Phenazopyridine-Induced Methemoglobinemia in a Jehovah’s Witness Treated with High-Dose Ascorbic Acid Due to Methylene Blue Contradictions: A Case Report and Review of the Literature
Abstract
1. Introduction
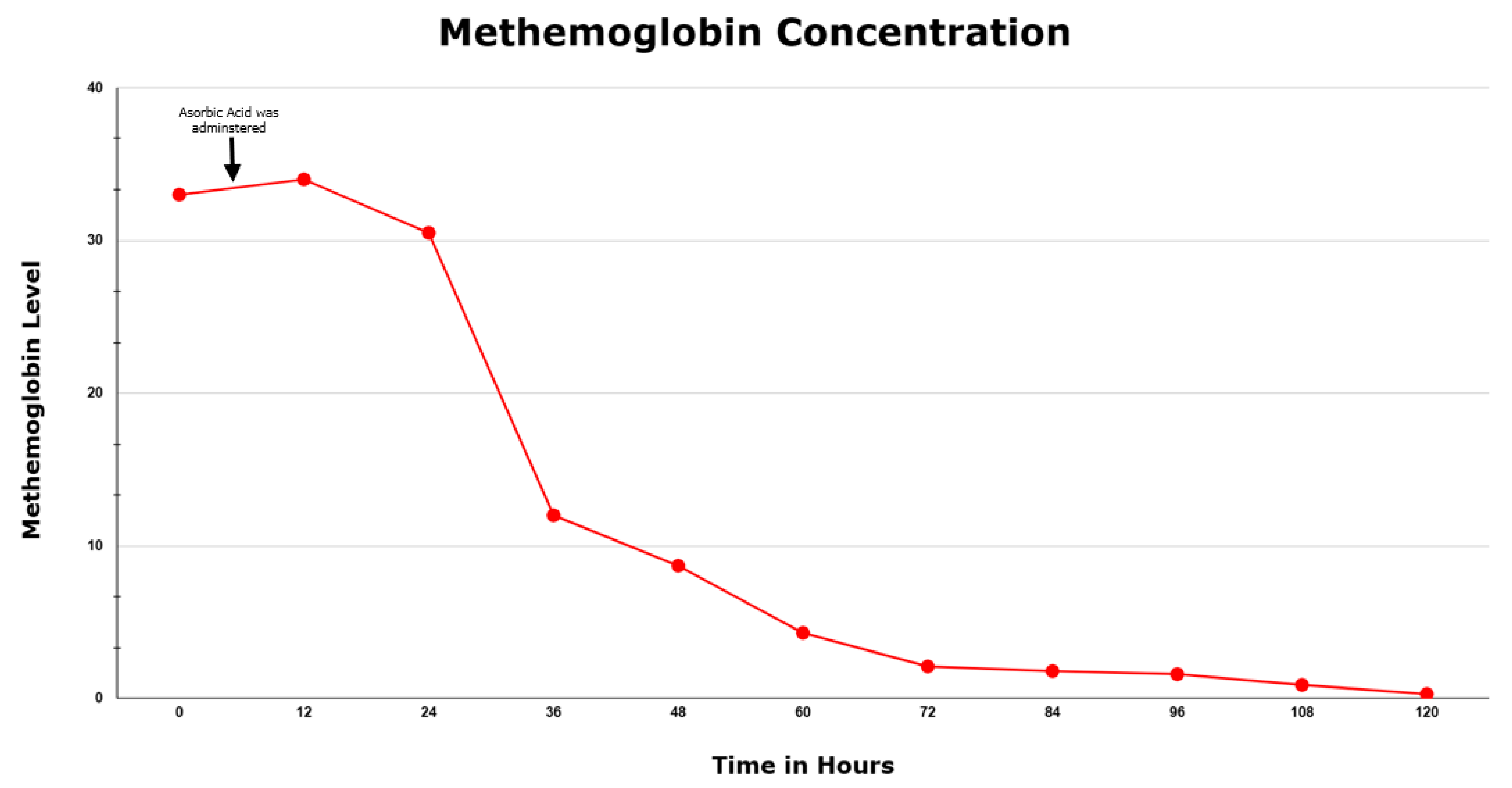
2. Case Report
3. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Iolascon, A.; Bianchi, P.; Andolfo, I.; Russo, R.; Barcellini, W.; Fermo, E.; Toldi, G.; Ghirardello, S.; Rees, D.; Van Wijk, R.; et al. Recommendations for diagnosis and treatment of methemoglobinemia. Am. J. Hematol. 2021, 96, 1666–1678. [Google Scholar] [CrossRef] [PubMed]
- Ludlow, J.T.; Wilkerson, R.G.; Nappe, T.M. Methemoglobinemia. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK537317/ (accessed on 17 December 2022).
- Crowley, M.A.; Mollan, T.L.; Abdulmalik, O.Y.; Butler, A.D.; Goodwin, E.F.; Sarkar, A.; Stolle, C.A.; Gow, A.J.; Olson, J.S.; Weiss, M.J. A hemoglobin variant associated with neonatal cyanosis and anemia. N. Engl. J. Med. 2011, 364, 1837. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.M.; Prchal, J.T. Methemoglobinemia and other dyshemoglobinemias. In Williams Hematology, 9e; Kaushansky, K., Lichtman, M.A., Prchal, J.T., Levi, M.M., Press, O.W., Burns, L.J., Caligiuri, M., Eds.; McGraw Hill: New York, NY, USA, 2015; p. 789. [Google Scholar]
- Wright, R.O.; Lewander, W.J.; Woolf, A.D. Methemoglobinemia: Etiology, pharmacology, and clinical management. Ann. Emerg. Med. 1999, 34, 646–656. [Google Scholar] [CrossRef] [PubMed]
- Wilkerson, R.G. Getting the blues at a rock concert: A case of severe methaemoglobinaemia. Emerg. Med. Australas. 2010, 22, 466–469. [Google Scholar] [CrossRef]
- Murphy, T.; Fernandez, M. Acquired methemoglobinemia from phenazopyridine use. Int. J. Emerg. Med. 2018, 11, 45. [Google Scholar] [CrossRef]
- Shahani, L.; Sattovia, S. Acquired methaemoglobinaemia related to phenazopyridine ingestion. BMJ Case Rep. 2012, 2012, 1–2. [Google Scholar] [CrossRef]
- Yu, C.H.; Wang, C.H.; Chang, C.C. Chocolate-colored blood with normal artery oxygen: Methemoglobinemia related to phenazopyridine. Am. J. Med. Sci. 2011, 341, 337. [Google Scholar] [CrossRef]
- Kc, O.; Subedi, A.; Sharma, R.; Dahal, P.H.; Koirala, M. A Case of Severe Hypoxia Caused by Phenazopyridine-Induced Methemoglobinemia: A near Fatal Event from Over-the-Counter Medication Use. Clin. Pract. 2022, 12, 845–851. [Google Scholar] [CrossRef]
- Hamza, A.; Nasrullah, A.; Singh, R.; DiSilvio, B. Phenazopyridine-Induced Methaemoglobinaemia The Aftermath of Dysuria Treatment. Eur. J. Case Rep. Intern. Med. 2022, 9, 003191. [Google Scholar] [CrossRef]
- Shah, P.K.; Kraft, K.H. Perioperative Hypoxia Secondary to Phenazopyridine-induced Methemoglobinemia in an Adolescent Patient Without Renal Insufficiency or Overdose: An Unusual Case. Urology 2019, 130, 142–143. [Google Scholar] [CrossRef]
- Kozik, S.; Kirkham, C.; Sudario, G. Acquired Methemoglobinemia in a Ketamine-induced Ulcerative Cystitis Patient: A Case Report. Clin. Pract. Cases Emerg. Med. 2022, 6, 137–140. [Google Scholar] [CrossRef] [PubMed]
- Chiu, D.; Lubin, B. Oxidative hemoglobin denaturation and RBC destruction: The effect of heme on red cell membranes. Semin. Hematol. 1989, 26, 128. [Google Scholar] [PubMed]
- Abe, K.; Sugita, Y. Properties of cytochrome b5 and methemoglobin reduction in human erythrocytes. Eur. J. Biochem. 1979, 101, 423–428. [Google Scholar] [CrossRef] [PubMed]
- Yubisui, T.; Takeshita, M.; Yoneyama, Y. Reduction of methemoglobin through flavin at the physiological concentration by NADPH-flavin reductase of human erythrocytes. J. Biochem. 1980, 87, 1715. [Google Scholar] [CrossRef] [PubMed]
- Rosen, P.J.; Johnson, C.; McGehee, W.G.; Beutler, E. Failure of methylene blue treatment in toxic methemoglobinemia. Association with glucose-6-phosphate dehydrogenase deficiency. Ann. Intern. Med. 1971, 75, 83. [Google Scholar] [CrossRef]
- Rehman, A.; Shehadeh, M.; Khirfan, D.; Jones, A. Severe acute haemolytic anaemia associated with severe methaemoglobinaemia in a G6PD-deficient man. BMJ Case Rep. 2018, 2018, bcr-2017. [Google Scholar] [CrossRef] [PubMed]
- Cheah, C.Y.; Lew, T.E.; Seymour, J.F.; Burbury, K. Rasburicase causing severe oxidative hemolysis and methemoglobinemia in a patient with previously unrecognized glucose-6-phosphate dehydrogenase deficiency. Acta Haematol. 2013, 130, 254–259. [Google Scholar] [CrossRef]
- El Hamzaoui, H.; Chajai, I.; El Ouazzani, M.C.; Benhalima, A.; El Arfaoui, M.; Alilou, M. Acute dapsone poisoning with methemoglobinemia: A case report. Pan Afr. Med. J. 2022, 43, 20. [Google Scholar] [CrossRef]
- Kabir, H.; Lakshmanan, R.; Gopinath, S.; Bhonagiri, D. Dapsone-induced methemoglobinemia-A case report. Clin. Case Rep. 2021, 9, e04054. [Google Scholar] [CrossRef]
- Reeves, D.J.; Saum, L.M.; Birhiray, R.I.V. ascorbic acid for treatment of apparent rasburicase-induced methemoglobinemia in a patient with acute kidney injury and assumed glucose-6-phosphate dehydrogenase deficiency. Am. J. Health Syst. Pharm. 2016, 73, e238–e242. [Google Scholar] [CrossRef]
- Sahu, K.K.; Dhibar, D.P.; Gautam, A.; Kumar, Y.; Varma, S.C. Role of ascorbic acid in the treatment of methemoglobinemia. Turk. J. Emerg. Med. 2016, 16, 119–120. [Google Scholar] [CrossRef] [PubMed]
- De Crem, N.; Verleden, G.M.; Godinas, L.; Vos, R. Once in a blue moon: Primaquine-induced methemoglobinemia—A case report. Respir. Med. Case Rep. 2022, 38, 101675. [Google Scholar] [CrossRef]
- Topal, H.; Topal, Y. Toxic methemoglobinemia treated with ascorbic Acid: Case report. Iran. Red. Crescent. Med. J. 2013, 15, e12718. [Google Scholar] [CrossRef] [PubMed]
- Powell, T.G.; Choi, E.; Vuppala, S.; Hayek, H. Lava Lamp-Induced Methemoglobinemia in Dementia: A Case Report. Cureus 2022, 14, e29441. [Google Scholar] [CrossRef] [PubMed]
- Asif, A.; Preetham, C.; Mahajyoti, C.; Nibedita, M. Clofazimine-induced methemoglobinemia: A rare incidence. J. Fam. Med. Prim. Care 2018, 7, 1573–1575. [Google Scholar] [CrossRef]
- Kilicli, E.; Aksel, G.; Akbuga Ozel, B.; Kavalci, C.; Suveren Artuk, D. Cost of Beauty; Prilocaine Induced Methemoglobinemia. Turk. J. Emerg. Med. 2016, 14, 185–187. [Google Scholar] [CrossRef]
- Park, S.Y.; Lee, K.W.; Kang, T.S. High-dose vitamin C management in dapsone-induced methemoglobinemia. Am. J. Emerg. Med. 2014, 32, 684.e1. [Google Scholar] [CrossRef]
- Singh, P.; Rakesh, K.; Agarwal, R.; Tripathi, P.P.; Dhooria, S.; Sehgal, I.S.; Prasad, K.T.; Hans, R.; Sharma, R.; Sharma, N.; et al. Therapeutic whole blood exchange in the management of methaemoglobinemia: Case series and systematic review of literature. Transfus. Med. 2020, 30, 231. [Google Scholar] [CrossRef]
- Cho, Y.; Park, S.W.; Han, S.-K.; bin Kim, H.; Yeom, S.R. A Case of Methemoglobinemia Successfully Treated with Hyperbaric Oxygenation Monotherapy. J. Emerg. Med. 2017, 53, 685. [Google Scholar] [CrossRef]
| Author | Cause of Methemoglobinemia | Initial Methemoglobinemia Percentage | Ascorbic Acid Dosing and Treatment | Why Methylene Blue Was Not Utilized |
|---|---|---|---|---|
| Hamzaoui et al. [21] | Dapsone | 13.70% | 1 g of oral ascorbic acid every 12 h for 2 days and a loading dose of 50 g of activated charcoal orally, followed by 25 g every 6 h orally | It was not available |
| Kabir et al. [22] | Dapsone | 17.70% | 10 g of IV ascorbic acid every 6 h for 4 days, and was then switched to oral vitamin C | Unknown glucose-6-phosphate dehydrogenase status |
| Reeves et al. [23] | Rasburicase-induced | 14.50% | 5 g of IV ascorbic acid every 6 h for 3 days | Glucose-6-phosphate dehydrogenase deficiency |
| Sahu et al. [24] | Dapsone | 18.30% | 1 g of IV ascorbic acid every 12 h for 7 days | It was not available |
| De Crem et al. [25] | Primaquine | 33.70% | 1 g of IV ascorbic acid 4 times daily for 7 days | Patient was taking trazodone |
| Topal et al. [26] | Pilocarpine | 24.50% | 3 g of IV ascorbic acid over the course of 24 h | It was not available |
| Powell et al. [27] | Lava lamp poisoning | Over 30% | 5 g of IV ascorbic acid over the course of 24 h | Patient was taking trazodone and duloxetine |
| Asif et al. [28] | Clofazimine | 26.70% | 0.5 g of oral ascorbic acid every 6 h (2000 mg/day) and 600 mg of oral N-acetylcysteine 600 mg every 8 h | It was not available |
| Kilicli et al. [29] | Prilocaine | 14.10% | 3 g of IV ascorbic acid over the course of 24 h | It was not available |
| Menakuru et al. | Phenazopyridine | 33.0% | 5 g of IV ascorbic acid every 12 h for 2.5 days | Patient was taking citalopram |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Menakuru, S.R.; Dhillon, V.S.; Atta, M.; Mann, K.; Salih, A. Phenazopyridine-Induced Methemoglobinemia in a Jehovah’s Witness Treated with High-Dose Ascorbic Acid Due to Methylene Blue Contradictions: A Case Report and Review of the Literature. Hematol. Rep. 2023, 15, 325-330. https://doi.org/10.3390/hematolrep15020034
Menakuru SR, Dhillon VS, Atta M, Mann K, Salih A. Phenazopyridine-Induced Methemoglobinemia in a Jehovah’s Witness Treated with High-Dose Ascorbic Acid Due to Methylene Blue Contradictions: A Case Report and Review of the Literature. Hematology Reports. 2023; 15(2):325-330. https://doi.org/10.3390/hematolrep15020034
Chicago/Turabian StyleMenakuru, Sasmith R., Vijaypal S. Dhillon, Mona Atta, Keeret Mann, and Ahmed Salih. 2023. "Phenazopyridine-Induced Methemoglobinemia in a Jehovah’s Witness Treated with High-Dose Ascorbic Acid Due to Methylene Blue Contradictions: A Case Report and Review of the Literature" Hematology Reports 15, no. 2: 325-330. https://doi.org/10.3390/hematolrep15020034
APA StyleMenakuru, S. R., Dhillon, V. S., Atta, M., Mann, K., & Salih, A. (2023). Phenazopyridine-Induced Methemoglobinemia in a Jehovah’s Witness Treated with High-Dose Ascorbic Acid Due to Methylene Blue Contradictions: A Case Report and Review of the Literature. Hematology Reports, 15(2), 325-330. https://doi.org/10.3390/hematolrep15020034





