Possible Role of Carbocysteine Syrup in the Deflation of Percutaneous Endoscopic Gastrostomy Balloons
Abstract
1. Introduction
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rahnemai-Azar, A.A.; Rahnemaiazar, A.A.; Naghshizadian, R.; Kurtz, A.; Farkas, D.T. Percutaneous endoscopic gastrostomy: Indications, technique, complications and management. World J. Gastroenterol. 2014, 20, 7739–7751. [Google Scholar] [CrossRef] [PubMed]
- Welbank, T.; Kurien, M. To PEG or not to PEG that is the question. Proc. Nutr. Soc. 2021, 80, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Roche, K.F.; Bower, K.L.; Collier, B.; Neel, D.; Esry, L. When should the appropriateness of PEG be questioned? Curr. Gastroenterol. Rep. 2023, 25, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Du, G.; Liu, F.; Ma, X.; Chen, S.; Dai, M.; Wei, L.; Liu, Z. Comparison between percutaneous endoscopic gastrostomy and nasogastric feeding in 160 patients with swallowing disturbances: A two-year follow-up study. Clin. Interv. Aging 2022, 17, 1803–1810. [Google Scholar] [CrossRef] [PubMed]
- Hucl, T.; Spicak, J. Complications of percutaneous endoscopic gastrostomy. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 769–781. [Google Scholar] [CrossRef] [PubMed]
- Villela, E.L.; Sakai, P.; Almeida, M.R.; Moura, E.G.; Faintuch, J. Endoscopic gastrostomy replacement tubes: Long-term randomized trial with five silicone commercial models. Clin. Nutr. 2014, 33, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.; Shahidullah, A.; Richards, S. Reducing the unintended dislodgement of gastrostomy tubes in a long-term acute care Hospital: A QA/QI pilot study. Gastroenterol. Res. 2018, 11, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Hooper, C.; Calvert, J. The role for S-carboxymethylcysteine (carbocisteine) in the management of chronic obstructive pulmonary disease. Int. J. Chron. Obstruct. Pulmon. Dis. 2008, 3, 659–669. [Google Scholar] [PubMed]
- Kohli, D.R.; Radadiya, D.K.; Patel, H.; Sharma, P.; Desai, M. Comparative outcomes of endoscopic and radiological gastrostomy tube placement: A systematic review and meta-analysis with GRADE analysis. Ann. Gastroenterol. 2022, 35, 592–602. [Google Scholar] [CrossRef] [PubMed]
- Cominardi, A.; Lisotti, A.; Teci, E.; Mangano, G.; Fusaroli, P. Elective home replacement of gastrostomy feeding tubes is safe and cost-effective. Has hospital referral become obsolete? Dig. Liver. Dis. 2021, 53, 620–624. [Google Scholar] [CrossRef] [PubMed]
- Boeykens, K.; Duysburgh, I. Prevention and management of major complications in percutaneous endoscopic gastrostomy. BMJ Open Gastroenterol. 2021, 8, e000628. [Google Scholar] [CrossRef] [PubMed]
- Cutrín-Gómez, E.; Anguiano-Igea, S.; Delgado-Charro, M.B.; Gómez-Amoza, J.L.; Otero-Espinar, F.J. Effect of penetration enhancers on drug nail permeability from cyclodextrin/poloxamer-soluble polypseudorotaxane-based nail lacquers. Pharmaceutics 2018, 10, 273. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, R.; Targhotra, M.; Kumar, B.; Sahoo, P.K.; Chauhan, M.K. Treatment and management strategies of onychomycosis. J. Mycol. Med. 2020, 30, 100949. [Google Scholar] [CrossRef] [PubMed]
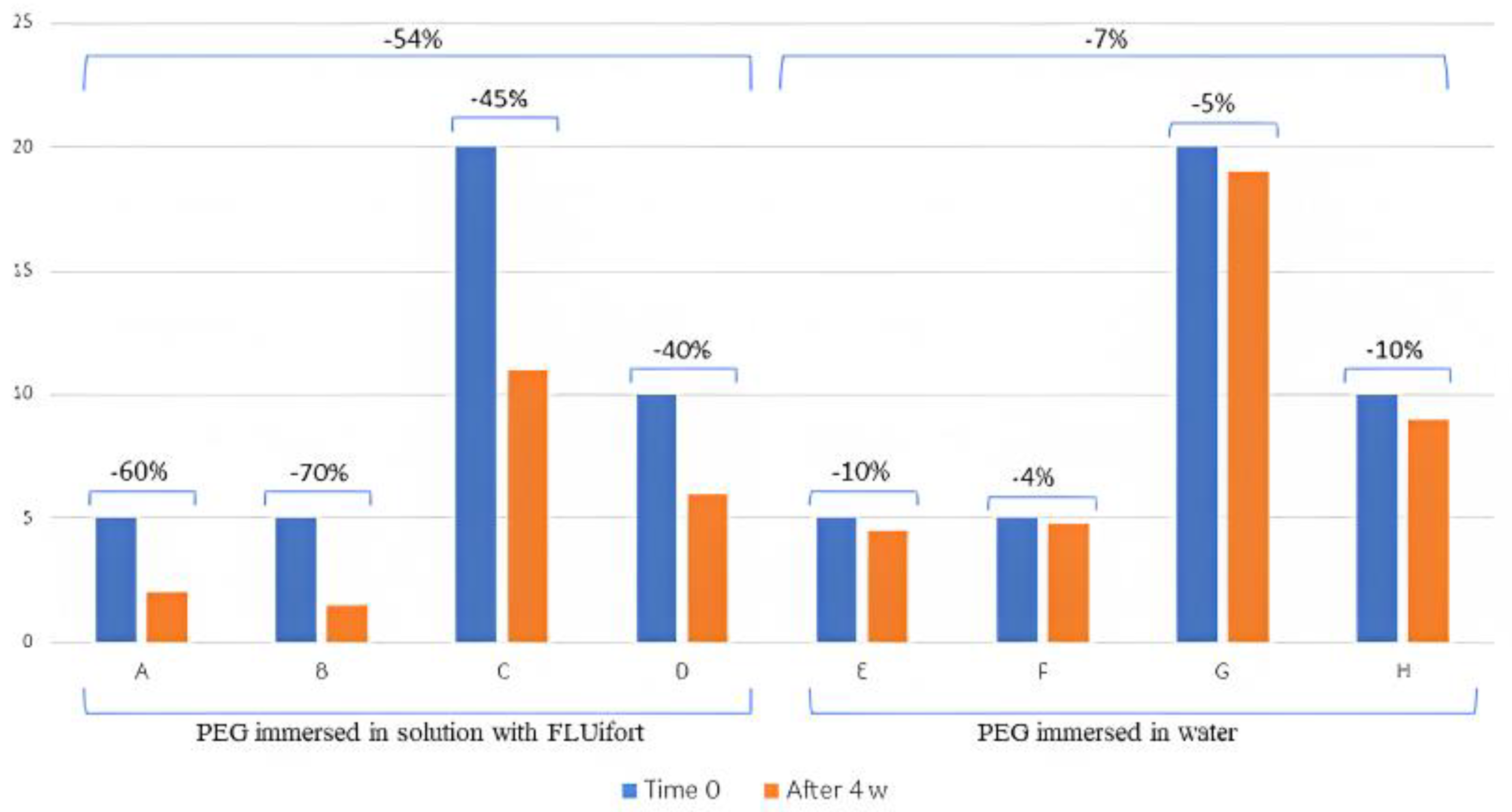
| PEG Immersed in Solution with FLUifort® | PEG Immersed in Water | |||||||
|---|---|---|---|---|---|---|---|---|
| A | B | C | D | E | F | G | H | |
| Time 0 | 5 mL | 5 mL | 20 mL | 10 mL | 5 mL | 5 mL | 20 mL | 10 mL |
| After 4 w | 2 mL | 1.5 mL | 11 mL | 6 mL | 4.5 mL | 4.8 mL | 19 mL | 9 mL |
| Difference | −60% | −70% | −45% | −40% | −10% | −4% | −5% | −10% |
| Average | −54% | −7% | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bassotti, G.; Castellani, D. Possible Role of Carbocysteine Syrup in the Deflation of Percutaneous Endoscopic Gastrostomy Balloons. Clin. Pract. 2023, 13, 483-486. https://doi.org/10.3390/clinpract13020043
Bassotti G, Castellani D. Possible Role of Carbocysteine Syrup in the Deflation of Percutaneous Endoscopic Gastrostomy Balloons. Clinics and Practice. 2023; 13(2):483-486. https://doi.org/10.3390/clinpract13020043
Chicago/Turabian StyleBassotti, Gabrio, and Danilo Castellani. 2023. "Possible Role of Carbocysteine Syrup in the Deflation of Percutaneous Endoscopic Gastrostomy Balloons" Clinics and Practice 13, no. 2: 483-486. https://doi.org/10.3390/clinpract13020043
APA StyleBassotti, G., & Castellani, D. (2023). Possible Role of Carbocysteine Syrup in the Deflation of Percutaneous Endoscopic Gastrostomy Balloons. Clinics and Practice, 13(2), 483-486. https://doi.org/10.3390/clinpract13020043







