The Behavior of Terbuthylazine, Tebuconazole, and Alachlor during Denitrification Process
Abstract
:1. Introduction
2. Materials and Methods
2.1. Principles of the Assays
- Control treatment (C): Denitrification is in progress without disturbance;
- Treatment 1: Denitrification process is affected by the presence of the tested pesticide; at the same time, the conditions for both biotic and abiotic loss of the tested pesticide are created;
- Treatment 2: Biological processes (denitrification and biotic loss of the tested pesticide) are stopped, while abiotic loss of the tested pesticide is in progress.
2.2. Chemicals and Organic Carrier
2.3. Analytical Methods
3. Results and Discussion
3.1. Conditions of Laboratory Denitrification Assays
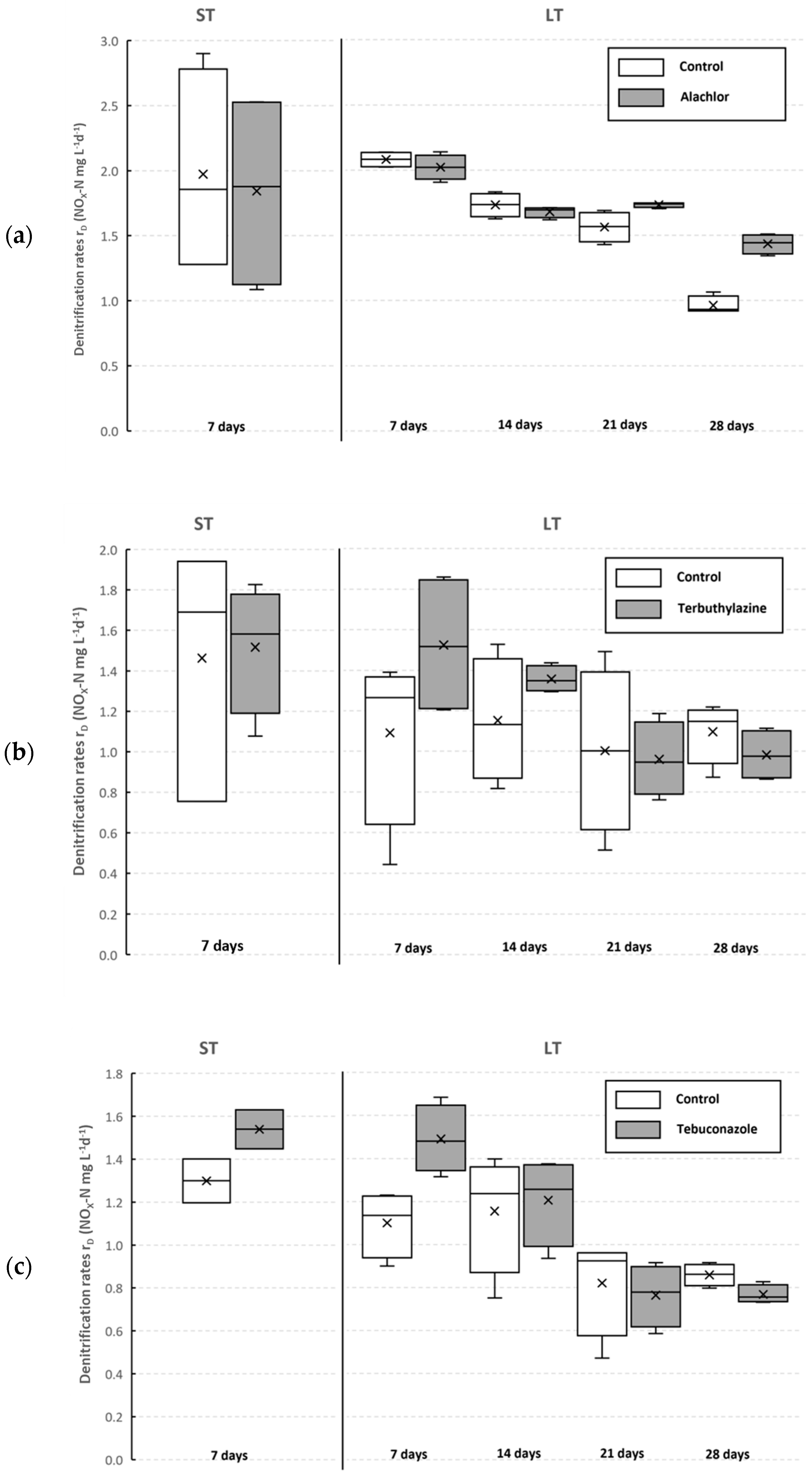
3.2. Inhibition of Denitrification Process
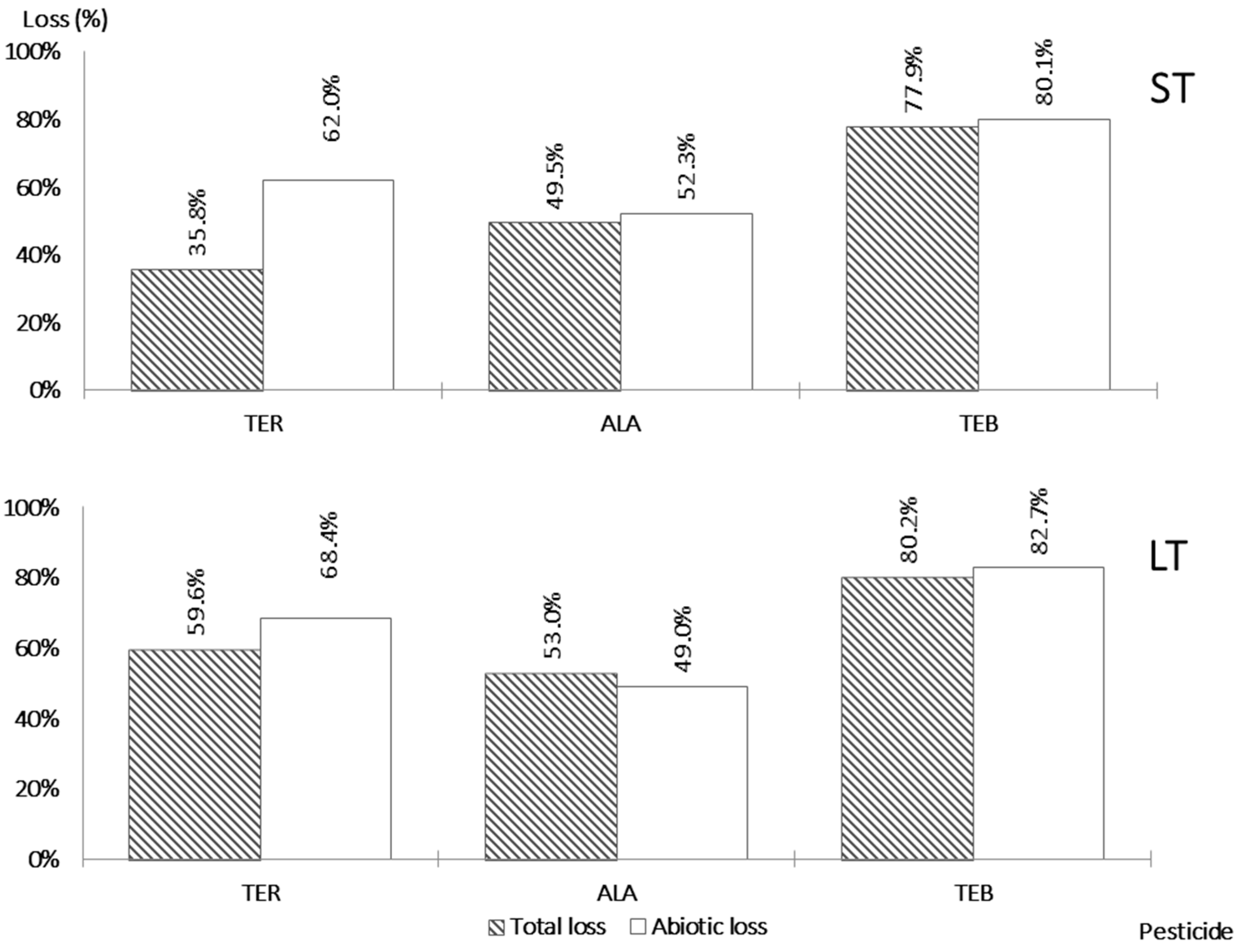
3.3. Removal of the Pesticides during Denitrification Process
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO (Food and Agriculture Organization of the United Nations). Pesticides Use. 2022. Available online: http://www.fao.org/faostat/en/#data/RP/visualize (accessed on 27 August 2023).
- European Commission. Active Substances, Safeners and Synergists. 2023. Available online: https://ec.europa.eu/food/plant/pesticides/eu-pesticides-database/start/screen/active-substances (accessed on 27 August 2023).
- de Souza, R.M.; Seibert, D.; Quesada, H.B.; de Jesus Bassetti, F.; Fagundes-Klen, M.R.; Bergamasco, R. Occurrence, impacts and general aspects of pesticides in surface water: A review. Process Saf. Environ. Prot. 2020, 135, 22–37. [Google Scholar] [CrossRef]
- Pino, N.; Peñuela, G. Simultaneous degradation of the pesticides methyl parathion and chlorpyrifos by an isolated bacterial consortium from a contaminated site. Int. Biodeterior. Biodegrad. 2011, 65, 827–831. [Google Scholar] [CrossRef]
- Schulz, R. Field Studies on Exposure, Effects, and Risk Mitigation of Aquatic Nonpoint-Source Insecticide Pollution: A Review. J. Environ. Qual. 2004, 33, 419–448. [Google Scholar] [CrossRef] [PubMed]
- Caldas, S.S.; Demoliner, A.; Costa, F.P.; D’Oca, M.G.M.; Primel, E.G. Pesticide residue determination in groundwater using solid-phase extraction and high-performance liquid chromatography with diode array detector and liquid chromatography-tandem mass spectrometry. J. Braz. Chem. Soc. 2010, 21, 642–650. [Google Scholar] [CrossRef]
- Nasiri, M.; Ahmadzadeh, H.; Amiri, A. Sample preparation and extraction methods for pesticides in aquatic environments: A review. TrAC Trends Anal. Chem. 2020, 123, 115772. [Google Scholar] [CrossRef]
- Barra Caracciolo, A.; Fajardo, C.; Grenni, P.; SaccÃ, M.L.; Amalfitano, S.; Ciccoli, R.; Martin, M.; Gibello, A. The role of a groundwater bacterial community in the degradation of the herbicide terbuthylazine. FEMS Microbiol. Ecol. 2010, 71, 127–136. [Google Scholar] [CrossRef]
- Kodeš, V. Problematika pesticidů v ochraně vod—Jaká data máme k dispozici a co nám říkají? In Proceedings of the Sborník Přednášek a Posterových Sdělení z 13. Bienální Konference a Výstavy VODA. Poděbrady, Czech Republic, 18–19 September 2019. [Google Scholar]
- Moulisová, A.; Bendakovská, L.; Kožíšek, F.; Vavrouš, A.; Jeligová, H.; Kotal, F. Pesticidy a jejich metabolity v pitné vodě. Vodn. Hospodářství Spec. Vědeckotechnický Časopis Pro Proj. Realiz. Plánování Vodn. Hospodářství Souvisejích Oborech Zivotn. Prostředí 2018, 68, 4–10. [Google Scholar]
- Fenner, K.; Canonica, S.; Wackett, L.P.; Elsner, M. Evaluating Pesticide Degradation in the Environment: Blind Spots and Emerging Opportunities. Science 2013, 341, 752–758. [Google Scholar] [CrossRef]
- Navarro, S.; Vela, N.; José Giménez, M.; Navarro, G. Persistence of four s-triazine herbicides in river, sea and groundwater samples exposed to sunlight and darkness under laboratory conditions. Sci. Total Environ. 2004, 329, 87–97. [Google Scholar] [CrossRef]
- Chen, Z.; Chen, Y.; Vymazal, J.; Kule, L.; Koželuh, M. Dynamics of chloroacetanilide herbicides in various types of mesocosm wetlands. Sci. Total Environ. 2017, 577, 386–394. [Google Scholar] [CrossRef]
- Howarth, R.W. Coastal nitrogen pollution: A review of sources and trends globally and regionally. Harmful Algae 2008, 8, 14–20. [Google Scholar] [CrossRef]
- Ju, X.T.; Zhang, C. Nitrogen cycling and environmental impacts in upland agricultural soils in North China: A review. J. Integr. Agric. 2017, 16, 2848–2862. [Google Scholar] [CrossRef]
- WHO. Guidelines for Drinking-Water Quality, 3rd ed.; WHO: Geneva, Switzerland, 2004. [Google Scholar]
- Capodici, M.; Avona, A.; Laudicina, V.A.; Viviani, G. Biological groundwater denitrification systems: Lab-scale trials aimed at nitrous oxide production and emission assessment. Sci. Total Environ. 2018, 630, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Hiscock, K.M.; Lloyd, J.W.; Lerner, D.N. Review of natural and artificial denitrification of groundwater. Water Res. 1991, 25, 1099–1111. [Google Scholar] [CrossRef]
- Michel, C.; Baran, N.; André, L.; Charron, M.; Joulian, C. Side Effects of Pesticides and Metabolites in Groundwater: Impact on Denitrification. Front. Microbiol. 2021, 12, 662727. [Google Scholar] [CrossRef]
- Pániková, K.; Weigelhofer, G.; Bílková, Z.; Malá, J. Denitrification Assays for Testing Effects of Xenobiotics on Aquatic Denitrification and Their Degradation in Aquatic Environments. Water 2023, 15, 2536. [Google Scholar] [CrossRef]
- Gikas, G.D.; Pérez-Villanueva, M.; Tsioras, M.; Alexoudis, C.; Pérez-Rojas, G.; Masís-Mora, M.; Lizano-Fallas, V.; Rodríguez-Rodríguez, C.E.; Vryzas, Z.; Tsihrintzis, V.A. Low-cost approaches for the removal of terbuthylazine from agricultural wastewater: Constructed wetlands and biopurification system. Chem. Eng. J. 2018, 335, 647–656. [Google Scholar] [CrossRef]
- Elsayed, O.F.; Maillard, E.; Vuilleumier, S.; Millet, M.; Imfeld, G. Degradation of chloroacetanilide herbicides and bacterial community composition in lab-scale wetlands. Sci. Total Environ. 2015, 520, 222–231. [Google Scholar] [CrossRef]
- Colosio, C.; Rubino, F.M.; Moretto, A. Pesticides. In International Encyclopedia of Public Health; Elsevier: Amsterdam, The Netherlands, 2017; pp. 454–462. [Google Scholar] [CrossRef]
- Cao, D.; Wu, R.; Dong, S.; Wang, F.; Ju, C.; Yu, S.; Xu, S.; Fang, H.; Yu, Y. Triazole resistance in Aspergillus fumigatus in crop plant soil after tebuconazole applications. Environ. Pollut. 2020, 266, 115124. [Google Scholar] [CrossRef]
- European Food Safety Authority. Conclusion regarding the peer review of the pesticide risk assessment of the active substance tebuconazole. EFSA Sci. Rep. 2008, 4–6. Available online: https://efsa.onlinelibrary.wiley.com/doi/epdf/10.2903/j.efsa.2008.176r (accessed on 27 August 2023).
- Herrero-Hernández, E.; Andrades, M.S.; Marín-Benito, J.M.; Sánchez-Martín, M.J.; Rodríguez-Cruz, M.S. Field-scale dissipation of tebuconazole in a vineyard soil amended with spent mushroom substrate and its potential environmental impact. Ecotoxicol. Environ. Saf. 2011, 74, 1480–1488. [Google Scholar] [CrossRef]
- Fresenius, W.; Quentin, K.; Schneider, W. Chemical and Microbiological Water Examination and Quality Assurance. In Water Analysis—A Practical Guide to Physico-Chemical, 1st ed.; Springer-Verlag: Berlin Heidelberg, Germany, 1988. [Google Scholar]
- Zhao, J.; He, Q.; Chen, N.; Peng, T.; Feng, C. Denitrification behavior in a woodchip-packed bioreactor with gradient filling for nitrate-contaminated water treatment. Biochemical Engineering Journal 2020, 154. [Google Scholar] [CrossRef]
- Paul, E.A. Soil Microbiology, Ecology, and Biochemistry, 3rd ed.; Elsevier Inc.: Oxford, UK, 2007. [Google Scholar]
- Oh, J.; Silverstein, J.A. Acetate Limitation and Nitrite Accumulation during Denitrification. J. Environ. Eng. 1999, 125, 234–242. [Google Scholar] [CrossRef]
- Hu, R.; Zheng, X.; Zheng, T.; Xin, J.; Wang, H.; Sun, Q. Effects of carbon availability in a woody carbon source on its nitrate removal behavior in solid-phase denitrification. J. Environ. Manag. 2019, 246, 832–839. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Ma, B.; Li, X.; Zhang, Q.; Peng, Y. Long-term effect of pH on denitrification: High pH benefits achieving partial-denitrification. Bioresour. Technol. 2019, 278, 444–449. [Google Scholar] [CrossRef]
- Chung, J.; Bae, W. Nitrite reduction by a mixed culture under conditions relevant to shortcut biological nitrogen removal. Biodegradation 2002, 13, 163–170. [Google Scholar] [CrossRef]
- Lahdhiri, A.; Lesage, G.; Hannachi, A.; Heran, M. Minimum COD needs for denitrification: From biological models to experimental set-up. Desalination Water Treat. 2017, 61, 326–334. [Google Scholar] [CrossRef]
- Novak, P.J.; Christ, S.J.; Parkin, G.F. Kinetics of alachlor transformation and identification of metabolites under anaerobic conditions. Water Res. 1997, 31, 3107–3115. [Google Scholar] [CrossRef]
- Pozo, C.; Salmeron, V.; Rodelas, B.; Martinez-Toledo, M.V.; Gonzalez-Lopez, J. Effects of the herbicide alachlor on soil microbial activities. Ecotoxicology 1994, 3, 4–10. [Google Scholar] [CrossRef]
- Cycoń, M.; Piotrowska-Seget, Z.; Kaczyńska, A.; Kozdrój, J. Microbiological characteristics of a sandy loam soil exposed to tebuconazole and λ-cyhalothrin under laboratory conditions. Ecotoxicology 2006, 15, 639–646. [Google Scholar] [CrossRef]
- Graham, D.W.; Miley, M.K.; deNoyelles, F.; Smith, V.H.; Thurman, E.M.; Carter, R. Alachlor transformation patterns in aquatic field mesocosms under variable oxygen and nutrient conditions. Water Res. 2000, 34, 4054–4062. [Google Scholar] [CrossRef]
- Dehghani, M.; Nasseri, S.; Zamanian, Z. Biodegradation of alachlor in liquid and soil cultures under variable carbon and nitrogen sources by bacterial consortium isolated from corn field soil. Iran. J. Environ. Health Sci. Eng. 2013, 10, 21. [Google Scholar] [CrossRef] [PubMed]
- Čadková, E.; Komárek, M.; Kaliszová, R.; Vaněk, A.; Balíková, M. Tebuconazole Sorption in Contrasting Soil Types. Soil Sediment Contam. Int. J. 2013, 22, 404–414. [Google Scholar] [CrossRef]
- Ilhan, Z.E.; Ong, S.K.; Moorman, T.B. Dissipation of Atrazine, Enrofloxacin, and Sulfamethazine in Wood Chip Bioreactors and Impact on Denitrification. J. Environ. Qual. 2011, 40, 1816–1823. [Google Scholar] [CrossRef] [PubMed]
- Krause Camilo, B. Bioreactor reduces atrazine and nitrate in tile drain waters. Ecol. Eng. 2016, 86, 269–278. [Google Scholar] [CrossRef]
- Ahmad, K.S. Evaluating the Adsorption Potential of Alachlor and Its Subsequent Removal from Soils via Activated Carbon. Soil Sediment Contam. Int. J. 2018, 27, 249–266. [Google Scholar] [CrossRef]

| Wood Shavings | Initial Liquid Medium, Solution in DIW | Treatment | Additional Reagents Added after 48 h | |
|---|---|---|---|---|
| c (NOX-N) | c (NaHCO3) | |||
| 25 g per bottle (COD > 100 mg L−1) | ST 30 mg L−1 LT 15 mg L−1 | 0.5 g L−1 | C | 0.1 mL pure methanol per L sample |
| 1 | 0.1 mL of the tested pesticide per L sample (conc. 1000 mg L−1 in pure methanol solution) | |||
| 2 | 0.1 mL of the tested pesticide per L sample (conc. 1000 mg L−1 in pure methanol solution) + 3.8 mL HgCl2 (123.5 mg L−1) | |||
| Parameter | pH (-) | DO (mg L−1) | NO2-N (mg L−1) | COD (mg L−1) | |||||
|---|---|---|---|---|---|---|---|---|---|
| Treatment | C | 1 | C | 1 | C | 1 | C | 1 | |
| TER | ST | 7.26 ± 0.03 | 7.20 ± 0.11 | 0.45 ± 0.05 | 0.33 ± 0.07 | 0.174 ± 0.090 | 0.227 ± 0.015 | 260 ± 33 | 260 ± 29 |
| LT | 7.70 ± 0.13 | 7.78 ± 0.09 | 1.51 ± 0.79 | 2.50 ± 1.13 | 0.050 ± 0.004 | 0.076 ± 0.012 | 130 ± 18 | 120 ± 25 | |
| ALA | ST | 7.51 ± 0.06 | 7.41 ± 0.17 | 0.39 ± 0.07 | 0.29 ± 0.02 | 3.596 ± 3.576 | 6.119 ± 2.165 | 290 ± 22 | 290 ± 41 |
| LT | 7.71 ± 0.11 | 7.71 ± 0.14 | 1.89 ± 0.63 | 1.07 ± 0.27 | 0.120 ± 0.005 | 0.120 ± 0.006 | 110 ± 17 | 120 ± 19 | |
| TEB | ST | 7.47 ± 0.05 | 7.48 ± 0.01 | 0.49 ± 0.08 | 0.35 ± 0.02 | 1.527 ± 1.421 | 6.528 ± 2.506 | 220 ± 0 | 270 ± 45 |
| LT | 7.73 ± 0.08 | 7.77 ± 0.06 | 0.91 ± 0.22 | 1.89 ± 0.33 | 0.057 ± 0.022 | 0.062 ± 0.006 | 130 ± 11 | 100 ± 8 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pániková, K.; Bílková, Z.; Malá, J. The Behavior of Terbuthylazine, Tebuconazole, and Alachlor during Denitrification Process. J. Xenobiot. 2023, 13, 560-571. https://doi.org/10.3390/jox13040036
Pániková K, Bílková Z, Malá J. The Behavior of Terbuthylazine, Tebuconazole, and Alachlor during Denitrification Process. Journal of Xenobiotics. 2023; 13(4):560-571. https://doi.org/10.3390/jox13040036
Chicago/Turabian StylePániková, Kristína, Zuzana Bílková, and Jitka Malá. 2023. "The Behavior of Terbuthylazine, Tebuconazole, and Alachlor during Denitrification Process" Journal of Xenobiotics 13, no. 4: 560-571. https://doi.org/10.3390/jox13040036
APA StylePániková, K., Bílková, Z., & Malá, J. (2023). The Behavior of Terbuthylazine, Tebuconazole, and Alachlor during Denitrification Process. Journal of Xenobiotics, 13(4), 560-571. https://doi.org/10.3390/jox13040036






