Case Series of Multisystem Inflammatory Syndrome (MIS-C) in Children during the SARS-CoV-2 Pandemic in Latvia
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Case Reports
3.1.1. Demographic Characteristics and Underlying Medical Conditions
3.1.2. Initial Signs and Symptoms
3.1.3. Laboratory Markers and Additional Diagnostics
3.1.4. Link to SARS-CoV-2
3.1.5. Treatment and Clinical Course
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Concerning the State of Emergency Declared in Latvia. Available online: https://www.mfa.gov.lv/en/consular-information/news/66931-concerning-the-state-of-emergency-declared-in-latvia (accessed on 12 March 2020).
- News about COVID-19. Available online: https://www.spkc.gov.lv/lv/covid-19 (accessed on 7 March 2021).
- Rafferty, M.S.; Burrows, H.; Joseph, J.P.; Leveille, J.; Nihtianova, S.; Amirian, E.S. Multisystem inflammatory syndrome in children (MIS-C) and the coronavirus pandemic: Current knowledge and implications for public health. J. Infect. Public Health 2021, 14, 484–494. [Google Scholar] [CrossRef]
- Junior, H.S.; Sakano, T.M.S.; Rodrigues, R.M.; Eisencraft, A.P.; de Carvalho, V.E.L.; Schvartsman, C.; Reis, A.G.A.D.C. Multisystem inflammatory syndrome associated with COVID-19 from the pediatric emergency physician’s point of view. J. Pediatr. 2021, 97, 140–159. [Google Scholar] [CrossRef] [PubMed]
- Kwak, J.H.; Lee, S.-Y.; Choi, J.-W.; Korean Society of Kawasaki Disease. Clinical features, diagnosis, and outcomes of multisystem inflammatory syndrome in children associated with coronavirus disease 2019. Clin. Exp. Pediatr. 2021, 64, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Abbas, M.; Törnhage, C.-J. Family Transmission of COVID-19 Including a Child with MIS-C and Acute Pancreatitis. Int. Med. Case Rep. J. 2021, 5, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Cirks, B.T.; Geracht, J.C.; Jones, O.Y.; May, J.W.; Mikita, C.P.; Rajnik, M.; Helfrich, A.M. Multisystem Inflammatory Syndrome in Children during the COVID-19 Pandemic: A Case Report on Managing the Hyperinflammation. Mil. Med. 2021, 26, e270–e276. [Google Scholar] [CrossRef] [PubMed]
- Henderson, L.A.; Canna, S.W.; Friedman, K.G.; Gorelik, M.; Lapidus, S.K.; Bassiri, H.; Behrens, E.M.; Ferris, A.; Kernan, K.F.; Schulert, G.S.; et al. American College of Rheumatology Clinical Guidance for Multisystem Inflammatory Syndrome in Children Associated with SARS–CoV-2 and Hyperinflammation in Pediatric COVID-19: Version 1. Arthritis Rheumatol. 2020, 72, 1791–1805. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention Health Alert Network (HAN). Multisystem Inflammatory Syndrome in Children (MIS-C) Associated with Coronavirus Disease 2019 (COVID-19). Available online: https://emergency.cdc.gov/han/2020/han00432.asp (accessed on 30 August 2020).
- Belot, A.; Antona, D.; Renolleau, S.; Javouhey, E.; Hentgen, V.; Angoulvant, F.; Delacourt, C.; Iriart, X.; Ovaert, C.; Bader-Meunier, B.; et al. SARS-CoV-2-related paediatric inflammatory multisystem syndrome, an epidemiological study, France, 1 March to 17 May 2020. Eurosurveillance 2020, 25, 2001010. [Google Scholar] [CrossRef]
- Remppis, J.; Ganzenmueller, T.; Vasconcelos, M.K.; Heinzel, O.; Handgretinger, R.; Renk, H. A case series of children and young people admitted to a tertiary care hospital in Germany with COVID-19. BMC Infect. Dis. 2021, 21, 1–6. [Google Scholar] [CrossRef]
- Morris, S.B.; Schwartz, N.G.; Patel, P.; Abbo, L.; Beauchamps, L.; Balan, S.; Lee, E.H.; Paneth-Pollak, R.; Geevarughese, A.; Lash, M.K.; et al. Case Series of Multisystem Inflammatory Syndrome in Adults Associated with SARS-CoV-2 Infection—United Kingdom and United States, March–August 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 9, 1450–1456. [Google Scholar] [CrossRef]
- Fouriki, A.; Fougère, Y.; De Camaret, C.; Rohner, G.B.; Grazioli, S.; Wagner, N.; Relly, C.; Schmid, J.P.; Trück, J.; Kottanatu, L.; et al. Case Report: Case Series of Children With Multisystem Inflammatory Syndrome Following SARS-CoV-2 Infection in Switzerland. Front. Pediatr. 2021, 5, 594127. [Google Scholar] [CrossRef]
- Torres, J.P.; Izquierdo, G.; Acuña, M.; Pavez, D.; Reyes, F.; Fritis, A.; González, R.; Rivacoba, C.; Contardo, V.; Tapia, L.I. Multisystem inflammatory syndrome in children (MIS-C): Report of the clinical and epidemiological characteristics of cases in Santiago de Chile during the SARS-CoV-2 pandemic. Int. J. Infect. Dis. 2020, 100, 75–81. [Google Scholar] [CrossRef]
- Mania, A.; Mazur-Melewska, K.; Lubarski, K.; Kuczma-Napierała, J.; Mazurek, J.; Jończyk-Potoczna, K.; Służewski, W.; Figlerowicz, M. Wide spectrum of clinical picture of COVID-19 in children—From mild to severe disease. J. Infect. Public Health 2021, 29, 374–379. [Google Scholar] [CrossRef]
- Dufort, E.M.; Koumans, E.H.; Chow, E.J.; Rosenthal, E.M.; Muse, A.; Rowlands, J.; Barranco, M.A.; Maxted, A.M.; Rosenberg, E.S.; Easton, D.; et al. Multisystem Inflammatory Syndrome in Children in New York State. N. Engl. J. Med. 2020, 23, 347–358. [Google Scholar] [CrossRef]
- Lee, P.Y.; Day-Lewis, M.; Henderson, L.A.; Friedman, K.G.; Lo, J.; Roberts, J.E.; Lo, M.S.; Platt, C.D.; Chou, J.; Hoyt, K.J.; et al. Distinct clinical and immunological features of SARS–CoV-2–induced multisystem inflammatory syndrome in children. J. Clin. Investig. 2020, 130, 5942–5950. [Google Scholar] [CrossRef]
- Grimaud, M.; Starck, J.; Levy, M.; Marais, C.; Chareyre, J.; Khraiche, D.; Leruez-Ville, M.; Quartier, P.; Léger, P.L.; Geslain, G.; et al. Acute myocarditis and multisystem inflammatory emerging disease following SARS-CoV-2 infection in critically ill children. Ann. Intensiv. Care 2020, 1, 69. [Google Scholar] [CrossRef]
- Belhadjer, Z.; Méot, M.; Bajolle, F.; Khraiche, D.; Legendre, A.; Abakka, S.; Auriau, J.; Grimaud, M.; Oualha, M.; Beghetti, M.; et al. Acute Heart Failure in Multisystem Inflammatory Syndrome in Children in the Context of Global SARS-CoV-2 Pandemic. Circulation 2020, 4, 429–436. [Google Scholar] [CrossRef]
- Jackson, R.J.; Chavarria, H.D.; Hacking, S.M. A Case of Multisystem Inflammatory Syndrome in Children Mimicking Acute Appendicitis in a COVID-19 Pandemic Area. Cureus 2020, 29, e10722. [Google Scholar] [CrossRef]
- Ramcharan, T.; Nolan, O.; Lai, C.Y.; Prabhu, N.; Krishnamurthy, R.; Richter, A.G.; Jyothish, D.; Kanthimathinathan, H.K.; Welch, S.B.; Hackett, S.; et al. Paediatric Inflammatory Multisystem Syndrome: Temporally Associated with SARS-CoV-2 (PIMS-TS): Cardiac Features, Management and Short-Term Outcomes at a UK Tertiary Paediatric Hospital. Pediatr. Cardiol. 2020, 41, 1391–1401. [Google Scholar] [CrossRef]
- Abrams, J.Y.; Godfred-Cato, S.E.; Oster, M.E.; Chow, E.; Koumans, E.H.; Bryant, B.; Leung, J.W.; Belay, E.D. Multisystem Inflammatory Syndrome in Children Associated with Severe Acute Respiratory Syndrome Coronavirus 2: A Systematic Review. J. Pediatr. 2020, 226, 45–54.e1. [Google Scholar] [CrossRef]
- Godfred-Cato, S.; Bryant, B.; Leung, J.; Oster, M.E.; Conklin, L.; Abrams, J.; Roguski, K.; Wallace, B.; Prezzato, E.; Koumans, E.H.; et al. COVID-19–Associated Multisystem Inflammatory Syndrome in Children—United States, March–July 2020. MMWR Morb. Mortal. Wkly. Rep. 2020, 14, 1074–1080. [Google Scholar] [CrossRef]
- Radia, T.; Williams, N.; Agrawal, P.; Harman, K.; Weale, J.; Cook, J.; Gupta, A. Multi-system inflammatory syndrome in children & adolescents (MIS-C): A systematic review of clinical features and presentation. Paediatr. Respir. Rev. 2020, 11, 51–57. [Google Scholar] [CrossRef]
- Children’s Hospital of Philadelphia. Emergency Department, ICU and Inpatient Clinical Pathway for Evaluation of Possible Multisystem Inflammatory Syndrome (MIS-C). Available online: https://www.chop.edu/clinical-pathway/multisystem-inflammatory-syndrome-mis-c-clinical-pathway (accessed on 28 May 2020).
- Hennon, T.R.; Penque, M.D.; Abdul-Aziz, R.; Alibrahim, O.S.; McGreevy, M.B.; Prout, A.J.; Schaefer, B.A.; Ambrusko, S.J.; Pastore, J.V.; Turkovich, S.J.; et al. COVID-19 associated Multisystem Inflammatory Syndrome in Children (MIS-C) guidelines; A Western New York approach. Prog. Pediatr. Cardiol. 2020, 57, 101232. [Google Scholar] [CrossRef]
- Cattalini, M.; On Behalf of the Rheumatology Study Group of the Italian Society of Pediatrics; Taddio, A.; Bracaglia, C.; Cimaz, R.; Della Paolera, S.; Filocamo, G.; La Torre, F.; Lattanzi, B.; Marchesi, A.; et al. Childhood multisystem inflammatory syndrome associated with COVID-19 (MIS-C): A diagnostic and treatment guidance from the Rheumatology Study Group of the Italian Society of Pediatrics. Ital. J. Pediatr. 2021, 47, 1–6. [Google Scholar] [CrossRef]
- Tolunay, O.; Çelik, Ü.; Arslan, İ.; Orgun, A.; Demir, H.; Demir, O.; Dağdelen, E.Ç. Multisystem Inflammatory Syndrome in Children (MIS-C) Associated with COVID-19: A Case Series Experience in a Tertiary Care Hospital of Southern Turkey. J. Trop. Pediatr. 2021. [Google Scholar] [CrossRef]
| Characteristic | Total |
|---|---|
| Sex, n: Female: Male: | 2/13 11/13 |
| Age in years, range, median | 4–17, 8.8 |
| Comorbidities, n | 3/13 |
| Days in hospital, median (IQR) | 13 (11–18) |
| Outcome | Recovery |
| PICU admission, n Days in PICU, median (IQR) | 7/13 2 (1.25–2.75) |
| Clinical characteristic: | |
| Duration of symptoms at admission, median days (IQR) | 5 (4–6) |
| Days with fever, median (IQR) | 7 (5.75–7.25) |
| No. of organ systems involved, n: 2–3 4–5 ≥6 | 0/13 13/13 0/13 |
| Clinical manifestations, n: Fever | 13/13 |
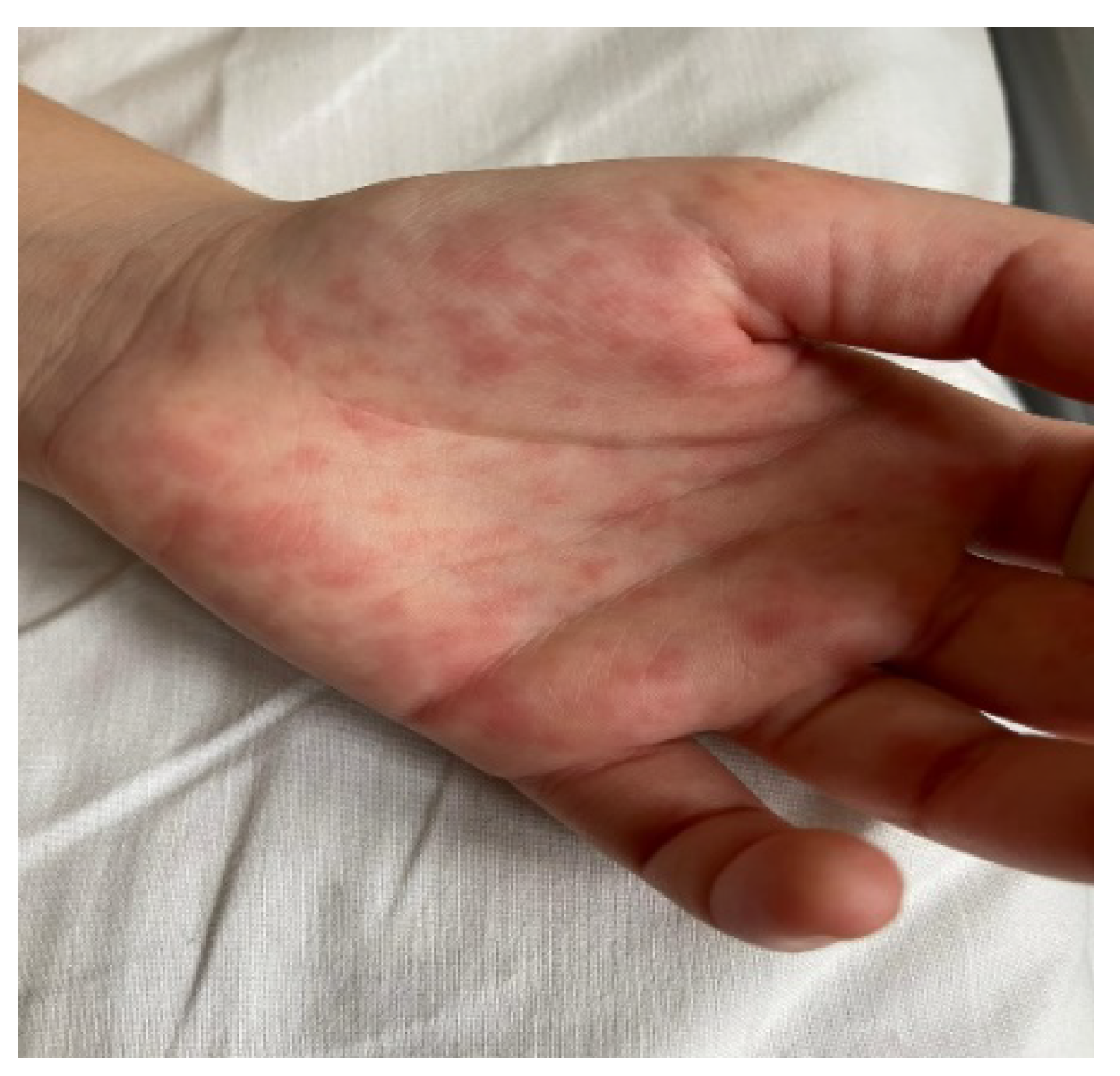
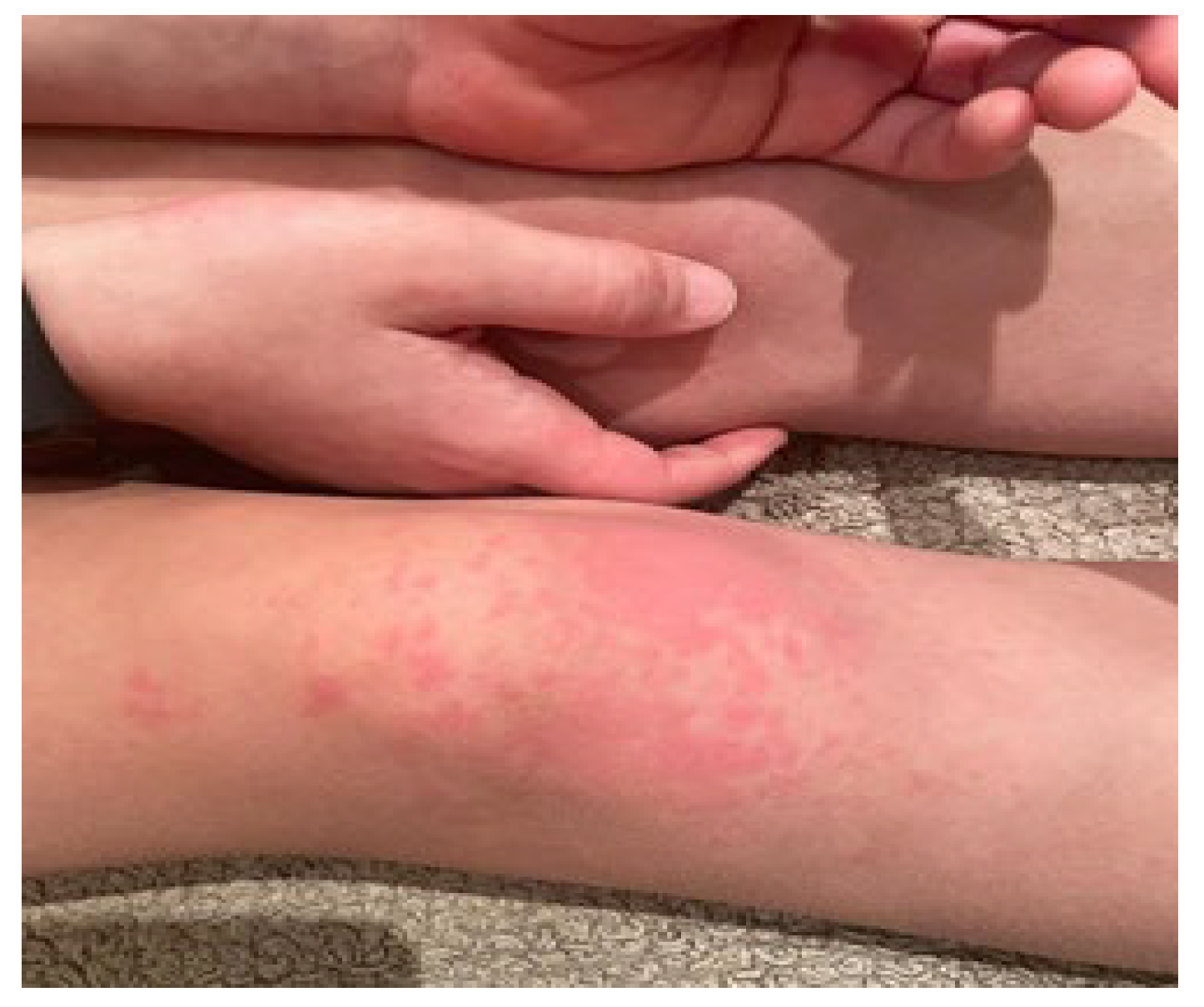
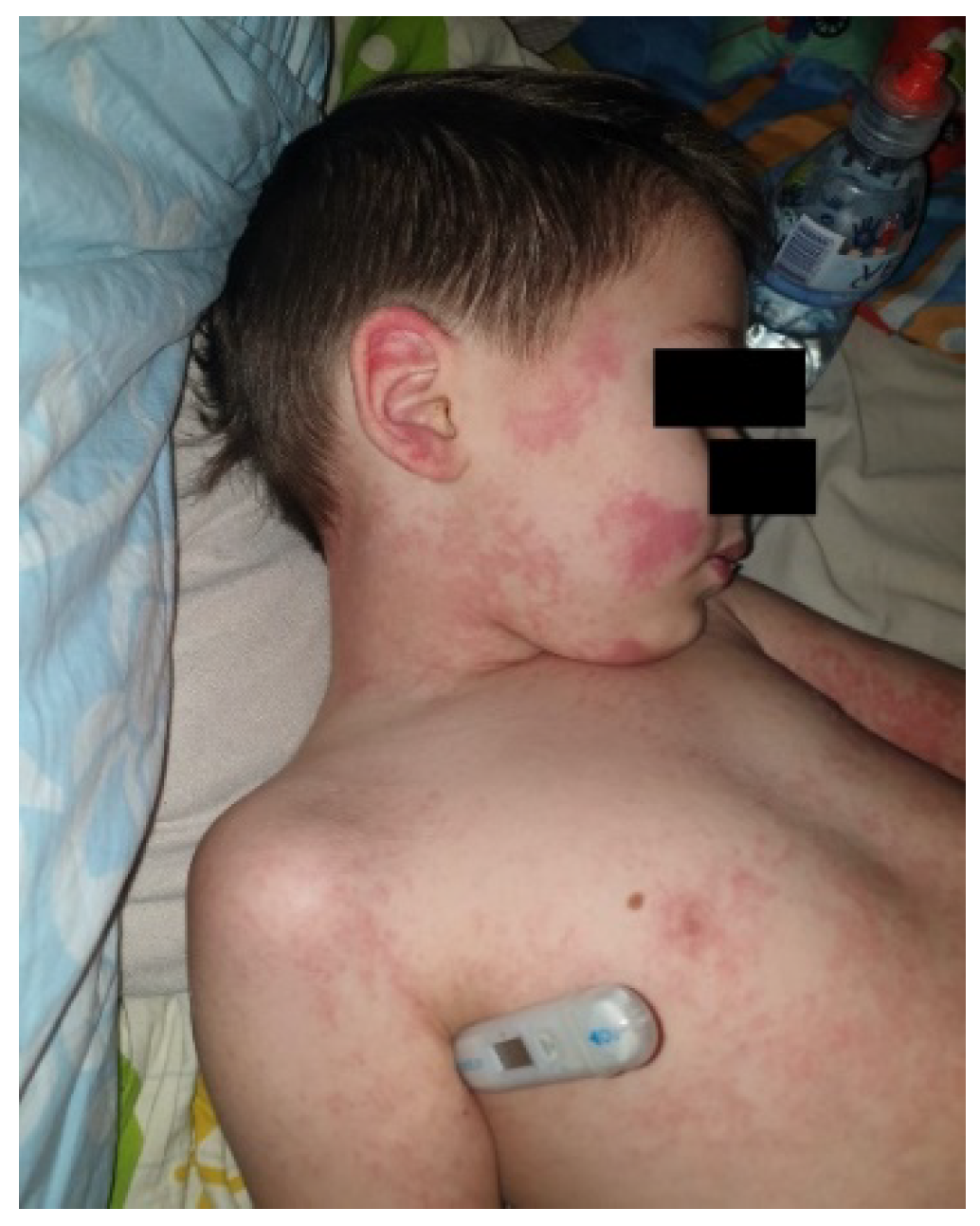
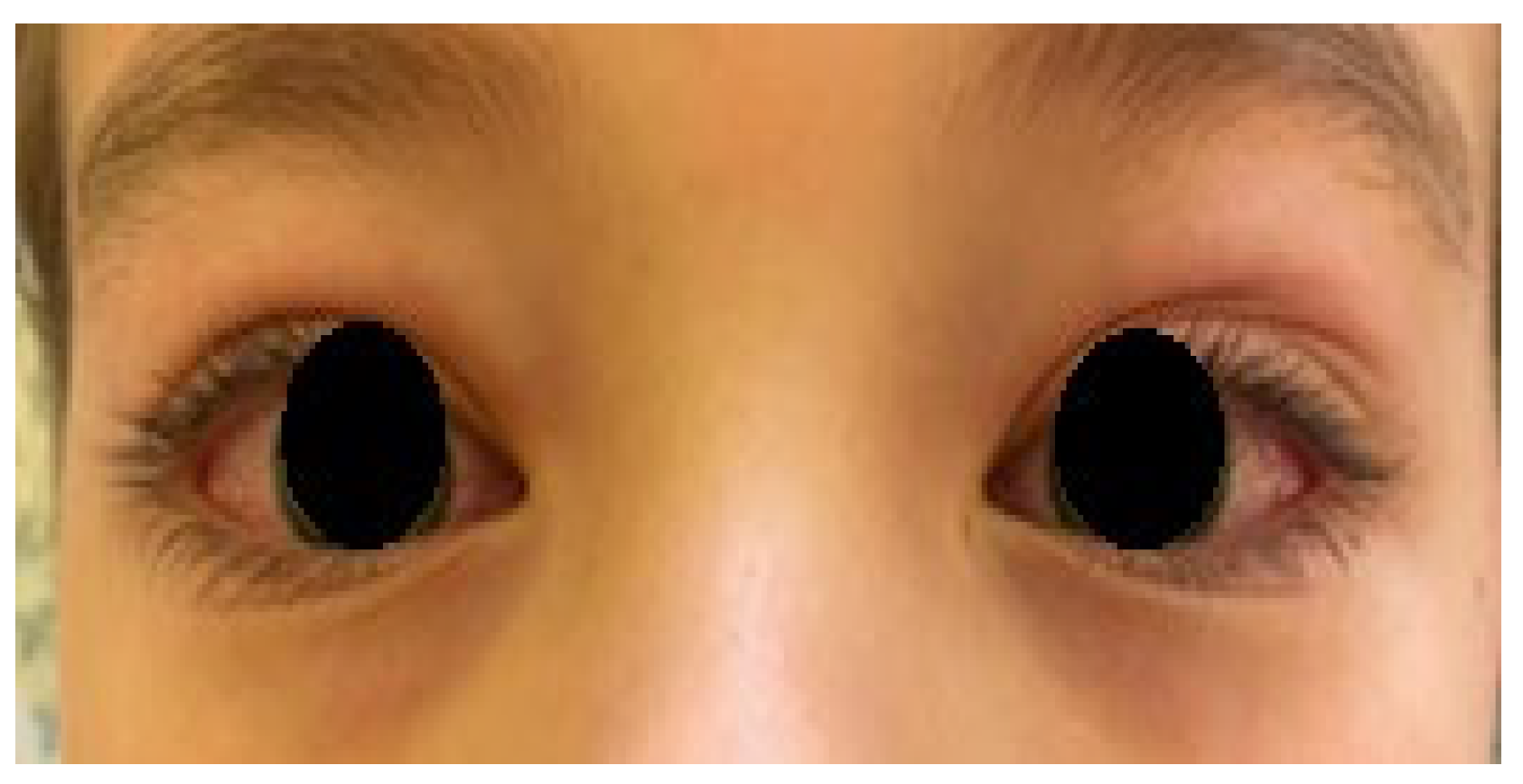
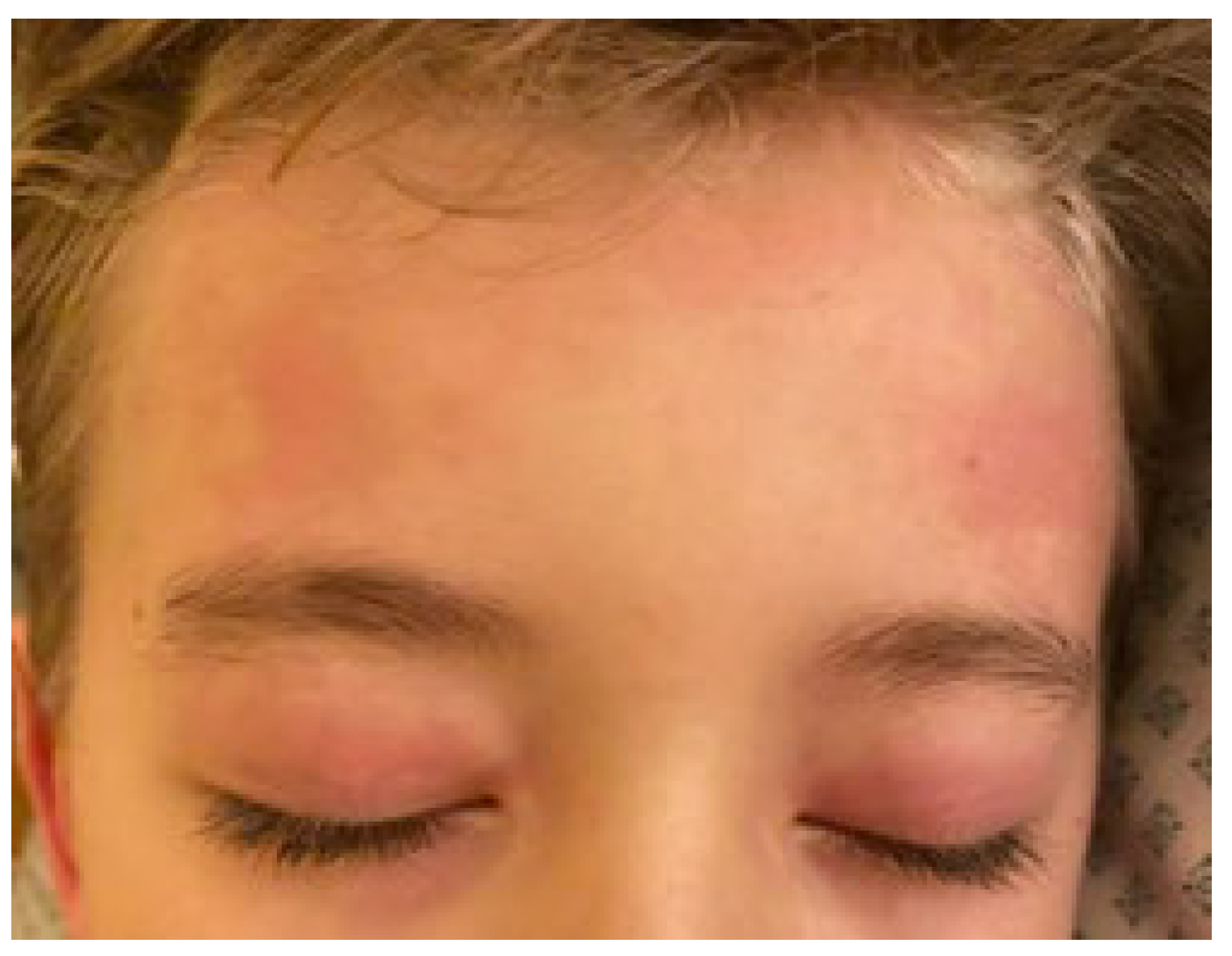
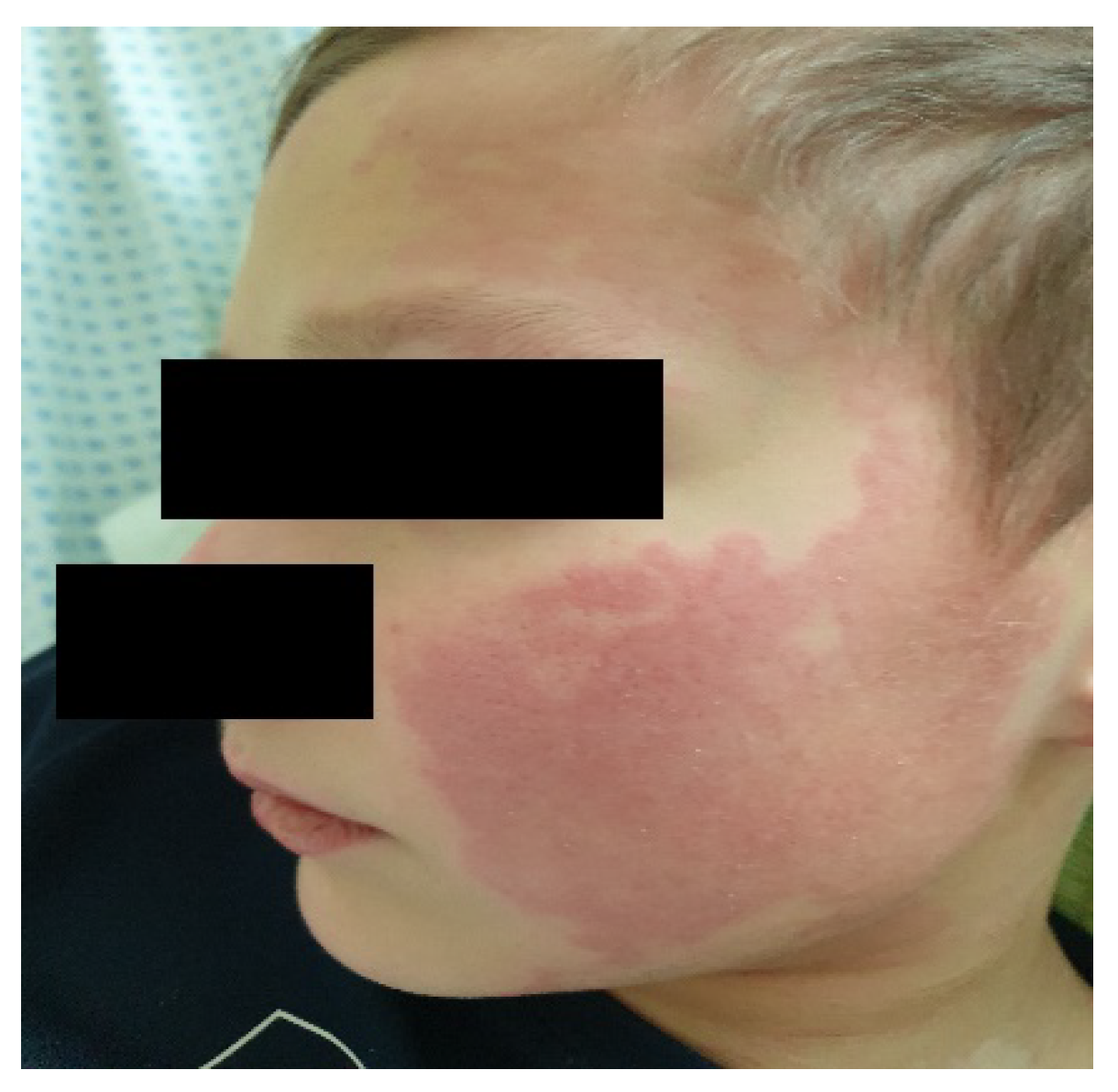
Rash:
| 10/13 2/13 |
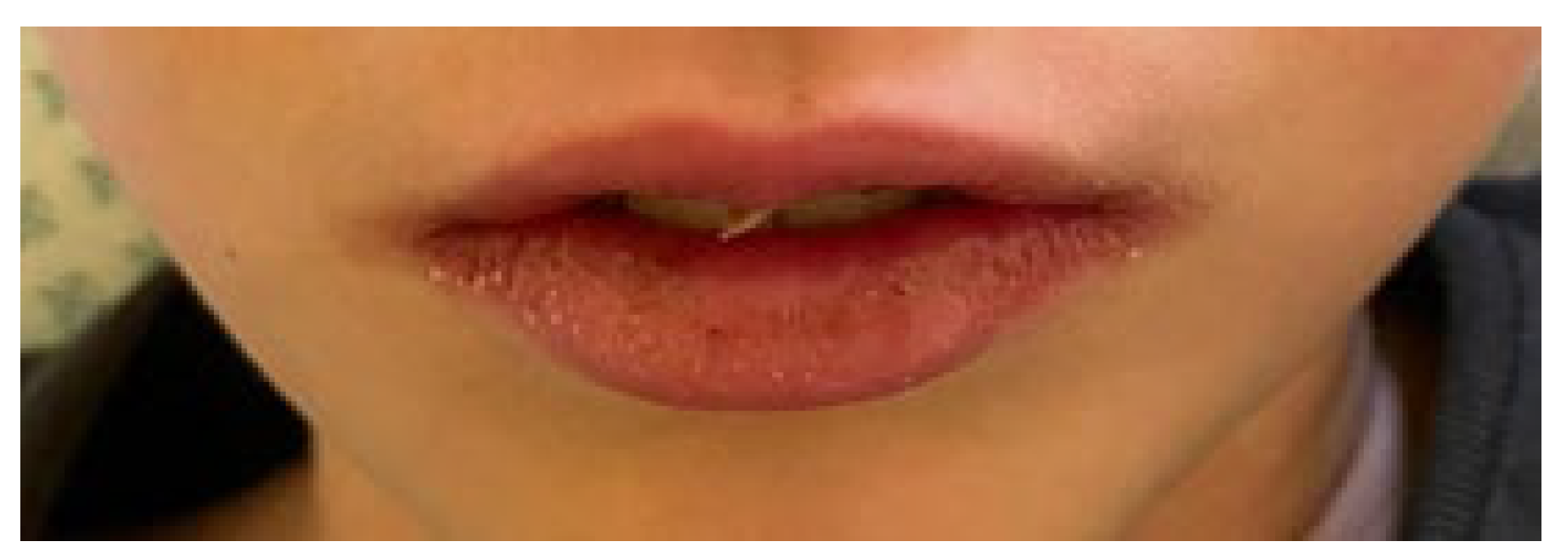
Mucocutaneous lesions:
| 12/13 2/13 9/13 11/13 |
Extremity changes:
Gastrointestinal:
| 9/13 7/13 11/13 3/13 9/13 6/13 |
Cardiovascular:
| 5/13 13/13 1/13 2/13 10/13 |
Respiratory:
| 10/13 6/13 3/13 2/13 6/13 8/13 |
Neurologic:
| 6/13 3/13 4/13 2/13 2/13 1/13 |
Other:
| 8/13 2/13 6/13 3/13 |
| Characteristic | Results |
|---|---|
| The Median of Peak Values (IQR) | |
Initial laboratory criteria, (reference ranges):
| 187.01 (135.61–249.15) 48 (40.5–65.5) 0.55 (0.44–0.65) 112 (96–134.25) 129.65 (126.8–131.25) 26.17 (24.3–32.12) |
Additional inflammatory markers:
| 194 (150–320) 583.2 (511.6–861.1) 332 (325.75–342.25) |
Cardiac biomarkers:
| 7218 (2434–17134) 93.4 (46.1–132.1) |
Coagulation parameters:
| 5.97 (3.31–10.47) 5.66 (4.81–7.01) |
| n | |
Chest X-ray:
| 3/13 3/13 4/13 4/13 1/13 |
Electrocardiography:
| 11/13 6/13 6/13 8/13 1/13 |
Echocardiography:
| 6/13 8/13 2/13 1/13 2/13 3/13 1/13 |
Abdominal ultrasonography:
| 2/13 4/13 3/13 1/13 1/13 4/13 |
| Pleural ultrasonography-effusion: | 9/13 |
Computed tomography of the lungs:
| 2/13 2/13 1/13 |
SARS-CoV-2 test results at the admission:
| 0/13 13/13 13/13 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Racko, I.; Smane, L.; Klavina, L.; Pucuka, Z.; Roge, I.; Pavare, J. Case Series of Multisystem Inflammatory Syndrome (MIS-C) in Children during the SARS-CoV-2 Pandemic in Latvia. Clin. Pract. 2021, 11, 363-373. https://doi.org/10.3390/clinpract11020051
Racko I, Smane L, Klavina L, Pucuka Z, Roge I, Pavare J. Case Series of Multisystem Inflammatory Syndrome (MIS-C) in Children during the SARS-CoV-2 Pandemic in Latvia. Clinics and Practice. 2021; 11(2):363-373. https://doi.org/10.3390/clinpract11020051
Chicago/Turabian StyleRacko, Iveta, Liene Smane, Lizete Klavina, Zanda Pucuka, Ieva Roge, and Jana Pavare. 2021. "Case Series of Multisystem Inflammatory Syndrome (MIS-C) in Children during the SARS-CoV-2 Pandemic in Latvia" Clinics and Practice 11, no. 2: 363-373. https://doi.org/10.3390/clinpract11020051
APA StyleRacko, I., Smane, L., Klavina, L., Pucuka, Z., Roge, I., & Pavare, J. (2021). Case Series of Multisystem Inflammatory Syndrome (MIS-C) in Children during the SARS-CoV-2 Pandemic in Latvia. Clinics and Practice, 11(2), 363-373. https://doi.org/10.3390/clinpract11020051





