Combination of Levamisole with Prednisone in Treating Recurrent Major Aphthous Ulcer in a Young Boy: A Case Report
Abstract
1. Introduction
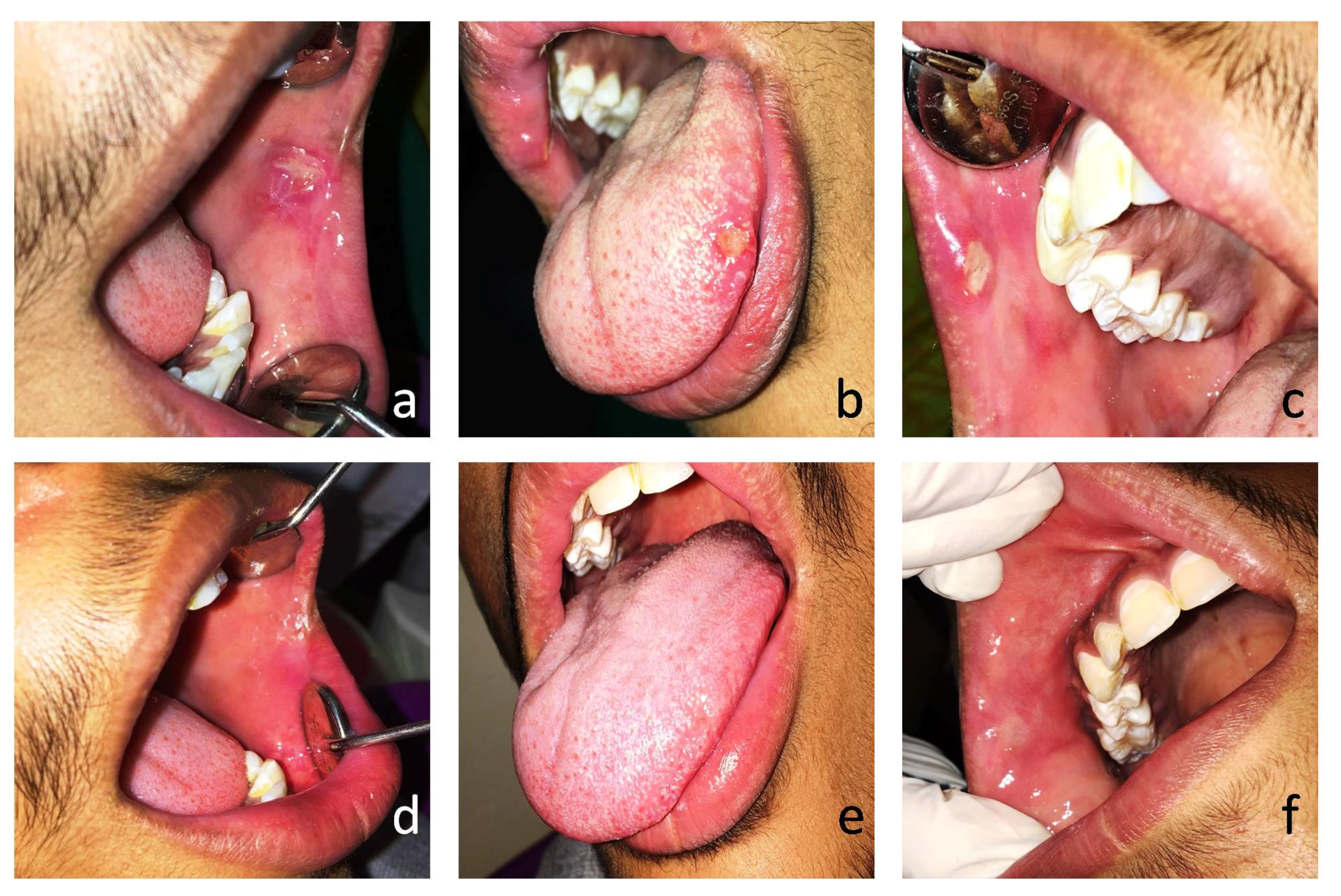
2. Case Report
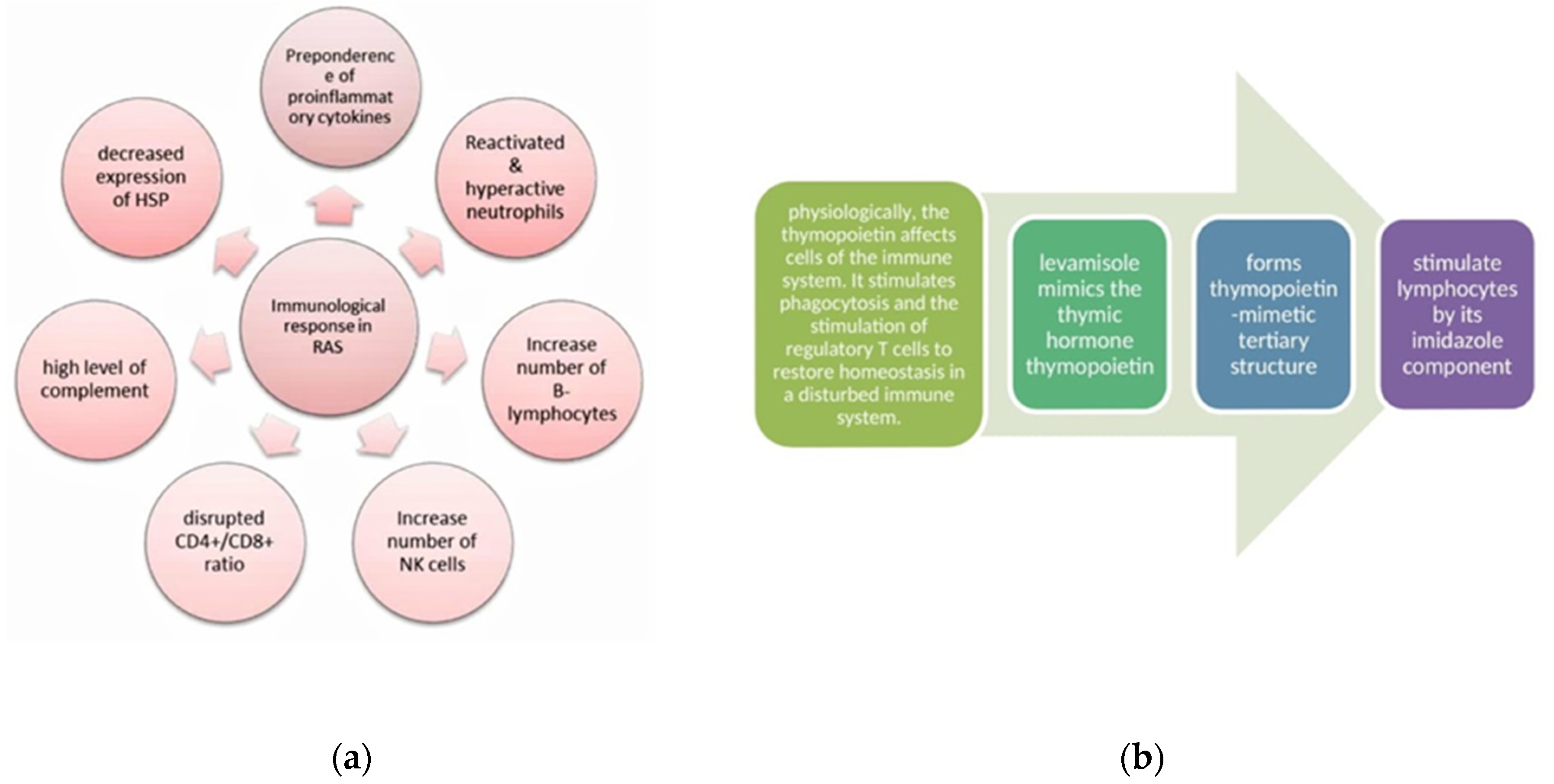
3. Discussion
- 150 mg daily for 3 consecutive days/weeks
- 150 mg for 3 consecutive days every other week
- 50 mg 3 times daily for 2 consecutive days every week
- 150 mg three times daily for 3 consecutive days/weeks
- 150 mg daily for 3 consecutive days/weeks with an interval of 2 weeks [20].
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Consent for Publication
Conflicts of Interest
References
- Tarakji, B.; Gazal, G.; Al-Maweri, S.A.; Azzeghaiby, S.N.; Alaizari, N. Guideline for the diagnosis and treatment of recurrent aphthous stomatitis for dental practitioners. JIOH 2015, 7, 74. [Google Scholar] [PubMed]
- Chavan, M.; Jain, H.; Diwan, N.; Khedkar, S.; Shete, A.; Durkar, S. Recurrent aphthous stomatitis: A review. J. Oral Pathol. Med. 2012, 41, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Gupta, P.; Ashok, L.; Naik, S.R. Assessment of serum interleukin-8 as a sensitive serological marker in monitoring the therapeutic effect of levamisole in recurrent aphthous ulcers: A randomized control study. Indian J. Dent. Res. 2014, 25, 284. [Google Scholar] [CrossRef]
- Vujevich, J.; Zirwas, M. Treatment of severe, recalcitrant, major aphthous stomatitis with adalimumab. Cutis N. Y. 2005, 76, 129–132. [Google Scholar]
- Picciani, B.L.S.; Silva-Junior, G.O.; Barbirato, D.S.; Ramos, R.T.; Cantisano, M.H. Regression of major recurrent aphthous ulcerations using a combination of intralesional corticosteroids and levamisole: A case report. Clinics 2010, 65, 650–652. [Google Scholar] [CrossRef] [PubMed]
- Darshan, D.D.; Kumar, C.N.; Kumar, A.D.; Manikantan, N.S.; Balakrishnan, D.; Uthkal, M.P. Clinical study to know the efficacy of Amlexanox 5% with other topical Antiseptic, Analgesic and Anesthetic agents in treating minor RAS. JIOH 2014, 6, 5–11. [Google Scholar] [PubMed]
- Katti, G.; Darshan, D.D. Amlexanox in the Treatment of Recurrent Minor Aphthous Ulcers. Int. J. Dent. Clin. 2011, 3, 23–26. [Google Scholar]
- Sabbagh, A.H.; Felemban, M.F. Therapeutic management of recurrent aphthous stomatitis: A review of the growing knowledge. Ann. Int. Med. Den. Res. 2016, 2. [Google Scholar] [CrossRef]
- Ślebioda, Z.; Szponar, E.; Kowalska, A. Etiopathogenesis of recurrent aphthous stomatitis and the role of immunologic aspects: A literature review. Arch. Immunol. Ther. Exp. 2014, 62, 205–215. [Google Scholar] [CrossRef]
- Padmaja Bathina, S.S.K.; Kudagi, B.L.; Chandra, B.; Pathapati, R.M.; Buchineni, M. A study of levamisole in treating recurrent aphthous stomatitis (RAS) in a tertiary care teaching hospital. Int. J. Inst. Pharm. Life Sci. 2015, 5, 344–350. [Google Scholar]
- Rajendran, P. Effect and tolerability of Levamisole in the management of aphthous ulcers—A cross-sectional study. IAIM 2015, 2, 27–31. [Google Scholar]
- Tripathi, K. Essentials of Medical Pharmacology; JP Medical Ltd.: New Delhi, India, 2013. [Google Scholar]
- Shivhare, P.; Shankarnarayan, L.; Singh, A.; Patil, S.T.; Yadav, M. Role of Immunomodulators in Oral Diseases. Int. J. Oral Health Med. Res. 2015, 2, 73–80. [Google Scholar]
- Parvathi Devi, M.K.; Ramesh, D.N.; Koppal, S.; Byatnal, A.R.; Rukmangada, T.; Byatnal, A.A. Efficacy of rebamipide and levamisole in the treatment of patients with recurrent aphthous ulcer-a comparative study. J. Clin. Diagn. Res. 2014, 8, ZC119. [Google Scholar]
- Symoens, J.; Brugmans, J. Letter: Treatment of recurrent aphthous stomatitis and herpes with levamisole. Br. Med. J. 1974, 4, 592–596. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Meyer, J.D.; Degraeve, M.; Clarysse, J.; De Loose, F.; Peremans, W. Levamisole in aphthous stomatitis: Evaluation of three regimens. Br. Med. J. 1977, 1, 671–674. [Google Scholar] [CrossRef]
- de Heyning Van, J. Levamisole in the treatment of recurrent aphthous stomatitis. Laryngoscope 1978, 88, 522–527. [Google Scholar] [CrossRef] [PubMed]
- Weckx, L.L.M.; Hirata, C.H.W.; de Abreu, M.A.M.M.; Fillizolla, V.C.; de Silva, O.M.P. Levamisole does not prevent lesions of recurrent aphthous stomatitis: A double-blind placebo-controlled clinical trial. Rev. Assoc. Med. Bras. 2009, 55, 132–138. [Google Scholar] [CrossRef]
- Sharda, N.; Shashikanth, M.; Kant, P.; Jain, M. Levamisole and low-dose prednisolone in the treatment of recurrent aphthous stomatitis. J. Oral Pathol. Med. 2014, 43, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Chamani, G.; Rad, M.; Zarei, M.R.; Hashemimanesh, S.R.; Abbaszadeh, E.; Sadeghi, M. Effect of levamisole on the treatment of recurrent aphthous stomatitis: A systematic review and meta-analysis. J. Oral Health Oral Epidemiol. 2016, 5, 70–77. [Google Scholar]
- Sharma, S.; Ali, F.M.; Saraf, K.; Mudhol, A. Antihelminthic drugs in recurrent aphthous stomatitis: A short review. J. Pharm. Bioallied Sci. 2014, 6, 65–69. [Google Scholar]
- Edgar, N.R.; Saleh, D.; Miller, R.A. Recurrent aphthous stomatitis: A review. J. Clin. Aesthet. Dermatol. 2017, 10, 26–29. [Google Scholar] [PubMed]
- Lalabonova, H.; Daskalov, H. Clinical assessment of the therapeutic effect of low-level laser therapy on chronic recurrent aphthous stomatitis. Biotechnol. Biotechnol. Equip. 2014, 28, 929–933. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.C.; Jeong, S.J.; Eun, Y.G.; Song, R.; Oh, I.H. Risk of autoimmune diseases in recurrent aphthous ulcer patients: A nationwide population study. Oral Dis. 2020, 26, 1–8. [Google Scholar] [CrossRef] [PubMed]
| Character | Minor RAS | Major RAS | Herpetiform Ulcers |
|---|---|---|---|
| Peak age (decade) | 2nd | 1st and 2nd | 3rd |
| Number of ulcers | 1–5 | 1–3 | 5–20 (up to 100) |
| Size of ulcers (mm) | Less than 10 mm | More than 10 mm | 1–2 mm |
| Duration | 7–14 days | 2 weeks–3 months | 7–14 days |
| Healing with scarring | No | Yes | No |
| Site | Non-keratinized mucosa—especially labial/buccal mucosa; dorsum and lateral borders of the tongue | Keratinized and non-keratinized mucosa, particularly the soft palate | Non-keratinized Mucosa, but Particularly the floor of the mouth and the ventral surface of the tongue |
| Percentage of cases | 85% of all cases | 10–15% of all cases | 5–10% of all cases |
| Author | Year | Index of Improvement |
|---|---|---|
| J. Symoens [15] | 1974 | Reduction in pain, number, frequency |
| J DE Meyer et al. [16] | 1977 | Reduction in number, frequency and duration |
| Van de Heyning [17] | 1978 | Reduction in pain, number, duration |
| Weckx et al. [18] | 2009 | Reduction in size, number, frequency and duration |
| Picciani BLS [5] | 2010 | Reduction in number, frequency and duration |
| Sharda et al. [19] | 2014 | Reduction in pain, number, frequency and duration |
| Bathina P et al. [10] | 2015 | Reduction in pain, number, duration |
| TAR Raja [11] | 2015 | Reduction in pain, number, frequency |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prajapat, J.; Prajapat, R.; Khanagar, S.B.; Vishwanathaiah, S.; Naik, S.; Jhugroo, C.; Devang Divakar, D. Combination of Levamisole with Prednisone in Treating Recurrent Major Aphthous Ulcer in a Young Boy: A Case Report. Clin. Pract. 2021, 11, 250-256. https://doi.org/10.3390/clinpract11020038
Prajapat J, Prajapat R, Khanagar SB, Vishwanathaiah S, Naik S, Jhugroo C, Devang Divakar D. Combination of Levamisole with Prednisone in Treating Recurrent Major Aphthous Ulcer in a Young Boy: A Case Report. Clinics and Practice. 2021; 11(2):250-256. https://doi.org/10.3390/clinpract11020038
Chicago/Turabian StylePrajapat, Jyoti, Rajesh Prajapat, Sanjeev B. Khanagar, Satish Vishwanathaiah, Sachin Naik, Chitra Jhugroo, and Darshan Devang Divakar. 2021. "Combination of Levamisole with Prednisone in Treating Recurrent Major Aphthous Ulcer in a Young Boy: A Case Report" Clinics and Practice 11, no. 2: 250-256. https://doi.org/10.3390/clinpract11020038
APA StylePrajapat, J., Prajapat, R., Khanagar, S. B., Vishwanathaiah, S., Naik, S., Jhugroo, C., & Devang Divakar, D. (2021). Combination of Levamisole with Prednisone in Treating Recurrent Major Aphthous Ulcer in a Young Boy: A Case Report. Clinics and Practice, 11(2), 250-256. https://doi.org/10.3390/clinpract11020038






