Audiometric Characteristics and Tinnitus Features in a Cohort of 170 Spanish Patients
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Audiometric Measurements
- 1.
- Hearing impaired (HI) subgroup: subjects with any (HL(fi)) ≥ 40, AAT ≥ 30, or
- 2.
- Normal hearing (NH) subgroup: all other subjects.
2.3. Tinnitus Assessment
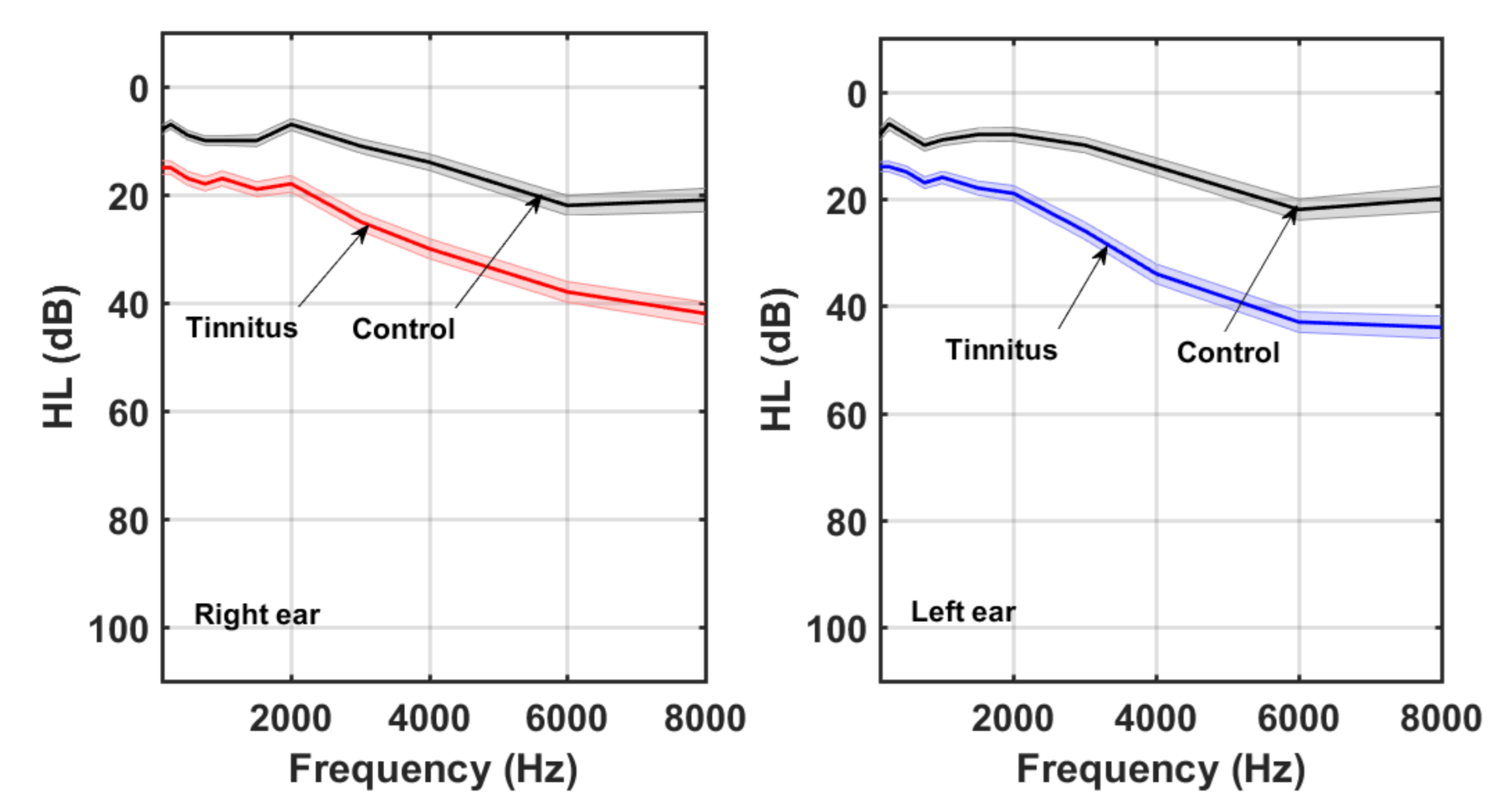
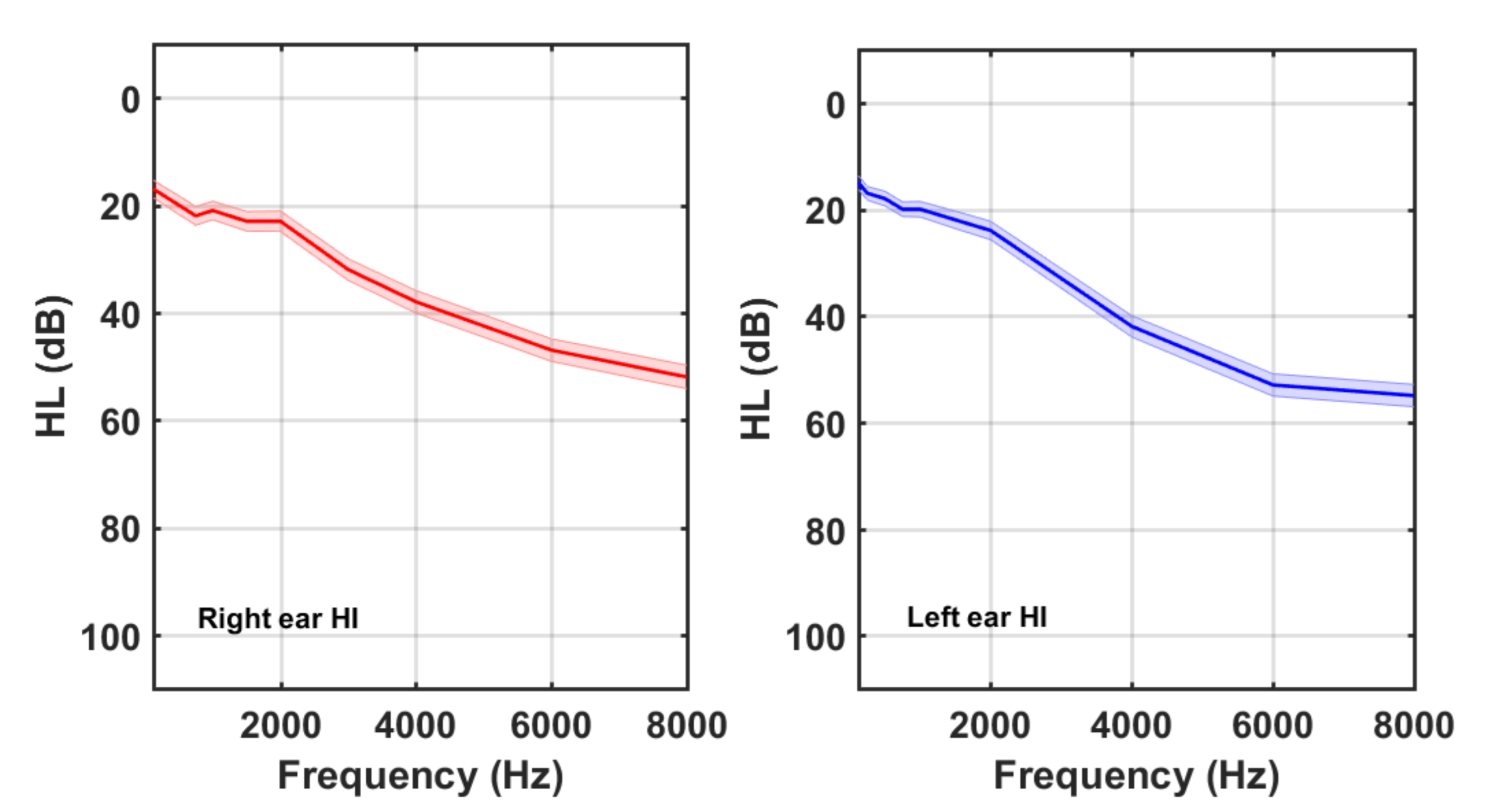
3. Results
4. Discussion
5. Conclusions
- In this cohort, 35% of subjects allocated their tinnitus to the left ear, and 16% assigned their tinnitus to the right ear, whilst, in 49% of subjects, the tinnitus was bilateral.
- The most frequent tinnitus sound was hissing (36%), followed by tonal (34%) and ringing (30%).
- The predominant tinnitus etiology (35%) was emotional troubles (stress, depression, anxiety, and obsessive–compulsive disorder), followed by HL (25%), overexposure to noise (15%), tube dysfunction (5%), ear surgery (4%), idiopathic (4%), and others (12%, including head trauma, ototoxicity, otitis, rhinitis and sinusitis, barotrauma, cervical troubles, Meniere, and hydrocephaly).
- The subjective outcome of tinnitus distress (THI) was not correlated with the objective measure of hearing loss (AAT), whether for the HI or NH subgroups. Therefore, it seems that hearing loss can become a risk factor for triggering tinnitus, although the grade of tinnitus-related distress may be more determined by emotional aspects of the patient response.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jastreboff, P.J. Phantom auditory perception (Tinnitus): Mechanisms of generation and perception. Neurosci. Res. 1990, 8, 221–254. [Google Scholar] [CrossRef]
- Eggermont, J.J.; Tass, P.A. Maladaptive neural synchrony in tinnitus: Origin and restoration. Front. Neurol. 2015, 6, 29. [Google Scholar] [CrossRef] [Green Version]
- McFerran, D.J.; Stockdale, D.; Holme, R.; Large, C.H.; Baguley, D.M. Why is there no cure for tinnitus? Front Neurosci. 2019, 13, 802. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van de Heyning, P.; Meeus, O.; Blaivie, C.; Vermeire, K.; Boudewyns, A.; De Ridder, D. Tinnitus: A multidisciplinary clinical approach. B-ENT 2007, 3, 3–10. [Google Scholar] [PubMed]
- Cederroth, C.R.; Gallus, S.; Hall, D.; Kleinjung, T.; Langguth, B.; Maruotti, A. Editorial: Towards an understanding of tinnitus heterogeneity. Front. Aging. Neurosci. 2019, 11, 53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Searchfield, G.D.; Durai, M.; Linford, T. A state-of-the-art review: Personalization of tinnitus sound therapy. Front. Psychol. 2017, 8, 1599. [Google Scholar] [CrossRef] [Green Version]
- Pienkowski, M. Rationale and efficacy of sound therapies for tinnitus and hyperacusis. Neuroscience 2019, 407, 120–134. [Google Scholar] [CrossRef]
- Cima, R.F.; Maes, I.H.; Joore, M.A.; Scheyen, D.J.; El Refaie, A.; Baguley, D.M.; Anteunis, L.J.; van Breukelen, G.J.; Vlaeyen, J.W. Specialised treatment based on cognitive behaviour therapy versus usual care for tinnitus: A randomised controlled trial. Lancet 2012, 379, 1951–1959. [Google Scholar] [CrossRef]
- Theodoroff, S.M.; Griest, S.E.; Folmer, R.L. Transcranial magnetic stimulation for tinnitus: Using the Tinnitus Functional Index to predict benefit in a randomized controlled trial. Trials 2017, 18, 64. [Google Scholar] [CrossRef] [Green Version]
- Jastreboff, P.J. 25 years of tinnitus retraining therapy. HNO 2015, 63, 307–311. [Google Scholar] [CrossRef]
- Marks, K.L.; Martel, D.T.; Wu, C.; Basura, G.J.; Roberts, L.E.; Schvartz-Leyzac, K.C.; Shore, S.E. Auditory-somatosensory bimodal stimulation desynchronizes brain circuitry to reduce tinnitus in guinea pigs and humans. Sci. Transl. Med. 2018, 10, eaal3175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eggermont, J.J. Noise and the Brain; Academic Press: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Eggermont, J.J. The Neuroscience of Tinnitus; Oxford University Press: Oxford, UK, 2012. [Google Scholar]
- Lindblad, A.C.; Rosenhall, U.; Oloffson, A.; Hagerman, B. Tinnitus and other auditory problems–Occupational noise exposure below risk limits may cause inner ear dysfunction. PLoS ONE 2014, 9, e97377. [Google Scholar] [CrossRef] [Green Version]
- Weisz, N.; Hartmann, T.; Dohrmann, K.; Schlee, W.; Noreña, A. High-frequency tinnitus without hearing loss does not mean absence of deafferentation. Hear Res. 2006, 222, 108–114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liberman, M.C.; Kujawa, S.G. Cochlear synaptopathy in acquired sensorineural hearing loss: Manifestations and mechanisms. Hear Res. 2017, 349, 138–147. [Google Scholar] [CrossRef]
- Xiong, B.; Liu, Z.; Liu, Q.; Peng, Y.; Wu, H.; Lin, Y.; Zhao, X.; Sun, W. Missed hearing loss in tinnitus patients with normal audiograms. Hear Res. 2019, 384, 107826. [Google Scholar] [CrossRef] [PubMed]
- Cuesta, M.; Cobo, P. Relating tinnitus features and audiometric characteristics in a cohort of 34 tinnitus subjects. Loquens 2018, 5, e054. [Google Scholar] [CrossRef] [Green Version]
- Herráiz, C.; Hernández Calvín, F.J.; Plaza, G.; Tapia, M.C.; De los Santos, G. Evaluación de la incapacidad en los pacientes con acúfenos (Evaluation of handicap in tinnitus patients). Acta Otorrinolaringol. Esp. 2001, 52, 142–145. [Google Scholar] [CrossRef]
- Diges, I.; Simón, F.; Cobo, P. Assessing auditory processing deficits in tinnitus and hearing impaired patients with the Auditory Behavior Questionnaire. Front. Neurosci. 2017, 11, 187. [Google Scholar] [CrossRef] [Green Version]
- Manche, S.K.; Madhavi, J.; Meganadh, K.R.; Jyothi, A. Association of tinnitus and hearing loss in otological disorders: A decade-long epidemiological study in a South Indian population. Braz. J. Otorhinolaryngol. 2016, 82, 643–649. [Google Scholar] [CrossRef] [Green Version]
- Wallhäusser-Franke, E.; D’Amelio, R.; Glauner, A.; Delb, W.; Servais, J.J.; Hörmann, K.; Repik, I. Transition from acute to chronic Tinnitus: Predictors for the Development of chronic Distressing Tinnitus. Front. Neurol. 2017, 8, 605. [Google Scholar] [CrossRef] [Green Version]
- Chalder, T.; Willis, C. ‘‘Lumping’’ and ‘‘splitting’’ medically unexplained symptoms: Is there a role for a transdiagnostic approach? J. Ment. Health 2017, 26, 187–191. [Google Scholar] [CrossRef]
- Schecklmann, M.; Vielsmeier, V.; Steffens, T.; Landgrebe, M.; Langguth, B.; Kleinjung, T. Relationship between audiometric slope and tinnitus pitch in tinnitus patients: Insights into the mechanisms of tinnitus generation. PLoS ONE 2012, 7, e34878. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, A.; Munjal, S.; Panda, N.; Mohanty, M. Demographic variations in tinnitus subjects with and without hearing loss: A study of 175 subjects. Int. Tinnitus J. 2018, 22, 77–83. [Google Scholar] [CrossRef] [Green Version]
- Shekhawat, G.S.; Searchfield, G.D.; Stinear, C.M. The relationship between tinnitus pitch and hearing sensitivity. Eur. Arch. Otolaryngol. 2014, 271, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Available online: http://www.tinnitusarchive.org/dataSets/1/tinnitusAttribute/predominantTinnitusSounds/index.html (accessed on 18 June 2021).
- Hébert, S.; Lupien, S.J. The sound of stress: Blunted cortisol reactivity to psychosocial stress in tinnitus sufferers. Neuroscience Letters 2007, 411, 138–142. [Google Scholar] [CrossRef]
- Pinto, P.C.L.; Sanchez, T.G.; Tomita, S. The impact of gender, age and hearing loss on tinnitus severity. Braz. J. Otorhinolaryngol. 2010, 76, 18–24. [Google Scholar] [CrossRef] [Green Version]
- Martines, F.; Bentivegna, D.; Martines, E.; Scciaca, V.; Martinciglio, G. Assessing audiological, pathophysiological and psychological variables in tinnitus patients with or without hearing loss. Eur. Arch. Otorhinolaryngol. 2010, 267, 1685–1693. [Google Scholar] [CrossRef]
- Weidt, S.; Delsignore, A.; Meyer, M.; Rufer, M.; Peter, N.; Drabe, N.; Kleinjung, T. Which tinnitus-related characteristics affect current health-related quality of life and depression? A cross-sectional cohort study. Psychiatry Res. 2016, 237, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Aazh, H.; Salvi, R. The relationship between severity of hearing loss and subjective tinnitus loudness among patients seen in a specialist tinnitus and hyperacusis therapy clinic in UK. J. Am. Acad. Audiol. 2019, 30, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Mores, J.T.; Bozza, A.; Magni, C.; Casali, R.L.; Ramos do Amoral, M.I. Clinical profile and implications of tinnitus in individuals with and without hearing loss. Codas 2019, 31, e20180029. [Google Scholar] [CrossRef] [PubMed]
- Savastano, M. Tinnitus with or without hearing loss: Are its characteristics different? Eur. Arch. Otorhinolaryngol. 2008, 265, 1295–1300. [Google Scholar] [CrossRef] [PubMed]
- Nicolas-Puel, C.; Faulconbridge, R.L.; Guitton, M.; Puel, J.L.; Mondain, M.; Uziel, A. Characteristics of tinnitus and etiology of associated hearing loss: A study of 123 patients. Int. Tinnitus J. 2002, 8, 37–44. [Google Scholar]
- Biehl, R.; Boecking, B.; Brueggemann, P.; Grosse, R.; Mazurek, B. Personality Traits, Perceived Stress, and Tinnitus-Related Distress in Patients With Chronic Tinnitus: Support for a Vulnerability-Stress Model. Front. Psychol. 2020, 10, 3093. [Google Scholar] [CrossRef] [PubMed]
- Niemann, U.; Boecking, B.; Brueggemann, P.; Mebus, W.; Mazurek, B.; Spiliopoulou, M. Tinnitus-related distress after multimodal treatment can be characterized using a key subset of baseline variables. PLoS ONE 2020, 15, e0228037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hiller, W.; Janca, A.; Buerke, K.C. Association between tinnitus and somatoform disorders. J. Psychosom. Res. 1997, 43, 613–624. [Google Scholar] [CrossRef]
- Hiller, W.; Goebel, G.; Svitak, M.; Schätz, M.; Janca, A. Association between tinnitus and the diagnostic concept of somatoform disorders. In Proceedings of the Sixth International Tinnitus Seminar, Cambridge, UK, 5–9 September 1999; The Tinnitus and Hyperacusis Center: London, UK, 1999; pp. 373–377. [Google Scholar]
- Boecking, B.; Biehl, R.; Brueggemann, P.; Mazurek, B. Health-Related Quality of Life, Depressive Symptoms, Anxiety, and Somatization Symptoms in Male and Female Patients with Chronic Tinnitus. J. Clin. Med. 2021, 10, 2798. [Google Scholar] [CrossRef] [PubMed]


| Tinnitus Group | Control Group | |||||
|---|---|---|---|---|---|---|
| Total | Males | Females | Total | Males | Females | |
| N | 170 | 110 | 60 | 85 | 38 | 47 |
| Age (mean) | 50 | 51 | 49 | 44 | 47 | 43 |
| Age (SD) | 11 | 11 | 10 | 13 | 13 | 12 |
| NH Subgroup | HI Subgroup | |
|---|---|---|
| Total | 49 (29%) | 121 (71%) |
| Males | 26 (53%) | 84 (69%) |
| Females | 23 (47%) | 37 (31%) |
| Total | NH Subgroup | HI Subgroup | ||
|---|---|---|---|---|
| Duration, in months (mean, SD) | 75, 102 | 66, 110 | 79, 99 | |
| Pitch, in Hz (mean, SD) | 5200, 3016 | 5085, 3764 | 5262, 2670 | |
| THI, % (mean, SD) | 47, 25 | 45, 26 | 47, 25 | |
| Lateralization | Bilateral, N (%) | 84 (49%) | 26 (53%) | 59 (49%) |
| Left ear, N (%) | 59 (35%) | 17 (35%) | 42 (35%) | |
| Right ear, N (%) | 27 (16%) | 6 (12%) | 20 (16%) | |
| Tinnitus sound | Hissing, N (%) | 62 (36%) | 16 (33%) | 47 (39%) |
| Ringing, N (%) | 50 (30%) | 16 (33%) | 34 (28%) | |
| Tonal, N (%) | 58 (34%) | 17 (34%) | 40 (33%) | |
| AAT (All) | AAT (NH) | AAT (HI) | ||||
|---|---|---|---|---|---|---|
| Left Ear | Right Ear | Left Ear | Right Ear | Left Ear | Right Ear | |
| THI (All) | 0.06 | 0.094 | ||||
| THI (NH) | −0.078 | −0.06 | ||||
| THI (HI) | 0.057 | 0.10 | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cuesta, M.; Cobo, P. Audiometric Characteristics and Tinnitus Features in a Cohort of 170 Spanish Patients. Audiol. Res. 2021, 11, 594-602. https://doi.org/10.3390/audiolres11040053
Cuesta M, Cobo P. Audiometric Characteristics and Tinnitus Features in a Cohort of 170 Spanish Patients. Audiology Research. 2021; 11(4):594-602. https://doi.org/10.3390/audiolres11040053
Chicago/Turabian StyleCuesta, María, and Pedro Cobo. 2021. "Audiometric Characteristics and Tinnitus Features in a Cohort of 170 Spanish Patients" Audiology Research 11, no. 4: 594-602. https://doi.org/10.3390/audiolres11040053
APA StyleCuesta, M., & Cobo, P. (2021). Audiometric Characteristics and Tinnitus Features in a Cohort of 170 Spanish Patients. Audiology Research, 11(4), 594-602. https://doi.org/10.3390/audiolres11040053







