The Role of Serial Liquid Biopsy in the Management of Metastatic Non-Small Cell Lung Cancer (NSCLC)
Abstract
:1. Introduction
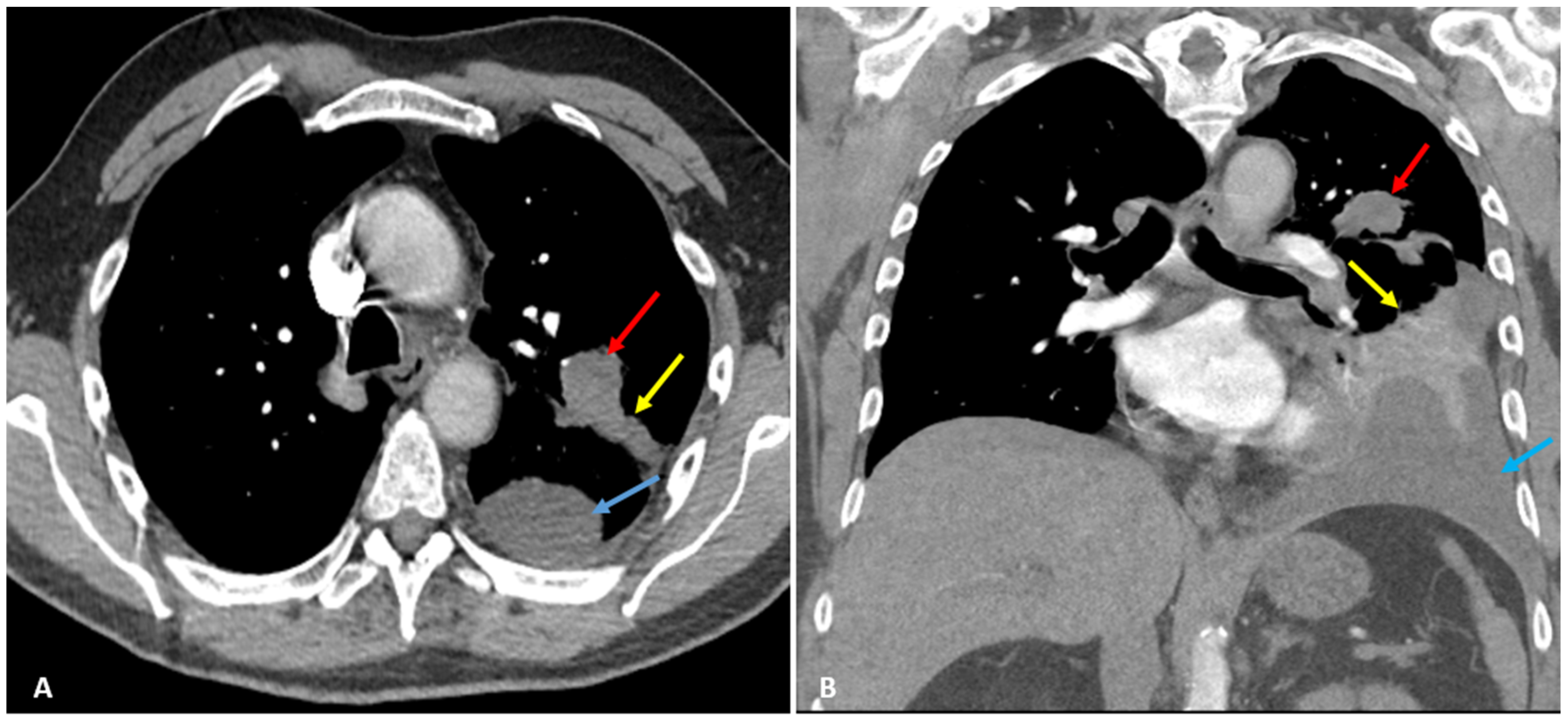
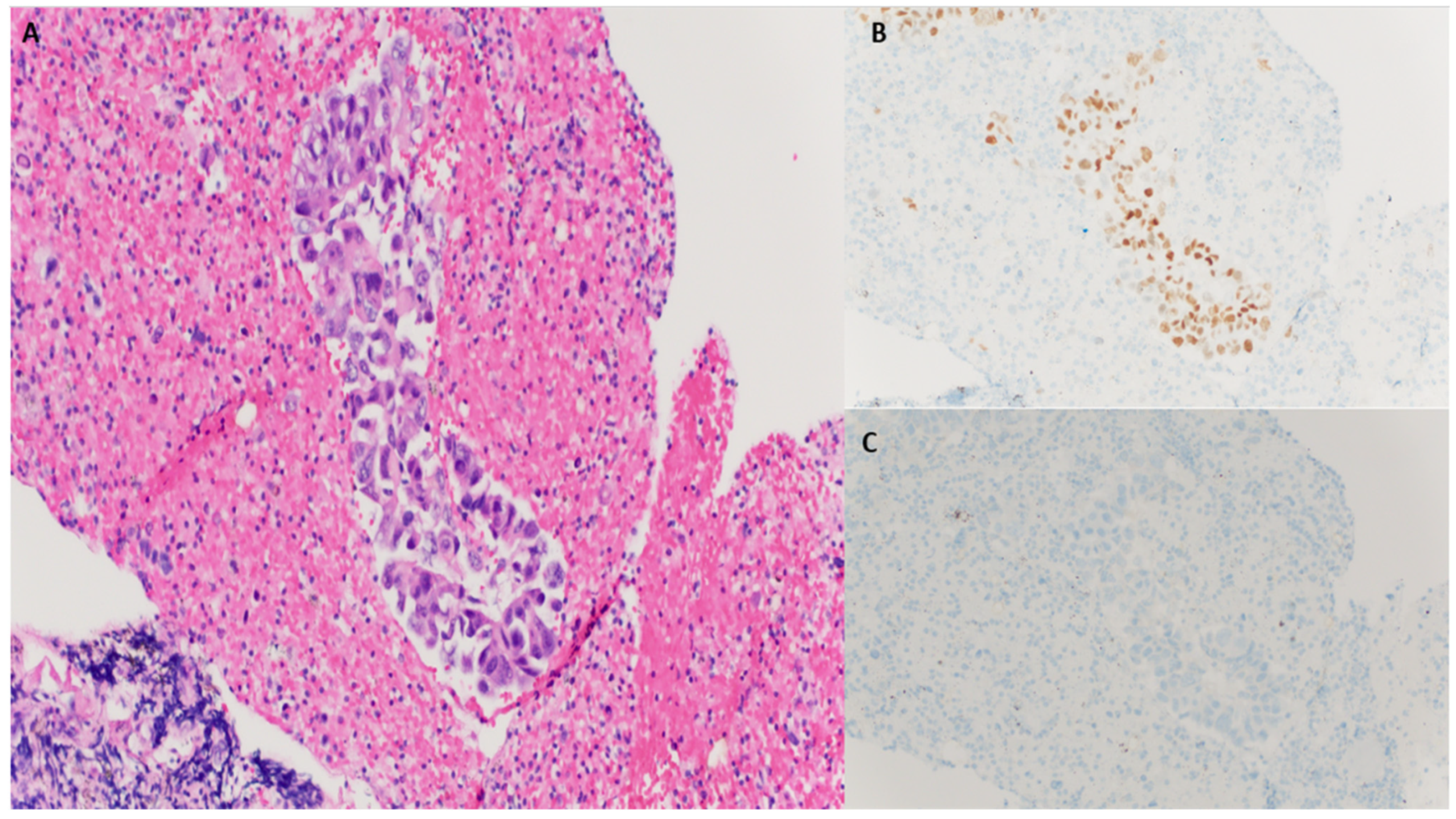
2. Case Presentation
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Navada, S.; Lai, P.; Schwartz, A.G.; Kalemkerian, G.P. Temporal trends in small cell lung cancer: Analysis of the national Surveillance, Epidemiology, and End-Results (SEER) database. J. Clin. Oncol. 2006, 24 (Suppl. S18), 7082. [Google Scholar] [CrossRef]
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Murray, T.; Thun, M.J. Cancer Statistics, 2008. CA A Cancer J. Clin. 2008, 58, 71–96. [Google Scholar] [CrossRef]
- Hackshaw, A.K.; Law, M.R.; Wald, N.J. The accumulated evidence on lung cancer and environmental tobacco smoke. BMJ 1997, 315, 980–988. [Google Scholar] [CrossRef] [Green Version]
- Ruiz-Cordero, R.; Devine, W.P. Targeted Therapy and Checkpoint Immunotherapy in Lung Cancer. Surg. Pathol. Clin. 2020, 13, 17–33. [Google Scholar] [CrossRef]
- Guardant360—Clinical Test—NIH Genetic Testing Registry (GTR)—NCBI. Available online: https://www.ncbi.nlm.nih.gov/gtr/tests/527948/methodology/ (accessed on 6 June 2022).
- Alix-Panabières, C.; Pantel, K. Circulating tumor cells: Liquid biopsy of cancer. Clin. Chem. 2013, 59, 110–118. [Google Scholar] [CrossRef]
- Russo, A.; Perez, D.D.M.; Gunasekaran, M.; Scilla, K.; Lapidus, R.; Cooper, B.; Mehra, R.; Adamo, V.; Malapelle, U.; Rolfo, C. Liquid biopsy tracking of lung tumor evolutions over time. Expert Rev. Mol. Diagn. 2019, 19, 1099–1108. [Google Scholar] [CrossRef]
- Hochmair, M.J.; Buder, A.; Schwab, S.; Burghuber, O.C.; Prosch, H.; Hilbe, W.; Cseh, A.; Fritz, R.; Filipits, M. Liquid-Biopsy-Based Identification of EGFR T790M Mutation-Mediated Resistance to Afatinib Treatment in Patients with Advanced EGFR Mutation-Positive NSCLC, and Subsequent Response to Osimertinib. Target. Oncol. 2019, 14, 75–83. [Google Scholar] [CrossRef] [Green Version]
- Ito, K.; Suzuki, Y.; Saiki, H.; Sakaguchi, T.; Hayashi, K.; Nishii, Y.; Watanabe, F.; Hataji, O. Utility of Liquid Biopsy by Improved PNA-LNA PCR Clamp Method for Detecting EGFR Mutation at Initial Diagnosis of Non-Small-Cell Lung Cancer: Observational Study of 190 Consecutive Cases in Clinical Practice. Clin. Lung Cancer 2018, 19, 181–190. [Google Scholar] [CrossRef]
- Passiglia, F.; Rizzo, S.; Di Maio, M.; Galvano, A.; Badalamenti, G.; Listì, A.; Gulotta, L.; Castiglia, M.; Fulfaro, F.; Bazan, V.; et al. Publisher Correction: The diagnostic accuracy of circulating tumor DNA for the detection of EGFR-T790M mutation in NSCLC: A systematic review and meta-analysis. Sci. Rep. 2018, 8, 17270. [Google Scholar] [CrossRef]
- Drilon, A.; Rekhtman, N.; Arcila, M.; Wang, L.; Ni, A.; Albano, M.; Van Voorthuysen, M.; Somwar, R.; Smith, R.S.; Montecalvo, J.; et al. Cabozantinib in patients with advanced RET-rearranged non-small-cell lung cancer: An open-label, single-centre, phase 2, single-arm trial. Lancet Oncol. 2016, 17, 1653–1660. [Google Scholar] [CrossRef] [Green Version]
- Suppiah, R.; Gershenhorn, B.; Markman, M. A Case Report Demonstrating the Potential Clinical Relevance of Liquid Tumor Biopsies in Lung Cancer. Case Rep. Oncol. 2016, 9, 714–717. [Google Scholar] [CrossRef]
- Dietz, S.; Christopoulos, P.; Gu, L.; Volckmar, A.L.; Endris, V.; Yuan, Z.; Ogrodnik, S.J.; Zemojtel, T.; Heussel, C.P.; Schneider, M.A.; et al. Serial liquid biopsies for detection of treatment failure and profiling of resistance mechanisms in KLC1-ALK-rearranged lung cancer. Cold Spring Harb. Mol. Case Stud. 2019, 5, a004630. [Google Scholar] [CrossRef] [Green Version]
- Bordi, P.; Tiseo, M.; Rofi, E.; Petrini, I.; Restante, G.; Danesi, R.; Del Re, M. Detection of ALK and KRAS Mutations in Circulating Tumor DNA of Patients With Advanced ALK-Positive NSCLC With Disease Progression During Crizotinib Treatment. Clin Lung Cancer 2017, 18, 692–697. [Google Scholar] [CrossRef]
- Shepherd, F.A.; Dancey, J.; Ramlau, R.; Mattson, K.; Gralla, R.; O’Rourke, M.; Levitan, N.; Gressot, L.; Vincent, M.; Burkes, R.; et al. Prospective Randomized Trial of Docetaxel Versus Best Supportive Care in Patients With Non–Small-Cell Lung Cancer Previously Treated With Platinum-Based Chemotherapy. J. Clin. Oncol. 2016, 18, 2095–2103. [Google Scholar] [CrossRef]
- Simanshu, D.K.; Nissley, D.V.; McCormick, F. RAS Proteins and Their Regulators in Human Disease. Cell 2017, 170, 17–33. [Google Scholar] [CrossRef] [Green Version]
- Cox, A.D.; Fesik, S.W.; Kimmelman, A.C.; Luo, J.; Der, C.J. Drugging the undruggable RAS: Mission possible? Nat. Rev. Drug Discov. 2014, 13, 828–851. [Google Scholar] [CrossRef] [Green Version]
- Ostrem, J.M.; Peters, U.; Sos, M.L.; Wells, J.A.; Shokat, K.M. K-Ras(G12C) inhibitors allosterically control GTP affinity and effector interactions. Nature 2013, 503, 548–551. [Google Scholar] [CrossRef] [Green Version]
- Skoulidis, F.; Li, B.T.; Dy, G.K.; Price, T.J.; Falchook, G.S.; Wolf, J.; Italiano, A.; Schuler, M.; Borghaei, H.; Barlesi, F.; et al. Sotorasib for Lung Cancers with KRAS p.G12C Mutation. N. Engl. J. Med. 2021, 384, 2371–2381. [Google Scholar] [CrossRef]
- Hong, D.S.; Fakih, M.G.; Strickler, J.H.; Desai, J.; Durm, G.A.; Shapiro, G.I.; Falchook, G.S.; Price, T.J.; Sacher, A.; Denlinger, C.S.; et al. KRAS G12C Inhibition with Sotorasib in Advanced Solid Tumors. N. Engl. J. Med. 2020, 383, 1207–1217. [Google Scholar] [CrossRef]
- Zulato, E.; Attili, I.; Pavan, A.; Nardo, G.; Del Bianco, P.; Boscolo Bragadin, A.; Verza, M.; Pasqualini, L.; Pasello, G.; Fassan, M.; et al. Early assessment of KRAS mutation in cfDNA correlates with risk of progression and death in advanced non-small-cell lung cancer. Br. J. Cancer 2020, 123, 81–91. [Google Scholar] [CrossRef]
- Heitzer, E.; Van Den Broek, D.; Denis, M.G.; Hofman, P.; Hubank, M.; Mouliere, F.; Paz-Ares, L.; Schuuring, E.; Sültmann, H.; Vainer, G.; et al. Recommendations for a practical implementation of circulating tumor DNA mutation testing in metastatic non-small-cell lung cancer. ESMO Open 2022, 7, 100399. [Google Scholar] [CrossRef]
- Wang, S.; Luo, R.; Shi, Y.; Han, X. The impact of the ALK fusion variant on clinical outcomes in EML4-ALK patients with NSCLC: A systematic review and meta-analysis. Future Oncol. 2021, 18, 385–402. [Google Scholar] [CrossRef]
- Schmid, S.; Gautschi, O.; Rothschild, S.; Mark, M.; Froesch, P.; Klingbiel, D.; Reichegger, H.; Jochum, W.; Diebold, J.; Früh, M. Clinical outcome of ALK-positive non–small cell lung cancer (NSCLC) patients with De Novo EGFR or KRAS co-mutations receiving tyrosine kinase inhibitors (TKIs). J. Thorac. Oncol. 2017, 12, 681–688. [Google Scholar] [CrossRef] [Green Version]
- Noordhof, A.L.; Damhuis, R.A.M.; Hendriks, L.E.L.; de Langen, A.J.; Timens, W.; Venmans, B.J.W.; van Geffen, W.H. Prognostic impact of KRAS mutation status for patients with stage IV adenocarcinoma of the lung treated with first-line pembrolizumab monotherapy. Lung Cancer 2021, 155, 163–169. [Google Scholar] [CrossRef]
| Pathologic Diagnosis and Molecular Profiling | Oncology Treatment | Treatment Duration |
|---|---|---|
| Tissue biopsy in August 2020 with ALK rearrangement | Alectinib | September 2020–November 2020 |
| Liquid biopsy in November 2020 with KRAS p.G12C mutation | Bevacizumab/atezolizumab/carboplatin/paclitaxel | November 2020–January 2021 |
| Bosutinib/pemetrexed via Phase 1 clinical trial | January 2021–July 2021 | |
| Sotorasib after FDA approval | July 2021–November 2021 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sama, S.; Le, T.; Ullah, A.; Elhelf, I.A.; Kavuri, S.K.; Karim, N.A. The Role of Serial Liquid Biopsy in the Management of Metastatic Non-Small Cell Lung Cancer (NSCLC). Clin. Pract. 2022, 12, 419-424. https://doi.org/10.3390/clinpract12030046
Sama S, Le T, Ullah A, Elhelf IA, Kavuri SK, Karim NA. The Role of Serial Liquid Biopsy in the Management of Metastatic Non-Small Cell Lung Cancer (NSCLC). Clinics and Practice. 2022; 12(3):419-424. https://doi.org/10.3390/clinpract12030046
Chicago/Turabian StyleSama, Srikar, Thuy Le, Asad Ullah, Islam A. Elhelf, Sravan K. Kavuri, and Nagla Abdel Karim. 2022. "The Role of Serial Liquid Biopsy in the Management of Metastatic Non-Small Cell Lung Cancer (NSCLC)" Clinics and Practice 12, no. 3: 419-424. https://doi.org/10.3390/clinpract12030046
APA StyleSama, S., Le, T., Ullah, A., Elhelf, I. A., Kavuri, S. K., & Karim, N. A. (2022). The Role of Serial Liquid Biopsy in the Management of Metastatic Non-Small Cell Lung Cancer (NSCLC). Clinics and Practice, 12(3), 419-424. https://doi.org/10.3390/clinpract12030046







