Fast Track Protocols and Early Rehabilitation after Surgery in Total Hip Arthroplasty: A Narrative Review
Abstract
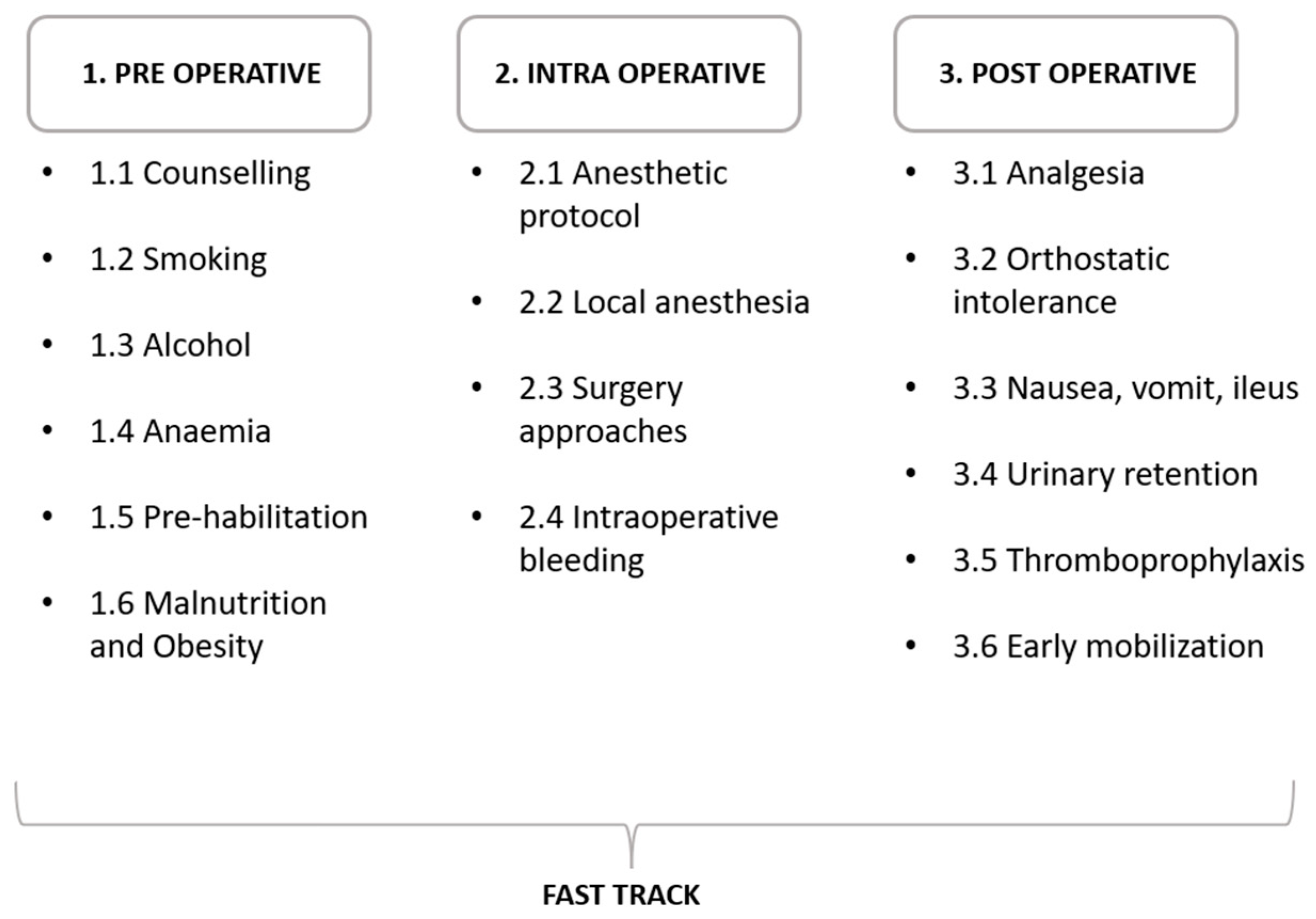
1. Introduction
2. Pre-Operative Optimization
2.1. Counselling
2.2. Smoking
2.3. Alcohol Consumption
2.4. Anemia
2.5. Pre-Habilitation
2.6. Malnutrition/Obesity
3. Peri-Operative Optimization
3.1. Anesthetic Protocol
3.2. Block/Local Anesthesia
3.3. Surgical Approaches
3.4. Intraoperative Bleeding
4. Post-Operative Optimization
4.1. Analgesia
4.2. Orthostatic Intolerance
4.3. Nausea, Vomit, Ileus
4.4. Postoperative Urinary Retention
4.5. Thromboprophylaxis
4.6. Early Mobilization
5. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Kehlet, H. Multimodal approach to control postoperative pathophysiology and rehabilitation. Br. J. Anaesth. 1997, 78, 606–617. [Google Scholar] [CrossRef] [PubMed]
- Bassetty, K.C.; Thomas, D.S.; Sebastian, A.; Thomas, A.; Chandy, R.; Peedicayil, A.; Thomas, V. ERAS: An Audit of Existing Practices. J. Obstet. Gynecol. India 2022, 72, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Nöth, U.; Geiser, T.; Kranich, T.; von Rottkay, E.; Reichert, J.C.; Reyle-Hahn, M.; Rackwitz, L. Fast track strategies in hip arthroplasty. Orthopade 2019, 48, 330–336. [Google Scholar] [CrossRef] [PubMed]
- Hansen, T.B. Fast track in hip arthroplasty. EFORT Open Rev. 2017, 2, 179–188. [Google Scholar] [CrossRef]
- Zhong, M.; Liu, D.; Tang, H.; Zheng, Y.; Bai, Y.; Liang, Q.; Yang, X. Impacts of the perioperative fast track surgery concept on the physical and psychological rehabilitation of total hip arthroplasty: A prospective cohort study of 348 patients. Medicine 2021, 100, e26869. [Google Scholar] [CrossRef]
- Frassanito, L.; Vergari, A.; Nestorini, R.; Cerulli, G.; Placella, G.; Pace, V.; Rossi, M. Enhanced recovery after surgery (ERAS) in hip and knee replacement surgery: Description of a multidisciplinary program to improve management of the patients undergoing major orthopedic surgery. Musculoskelet. Surg. 2020, 104, 87–92. [Google Scholar] [CrossRef]
- Büttner, M.; Mayer, A.M.; Büchler, B.; Betz, U.; Drees, P.; Susanne, S. Economic analyses of fast-track total hip and knee arthroplasty: A systematic review. Eur. J. Orthop. Surg. Traumatol. Orthop. Traumatol. 2020, 30, 67–74. [Google Scholar] [CrossRef]
- Husted, H. Fast-track hip and knee arthroplasty: Clinical and organizational aspects. Acta Orthop. Suppl. 2012, 83, 1–39. [Google Scholar] [CrossRef]
- Husted, H.; Holm, G. Fast track in total hip and knee arthroplasty - experiences from Hvidovre University Hospital, Denmark. Injury 2006, 37 (Suppl. S5), S31–S35. [Google Scholar] [CrossRef]
- Husted, H.; Jensen, C.M.; Solgaard, S.; Kehlet, H. Reduced length of stay following hip and knee arthroplasty in Denmark 2000–2009: From research to implementation. Arch. Orthop. Trauma Surg. 2011, 132, 101–104. [Google Scholar] [CrossRef]
- Winther, S.B.; Foss, O.A.; Wik, T.S.; Davis, S.P.; Engdal, M.; Jessen, V.; Husby, O.S. 1-year follow-up of 920 hip and knee arthroplasty patients after implementing fast-track. Acta Orthop. 2015, 86, 78–85. [Google Scholar] [CrossRef] [PubMed]
- De Ladoucette, A.; Mertl, P.; Henry, M.-P.; Bonin, N.; Tracol, P.; Courtin, C.; Jenny, J.-Y. Fast track protocol for primary total hip arthroplasty in non-trauma cases reduces the length of hospital stay: Prospective French multicenter study. Orthop. Traumatol. Surg. Res. 2020, 106, 1527–1531. [Google Scholar] [CrossRef] [PubMed]
- Pamilo, K.J.; Torkki, P.; Peltola, M.; Pesola, M.; Remes, V.; Paloneva, J. Reduced length of uninterrupted institutional stay after implementing a fast-track protocol for primary total hip replacement. Acta Orthop. 2018, 89, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Glassou, E.N.; Pedersen, A.B.; Hansen, T.B. Risk of re-admission, reoperation, and mortality within 90 days of total hip and knee arthroplasty in fast-track departments in Denmark from 2005 to 2011. Acta Orthop. 2014, 85, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Hartog, Y.M.D.; Mathijssen, N.M.C.; Vehmeijer, S.B.W. Reduced length of hospital stay after the introduction of a rapid recovery protocol for primary THA procedures. Acta Orthop. 2013, 84, 444–447. [Google Scholar] [CrossRef]
- Jorgensen, C.C.; Kjaersgaard-Andersen, P.; Solgaard, S.; Kehlet, H.; Group LFC for FH and KRC. Hip dislocations after 2734 elective unilateral fast-track total hip arthroplasties: Incidence, circumstances and predisposing factors. Arch. Orthop. Trauma Surg. 2014, 134, 1615–1622. [Google Scholar] [CrossRef]
- Gromov, K.; Troelsen, A.; Otte, K.S.; Orsnes, T.; Ladelund, S.; Husted, H. Removal of restrictions following primary THA with posterolateral approach does not increase the risk of early dislocation. Acta Orthop. 2015, 86, 463–468. [Google Scholar] [CrossRef]
- Ammarullah, M.I.; Santoso, G.; Sugiharto, S.; Supriyono, T.; Wibowo, D.B.; Kurdi, O.; Tauviqirrahman, M.; Jamari, J. Minimizing Risk of Failure from Ceramic-on-Ceramic Total Hip Prosthesis by Selecting Ceramic Materials Based on Tresca Stress. Sustainability 2022, 14, 13413. [Google Scholar] [CrossRef]
- Zhu, S.; Qian, W.; Jiang, C.; Ye, C.; Chen, X. Enhanced recovery after surgery for hip and knee arthroplasty: A systematic review and meta-analysis. Postgrad. Med. J. 2017, 93, 736–742. [Google Scholar] [CrossRef]
- Wainwright, T.W.; Memtsoudis, S.G.; Kehlet, H. Fast-track hip and knee arthroplasty...how fast? Br. J. Anaesth. 2021, 126, 348–349. [Google Scholar] [CrossRef]
- Kehlet, H. History and future challenges in fast-track hip and knee arthroplasty. Orthopäde 2020, 49, 290–292. [Google Scholar] [CrossRef] [PubMed]
- Aasvang, E.K.; Luna, I.E.; Kehlet, H. Challenges in postdischarge function and recovery: The case of fast-track hip and knee arthroplasty. Br. J. Anaesth. 2015, 115, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Wainwright, T.W.; Gill, M.; A McDonald, D.; Middleton, R.G.; Reed, M.; Sahota, O.; Yates, P.; Ljungqvist, O. Consensus statement for perioperative care in total hip replacement and total knee replacement surgery: Enhanced Recovery After Surgery (ERAS®) Society recommendations. Acta Orthop. 2020, 91, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Sivaloganathan, S.; Blakeney, W.G.; Vendittoli, P.-A. Modernizing Total Hip Arthroplasty Perioperative Pathways: The Implementation of ERAS-Outpatient Protocol. J. Clin. Med. 2022, 11, 3293. [Google Scholar] [CrossRef] [PubMed]
- Radetzki, F.; Körber, T.; Wohlrab, D.; Delank, K.S. Risk analysis of restrictive factors for fast-track hip arthroplasty - a retrospective study of 1138 patients. Acta Orthop. Belg. 2021, 87, 601–606. [Google Scholar] [CrossRef]
- Petersen, P.B.; Jørgensen, C.C.; Kehlet, H.; Lundbeck Foundation Centre for Fast-Track Hip and Knee Replacement Collaborative Group. Fast-track hip and knee arthroplasty in older adults—a prospective cohort of 1,427 procedures in patients ≥85 years. Age Ageing 2020, 49, 425–431. [Google Scholar] [CrossRef]
- Ayers, D.; Franklin, P.; Ring, D. The role of emotional health in functional outcomes after orthopaedic surgery: Extending the biopsychosocial model to orthopaedics: AOA critical issues. J. Bone Jt. Surg. Am. 2013, 95, e165. [Google Scholar] [CrossRef]
- Sjöling, M.; Nordahl, G.; Olofsson, N.; Asplund, K. The impact of preoperative information on state anxiety, postoperative pain and satisfaction with pain management. Patient. Educ. Couns. 2003, 51, 169–176. [Google Scholar] [CrossRef]
- McDonald, S.; Page, M.; Beringer, K.; Wasiak, J.; Sprowson, A. Preoperative education for hip or knee replacement. Cochrane Database Syst. Rev. 2014, 2014, CD003526. [Google Scholar] [CrossRef]
- Frederiks, S.; Sidani, S.; Shugurensky, D. The effect of anxiety on learning outcomes post-CABG. Can. J. Nurs. Res. 2008, 40, 127–140. [Google Scholar]
- Husted, H.; Solgaard, S.; Hansen, T.B.; Søballe, K.; Kehlet, H. Care principles at four fast-track arthroplasty departments in Denmark. Dan. Med. Bull. 2010, 57, A4166. [Google Scholar] [PubMed]
- Ibrahim, M.S.; A Khan, M.; Nizam, I.; Haddad, F.S. Peri-operative interventions producing better functional outcomes and enhanced recovery following total hip and knee arthroplasty: An evidence-based review. BMC Med. 2013, 11, 37. [Google Scholar] [CrossRef] [PubMed]
- Kehlet, H.; Wilmore, D. Evidence-based surgical care and the evolution of fast-track surgery. Ann. Surg. 2008, 248, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Kehlet, H.; Søballe, K. Fast-track hip and knee replacement—What are the issues? Acta Orthop. 2010, 81, 271–272. [Google Scholar] [CrossRef] [PubMed]
- Lindberg-Larsen, M.; Varnum, C.; Jakobsen, T.; Andersen, M.R.; Sperling, K.; Overgaard, S.; Hansen, T.B.; Jørgensen, C.C.; Kehlet, H.; Gromov, K. Study protocol for discharge on day of surgery after hip and knee arthroplasty from the Center for Fast-track Hip and Knee Replacement. Acta Orthop. 2023, 94, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Husted, H.; Lunn, T.H.; Troelsen, A.; Gaarn-Larsen, L.; Kristensen, B.B.; Kehlet, H. Why still in hospital after fast-track hip and knee arthroplasty? Acta Orthop. 2011, 82, 679–684. [Google Scholar] [CrossRef]
- Singh, J.A. Smoking and Outcomes After Knee and Hip Arthroplasty: A Systematic Review. J. Rheumatol. 2011, 38, 1824–1834. [Google Scholar] [CrossRef]
- Yue, C.; Cui, G.; Ma, M.; Tang, Y.; Li, H.; Liu, Y.; Zhang, X. Associations between smoking and clinical outcomes after total hip and knee arthroplasty: A systematic review and meta-analysis. Front. Surg. 2022, 9, 970537. [Google Scholar] [CrossRef]
- Debbi, E.M.; Rajaee, S.S.; Spitzer, A.I.; Paiement, G.D. Smoking and Total Hip Arthroplasty: Increased Inpatient Complications, Costs, and Length of Stay. J. Arthroplast. 2019, 34, 1736–1739. [Google Scholar] [CrossRef]
- Matharu, G.S.; Mouchti, S.; Twigg, S.; Delmestri, A.; Murray, D.W.; Judge, A.; Pandit, H.G. The effect of smoking on outcomes following primary total hip and knee arthroplasty: A population-based cohort study of 117,024 patients. Acta Orthop. 2019, 90, 559–567. [Google Scholar] [CrossRef]
- Møller, A.M.; Villebro, N.; Pedersen, T.; Tønnesen, H. Effect of preoperative smoking intervention on postoperative complications: A randomised clinical trial. Lancet 2002, 359, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, C.C.; Kehlet, H.; Lundbeck Foundation Centre for Fast-Track Hip and Knee Replacement Collaborative Group. Outcomes in smokers and alcohol users after fast-track hip and knee arthroplasty. Acta Anaesthesiol. Scand. 2013, 57, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, A.M.; Farley, K.X.; Boden, S.H.; Wilson, J.M.; Daly, C.A.; Gottschalk, M.B.; Wagner, E.R. The use of tobacco is a modifiable risk factor for poor outcomes and readmissions after shoulder arthroplasty. Bone Jt. J. 2020, 102-B, 1549–1554. [Google Scholar] [CrossRef] [PubMed]
- Tønnesen, H. The alcohol patient and surgery. Alcohol Alcohol. 1999, 34, 148–152. [Google Scholar] [CrossRef]
- Tønnesen, H.; Kehlet, H. Preoperative alcoholism and postoperative morbidity. Br. J. Surg. 1999, 86, 869–874. [Google Scholar] [CrossRef]
- Nneson, H.T.O.; Petersen, K.; Nielsen, H.; Kehlet, H.; Jgaard, L.H.O.; Stokholm, K.; Knigge, U. Postoperative morbidity among symptom-free alcohol misusers. Lancet 1992, 340, 334–337. [Google Scholar] [CrossRef]
- Horn, A.R.; Diamond, K.B.; Ng, M.K.; Vakharia, R.M.; Mont, M.A.; Erez, O. The Association of Alcohol Use Disorder with Perioperative Complications following Primary Total Hip Arthroplasty. Hip Pelvis 2021, 33, 231–238. [Google Scholar] [CrossRef]
- Lavernia, C.J.; Villa, J.M.; Contreras, J.S. Alcohol Use in Elective Total Hip Arthroplasty: Risk or Benefit? Clin. Orthop. 2013, 471, 504–509. [Google Scholar] [CrossRef]
- Bisbe, E.; Basora, M.; Colomina, M.J.; Spanish Best Practice in Peri-operative Anaemia Optimisation Panel. Peri-operative treatment of anaemia in major orthopaedic surgery: A practical approach from Spain. Blood Transfus Trasfus Sangue 2017, 15, 296–306. [Google Scholar] [CrossRef]
- Trunfio, T.A.; Borrelli, A.; Improta, G. Is It Possible to Predict the Length of Stay of Patients Undergoing Hip-Replacement Surgery? Int. J. Environ. Res. Public Health 2022, 19, 6219. [Google Scholar] [CrossRef]
- Varghese, V.D.; Liu, D.; Ngo, D.; Edwards, S. Efficacy and cost-effectiveness of universal pre-operative iron studies in total hip and knee arthroplasty. J. Orthop. Surg. Res. 2021, 16, 536. [Google Scholar] [CrossRef]
- Munting, K.E.; Klein, A. Optimisation of pre-operative anaemia in patients before elective major surgery - why, who, when and how? Anaesthesia 2019, 74 (Suppl. S1), 49–57. [Google Scholar] [CrossRef] [PubMed]
- Goodnough, L.; Maniatis, A.; Earnshaw, P.; Benoni, G.; Beris, P.; Bisbe, E.; Fergusson, D.; Gombotz, H.; Habler, O.; Monk, T.; et al. Detection, evaluation, and management of preoperative anaemia in the elective orthopaedic surgical patient: NATA guidelines. Br. J. Anaesth. 2011, 106, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Jans, Ø.; Jørgensen, C.; Kehlet, H.; Johansson, P.I.; Lundbeck Foundation Centre for Fast-track Hip and Knee Replacement Collaborative Group. Role of preoperative anemia for risk of transfusion and postoperative morbidity in fast-track hip and knee arthroplasty. Transfusion 2014, 54, 717–726. [Google Scholar] [CrossRef] [PubMed]
- Matassi, F.; Duerinckx, J.; Vandenneucker, H.; Bellemans, J. Range of motion after total knee arthroplasty: The effect of a preoperative home exercise program. Knee Surg. Sport. Traumatol. Arthrosc. 2012, 22, 703–709. [Google Scholar] [CrossRef]
- Mak, J.C.S.; Fransen, M.; Jennings, M.; March, L.; Mittal, R.; Harris, I.A. Evidence-based review for patients undergoing elective hip and knee replacement. ANZ J. Surg. 2014, 84, 17–24. [Google Scholar] [CrossRef]
- Widmer, P.; Oesch, P.; Bachmann, S. Effect of Prehabilitation in Form of Exercise and/or Education in Patients Undergoing Total Hip Arthroplasty on Postoperative Outcomes—A Systematic Review. Medicina 2022, 58, 742. [Google Scholar] [CrossRef]
- Wang, L.; Lee, M.; Zhang, Z.; Moodie, J.; Cheng, D.; Martin, J. Does preoperative rehabilitation for patients planning to undergo joint replacement surgery improve outcomes? A systematic review and meta-analysis of randomised controlled trials. BMJ Open 2016, 6, e009857. [Google Scholar] [CrossRef]
- Thornqvist, C.; Gislason, G.H.; Køber, L.; Jensen, P.F.; Torp-Pedersen, C.; Andersson, C. Body mass index and risk of perioperative cardiovascular adverse events and mortality in 34,744 Danish patients undergoing hip or knee replacement. Acta Orthop. 2014, 85, 456–462. [Google Scholar] [CrossRef]
- Nicholson, J.A.; Dowrick, A.S.; Liew, S.M. Nutritional Status and Short-Term Outcome of Hip Arthroplasty. J. Orthop. Surg. 2012, 20, 331–335. [Google Scholar] [CrossRef]
- Husted, H.; Jørgensen, C.C.; Gromov, K.; Kehlet, H. Lundbeck Foundation Center for Fast-track Hip and Knee Replacement Collaborative Group. Does BMI influence hospital stay and morbidity after fast-track hip and knee arthroplasty? Acta Orthop. 2016, 87, 466–472. [Google Scholar] [CrossRef] [PubMed]
- Haverkamp, D.; Klinkenbijl, M.N.; Somford, M.P.; Albers, G.H.; van der Vis, H.M. Obesity in total hip arthroplasty—Does it really matter? A meta-analysis. Acta Orthop. 2011, 82, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Jameson, S.S.; Mason, J.M.; Baker, P.N.; Elson, D.W.; Deehan, D.J.; Reed, M.R. The impact of body mass index on patient reported outcome measures (PROMs) and complications following primary hip arthroplasty. J. Arthroplast. 2014, 29, 1889–1898. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Wahafu, T.; Cheng, M.; Cheng, T.; Zhang, Y.; Zhang, X. The infl uence of obesity on primary total hip arthroplasty outcomes: A meta-analysis of prospective cohort studies. Orthop. Traumatol. Surg. Res. 2015, 101, 289–296. [Google Scholar] [CrossRef] [PubMed]
- Pu, X.; Sun, J.-M. General anesthesia vs spinal anesthesia for patients undergoing total-hip arthroplasty: A meta-analysis. Medicine 2019, 98, e14925. [Google Scholar] [CrossRef]
- Oseka, L.; Pecka, S. Anesthetic Management in Early Recovery After Surgery Protocols for Total Knee and Total Hip Arthroplasty. AANA J. 2018, 86, 32–39. [Google Scholar]
- Bailey, J.G.; Miller, A.; Richardson, G.; Hogg, T.; Uppal, V. Cost comparison between spinal versus general anesthesia for hip and knee arthroplasty: An incremental cost study. Can. J. Anaesth. 2022, 69, 1349–1359. [Google Scholar] [CrossRef]
- Kelly, M.E.; Turcotte, J.J.; Aja, J.M.; MacDonald, J.H.; King, P.J. General vs Neuraxial Anesthesia in Direct Anterior Approach Total Hip Arthroplasty: Effect on Length of Stay and Early Pain Control. J. Arthroplast. 2021, 36, 1013–1017. [Google Scholar] [CrossRef]
- Indelli, P.F.; A Grant, S.; Nielsen, K.; Vail, T.P. Regional Anesthesia in Hip Surgery. Clin. Orthop. 2005, 441, 250–255. [Google Scholar] [CrossRef]
- Fu, G.; Li, H.; Wang, H.; Zhang, R.; Li, M.; Liao, J.; Ma, Y.; Zheng, Q.; Li, Q. Comparison of Peripheral Nerve Block and Spinal Anesthesia in Terms of Postoperative Mortality and Walking Ability in Elderly Hip Fracture Patients—A Retrospective, Propensity-Score Matched Study. Clin. Interv. Aging 2021, 16, 833–841. [Google Scholar] [CrossRef]
- Memtsoudis, S.G.; Cozowicz, C.; Bekeris, J.; Bekere, D.; Liu, J.; Soffin, E.M.; Mariano, E.R.; Johnson, R.L.; Hargett, M.J.; Lee, B.H.; et al. Anaesthetic care of patients undergoing primary hip and knee arthroplasty: Consensus recommendations from the International Consensus on Anaesthesia-Related Outcomes after Surgery group (ICAROS) based on a systematic review and meta-analysis. Br. J. Anaesth. 2019, 123, 269–287. [Google Scholar] [CrossRef]
- Turcotte, J.J.; Stone, A.H.; Gilmor, R.J.; Formica, J.W.; King, P.J. The Effect of Neuraxial Anesthesia on Postoperative Outcomes in Total Joint Arthroplasty with Rapid Recovery Protocols. J. Arthroplast. 2020, 35, 950–954. [Google Scholar] [CrossRef] [PubMed]
- Paziuk, T.M.; Luzzi, A.J.; Fleischman, A.N.; Goswami, K.; Schwenk, E.S.; Levicoff, E.A.; Parvizi, J. General vs Spinal Anesthesia for Total Joint Arthroplasty: A Single-Institution Observational Review. J. Arthroplast. 2020, 35, 955–959. [Google Scholar] [CrossRef]
- Nishio, S.; Fukunishi, S.; Fukui, T.; Fujihara, Y.; Okahisa, S.; Takeda, Y.; Yoshiya, S. Comparison of continuous femoral nerve block with and without combined sciatic nerve block after total hip arthroplasty: A prospective randomized study. Orthop. Rev. 2017, 9, 7063. [Google Scholar] [CrossRef]
- Jørgensen, C.C.; Petersen, P.B.; Daugberg, L.O.; Jakobsen, T.; Gromov, K.; Varnum, C.; Andersen, M.R.; Palm, H.; Kehlet, H. Peripheral nerve-blocks and associations with length of stay and readmissions in fast-track total hip and knee arthroplasty. Acta Anaesthesiol. Scand. 2022, 67, 169–176. [Google Scholar] [CrossRef]
- Memtsoudis, S.G.; Cozowicz, C.; Bekeris, J.; Bekere, D.; Liu, J.; Soffin, E.M.; Mariano, E.R.; Johnson, R.L.; Go, G.; Hargett, M.J.; et al. Peripheral nerve block anesthesia/analgesia for patients undergoing primary hip and knee arthroplasty: Recommendations from the International Consensus on Anesthesia-Related Outcomes after Surgery (ICAROS) group based on a systematic review and meta-analysis of current literature. Reg. Anesthesia Pain Med. 2021, 46, 971–985. [Google Scholar] [CrossRef]
- Park, H.J.; Park, K.K.; Park, J.Y.; Lee, B.; Choi, Y.S.; Kwon, H.M. Peripheral Nerve Block for Pain Management after Total Hip Arthroplasty: A Retrospective Study with Propensity Score Matching. J. Clin. Med. 2022, 11, 5456. [Google Scholar] [CrossRef]
- Yang, R.; Liu, R.-H.; Xu, J.-N.; Xu, G.-H.; Jin, X.-B.; Xiao, R.; Mei, B. Effects of Different Local Analgesic Techniques on Postoperative Quality of Life and Pain in Patients Undergoing Total Hip Arthroplasty Under General Anesthesia: A Randomized Controlled Trial. J. Pain Res. 2021, 14, 527–536. [Google Scholar] [CrossRef]
- Aliste, J.; Layera, S.; Bravo, D.; Jara, Á.; Muñoz, G.; Barrientos, C.; Wulf, R.; Brañez, J.; Finlayson, R.J.; Tran, D.Q. Randomized comparison between pericapsular nerve group (PENG) block and suprainguinal fascia iliaca block for total hip arthroplasty. Reg. Anesth. Pain Med. 2021, 46, 874–878. [Google Scholar] [CrossRef]
- Pascarella, G.; Costa, F.; Del Buono, R.; Pulitanò, R.; Strumia, A.; Piliego, C.; De Quattro, E.; Cataldo, R.; Agrò, F.E.; Carassiti, M.; et al. Impact of the pericapsular nerve group (PENG) block on postoperative analgesia and functional recovery following total hip arthroplasty: A randomised, observer-masked, controlled trial. Anaesthesia 2021, 76, 1492–1498. [Google Scholar] [CrossRef]
- Larsen, K.; Hansen, T.B.; Søballe, K.; Kehlet, H. Patient-reported outcome after fast-track hip arthroplasty: A prospective cohort study. Heal. Qual. Life Outcomes 2010, 8, 144. [Google Scholar] [CrossRef] [PubMed]
- Bernaus, M.; Novellas, M.; Bartra, A.; Núñez, J.H.; Anglès, F. Local infiltration analgesia does not have benefits in fast-track hip arthroplasty programmes: A double-blind, randomised, placebo-controlled, clinical trial. HIP Int. J. Clin. Exp. Res. Hip. Pathol. Ther. 2022, 32, 711–716. [Google Scholar] [CrossRef]
- Stratos, I.; Heller, K.-D.; Rudert, M. German surgeons’ technical preferences for performing total hip arthroplasties: A survey from the National Endoprosthesis Society. Int. Orthop. 2022, 46, 733–739. [Google Scholar] [CrossRef]
- Eingartner, C. Current trends in total hip arthroplasty. Ortop. Traumatol. Rehabil. 2007, 9, 8–14. [Google Scholar]
- Peters, R.M.; Have, B.L.E.F.T.; Rykov, K.; Van Steenbergen, L.; Putter, H.; Rutgers, M.; Vos, S.; Van Steijnen, B.; Poolman, R.W.; Vehmeijer, S.B.W.; et al. The learning curve of the direct anterior approach is 100 cases: An analysis based on 15,875 total hip arthroplasties in the Dutch Arthroplasty Register. Acta Orthop. 2022, 93, 775–782. [Google Scholar] [CrossRef]
- Godoy-Monzon, D.; Buttaro, M.; Comba, F.; Piccaluga, F.; Cid-Casteulani, A.; Ordas, A. Comparative study of radiological and functional outcomes following a direct anterior approach versus to a posterolateral approach to the hip. Rev. Esp. Cirugia Ortop. Traumatol. Engl. Ed. 2019, 63, 370–375. [Google Scholar] [CrossRef]
- Zhu, L.; Ma, J.; Sang, W.; Lu, H.; Wang, C.; Jiang, Y. Mid-term effectiveness of total hip arthroplasty by direct anterior approach. Chin. J. Reparative Reconstr. Surg. 2017, 31, 1031–1035. [Google Scholar] [CrossRef]
- Barrett, W.P.; Turner, S.E.; Leopold, J.P. Prospective Randomized Study of Direct Anterior vs Postero-Lateral Approach for Total Hip Arthroplasty. J. Arthroplast. 2013, 28, 1634–1638. [Google Scholar] [CrossRef]
- Higgins, B.T.; Barlow, D.R.; Heagerty, N.E.; Lin, T.J. Anterior vs. Posterior Approach for Total Hip Arthroplasty, a Systematic Review and Meta-analysis. J. Arthroplast. 2015, 30, 419–434. [Google Scholar] [CrossRef]
- Krieger, D.M.; Elias, I.; Hartmann, T. ‘Hip-in-a-Day’. Orthopade 2020, 49, 324–333. [Google Scholar] [CrossRef]
- Coenders, M.J.; Mathijssen, N.M.C.; Vehmeijer, S.B.W. Three and a half years’ experience with outpatient total hip arthroplasty. Bone Jt. J. 2020, 102-B, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Soffin, E.M.; Gibbons, M.M.; Ko, C.Y.; Kates, S.L.; Wick, E.C.; Cannesson, M.; Scott, M.J.; Wu, C.L. Evidence Review Conducted for the Agency for Healthcare Research and Quality Safety Program for Improving Surgical Care and Recovery. Obstet. Anesthesia Dig. 2019, 128, 454–465. [Google Scholar] [CrossRef]
- Moskal, J.T.; Capps, S.G. Meta-analysis of Intravenous Tranexamic Acid in Primary Total Hip Arthroplasty. Orthopedics 2016, 39, e883–e892. [Google Scholar] [CrossRef] [PubMed]
- Shichman, I.; Shaked, O.; Ashkenazi, I.; Schwarzkopf, R.; Warschawski, Y.; Snir, N. Tranexamic acid in non-elective primary total hip arthroplasty. Injury 2021, 52, 1544–1548. [Google Scholar] [CrossRef] [PubMed]
- Remérand, F.; Cotten, M.; N’guessan, Y.; Couvret, C.; Rosset, P.; Favard, L.; Laffon, M.; Fusciardi, J. Tranexamic acid decreases risk of haematomas but not pain after hip arthroplasty. Orthop. Traumatol. Surg. Res. 2013, 99, 667–673. [Google Scholar] [CrossRef]
- Debre, J.; Štěpán, Z.; Dupal, J. Tranexamic Acid Reduces the Incidence of Heterotopic Ossifications after Elective Primary Total Hip Arthroplasty. Acta Chir. Orthop. Traumatol. Cech. 2021, 88, 13–17. [Google Scholar]
- El Beheiry, H.; Lubberdink, A.; Clements, N.; Dihllon, K.; Sharma, V. Tranexamic acid administration to older patients undergoing primary total hip arthroplasty conserves hemoglobin and reduces blood loss. Can. J. Surg. J. Can. Chir. 2018, 61, 177–184. [Google Scholar] [CrossRef]
- Changjun, C.; Xin, Z.; Yue, L.; Chengcheng, Z.; Qiuru, W.; Qianhao, L.; Pengde, K. Tranexamic acid attenuates early post-operative systemic inflammatory response and nutritional loss and avoids reduction of fibrinogen in total hip arthroplasty within an enhanced recovery after surgery pathway. Int. Orthop. 2021, 45, 2811–2818. [Google Scholar] [CrossRef]
- Levy, J.H.; Koster, A.; Quinones, Q.J.; Milling, T.J.; Key, N.S. Antifibrinolytic Therapy and Perioperative Considerations. Anesthesiology 2018, 128, 657–670. [Google Scholar] [CrossRef]
- Pitchon, D.N.; Dayan, A.C.; Schwenk, E.S.; Baratta, J.L.; Viscusi, E.R. Updates on Multimodal Analgesia for Orthopedic Surgery. Anesthesiol. Clin. 2018, 36, 361–373. [Google Scholar] [CrossRef]
- Beverly, A.; Kaye, A.D.; Ljungqvist, O.; Urman, R.D. Essential Elements of Multimodal Analgesia in Enhanced Recovery After Surgery (ERAS) Guidelines. Anesthesiol. Clin. 2017, 35, e115–e143. [Google Scholar] [CrossRef] [PubMed]
- Karlsen, A.P.H.; Geisler, A.; Petersen, P.L.; Mathiesen, O.; Dahl, J.B. Postoperative pain treatment after total hip arthroplasty: A systematic review. Pain 2015, 156, 8–30. [Google Scholar] [CrossRef] [PubMed]
- Singla, N.K.; Hale, M.E.; Davis, J.C.; Bekker, A.; Gimbel, J.; Jahr, J.; Royal, M.A.; Ang, R.Y.; Viscusi, E.R. IV Acetaminophen: Efficacy of a single dose for postoperative pain after hip arthroplasty: Subset data analysis of 2 unpublished randomized clinical trials. Am. J. Ther. 2015, 22, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Malviya, A.; Martin, K.; Harper, I.; Muller, S.D.; Emmerson, K.P.; Partington, P.F.; Reed, M.R. Enhanced recovery program for hip and knee replacement reduces death rate. Acta Orthop. 2011, 82, 577–581. [Google Scholar] [CrossRef] [PubMed]
- Hartman, J.; Khanna, V.; Habib, A.; Farrokhyar, F.; Memon, M.; Adili, A. Perioperative systemic glucocorticoids in total hip and knee arthroplasty: A systematic review of outcomes. J. Orthop. 2017, 14, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Kehlet, H.; Lindberg-Larsen, V. High-dose glucocorticoid before hip and knee arthroplasty: To use or not to use—That’s the question. Acta Orthop. 2018, 89, 477–479. [Google Scholar] [CrossRef]
- Broek, I.; Albajar, P. Clinical Guidelines: Diagnostic and Treatment Manual; for Curative Programmes in Hospitals and Dispensaries; Guidance for Prescribing, Edition 2010; Médecins Sans Frontières: Paris, France, 2010. [Google Scholar]
- Anekar, A.A.; Cascella, M. WHO Analgesic Ladder. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. [Google Scholar]
- Jans, Ø.; Bundgaard-Nielsen, M.; Solgaard, S.; Johansson, P.I.; Kehlet, H. Orthostatic intolerance during early mobilization after fast-track hip arthroplasty. Br. J. Anaesth. 2011, 108, 436–443. [Google Scholar] [CrossRef]
- Husted, H.; Otte, K.S.; Kristensen, B.B.; Ørsnes, T.; Kehlet, H. Readmissions after fast-track hip and knee arthroplasty. Arch. Orthop. Trauma Surg. 2010, 130, 1185–1191. [Google Scholar] [CrossRef]
- Smith, E.L.; Shah, A.; Son, S.J.; Niu, R.; Talmo, C.T.; Abdeen, A.; Ali, M.; Pinski, J.; Gordon, M.; Lozano-Calderon, S.; et al. Survivorship of Megaprostheses in Revision Hip and Knee Arthroplasty for Septic and Aseptic Indications: A Retrospective, Multicenter Study with Minimum 2-Year Follow-Up. Arthroplast. Today 2020, 6, 475–479. [Google Scholar] [CrossRef]
- Lindberg-Larsen, V.; Petersen, P.B.; Jans, Ø.; Beck, T.; Kehlet, H. Effect of pre-operative methylprednisolone on orthostatic hypotension during early mobilization after total hip arthroplasty. Acta Anaesthesiol. Scand. 2018, 62, 882–892. [Google Scholar] [CrossRef]
- Jans, Ø.; Mehlsen, J.; Kjærsgaard-Andersen, P.; Husted, H.; Solgaard, S.; Josiassen, J.; Lunn, T.H.; Kehlet, H. Oral Midodrine Hydrochloride for Prevention of Orthostatic Hypotension during Early Mobilization after Hip Arthroplasty. Anesthesiology 2015, 123, 1292–1300. [Google Scholar] [CrossRef] [PubMed]
- Roach, J.A.; George, C.J.; John, L.; Plocki, R. Assessing the Safe Resumption of Antihypertensive Medications Following Total Hip or Knee Arthroplasty Surgery. Orthop. Nurs. 2009, 28, 176–182. [Google Scholar] [CrossRef] [PubMed]
- Kehlet, H. Fast-track surgery—an update on physiological care principles to enhance recovery. Langenbeck Arch. Surg. 2011, 396, 585–590. [Google Scholar] [CrossRef]
- Kehlet, H.; Dahl, J.B. Anaesthesia, surgery, and challenges in postoperative recovery. Lancet 2003, 362, 1921–1928. [Google Scholar] [CrossRef]
- Fiore, J.F.; El-Kefraoui, C.; Chay, M.-A.; Nguyen-Powanda, P.; Do, U.; Olleik, G.; Rajabiyazdi, F.; Kouyoumdjian, A.; Derksen, A.; Landry, T.; et al. Opioid versus opioid-free analgesia after surgical discharge: A systematic review and meta-analysis of randomised trials. Lancet 2022, 399, 2280–2293. [Google Scholar] [CrossRef] [PubMed]
- Bjerregaard, L.S.; Jorgensen, C.C.; Kehlet, H.; Lundbeck Foundation Centre for Fast-Track Hip and Knee Replacement Collaborative Group. Serious renal and urological complications in fast-track primary total hip and knee arthroplasty; a detailed observational cohort study. Minerva Anestesiol. 2016; online ahead of print. [Google Scholar]
- Bjerregaard, L.S.; Bagi, P.; Kehlet, H. Postoperative urinary retention (POUR) in fast-track total hip and knee arthroplasty. Acta Orthop. 2014, 85, 8–10. [Google Scholar] [CrossRef]
- David, T.S.; Vrahas, M.S. Perioperative Lower Urinary Tract Infections and Deep Sepsis in Patients Undergoing Total Joint Arthroplasty. J. Am. Acad. Orthop. Surg. 2000, 8, 66–74. [Google Scholar] [CrossRef]
- Balderi, T.; Mistraletti, G.; D’Angelo, E.; Carli, F. Incidence of postoperative urinary retention (POUR) after joint arthroplasty and management using ultrasound-guided bladder catheterization. Minerva Anestesiol. 2011, 77, 1050–1057. [Google Scholar]
- Berend, K.R.; Lombardi, A.V.; Berend, M.E.; Adams, J.B.; Morris, M.J. The outpatient total hip arthroplasty: A paradigm change. Bone Jt. J. 2018, 100-B, 31–35. [Google Scholar] [CrossRef]
- Lawrie, C.M.; Ong, A.C.; Hernandez, V.H.; Rosas, S.; Post, Z.D.; Orozco, F.R. Incidence and Risk Factors for Postoperative Urinary Retention in Total Hip Arthroplasty Performed Under Spinal Anesthesia. J. Arthroplast. 2017, 32, 3748–3751. [Google Scholar] [CrossRef]
- Halawi, M.J.; Caminiti, N.; Cote, M.P.; Lindsay, A.D.; Williams, V.J. The Most Significant Risk Factors for Urinary Retention in Fast-track Total Joint Arthroplasty are Iatrogenic. J. Arthroplast. 2019, 34, 136–139. [Google Scholar] [CrossRef] [PubMed]
- Bjerregaard, L.S.; Bogø, S.; Raaschou, S.; Troldborg, C.; Hornum, U.; Poulsen, A.M.; Bagi, P.; Kehlet, H. Incidence of and risk factors for postoperative urinary retention in fast-track hip and knee arthroplasty. Acta Orthop. 2014, 86, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Scholten, R.; Kremers, K.; van de Groes, S.A.; Somford, D.M.; Koëter, S. Incidence and Risk Factors of Postoperative Urinary Retention and Bladder Catheterization in Patients Undergoing Fast-Track Total Joint Arthroplasty: A Prospective Observational Study on 371 Patients. J. Arthroplast. 2018, 33, 1546–1551. [Google Scholar] [CrossRef] [PubMed]
- Nyman, M.H.; Gustafsson, M.; Langius-Eklöf, A.; Johansson, J.-E.; Norlin, R.; Hagberg, L. Intermittent versus indwelling urinary catheterisation in hip surgery patients: A randomised controlled trial with cost-effectiveness analysis. Int. J. Nurs. Stud. 2013, 50, 1589–1598. [Google Scholar] [CrossRef]
- Falck-Ytter, Y.; Francis, C.W.; Johanson, N.A.; Curley, C.; Dahl, O.E.; Schulman, S.; Ortel, T.L.; Pauker, S.G.; Colwell, C.W., Jr. Prevention of VTE in Orthopedic Surgery Patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest 2012, 141, e278S–e325S. [Google Scholar] [CrossRef]
- Jameson, S.S.; Bottle, A.; Malviya, A.; Muller, S.D.; Reed, M.R. The impact of national guidelines for the prophylaxis of venous thromboembolism on the complications of arthroplasty of the lower limb. J. Bone Jt. Surg. Br. 2010, 92, 123–129. [Google Scholar] [CrossRef]
- Jørgensen, C.C.; Jacobsen, M.K.; Soeballe, K.; Hansen, T.B.; Husted, H.; Kjærsgaard-Andersen, P.; Hansen, L.T.; Laursen, M.B.; Kehlet, H. Thromboprophylaxis only during hospitalisation in fast-track hip and knee arthroplasty, a prospective cohort study. BMJ Open 2013, 3, e003965. [Google Scholar] [CrossRef]
- Petersen, P.B.; Kehlet, H.; Jørgensen, C.C.; on behalf of the Lundbeck Foundation Centre for Fast-track Hip and Knee Replacement Collaborative Group. Safety of In-Hospital Only Thromboprophylaxis after Fast-Track Total Hip and Knee Arthroplasty: A Prospective Follow-Up Study in 17,582 Procedures. Thromb. Haemost. 2018, 118, 2152–2161. [Google Scholar] [CrossRef]
- Talec, P.; Gaujoux, S.; Samama, C. Early ambulation and prevention of post-operative thrombo-embolic risk. J. Visc. Surg. 2016, 153, S11–S14. [Google Scholar] [CrossRef]
- Oberfeld, J.; von Hertzberg-Boelch, S.P.; Weissenberger, M.; Holzapfel, B.M.; Rudert, M.; Jakuscheit, A. Effect of Mobilization on the Day of Surgery After Total Hip Arthroplasty in Elderly, Obese, and Severely Diseased Patients. J. Arthroplast. 2021, 36, 3686–3691. [Google Scholar] [CrossRef]
- Okamoto, T.; Ridley, R.J.; Edmondston, S.J.; Visser, M.; Headford, J.; Yates, P.J. Day-of-Surgery Mobilization Reduces the Length of Stay After Elective Hip Arthroplasty. J. Arthroplast. 2016, 31, 2227–2230. [Google Scholar] [CrossRef] [PubMed]
- Hardy, A.; Courgeon, M.; Pellei, K.; Desmeules, F.; Loubert, C.; Vendittoli, P.-A. Improved clinical outcomes of outpatient enhanced recovery hip and knee replacements in comparison to standard inpatient procedures: A study of patients who experienced both. Orthop. Traumatol. Surg. Res. 2022, 108, 103236. [Google Scholar] [CrossRef] [PubMed]
- Vendittoli, P.-A.; Pellei, K.; Desmeules, F.; Massé, V.; Loubert, C.; Lavigne, M.; Fafard, J.; Fortier, L.-P. Enhanced recovery short-stay hip and knee joint replacement program improves patients outcomes while reducing hospital costs. Orthop. Traumatol. Surg. Res. 2019, 105, 1237–1243. [Google Scholar] [CrossRef] [PubMed]
- Maempel, J.F.; Clement, N.D.; Ballantyne, J.A.; Dunstan, E. Enhanced recovery programmes after total hip arthroplasty can result in reduced length of hospital stay without compromising functional outcome. Bone Jt. J. 2016, 98-B, 475–482. [Google Scholar] [CrossRef]
- de Carvalho Almeida, R.F.; Serra, H.O.; de Oliveira, L.P. Fast-track versus conventional surgery in relation to time of hospital discharge following total hip arthroplasty: A single-center prospective study. J. Orthop. Surg. 2021, 16, 488. [Google Scholar] [CrossRef] [PubMed]
- Huerfano, E.; Della Valle, A.G.; Shanaghan, K.; Girardi, F.; Memtsoudis, S.; Liu, J. Characterization of Re-admission and Emergency Department Visits Within 90 Days Following Lower-Extremity Arthroplasty. HSS J. Musculoskelet. J. Hosp. Spec. Surg. 2018, 14, 271–281. [Google Scholar] [CrossRef]
- Morrell, A.T.; Layon, D.R.; Scott, M.J.; Kates, S.L.; Golladay, G.J.; Patel, N.K. Enhanced Recovery after Primary Total Hip and Knee Arthroplasty: A Systematic Review. JBJS 2021, 103, 1938–1947. [Google Scholar] [CrossRef]
- Sutton, J.C.; Antoniou, J.; Epure, L.M.; Huk, O.L.; Zukor, D.J.; Bergeron, S.G. Hospital Discharge within 2 Days Following Total Hip or Knee Arthroplasty Does Not Increase Major-Complication and Readmission Rates. J. Bone Jt. Surg. 2016, 98, 1419–1428. [Google Scholar] [CrossRef]
- Hansen, T.B.; Bredtoft, H.K.; Larsen, K. Preoperative physical optimization in fast-track hip and knee arthroplasty. Dan. Med. J. 2012, 59, A4381. [Google Scholar]
- Jørgensen, C.; Kehlet, H.; Lundbeck Foundation Centre for Fast-track Hip and Knee Replacement Collaborative Group. Role of patient characteristics for fast-track hip and knee arthroplasty. Br. J. Anaesth. 2013, 110, 972–980. [Google Scholar] [CrossRef]
- Hansen, J.B.; Sørensen, J.F.L.; Glassou, E.N.; Homilius, M.; Hansen, T.B. Reducing patient–staff contact in fast-track total hip arthroplasty has no effect on patient-reported outcomes, but decreases satisfaction amongst patients with self-perceived complications: Analysis of 211 patients. Acta Orthop. 2022, 93, 264–270. [Google Scholar] [CrossRef] [PubMed]
- LeBrun, D.G.; Malfer, C.; Wilson, M.; Carroll, K.M.; Wang, V.; Mayman, D.J.; Cross, M.B.; Alexiades, M.M.; Jerabek, S.A.; Cushner, F.D.; et al. Telemedicine in an Outpatient Arthroplasty Setting During the COVID-19 Pandemic: Early Lessons from New York City. HSS J. Musculoskelet. J. Hosp. Spec. Surg. 2021, 17, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Nelson, M.; Bourke, M.; Crossley, K.; Russell, T. Telerehabilitation is non-inferior to usual care following total hip replacement—A randomized controlled non-inferiority trial. Physiotherapy 2020, 107, 19–27. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Martino, A.; Brunello, M.; Pederiva, D.; Schilardi, F.; Rossomando, V.; Cataldi, P.; D’Agostino, C.; Genco, R.; Faldini, C. Fast Track Protocols and Early Rehabilitation after Surgery in Total Hip Arthroplasty: A Narrative Review. Clin. Pract. 2023, 13, 569-582. https://doi.org/10.3390/clinpract13030052
Di Martino A, Brunello M, Pederiva D, Schilardi F, Rossomando V, Cataldi P, D’Agostino C, Genco R, Faldini C. Fast Track Protocols and Early Rehabilitation after Surgery in Total Hip Arthroplasty: A Narrative Review. Clinics and Practice. 2023; 13(3):569-582. https://doi.org/10.3390/clinpract13030052
Chicago/Turabian StyleDi Martino, Alberto, Matteo Brunello, Davide Pederiva, Francesco Schilardi, Valentino Rossomando, Piergiorgio Cataldi, Claudio D’Agostino, Rossana Genco, and Cesare Faldini. 2023. "Fast Track Protocols and Early Rehabilitation after Surgery in Total Hip Arthroplasty: A Narrative Review" Clinics and Practice 13, no. 3: 569-582. https://doi.org/10.3390/clinpract13030052
APA StyleDi Martino, A., Brunello, M., Pederiva, D., Schilardi, F., Rossomando, V., Cataldi, P., D’Agostino, C., Genco, R., & Faldini, C. (2023). Fast Track Protocols and Early Rehabilitation after Surgery in Total Hip Arthroplasty: A Narrative Review. Clinics and Practice, 13(3), 569-582. https://doi.org/10.3390/clinpract13030052








