Effects of Cognitive Sensory Motor Training on Lower Extremity Muscle Strength and Balance in Post Stroke Patients: A Randomized Controlled Study
Abstract
:1. Introduction
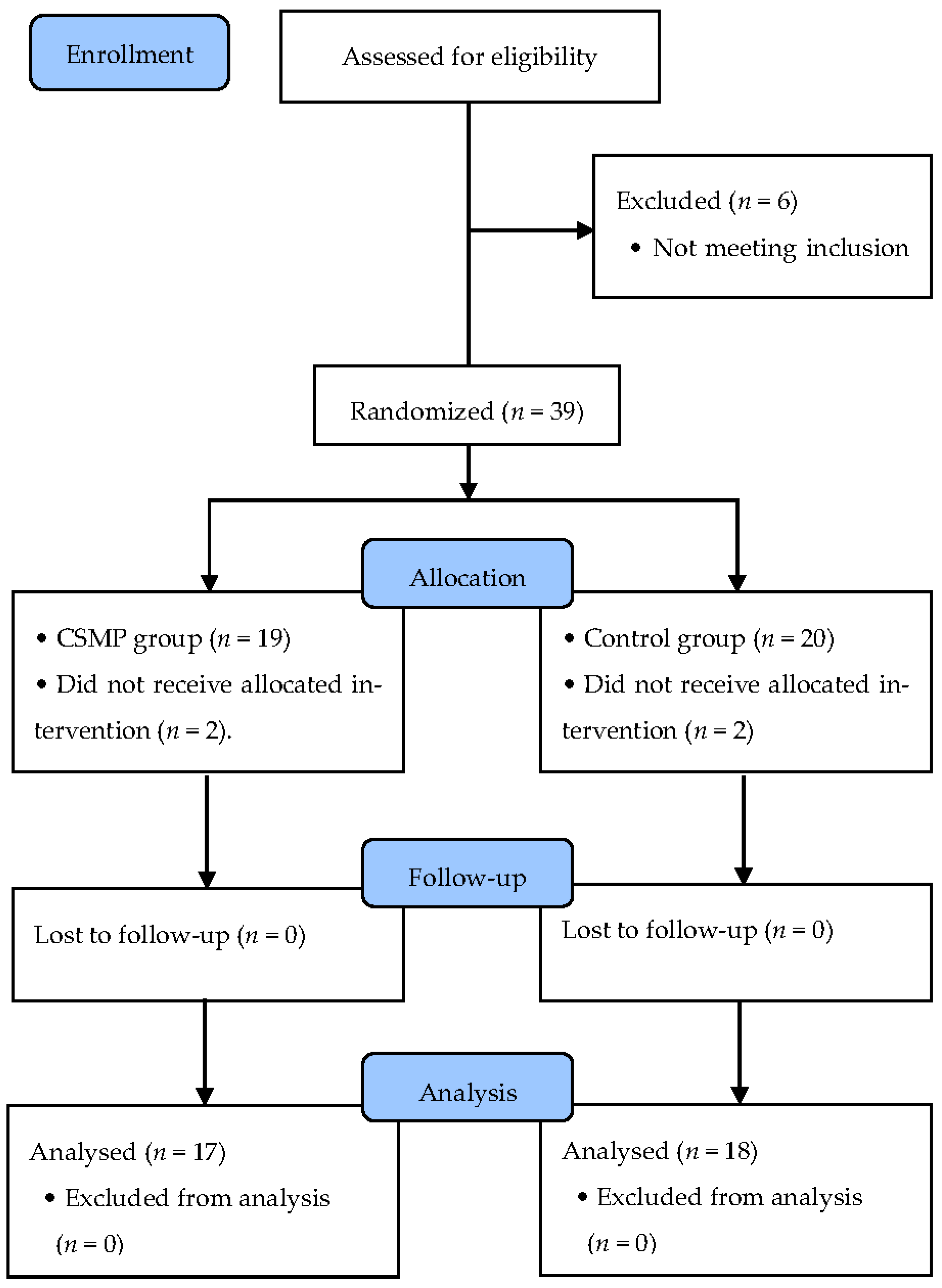
2. Materials and Methods
2.1. Participants
2.2. Procedure
2.3. Sample Size Calculation
2.4. Intervention
2.5. Measurement
2.5.1. Muscle Strength
2.5.2. Balance Test
2.5.3. Statistical Methods
3. Results
3.1. General Characteristics of Subjects
3.1.1. Comparison of LEMTA and MRC
3.1.2. Comparison of REOSA and REOAS
3.1.3. Comparison of RECSA and RECAS
3.1.4. Comparison of LOS
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rafsten, L.; Meirelles, C.; Danielsson, A.; Stibrant Sunnerhagen, K. Impaired motor function in the affected arm predicts impaired postural balance after stroke: A cross sectional study. Front. Neurol. 2019, 10, 912. [Google Scholar] [CrossRef] [Green Version]
- Mentiplay, B.F.; Clark, R.A.; Bower, K.J.; Williams, G.; Pua, Y.H. Five times sit-to-stand following stroke: Relationship with strength and balance. Gait. Posture 2020, 78, 35–39. [Google Scholar] [CrossRef] [PubMed]
- Perfetti, C. Der Hemiplegische Patient Cognitiv-Therapeutische Uegungen, 1st ed.; Pflaum: Munich, Germany, 1997; pp. 71–106. [Google Scholar]
- Birbamer, G.; Beise, U. Das Gehirn denkt nicht in musklekontraktionen. Ars Medici 2001, 6, 282–285. [Google Scholar]
- Chanubol, R.; Wongphaet, P.; Chavanich, N.; Werner, C.; Hesse, S.; Bardeleben, A.; Merholz, J. A randomized controlled trial of Cognitive Sensory Motor Training Therapy on the recovery of arm function in acute stroke patients. Clin. Rehabil. 2012, 26, 1096–1104. [Google Scholar] [CrossRef] [PubMed]
- Perfetti, C. Condotte Terapeutiche per Ia Reeducazione Motoria Dell’ Emiplegico; Ghedini Editore: Milano, Italy, 1986. [Google Scholar]
- Kurz, A.; Pohl, C.; Ramsenthaler, M.; Sorg, C. Cognitive rehabilitation in patients with mild cognitive impairment. Int. J. Geriat. Psy. 2009, 24, 163–168. [Google Scholar] [CrossRef]
- Patel, N.; Jankovic, J.; Hallett, M. Sensory aspects of movement disorders. Lancet. Neurol. 2014, 13, 100–112. [Google Scholar] [CrossRef] [Green Version]
- Anaya, M.A.; Branscheidt, M. Neurorehabilitation After Stroke. Stroke 2019, 50, e180–e182. [Google Scholar] [CrossRef] [Green Version]
- Wilson, R.D.; Bryden, A.M.; Kilgore, K.L.; Makowski, N.; Bourbeau, D.; Kowalski, K.E.; DiMarco, A.F.; Knutson, J.S. Neuromodulation for Functional Electrical Stimulation. Phys. Med. Rehabil. Clin. N. Am. 2019, 30, 301–318. [Google Scholar] [CrossRef]
- Kim, K.H.; Lee, K.B.; Bae, Y.H.; Fong, S.S.M.; Lee, S.M. Effects of progressive backward body weight supported treadmill training on gait ability in chronic stroke patients: A randomized controlled trial. Technol. Health. Care. 2017, 25, 867–876. [Google Scholar] [CrossRef] [Green Version]
- Derakhshanfar, M.; Raji, P.; Bagheri, H.; Jalili, M.; Tarhsaz, H. Sensory interventions on motor function, activities of daily living, and spasticity of the upper limb in people with stroke: A randomized clinical trial. J. Hand. Ther. 2020, 18. [Google Scholar] [CrossRef]
- Decety, J. Do imagined and executed actions share the same neural substrate? Brain. Res. Cogn. Brain. Res. 1996, 3, 87–93. [Google Scholar] [CrossRef]
- Thieme, H.; Bayn, M.; Wurg, M.; Zange, C.; Pohl, M.; Behrens, J. Mirror therapy for patients with severe arm paresis after stroke–A randomized controlled trial. Clin. Rehabil. 2013, 27, 314–324. [Google Scholar] [CrossRef]
- Jackson, P.L.; Doyon, J.; Richards, C.L.; Malouin, F. The efficacy of combined physical and mental practice in the learning of a foot-sequence task after stroke: A case report. Neurorehabil. Neural. Repair. 2004, 18, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Buccino, G.; Solodkin, A.; Small, S.L. Functions of the mirror neuron system: Implications for neurorehabilitation. Cogn. Behav. Neurol. 2006, 19, 55–63. [Google Scholar] [CrossRef]
- Lynch, E.A.; Hillier, S.L.; Stiller, K.; Campanella, R.R.; Fisher, P.H. Sensory retraining of the lower limb after acute stroke: A randomized controlled pilot trial. Arch. Phys. Med. Rehabil. 2007, 88, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Geiger, R.A.; Allen, J.B.; O’Keefe, J.; Hicks, R.R. Balance and mobility following stroke: Effects of physical therapy interventions with and without biofeedback/forceplate training. Phys. Ther. 2001, 81, 995–1005. [Google Scholar] [CrossRef]
- Haywood, K.; Getchell, N. Life Span Motor Development; Human Kinetics: Champaign, IL, USA, 2005. [Google Scholar]
- Schoene, D.; Lord, S.R.; Delbaere, K.; Severino, C.; Davies, T.A.; Smith, S.T. A randomized controlled pilot study of home based step training in older people using videogame technology. PLoS ONE 2013, 8, e57734. [Google Scholar]
- Wongphaet, P.; Sinlapawonsa, T.; Butrach, W.; Chira-Adisai, W.; Laothamatas, J.; Jitpraphai, C. Functional magnetic resonance imaging evidence of brain plasticity after cognitive sensory motor training in a stroke patient. Neurol. J. Thail. 2005, 5, 123–126. [Google Scholar]
- Kesar, T.M.; Reisman, D.S.; Perumal, R.; Jancosko, A.M.; Higginson, J.S.; Rudolph, K.S.; Binder-Macleod, S.A. Combination effects of fast treadmill walking and functional electrical stimulation on post-stroke gait. Gait. Posture 2011, 33, 309–313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cappellino, F.; Paolucci, T.; Zangrando, F.; Iosa, M.; Adriani, E.; Mancini, P.; Bellelli, A.; Saraceni, V.M. Neurocognitive rehabilitative approach effectiveness after anterior cruciate ligament reconstruction with patellar tendon. A randomized controlled trial. Eur. J. Phys. Rehabil. Med. 2012, 48, 17–30. [Google Scholar]
- Pohl, M.; Mehrholz, J.; Ritschel, C.; Rückriem, S. Speed-dependent treadmill training in ambulatory hemiparetic stroke patients A randomized controlled trial. Stroke 2002, 33, 553–558. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Y.R.; Chen, I.H.; Liao, K.K.; Huang, C.C.; Wang, R.Y. Cortical reorganization induced by body weight-supported treadmill training in patients with hemiparesis of different stroke durations. Arch. Phys. Med. Rehabil. 2010, 91, 513–518. [Google Scholar] [CrossRef] [PubMed]
- Koblbauer, I.F.; Lambrecht, Y.; van der Hulst, M.L.; Neeter, C.; Engelbert, R.H.; Poolman, R.W.; Scholtes, V.S. Reliability of maximal isometric knee strength testing with modified hand-held dynamometry in patients awaiting total knee arthroplasty: Useful in research and individual patient settings? A reliability study. BMC Musculoskelet. Disord. 2011, 12, 249. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carpenter, M.R.; Carpenter, R.L.; Peel, J.; Zukley, L.M. The reliability of isokinetic and isometric leg strength measures among individuals with symptoms of mild osteoarthritis. J. Sports Med. Phys. Fitness 2006, 46, 585–589. [Google Scholar] [CrossRef]
- Pradon, D.; Roche, N.; Enette, L.; Zory, R. Relationship between lower limb muscle strength and 6-minute walk test performance in stroke patients. J. Rehabil. Med. 2013, 45, 105–108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gregson, J.M.; Leathley, M.J.; Moore, A.P.; Smith, T.L.; Sharma, A.K.; Watkins, C.L. Reliability of measurements of muscle tone and muscle power in stroke patients. Age Ageing 2000, 29, 223–228. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swinnen, S.P.; Wenderoth, N. Two hands, one brain: Cognitive neuroscience of bimanual skill. Trends Cogn. Sci. 2004, 8, 18–25. [Google Scholar] [CrossRef]
- Wist, S.; Clivaz, K.; Sattelmayer, M. Muscle strengthening for hemiparesis after stroke: A meta-analysis. Ann. Phys. Rehabil. Med. 2016, 59, 114–124. [Google Scholar] [CrossRef]
- Saeys, W.; Vereeck, L.; Truijen, S.; Lafosse, C.; Wuyts, F.P.; van de Heyning, P. Randomized controlled trial of truncal exercises early after stroke to improve balance and mobility. Neurorehabil. Neural. Repair 2012, 26, 231–238. [Google Scholar] [CrossRef]
- Chen, X.; Liu, F.; Yan, Z.; Cheng, S.; Liu, X.; Li, H.; Li, Z. Therapeutic effects of sensory input training on motor function rehabilitation after stroke. Medicine 2018, 97, e13387. [Google Scholar] [CrossRef]
- Dai, S.; Piscicelli, C.; Clarac, E.; Baciu, M.; Hommel, M.; Pérennou, D. Balance, lateropulsion, and gait disorders in subacute stroke. Neurology 2021, 96, e2147–e2159. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhong, D.; Ye, J.; He, M.; Liu, X.; Zheng, H.; Jin, R.; Zhang, S.L. Rehabilitation for balance impairment in patients after stroke: A protocol of a systematic review and network meta-analysis. BMJ Open 2019, 9, e026844. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lim, C.G. Multi-Sensorimotor training improves proprioception and balance in subacute stroke patients: A randomized controlled pilot trial. Front Neurol. 2019, 10, 157. [Google Scholar] [CrossRef] [Green Version]
- Liu, T.W.; Ng, G.Y.F.; Ng, S.S.M. Effectiveness of a combination of cognitive behavioral therapy and task-oriented balance training in reducing the fear of falling in patients with chronic stroke: Study protocol for a randomized controlled trial. Trials 2018, 19, 168. [Google Scholar] [CrossRef] [Green Version]
- Kannan, L.; Vora, J.; Bhatt, T.; Hughes, S.L. Cognitive-motor exergaming for reducing fall risk in people with chronic stroke: A randomized controlled trial. NeuroRehabilitation 2019, 44, 493–510. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.Y.; Moon, Y.; Choi, J.D. Effects of Cognitive Task Training on Dynamic Balance and Gait of Patients with Stroke: A Preliminary Randomized Controlled Study. Med. Sci. Monit. Basic Res. 2020, 10, e925264. [Google Scholar] [CrossRef] [PubMed]
- Krukowska, J.; Bugajski, M.; Sienkiewicz, M.; Czernicki, J. The influence of NDT-Bobath and PNF methods on the field support and total path length measure foot pressure (COP) in patients after stroke. Neurol. Neurochi. Pol. 2016, 50, 449–454. [Google Scholar] [CrossRef]
| Intervention | Training Procedure |
|---|---|
| Proprioception and pressure training | Training was performed to improve the sense of position and sense of movement among proprioception senses in the first area and the last two areas after applying pressure to the ankle joint. |
| Tactile and pressure Stimulation training | CSMT was given using contact tasks that presented cognitive problems to distinguish the difference between the degree of pressure and surface material and the difference between sense of friction and weight. It was made possible to distinguish each sense by using visual sense and somatosensory during the task of distinguishing each sense. |
| Tactile and pressure Stimulation training | The trunk that presented cognitive problems was focused on in the training to distinguish the difference in the degree of sponge pressure in the sitting position. It was made possible to distinguish each sense using visual sense and somatosensory during the task of distinguishing each sense. |
| Proprioception and Spatial task training | It was divided into four sections after connecting the knee line of the affected lower limb and the center line to the line to train where the position of the foot of lower limb was located in space. |
| Two lines were made according to the angle of the knee while the subject’s heels were attached. The training was then conducted on the position of the leg in space. If this was undertaken completely, it was performed by adding a line. | |
| To recover spatial cognition, a movement was made to retrace an ellipse in front of the body. From the small position of a drawing board, the movement was induced along an increasingly large circle. |
| Variable | CSMT Group (n = 17) | Control Group (n = 18) | X2/t | p |
|---|---|---|---|---|
| Height (cm) | 165.58 (7.62) a | 166.54 (5.82) a | −0.421 c | 0.676 |
| Weight (kg) | 64.42 (8.53) a | 65.13 (10.81) a | −0.215 c | 0.831 |
| Age (year) | 51.75 (14.41) a | 58.22 (16.53) a | −1.340 c | 0.189 |
| Gender (Male/Female) | 9/8 | 10/8 | 0.024 b | 0.877 |
| Diagnosis (Infarction/Hemorrhage) | 8/9 | 11/7 | 0.696 b | 0.404 |
| Affected side (Left/Right) | 9/8 | 9/9 | 0.030 b | 0.862 |
| MMSE-K (score) | 26.65 (1.27) a | 26.89 (1.37) a | −0.541 c | 0.592 |
| Brunnstrom stage | 2.64 (0.49) a | 2.72 (0.46) a | −0.466 c | 0.644 c |
| Variable | CSMT Group (n = 17) | Control Group (n = 18) | F | p- Value | ||
|---|---|---|---|---|---|---|
| Pre-Test | Post-Test | Pre-Test | Post-Test | |||
| LEMTA (kg) | 3.70 (0.54) 1 | 4.07 (0.52) a,b | 3.88 (0.68) | 4.10 (0.61) a | 6.760 | 0.014 *,2 |
| MRC (score) | 13.35 (1.58) | 16.88 (1.83) a,b | 13.61 (1.72) | 14.78 (1.66) a | 96.243 | 0.000 *,2 |
| Variable | CSMT Group (n = 17) | Control Group (n = 18) | F | p- Value | ||
|---|---|---|---|---|---|---|
| Pre-Test | Post-Test | Pre-Test | Post-Test | |||
| REOSA (mm2) | 134.12 (39.00) 1 | 122.18 (39.72) a,b | 132.00 (36.22) | 126.28 (37.46) a | 10.066 | 0.003 *,2 |
| REOAS (cm/s) | 1.09 (0.28) | 0.89 (0.24) a,b | 1.07 (0.28) | 0.93 (0.27) a | 7.797 | 0.009 *,2 |
| RECSA (mm2) | 175.00 (24.99) | 161.24 (26.38) a,b | 173.87 (26.74) | 164.72 (28.03) a | 6.501 | 0.016 *,2 |
| RECAS (cm/s) | 1.42 (0.29) | 1.21 (0.28) a,b | 1.39 (0.36) | 1.23 (0.37) a | 19.372 | 0.000 *,2 |
| LOS (cm2) | 3086.95 (567.40) | 3507.00 (404.19) a,b | 3162.14 (396.33) | 3334.50 (375.42) a | 5.514 | 0.025 *,2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, K.-H.; Jang, S.-H. Effects of Cognitive Sensory Motor Training on Lower Extremity Muscle Strength and Balance in Post Stroke Patients: A Randomized Controlled Study. Clin. Pract. 2021, 11, 640-649. https://doi.org/10.3390/clinpract11030079
Kim K-H, Jang S-H. Effects of Cognitive Sensory Motor Training on Lower Extremity Muscle Strength and Balance in Post Stroke Patients: A Randomized Controlled Study. Clinics and Practice. 2021; 11(3):640-649. https://doi.org/10.3390/clinpract11030079
Chicago/Turabian StyleKim, Kyung-Hun, and Sang-Hun Jang. 2021. "Effects of Cognitive Sensory Motor Training on Lower Extremity Muscle Strength and Balance in Post Stroke Patients: A Randomized Controlled Study" Clinics and Practice 11, no. 3: 640-649. https://doi.org/10.3390/clinpract11030079
APA StyleKim, K.-H., & Jang, S.-H. (2021). Effects of Cognitive Sensory Motor Training on Lower Extremity Muscle Strength and Balance in Post Stroke Patients: A Randomized Controlled Study. Clinics and Practice, 11(3), 640-649. https://doi.org/10.3390/clinpract11030079





