidectomy previously, the serum levels of calcium were approaching the upper limit of normal range due to the chronic use of a high daily dosage of calcium-based phosphate binders. Secondary hyperparathyroidism in ESRD patients causes unique metabolic abnormalities and pathophysiology distinct from those observed in primary hyperparathyroidism [28]. Despite the high cure rate of acute pancreatitis by parathyroidectomy in patients with primary hyperparathyroidism, there is currently no available literature that examines whether treating secondary hyperparathyroidism can reduce the risk of acute pancreatitis in ESRD patients [29,30]. In exceptional situations like pica, the use of calcium acetate could potentially lead to hypercalcemia and necrotizing pancreatitis [31]. Whether this oral calcium intake can trigger pancreatitis in the absence of hypercalcemia requires further investigation. Further research is needed to clarify the potential association between treated secondary hyperparathyroidism and the occurrence of acute pancreatitis.
Considering the combination of contributing factors, this patient may be in the situation of a “perfect storm” for the development of acute pancreatitis. For this patient, acute pancreatitis following CABG appears to have been influenced by several factors. These include splanchnic ischemia caused by prolonged cardiopulmonary bypass, intraoperative blood loss, postoperative hypotension, a high ultrafiltration rate during dialysis, and episodes of intradialytic hypotension. Additionally, the patient’s medical history of ESRD may have rendered him more susceptible to the development of acute pancreatitis. The occurrence of a giant pancreatic pseudocyst in an ESRD patient after CABG surgery is exceptionally rare and noteworthy. This unique case emphasizes the utmost importance of vigilant monitoring and comprehensive postoperative care for ESRD patients receiving CABG. Prompt identification and management of any unusual complications is crucial to achieving the best outcomes for these patients.
Author Contributions
Conceptualization, C.-W.H. and M.-J.C.; methodology, C.-Y.H.; validation, Y.-J.S., C.-C.H. and W.-H.H.; investigation, C.-H.W.; resources, T.-H.Y.; writing—original draft preparation, M.-J.C. and Y.-J.S.; writing—review and editing, C.-Y.H. and C.-W.H.; visualization, M.-J.C. and Y.-J.S.; supervision, C.-W.H.; project administration, C.-W.H. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of Chang Gung Memorial Hospital (No. 201901044B0) on 01 July 2019.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available on reasonable request from the corresponding author. The data are not publicly available due to patient privacy.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Foley, R.N.; Parfrey, P.S.; Sarnak, M.J. Epidemiology of cardiovascular disease in chronic renal disease. J. Am. Soc. Nephrol. 1998, 9, S16–S23. [Google Scholar] [CrossRef] [PubMed]
- Bhatti, N.K.; Karimi Galougahi, K.; Paz, Y.; Nazif, T.; Moses, J.W.; Leon, M.B.; Stone, G.W.; Kirtane, A.J.; Karmpaliotis, D.; Bokhari, S.; et al. Diagnosis and Management of Cardiovascular Disease in Advanced and End-Stage Renal Disease. J. Am. Heart Assoc. 2016, 5, e003648. [Google Scholar] [CrossRef] [PubMed]
- K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am. J. Kidney Dis. 2005, 45, S1–S153.
- Liu, J.Y.; Birkmeyer, N.J.; Sanders, J.H.; Morton, J.R.; Henriques, H.F.; Lahey, S.J.; Dow, R.W.; Maloney, C.; DiScipio, A.W.; Clough, R.; et al. Risks of morbidity and mortality in dialysis patients undergoing coronary artery bypass surgery. Northern New England Cardiovascular Disease Study Group. Circulation 2000, 102, 2973–2977. [Google Scholar] [CrossRef]
- Lee, C.C.; Hsu, C.C.; Lin, M.H.; Sung, J.M.; Kuo, T.H. Healthcare utilization and expenditure among individuals with end-stage kidney disease in Taiwan. J. Formos. Med. Assoc. 2022, 121 (Suppl. 1), S47–S55. [Google Scholar] [CrossRef]
- Fortescue, E.B.; Kahn, K.; Bates, D.W. Development and validation of a clinical prediction rule for major adverse outcomes in coronary bypass grafting. Am. J. Cardiol. 2001, 88, 1251–1258. [Google Scholar] [CrossRef]
- Filsoufi, F.; Rahmanian, P.B.; Castillo, J.G.; Scurlock, C.; Legnani, P.E.; Adams, D.H. Predictors and outcome of gastrointestinal complications in patients undergoing cardiac surgery. Ann. Surg. 2007, 246, 323–329. [Google Scholar] [CrossRef]
- Rodriguez, F.; Nguyen, T.C.; Galanko, J.A.; Morton, J. Gastrointestinal complications after coronary artery bypass grafting: A national study of morbidity and mortality predictors. J. Am. Coll. Surg. 2007, 205, 741–747. [Google Scholar] [CrossRef]
- Fernández-del Castillo, C.; Harringer, W.; Warshaw, A.L.; Vlahakes, G.J.; Koski, G.; Zaslavsky, A.M.; Rattner, D.W. Risk factors for pancreatic cellular injury after cardiopulmonary bypass. N. Engl. J. Med. 1991, 325, 382–387. [Google Scholar] [CrossRef]
- Paajanen, H.; Nuutinen, P.; Harmoinen, A.; Pöyhönen, M.; Pitkänen, O.; Nordback, I.; Grönroos, J.; Nevalainen, T.J. Hyperamylasemia after cardiopulmonary bypass: Pancreatic cellular injury or impaired renal excretion of amylase? Surgery 1998, 123, 504–510. [Google Scholar] [CrossRef]
- de Waard, D.; Fagan, A.; Minnaar, C.; Horne, D. Management of patients after coronary artery bypass grafting surgery: A guide for primary care practitioners. CMAJ 2021, 193, E689–E694. [Google Scholar] [CrossRef] [PubMed]
- Lefor, A.T.; Vuocolo, P.; Parker, F.B., Jr.; Sillin, L.F. Pancreatic Complications Following Cardiopulmonary Bypass: Factors Influencing Mortality. Arch. Surg. 1992, 127, 1225–1231. [Google Scholar] [CrossRef] [PubMed]
- Nys, M.; Venneman, I.; Deby-Dupont, G.; Preiser, J.-C.; Vanbelle, S.; Albert, A.; Camus, G.; Damas, P.; Larbuisson, R.; Lamy, M. Pancreatic Cellular Injury after Cardiac Surgery with Cardiopulmonary Bypass: Frequency, Time Course and Risk Fa-ctors. Shock 2007, 27, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Krejci, V.; Hiltebrand, L.B.; Sigurdsson, G.H. Effects of epinephrine, norepinephrine, and phenylephrine on microcirculatory blood flow in the gastrointestinal tract in sepsis. Crit. Care Med. 2006, 34, 1456–1463. [Google Scholar] [CrossRef] [PubMed]
- Ohno, T.; Mutsuga, M.; Saito, S.; Tokuda, Y.; Nagai, K.; Umemoto, N.; Abe, T.; Usui, A. Incidence and clinical impact of silent pancreatitis after aortic arch surgery. Asian Cardiovasc. Thorac. Ann. 2023, 31, 303–311. [Google Scholar] [CrossRef]
- Elgharably, H.; Gamaleldin, M.; Ayyat, K.S.; Zaki, A.; Hodges, K.; Kindzelski, B.; Sharma, S.; Hassab, T.; Yongue, C.; Serna, S.; et al. Serious Gastrointestinal Complications After Cardiac Surgery and Associated Mortality. Ann. Thorac. Surg. 2021, 112, 1266–1274. [Google Scholar] [CrossRef]
- Marsoner, K.; Voetsch, A.; Lierzer, C.; Sodeck, G.H.; Fruhwald, S.; Dapunt, O.; Mischinger, H.J.; Kornprat, P. Gastrointestinal complications following on-pump cardiac surgery-A propensity matched analysis. PLoS ONE 2019, 14, e0217874. [Google Scholar] [CrossRef]
- Musleh, G.S.; Patel, N.C.; Grayson, A.D.; Pullan, D.M.; Keenan, D.J.; Fabri, B.M.; Hasan, R. Off-pump coronary artery bypass surgery does not reduce gastrointestinal complications. Eur. J. Cardiothorac. Surg. 2003, 23, 170–174. [Google Scholar] [CrossRef]
- Fitzgerald, T.; Kim, D.; Karakozis, S.; Alam, H.; Provido, H.; Kirkpatrick, J. Visceral ischemia after cardiopulmonary bypass. Am. Surg. 2000, 66, 623–626. [Google Scholar] [CrossRef]
- Yilmaz, A.T.; Arslan, M.; Demirkilç, U.; Ozal, E.; Kuralay, E.; Bingöl, H.; Oz, B.S.; Tatar, H.; Oztürk, O.Y. Gastrointestinal complications after cardiac surgery. Eur. J. Cardiothorac. Surg. 1996, 10, 763–767. [Google Scholar] [CrossRef]
- Hou, S.W.; Lee, Y.K.; Hsu, C.Y.; Lee, C.C.; Su, Y.C. Increased risk of acute pancreatitis in patients with chronic hemodialysis: A 4-year follow-up study. PLoS ONE 2013, 8, e71801. [Google Scholar] [CrossRef][Green Version]
- Sirinek, K.R.; O’Dorisio, T.M.; Gaskill, H.V.; Levine, B.A. Chronic renal failure: Effect of hemodialysis on gastrointestinal hormones. Am. J. Surg. 1984, 148, 732–735. [Google Scholar] [CrossRef]
- Yu, A.W.; Nawab, Z.M.; Barnes, W.E.; Lai, K.N.; Ing, T.S.; Daugirdas, J.T. Splanchnic erythrocyte content decreases during hemodialysis: A new compensatory mechanism for hypovolemia. Kidney Int. 1997, 51, 1986–1990. [Google Scholar] [CrossRef] [PubMed]
- Ookawara, S.; Ito, K.; Ueda, Y.; Minato, S.; Kaneko, S.; Hirata, M.; Kitano, T.; Miyazawa, H.; Hirai, K.; Morishita, Y. Factors affecting intradialytic hepatic oxygenation: Associations between ultrafiltration rate and changes in systemic blood pressure. Int. J. Artif. Organs 2023, 46, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Joglar, F.M.; Saadé, M. Outcome of pancreatitis in CAPD and HD patients. Perit. Dial. Int. 1995, 15, 264–266. [Google Scholar] [CrossRef]
- Sunkara, T.; Caughey, M.E.; Rawla, P.; Yarlagadda, K.S.; Gaduputi, V. Severe Acute Pancreatitis as an Index Clinical Manifestation of Parathyroid Adenoma. Cureus 2018, 10, e2445. [Google Scholar] [CrossRef]
- Frick, T.W. The role of calcium in acute pancreatitis. Surgery 2012, 152, S157–S163. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, J.; Locatelli, F.; Rodriguez, M. Secondary hyperparathyroidism: Pathogenesis, disease progression, and therapeutic options. Clin. J. Am. Soc. Nephrol. 2011, 6, 913–921. [Google Scholar] [CrossRef] [PubMed]
- Misgar, R.A.; Mathew, V.; Pandit, K.; Chowdhury, S. Primary hyperparathyroidism presenting as recurrent acute pancreatitis: A case report and review of literature. Indian J. Endocrinol. Metab. 2011, 15, 54–56. [Google Scholar] [CrossRef]
- Mixter, C.G.; Keynes, W.M.; Cope, O. Further Experience with Pancreatitis as a Diagnostic Clue to Hyperparathyroidism. N. Engl. J. Med. 1962, 266, 265–272. [Google Scholar] [CrossRef]
- Brener, Z.Z.; Bergman, M. Necrotizing pancreatitis due to hypercalcemia in a hemodialysis patient with pica. Clin. Kidney J. 2014, 7, 399–401. [Google Scholar] [CrossRef] [PubMed][Green Version]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).